Abstract
Background
A persistent infection by high-risk human papillomavirus (HR-HPV) is a prerequisite for the development of cervical neoplasms; however, most studies have focused on risk factors associated with HPV-16 and HPV-18 only.
Objectives
We assessed the association of risk factors with the prevalence of HPV-16, HPV-18, and non-16/18 HR-HPV infection and with the occurrence of cervical lesions in the baseline of a cohort study of HPV persistence in a Mexican population.
Methods
Cross-sectional study within the baseline of a 5-year dynamic cohort study of HR-HPV persistence in women with an abnormal cytology study result from 2015 to 2021. HPV DNA was detected using the Anyplex II HPV 28 kit. Data on lifestyle, sociodemographic, and reproductive factors were assessed using bivariate and multivariate analyses to determine the association of risk factors with HR-HPV infection status and histopathologic diagnosis.
Results
A total of 373 women were included in the study. The overall prevalence of HR-HPV infection was 69.97%. The most prevalent HR-HPV genotypes, including single and multiple infections, were HPV-53 (13.4%), HPV-16 (11.8%), HPV-58 (10.9%), HPV-31 (10.9%), and HPV-66 (10.7%). We found 90 multiple HR-HPV infection patterns, all of them with α-6 and -9 species. Significant associations of multiple HPV-16 and non-16/18 HR-HPV infections were found with marital status, number of lifetime sexual partners, and smoking history. The most prevalent genotype in CIN1 and CIN2 patients was HPV-16. No association was found between biological plausibility risk factors and cervical lesions.
Conclusions
The risk factors for non-16/18 HR-HPV multiple infections are no different than those linked to HPV-16 multiple infections.
Introduction
A persistent cervical infection with high-risk human papillomavirus (HR-HPV) is the main etiological factor for the development of precancerous lesions and their progression to cervical cancer. 1 HPV is one of the most common viral agents of sexually transmitted infections in women worldwide, and sexually active women have a 60%–70% lifetime risk of acquiring an HPV infection, without necessarily developing any symptoms. 2 About 90% of HPV infection cases will clear spontaneously within 12–24 months, and only a small fraction of those infections will persist or progress to malignancy. 3
The prevalence of HPV infection in cervical tissues varies depending on whether it is estimated in the open population or in sites with case concentration, such as dysplasia clinics. The worldwide prevalence of HPV infection in women with no cervical abnormalities is 11%–12%. 4 In Mexico, the prevalence reported in two recent studies based on large populations is 11%–13%,5,6 while the prevalence reported in dysplasia clinics is about 25%; 7 however, recent studies have reported a high unusual prevalence of up to 79.2%. 8
A woman’s age at first intercourse, 9 the number of lifetime sexual partners, 10 history of sexually transmitted infections (STIs), and other factors related to sexual activity are associated with exposure to the HPV infection. 11 Older age, high HPV viral load,12,13 and lower socioeconomic and educational status14-16 have been associated with an increased risk of disease in HPV-seropositive women.
Cofactors such as parity, use of hormonal contraceptives for more than 5 years—mainly diethylstilbestrol—17,18 smoking, 19 and concomitant STIs20,21 have been associated with a higher probability of HPV persistence and the appearance of invasive cervical cancer. 22
Most studies have reported factors associated with the prevalence of HPV infection in cervical tissues due to HPV strains with genotype 16 and 18. Meanwhile, little is known about sociodemographic and gynecological-obstetric cofactors associated with infection by other HR-HPV genotypes. Therefore, the objective of this study was to assess the prevalence of HR-HPV infection not only due to HPV-16 and -18, as well as of cervical lesions and risk factors associated, within the baseline cohort study on HPV persistence in the Mexican population.
Material and Methods
Study Design and Population
This is a cross-sectional study nested in the baseline of a 5-year dynamic cohort study of HPV persistence, conducted between 2015 and 2021 in women who attended the Women’s Health Care Center (CAPASAM) of the Health Services of the State of Morelos, Mexico, consecutively after having abnormal results in cytology studies issued by a referring health center. This study meets the STROBE guidelines for cross-sectional studies. 23 Recruitment and eligibility criteria were described in the study protocol, published in 2018.24,25 All women attending the CAPASAM were eligible for recruitment if they: (a) are 25 years old or older according to target population of the Mexican cervical cancer control program or younger women with cervical abnormalities; (b) have resided for 5 years or more in Morelos; (c) agree to participate in the study, sign an informed consent form, complete a questionnaire, and donate cervix exudate sample; (d) have a negative diagnosis for chronic inflammatory or autoimmune diseases at the beginning of the study and during follow-up, and a negative pregnancy status; and (e) received no previous anti-HPV vaccination, nor immunosuppressive treatment within 6 months before.
Assuming a power of 80% and a confidence level of 95%, a sample size of 73 was calculated, of the minimum population necessary to estimate the prevalence of HPV infection in the same population, taking as reference the estimated prevalence of HPV infection at the cervix level in population studies in Mexico of 11%–13%,5,6 with an alternate prevalence reported in dysplasia clinics in Mexico of 25%. (8) Thus, the inclusion of 373 women in the study was determined as sufficient. Calculations were done with Stata v. 16.0 (Stata-Corp, College Station, TX, USA).
Data Collection
At baseline (sampling 1), 373 women completed a structured questionnaire that included questions on lifestyle, sociodemographic, and reproductive factors. All participants agreed to participate in the study by signing an informed consent form. Information from each questionnaire was entered into a database. All data related to the patients were confidential and handled with a numerical code.
The following sociodemographic variables were included: Age (in years), marital status (single, married, concubinage, divorced or separated, widowed), indigenous language speaker (yes/no), educational level (literacy, elementary school, secondary school, high school, college, technical school), occupation (employee/unemployed).
The following variables describing a participants’ gynecological history were included: Age at menarche (age when a participant had her first menstrual period), sexual behavior (age at first intercourse [AFI]), number of lifetime sexual partners [LNSP], use of hormonal contraception (years of duration, current situation), and use of contraceptives (oral contraceptives [OC], injectable contraceptives [IC], condom, infrequent [<5%] contraceptive methods).
The following variables assessing a participant’s obstetric history were included: Parity, number of full-term pregnancies [FTPs]: nulliparous (no birth), primiparous (1 birth), multiparous (≥2 births), history of sexually transmitted diseases [STIs], previous HPV infection (yes/no), previous local treatment of cervical lesions, and familial history of cervical cancer—including consanguinity.
Finally, to explore lifestyle-related behaviors, smoking habit was included as a variable (years, number of cigarettes per day, whether the habit is old or current, previous years of smoking).
Procedures
Cervical swab samples were obtained from each participant who signed the informed consent form and completed the questionnaire, using a cervix brush (Rovers Medical Devices, Oss, Netherlands). The brush was immediately rinsed in a vial with PreservCyt solution (Cytyc Corporation, Marlborough, MA, USA), and the vial was placed in a ThinPrep Processor (Cytyc Corporation). DNA was extracted from cervical exudate following a previously described protocol.24,26 A colposcopic diagnosis was issued by a gynecologist at the consultation time, and pathological analysis was performed by the CAPASAM Pathology Unit.
HPV Genotyping and Pathological Examination
HPV DNA detection and virus load quantification were performed using the Seegene Anyplex™ II HPV 28 system (Seegene, Seoul, Korea), based on a real-time multiplex PCR assay that allows simultaneous amplification, detection, and differentiation of target nucleic acids of 19 high-risk HPV types (16, 18, 26, 31, 33, 35, 35, 39, 45,
Statistical Analysis
A bivariate analysis was performed to determine proportion differences in sociodemographic variables, sexual behavior, obstetric-gynecologic history, and frequency of symptoms between the groups with and without HR-HPV infection (overall HR-HPV prevalence and of each identified HR-HPV types, both in single and multiple infections), using a chi square test for categorical variables. While age and the number of sexual partners were first recorded as continuous, numeric variables, they were analyzed as categorical variables. History of hormonal contraception, smoking habit, symptoms, reason for follow-up, results of last Papanicolaou smear, and diagnosis of premalignant lesions in the population under study were both collected and analyzed as categorical variables. A test for trend across ordered groups (np chi square test) was used to verify whether the number of negative and positive HR-HPV cases followed a linear frequency trend in each group for the variables analyzed. Variables with frequency of less than 5 values were evaluated with Fischer´s exact test.
In the analysis, the subjects were classified as HR-HPV-positive when tested positive for any of the 19 HR-HPV types, including HPV-16 and -18. Positive subjects were then subclassified either as HPV-16- or HPV-18-positive when they were positive to either of both subtypes regardless of any other HR-HPV type, or as non-16/18-HR-HPV-positive when they were positive for other HR-HPV types (26, 31, 33, 35, 39, 45, 51, 52, 53, 56, 58, 59, 66, 68, 69, 73, 82) but not for HPV-16 nor HPV-18.
To determine the association between HR-HPV infection—either overall or segregated by genotype—with risk factors found to be differential distribution in the respective bivariate analysis, and the association of positivity to cervix premalignant lesion with risk factors found to be differential distribution in the respective bivariate analysis, a logistic regression analysis was performed, and 95% confidence intervals (CI) were calculated for odds ratios (OR), adjusting by variables with differential distribution in the bivariate analysis with the exception of exposition variable itself. The number of lifetime sexual partners, sexual behavior, oral contraceptive use, age, history of STIs, and current smoking were the variables used to fit the final model. All statistical analyses were performed with STATA v.16.0 (StataCorp, College Station, TX).
Results
Characteristics of Participants
Baseline Characteristics of Study Population.

n (% of total population, col).
HR-HPV positive n (col): number of cases with at least 1 high-risk HPV genotype detected, including cases with co-infection by 2 or more high-risk HPV genotypes.
HR-HPV prevalence (row), is defined as the percentage of subjects with at least 1 high-risk HPV genotype detected by specific stratum for each evaluated characteristic.
Use of other contraceptive method: coitus interruptus, intrauterine device, bilateral tubal occlusion, vasectomy, subdermal implant, or not using any.
Herpes simplex virus type 2 test was performed in a StepOne Rea-Time PCR system, using the SYBR select master mix.
Of these women, 76.4% reported not having used tobacco at any time in their lives. Smoking participants (23.5%) reported having smoked 1–5 cigarettes per week. The mean age at menarche was 12.7 ± 1.5 years; the average age at first intercourse was 17.6 ± 3.1 years; the mean number of lifetime sexual partners was 2.4 ± 1.8; and the mean number of pregnancies to term was 3.1 ± 2.04. Only 13.6% of participants had used hormonal contraceptives at any time. Interestingly, 9.3% of participants reported having had a sexually transmitted infection at some point in their lives, but a confirmatory test revealed that 28.2% of them were positive for herpes simplex virus type 2 (HSV-2); the prevalence of HSV-2 infection was 68.5% in HR-HPV positive women. About 56% of HR-HPV infected women had a moderate viral load, 10,000–49,000 total viral total copies. The highest proportion of women were overweight and obese, according to their body mass index (Table 1).
Overall HR-HPV Prevalence
The overall prevalence of HR-HPV infection, defined as the percentage of women with abnormal results in cervix cytology tests from the CAPASAM dysplasia clinic and with at least 1 high-risk HPV genotype detected, was 69.97%. The prevalence of single HR-HPV infection with any genotype was 37.5%, while the prevalence of coinfection by HR-HPV with any genotype was 32.9%. The prevalence of HPV-16, HPV-18, or non-16/18 HR-HPV single infection was 5.9%, 2.9%, and 27.6%, respectively. The prevalence of coinfection with HPV-16, HPV-18, or non-16/18 HR-HPV was 5.6%, 2.9%, and 24.4%, respectively (Figure 1). Distribution of high-risk HPV genotypes in single and multiple infection.
HR-HPV Prevalence by Age
The prevalence of HR-HPV with any genotype by age group shows a bimodal distribution with an increased prevalence for the women aged 15–29 years, while the number of positive cases decreased in middle-aged subjects and increased again in women older than 55 years (Figure 2). The age group with the highest prevalence of HR-HPV infection was 25–29 years (81.4%). Women aged 30–49 years had a higher prevalence of single HR-HPV infections (71.4%), whilst the highest prevalence of HR-HPV multiple infection was found in the group aged 15–29 years (41.8%) (Figure 2). Age-specific prevalence of any HR-HPV genotype. HR-HPV prevalence is defined as the percentage of women with at least one high-risk HPV genotype detected (including coinfections with other HR-HPV genotypes).
Distribution of HR-HPV Genotypes
The most prevalent HR-HPV genotypes in our study population, both as single infections and as coinfections, were HPV-53 (13.4%), HPV-16 (11.8%), HPV-58 (10.9%), HPV-31 (10.9%), and HPV-66 (10.7%). The HR-HPV genotypes with the highest prevalence in single infections were HPV-16 (5.8%), HPV-58 (4.5%), HPV-31 (3.2%), HPV-66 (3.2%), and HPV-18 (2.9%). The genotypes with the highest prevalence in coinfections were HPV-53 (11.5%), HPV-31 (7.7%), HPV-66 (7.5%), HPV-58 (6.4%), HPV-16 (5.6%), and HPV-59 (5.6%) (Figure 3). HPV-26 and HPV-69 were only found in single infections, while HPV-73 and HPV-82 were only found in coinfection. HR-HPV genotypes were found to infect the subjects in 90 possible combinations. The genotypes with the highest number of combinations were HPV-53, HPV-31, HPV-58, HPV-16, and HPV-66 (Figure 4). Distribution of prevalence of high-risk HPV genotypes for single and multiple infection. Heat map: Patterns of high-risk HPV infection. Heat map of the patterns of multiple HR-HPV infections. Horizontal and vertical axes represent the 28 high-risk genotypes that were evaluated in each patient. Blue cells indicate a higher number of combinations, and yellow cells indicate a lower number of combinations for each infection pattern. The color scale progresses from yellow to blue as the number of patterns per genotype increases. As shown, genotype 53 has the highest number of combinations.

Factors Related to Sociodemographic, Lifestyle, and Gynecological-Obstetric Information Associated with HR-HPV Prevalence
Clinical and Sex-Behavioral Characteristics in Genotype-Specific Prevalence of Single and Multiple HR-HPV Infections.

Use of other contraceptive methods: coitus interruptus, intrauterine device, bilateral tubal occlusion, vasectomy, subdermal implant, or not using any.
Herpes simplex virus type 2 test was performed in a StepOne Real-Time PCR system, using the SYBR select master mix.
Previous sexually transmitted diseases were self-reported by the patient.
HR-HPV positive n (%, col): number of cases with at least 1 high-risk HPV genotype detected, including cases with co-infection by 2 or more high-risk HPV genotypes.
HR-HPV prevalence (%, row), is defined as the percentage of subjects with at least 1 high-risk HPV genotype detected by specific stratum for each evaluated characteristic.
Values in bold type indicate significant P-values (P < .05) chi square test nptrend to determine differences in proportions between groups with and without HR-HPV infection for categorical variables.
Variables with cell <5 values were evaluated with Fischer´s exact test. Values in bold type indicate significant P-values (P < .05).
Bivariate analyses segregated by viral genotype showed a differential distribution in hormonal contraceptive use and current smoking habit in patients with single HPV-66 infections. Meanwhile, significant differences were observed in marital status among subjects with multiple infections by the genotypes HPV-16, non-16/18 HR-HPV, HR-HPV-58, HPV-51, and HPV-39. The number of sexual partners was significantly different in women with multiple infections by the genotypes HPV-16, non-16/18 HR-HPV, HPV-58, HPV-31, and HPV-53. A differential distribution of smoking history was found in patients coinfected with the genotypes HPV-16, non-16/18 HR-HPV, HPV-58, and HPV-53 with respect to negative subjects. Finally, the number of pregnancies was significantly different in patients coinfected with non-16/18 HR-HPV, HPV-58, and HPV-39 with respect to negative cases (Table S1).
Association of HR-HPV Infection With Sexual Health and Behavior-Related Variables.
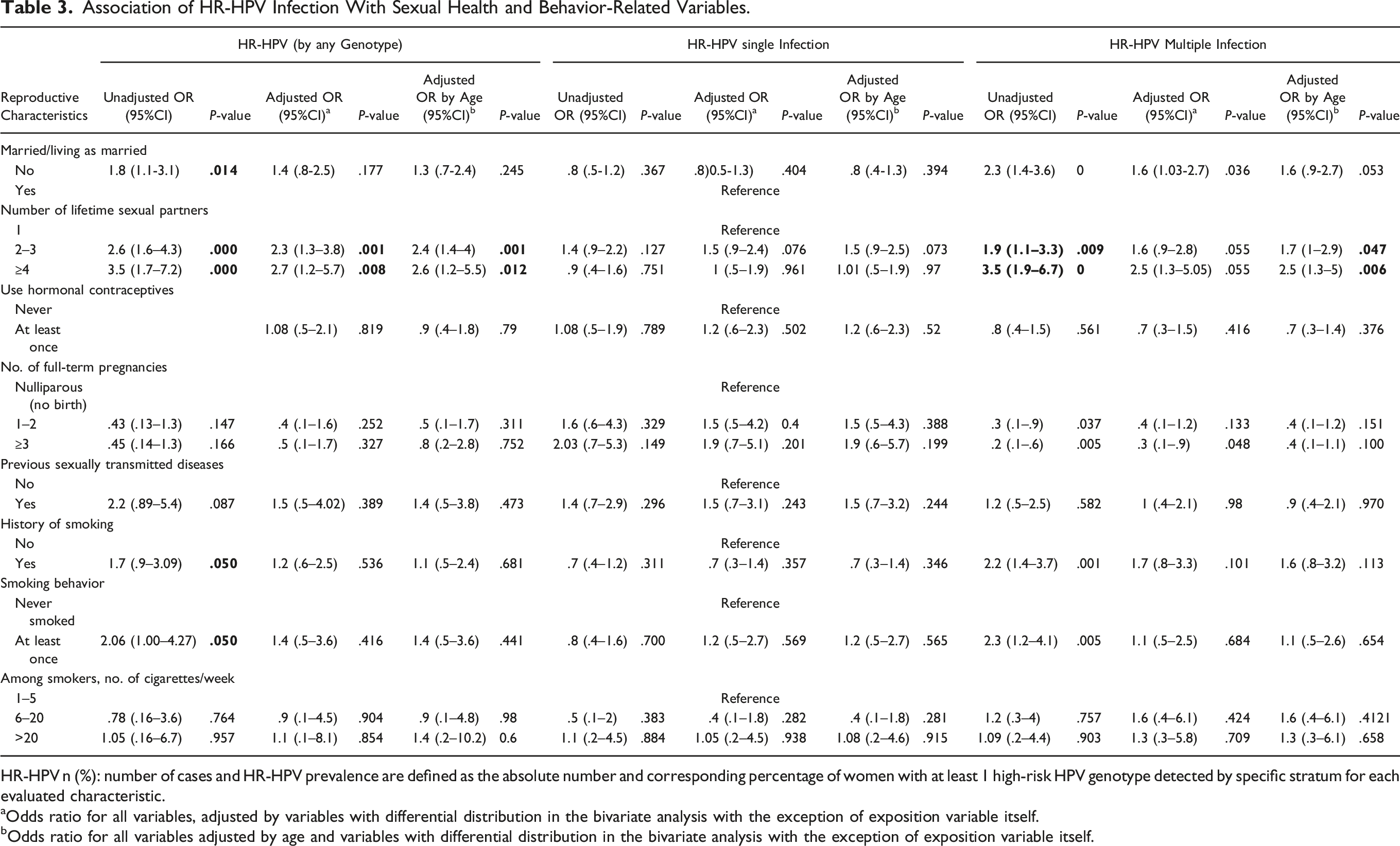
HR-HPV n (%): number of cases and HR-HPV prevalence are defined as the absolute number and corresponding percentage of women with at least 1 high-risk HPV genotype detected by specific stratum for each evaluated characteristic.
aOdds ratio for all variables, adjusted by variables with differential distribution in the bivariate analysis with the exception of exposition variable itself.
bOdds ratio for all variables adjusted by age and variables with differential distribution in the bivariate analysis with the exception of exposition variable itself.
Distribution and Association of HR-HPV Genotypes and Risk Factors per Histopathological Status
Prevalence of High-Risk HPV Infection by Histopathologic Diagnosis.
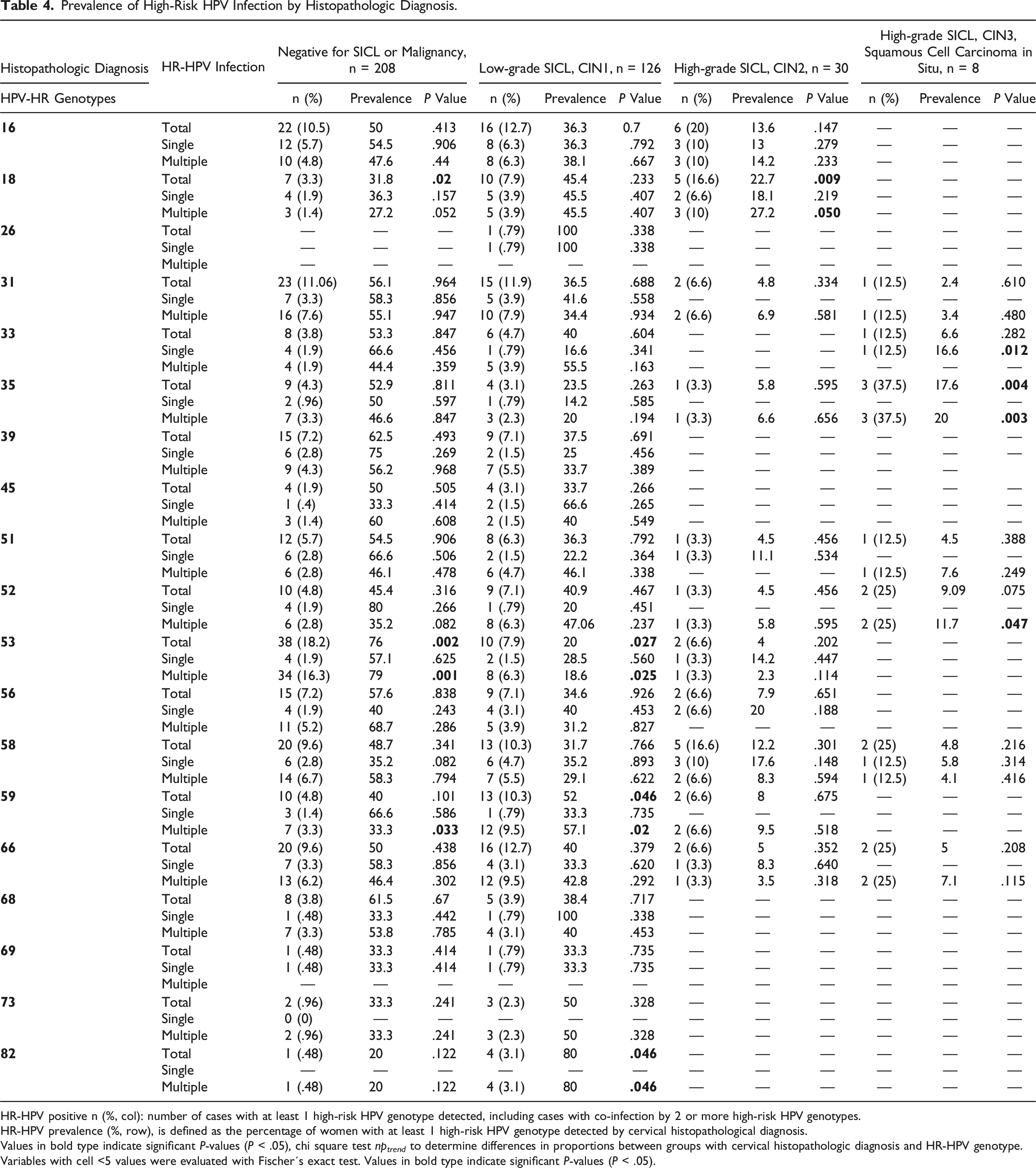
HR-HPV positive n (%, col): number of cases with at least 1 high-risk HPV genotype detected, including cases with co-infection by 2 or more high-risk HPV genotypes.
HR-HPV prevalence (%, row), is defined as the percentage of women with at least 1 high-risk HPV genotype detected by cervical histopathological diagnosis.
Values in bold type indicate significant P-values (P < .05), chi square test np trend to determine differences in proportions between groups with cervical histopathologic diagnosis and HR-HPV genotype.
Variables with cell <5 values were evaluated with Fischer´s exact test. Values in bold type indicate significant P-values (P < .05).
A bivariate analysis of histopathology diagnosis and coinfection by each genotype showed significant differences in HPV-53 and HPV59 positivity in patients with CIN1; significant differences were also observed in patients with CIN2 and with infection by HPV-18, and in patients with CIN3 with respect to infection by HPV-33, HPV35, and HPV-52. No significant differences in histopathology diagnosis were observed in patients with single infections (Table 4).
Sociodemographic, Sexual, and Reproductive Characteristics of Patients and Cervical Cancer Stage.
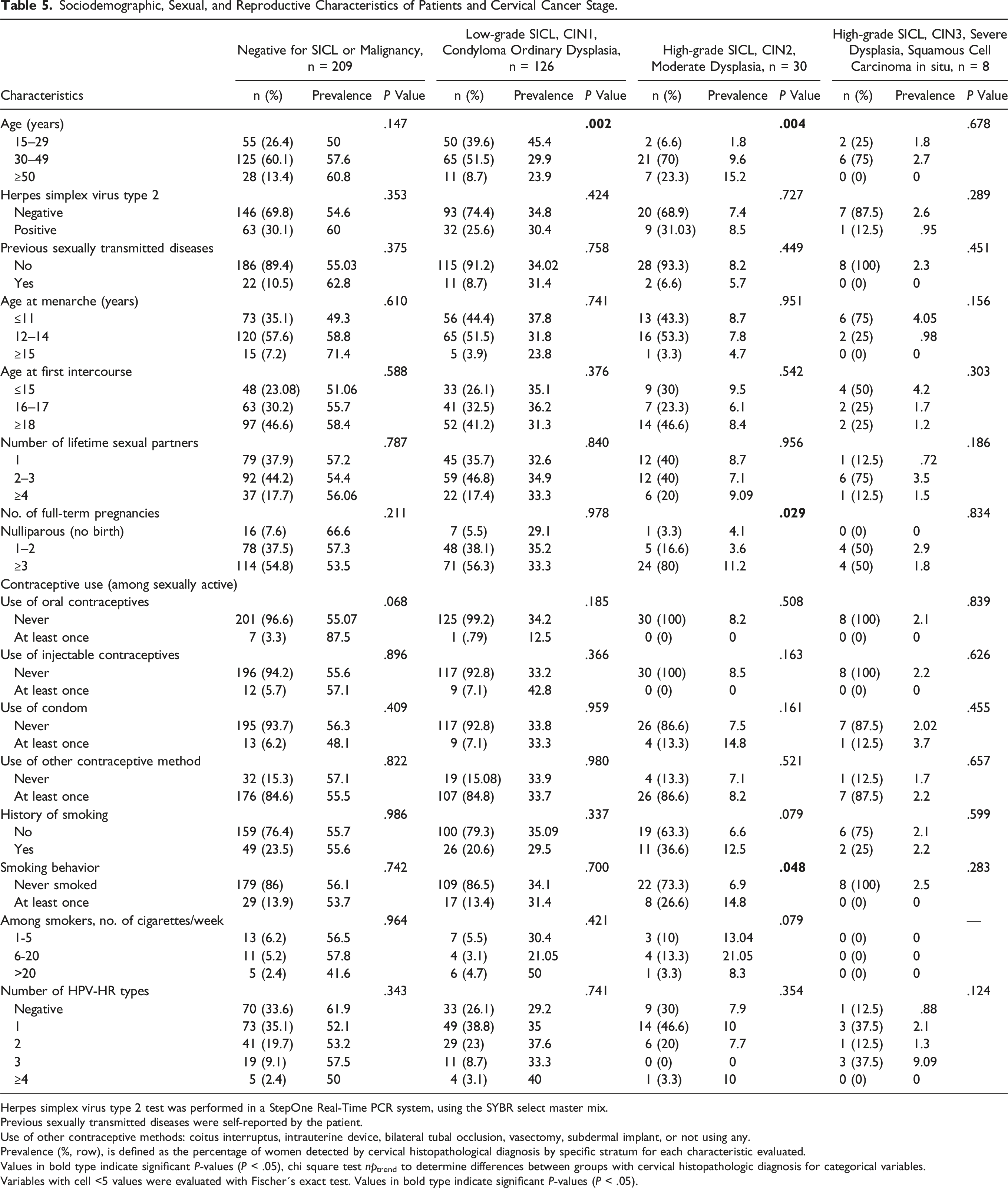
Herpes simplex virus type 2 test was performed in a StepOne Real-Time PCR system, using the SYBR select master mix.
Previous sexually transmitted diseases were self-reported by the patient.
Use of other contraceptive methods: coitus interruptus, intrauterine device, bilateral tubal occlusion, vasectomy, subdermal implant, or not using any.
Prevalence (%, row), is defined as the percentage of women detected by cervical histopathological diagnosis by specific stratum for each characteristic evaluated.
Values in bold type indicate significant P-values (P < .05), chi square test nptrend to determine differences between groups with cervical histopathologic diagnosis for categorical variables.
Variables with cell <5 values were evaluated with Fischer´s exact test. Values in bold type indicate significant P-values (P < .05).
Association Between Cervical Cancer Stage and Risk Factors.

HR-HPV n (%): Number of cases and HR-HPV prevalence are defined as the absolute number and the corresponding percentage of women with at least 1 high–risk HPV genotype detected by specific stratum for each characteristic evaluated.
Values in bold type indicate significant P-values (P < .05) for OR.
Statistically significant P-values ≤.05 of OR are marked in bold type.
aOdds ratio for all variables, adjusted by age.
Discussion
Cervical cancer was the fourth most important neoplastic disease in Mexico in females of all ages in 2020, according to GLOBOCAN. 27 Persistence of HR-HPV infection is a prerequisite for the modification of the squamous epithelium and progression to a preneoplastic lesion, which may result in cancer. 1 As the quadrivalent HPV vaccine (Gardasil, Merck Sharp & Dohme) only prevents infection by high-risk genotypes 16 and 18, observational studies evaluating the prevalence of other high-risk genotypes could help us to design new vaccines targeting most prevalent genotypes by region, increasing the coverage against non-16/18 genotypes. This study provided additional information on the relative distribution of HR-HPV genotypes other than HPV-16 and -18, and the risk factors associated in a Mexican population.
The overall prevalence of HR-HPV infection in women with precancerous lesions who were referred from first-level health centers was 69.8%, a value similar to that reported in other cross-sectional studies conducted in dysplasia clinics in some regions of Mexico (60%–90%);28-32 these rates are lower than those reported in studies with regional representativeness (24.78%), 7 but they are higher than the prevalence in the open population in Mexico.5,6 The prevalence of HR-HPV infection age-specific in our population is distributed as a “U-shaped” curve. In contrast with other studies in Mexico and worldwide,4,28,33,34 the first peak occurs at ages in the range 25–29 years, with a decline in the number of cases in middle-aged women (30–49 years); the second peak, observed after menopause, could be explained by the reactivation of a latent HR-HPV infection linked with a decline in the immune response.35,36 Infection rates are strongly dependent on the number of sexual partners and a patient’s sexual behavior. Higher transmission rates result from the increase in the number of cases in young women, aged 20–25 years. This peak is followed by a decline in the number of cases due to a spontaneous elimination of infection resulting from a successful immune response and a lesser exposure to new sexual partners as the age of the patients increases.11,37,38
The most prevalent HR-HPV genotypes in women attending the CAPASAM dysplasia clinic in 2015–2021 were HPV-53, HPV-16, HPV-58, HPV-31, HPV-66, and HPV-56. Our results showed that HPV-16 and HPV-18 were replaced by other HR-HPV types in the Mexican population. These results are comparable with other studies conducted in Mexico.4,28,31,39,40 The genotype distribution pattern showed that the most prevalent combinations were 31-53, 35-53, 53-58, and 33-35. Since these genotypes belong to the α-6 and α-9 species, this could suggest that phylogenetically related HR-HPV genotypes tend to cluster together, in agreement with reports by other authors.41-43 Senapati et al. reported that patients coinfected with phylogenetically related genotypes had a 2.9-fold increased risk of invasive cervical carcinoma with respect to other genotypes; thus, phylogenetically-driven, specific interactions between genotypes in multiple infections could increase or decrease the risk of cervical cancer. 44
Variables related to sexual behavior, such as marital status and the number of lifetime sexual partners, along with history of smoking, were associated with HR-HPV multiple infection by any genotype, in agreement with findings of other authors.45-47 However, no association was observed between these risk factors and single infection by any HR-HPV genotype.
When we evaluated the association of various risk factors with infections by HPV-16, HPV-18, and non-16/18 HR-HPV, no association was observed with HPV-18 positivity, neither as single nor multiple infection.
Single women had a higher risk of acquiring multiple infections by HPV-16 and oncogenic non-HPV-16/18 than married or cohabiting women. This result had already been reported by Kops et al 47 for multiple infections by the genotypes HPV-39, HPV-51, and HPV-58; in that study, the incidence of multiple infections was 1.29 times higher among single subjects compared with respect to married or cohabiting participants.
A positive association was found between having had more than 4 sexual partners and multiple infections by HPV-16 and non-16/18 HR-HPV, specifically HPV-31, HPV-53, and HPV-58; this is in line with the findings of other authors, who reported that unmarried subjects and those who had had a greater number of sexual partners over 5 years showed higher rates of multiple HPV infections. 47
The positive association between smoking history and multiple infections by HPV-16 and non-16/18 HR-HPV, specifically HPV-53 and HPV-58, has also been reported by other authors.48,49 Several components of cigarette smoke and their metabolites have been detected in cervical mucus and may interact with HPV. Benzo[a]pyrene increased the number of HPV genome copies, modulated HPV DNA replication, and accelerated the HPV replication cycle. 50 Nicotine and nicotine-derived nitrosamines such as 4-(methylnitrosamino)-1-(3-pyridyl)-1-butanone impair the defense mechanisms in cervical epithelium. 51 Xi et al. found that baseline viral load was higher among current smokers that in women who never smoked. 52
On the other hand, in our study, the number of pregnancies was a protective factor for positivity to multiple non-16/18 HR-HPV infections. Specifically, the risk of multiple HPV-39 and HPV-58 infections was lower in women who had at least 1 pregnancy. This could be explained by the fact that pregnant women are less likely to have new sexual partners, which decreases their exposure to HR-HPV infection. However, other authors have reported that having >7 full-term pregnancies increased the risk of invasive cervical carcinoma compared to women who had 1-2 full-term pregnancies. This has been attributed to the hormonal profile during pregnancy and eversion of the columnar epithelium over the ectocervix (ectopia) due to birth-related trauma, which favors the exposure of the squamocolumnar junction to HR-HPV infection.17,53 In agreement to this, no differences were observed between risk factors for HPV-16 infection or non-16/18 HR-HPV multiple infections.
With regard to the prevalence of HR-HPV infection by histopathological diagnosis, we found that the prevalence of infection due to any HR-HPV genotype increased directly with the severity of cervical lesions, which is consistent with other studies in Mexico.28,29,39,54 Interestingly, the lack of significant differences in the prevalence of single or multiple HR-HPV infections between patients with differing histopathological diagnoses in our study are in contrast with reports in other studies, that high-grade cervical lesions are linked with simple infections.31,55
The distribution of HR-HPV genotypes showed that HPV-16 predominated in patients diagnosed with CIN1 and CIN2, whereas the most prevalent genotype in patients with CIN3 was HPV-35. The higher prevalence of single and multiple HPV-16 infections in women with CIN1 and CIN2 indicates a higher oncogenic capacity than that of other genotypes, as reported by Padilla et al; according to that author, HPV-16 infection led to a 10.3-fold increase in the risk of developing cervical cancer. 31 Association with current smoking habit was found only in the CIN2 group. This association has also been observed by other authors, who reported that current smoking increases the risk of developing CIN2 by a factor of 1.89, as the rate of HR-HPV infection increased. 56
The main strengths of this study are the identification of risk factors associated with non-16/18 HPV infection, in addition to determining the prevalence of non-16/18 HPV infections in dysplasia clinics in central Mexico. The main limitation of our study is that the risk factors associated with premalignant lesions could be underestimated, since the sample size of women with premalignant lesions in the baseline study was small. Additionally, our population is not representative of other regions of the world, so these results are not generalizable to other populations.
In conclusion, the prevalence of single and multiple HR-HPV infections revealed a predominance of non-16/18 multiple HR-HPV infections. The risk factors for acquiring non-16/18 HR-HPV multiple infections are no different than those linked to HPV-16 multiple infections. Finally, HPV-16 was the most prevalent genotype in low- and high-grade cervical lesions. Our results describe the characteristics of a population in a dysplasia clinic, and these data could help us to determine the genotype circulation and identify women at a higher risk to develop cervical lesions.
Supplemental Material
Supplemental Material - Prevalence and Risk Factors for High-Risk Human Papillomavirus Infection and Cervical Disorders: Baseline Findings From an Human Papillomavirus Cohort Study
Supplemental Material for Prevalence and Risk Factors for High-Risk Human Papillomavirus Infection and Cervical Disorders: Baseline Findings From an Human Papillomavirus Cohort Study by Paula Saldaña-Rodríguez, Margarita Bahena-Román, Karina Delgado-Romero, Vicente Madrid-Marina, and Kirvis Torres-Poveda in Cancer Control
Supplemental Material
Supplemental Material - Prevalence and Risk Factors for High-Risk Human Papillomavirus Infection and Cervical Disorders: Baseline Findings From an Human Papillomavirus Cohort Study
Supplemental Material for Prevalence and Risk Factors for High-Risk Human Papillomavirus Infection and Cervical Disorders: Baseline Findings From an Human Papillomavirus Cohort Study by Paula Saldaña-Rodríguez, Margarita Bahena-Román, Karina Delgado-Romero, Vicente Madrid-Marina, and Kirvis Torres-Poveda in Cancer Control
Footnotes
Acknowledgments
The authors thank Juraj Lord for assistance in the preparation of the manuscript. This work was submitted in partial fulfillment of the requirements for the PhD degree of Paula Saldaña-Rodríguez from the Doctoral Program in Sciences of the School of Public Health of Mexico.
Author Contributions
KTP conceived the idea and had a full role in the conceptualization and design of the study, formal analysis, draft writing, and revision of the manuscript; PSR participated in the collection of clinical materials, analysis of the results, and manuscript draft. KDR participated in the selection of study population, gynecological specimen sampling, and patient care. MBR carried out HPV genotyping analyses. VMM participated in the analysis of the results and manuscript draft. All authors have read and agreed to the published version of the manuscript.
Conflict of Interest Statement
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Instituto Nacional de Salud Pública, Mexico, and grants from the Consejo Nacional de Ciencia y Tecnología (CONACYT, Mexico) to KTP (FONSEC SSA/IMSS/ISSSTE-2018 grant # A3-S-49099) and to VMM (grant CATEDRAS-2014-C01-#245520).
Ethical Statement
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
