Abstract
Background
Colorectal cancer is one of the most common cancers in the world, with about one million cases diagnosed annually. Various treatment methods can be used to treat colorectal cancer, including chemotherapy with different drug regimens. Considering the need to opt for more effective and less expensive drugs in the treatment of this disease, the present study aimed to compare the cost-effectiveness of FOLFOX6+Bevacizumab with FOLFOX6+Cetuximab in patients with stage IV colorectal cancer referred to medical centers in Shiraz, Iran, in 2021.
Materials and Methods
Using a decision tree, the cost-effectiveness and cost-utility of the 2 drug regimens were compared in all studied patients through the census method. Having a societal perspective, this study considered direct medical costs, direct non-medical costs, and indirect costs. The effectiveness indicators included the rate of major response to the drug combination used and the Quality-adjusted Life Year (QALY). The data were analyzed using Treeage 2011 and Excel 2016 software. In order to ensure the robustness of the results, one-way and probabilistic sensitivity analyses were performed as well.
Results
The results showed that the expected costs, the effectiveness (major response rate), and the QALYs of the FOLFOX6+Bevacizumab drug regimen were $16746.13(USD), .49, and .19, respectively, and those of the FOLFOX6+Cetuximab regimen were, respectively, $15191.05 (USD), .68, and .22. Therefore, FOLFOX6+Cetuximab compared to FOLFOX6+Bevacizumab was less costly and more effective and had a greater QALY, thus being considered as the dominant option. Also, the results of the sensitivity analyses showed that there was a bit of uncertainty.
Conclusion
Considering that the FOLFOX6+Cetuximab regimen was more cost-effective, it is suggested to be prioritized in preparing clinical guidelines for Iranian colorectal cancer patients. In addition, increasing the basic and supplementary insurance coverage for this drug combination as well as the use of remote technology to guide patients by oncologists can be solutions to reduce direct and indirect costs of the patients.
Background
Colorectal cancer is caused by the abnormal proliferation of intestinal mucosal cells and consequently, the interaction between environmental and genetic factors. 1 Exposure to these factors as well as the formation and persistence of defective DNA causes mutations and genetic changes. As a result, this process leads to cell transformation and growth, and eventually colorectal cancer. 2 In stage IV of this type of cancer, the disease metastasizes to distant parts and possibly affects the organs such as the liver and lungs. The liver is one of the most common sites of metastasis and the main cause of death in this disease. 3
Colorectal cancer is one of the most common cancers in the world, with almost one million people being diagnosed every year. 4 It is the third most common cancer in the world after lung and breast cancers, and the fourth cause of death from cancer after lung, liver, and stomach cancers. 5 Furthermore, colorectal cancer is the fourth most common cancer among Iranian men after stomach, prostate, and lung cancers, and the second after breast cancer among Iranian women. 6 The death rate from colorectal cancer is increasing in Iran, and at older ages, the death rate from this cancer increases as well. 7 The World Health Organization has predicted a 77% increase in the diagnosis of new cases and an 80% increase in deaths from colorectal cancer by 2030.8,9
On the other hand, the type of treatment method is selected based on the staging of the disease. Nowadays, chemotherapy and sometimes radiotherapy (in rectal cancers) are used in certain stages of the disease. 10 The treatment regimen to be selected depends on several factors, including the treatment previously received and the state of the tumor change. 11 In advanced colorectal cancer, liver metastases are non-resectable in 85% of patients due to the site of involvement as well as the progression of the disease. In the case of non-resectable liver metastases, palliative chemotherapy increases patient survival and quality of life. 3 In fact, it can be stated that the treatment goals for these patients are usually palliative rather than curative, with the exception of a small number of stage IV colorectal cancer cases in which metastasis is limited to the liver and may be treated with surgery. 12
In advanced colorectal cancer, available drugs are categorized into 3 main treatment groups as follows: 1- cytotoxic agents such as Fluoropyrimidines, Irinotecan, and Oxaliplatin, which are common active chemotherapy drugs; 2- angiogenesis inhibitors such as Bevacizumab; and 3- epidermal growth factor receptor (EGFR) antibodies such as Cetuximab and Panitumumab. 11
In this study, FOLFOX6+Bevacizumab and FOLFOX6+Cetuximab were investigated and compared.
FOLFOX consists of Folinic Acid (Leucovorin Calcium), Fluorouracil (5-FU), and Oxaliplatin. This drug shrinks cancerous tumors and helps reduce the symptoms of colorectal cancer. 13 It enters the bloodstream intravenously. 14 Bevacizumab is a recombinant human monoclonal antibiotic that inhibits the activities of all isoforms of the vascular endothelial growth factor (one of the main angiogenesis growth factors). Neutralization of the biological activity of the growth factor in the vascular layer reduces tumor angiogenesis and prevents its growth. Bevacizumab is slowly injected into the vein (intravenously). 11 Cetuximab is an immunoglobulin G1 monoclonal antibody that specifically targets EGFR and binds to the extracellular chain of EGFR, neutralizing the cause of cancer cells. Cetuximab is administered through intravenous injection. 15
On the other hand, the results of various studies indicated very high costs of treating colorectal cancer in stage IV.16-18 Chemotherapy drugs account for the largest share of the total costs paid by cancer patients.19-21 The fact that many chemotherapy drugs are imported and the effect of inflation and currency price changes are among the reasons for the high cost of chemotherapy drugs. 19 Therefore, it is necessary to choose more effective and less expensive drugs in the health sector and to determine priorities, which is possible by conducting economic evaluations. 22 An economic evaluation is a systematic and transparent analysis of various interventions in a field in order to examine and compare the costs and outcomes of those interventions. 23 Among the economic analysis tools used in the health sector are cost-effectiveness and cost-utility analyses. 22 In the cost-effectiveness analysis, program outcomes are measured in the most appropriate units of natural or physical effects, such as years of life gained or cases correctly diagnosed. 22 The cost-utility analysis is a form of cost-effectiveness analysis in which utility is used instead of natural units to measure the outcomes. This kind of analysis is performed when the health-related quality of life (HRQOL) is considered an important outcome. 24
Some studies addressed and compared the costs25-28 or effectiveness29-32 of the drugs investigated in this study separately. Some others investigated the cost-effectiveness of various drug combinations used in the treatment of stage IV colorectal cancer (not the drug combinations examined in the present study).33-37 However, there are few studies comparing the cost-effectiveness and cost-utility of the drug combinations studied in this research, one of which is the study of Shida et al. (2018) who conducted an economic evaluation of first-line drug regimens based on mFOLFOX6, mFOLFOX6+Bevacizumab, mFOLFOX6+Cetuximab, and mFOLFOX6+Panitumumab from the perspective of the public medical care. They suggested that the rates of overall survival and post-progression survival were higher for the patients receiving mFOLFOX6+Cetuximab, but mFOLFOX6+Pmab was the most cost-effective drug regimen in their study. 38
Therefore, since the researchers could not find any comprehensive research to determine and compare the cost-effectiveness of FOLFOX6+Bevacizumab and FOLFOX6+Cetuximab drug regimens from the societal perspective for the treatment of the patients with stage IV colorectal cancer, the present study aimed to compare the cost-effectiveness of the aforementioned drug regimens for the treatment of the patients with stage IV colorectal cancer who were referred to medical centers in Shiraz, Iran, in 2021 in order to determine more cost-effective drug combination for these patients and provide useful information to specialists and pharmacists as well as the managers and decision-makers of the health system.
Materials and Methods
Study Population/Patients
This is an applied, descriptive-analytical, and full economic evaluation study (cost-effectiveness study) conducted as a cross-sectional research on patients aged 18–85 years with K-RAS and N-RAS wild-type unresectable stage IV colorectal cancer who were referred to medical centers in Shiraz, Iran, in 2021. The research time period was one year, that is, 2021, and the follow-up period was up to 6 months. 39
The research population of this study comprised 88 individuals, all of whom were selected and studied through the census method. The patients were classified into 2 groups, patients treated with FOLFOX6+Bevacizumab (N = 47) and those treated with FOLFOX6+Cetuximab (N = 41). The patients who were continuously receiving treatment and consented to participate in the study were included in the research, and those with heart, liver, or kidney failure were excluded.
It should be noted that the reporting of this study conforms to CHEERS guidelines and the study has been reported according to this checklist. 40
Input Parameters
Costs
The present study was conducted from the societal perspective, and all direct medical, direct non-medical, and indirect costs were examined. The societal perspective is the most comprehensive perspective due to the inclusion of all direct medical costs and indirect costs and allows a full analysis of all costs attributable to the disease. In general, the societal perspective is preferred over other perspectives, because the disease does not only affect one person or organization that is directly involved; thus, this perspective deals with the patients, their families, the health system, insurance organizations, etc. that are involved. 41
In this study, the “bottom-up” method was used to calculate the costs. In the bottom-up approach, the minor activities and the data collected at the patient level are used to estimate the costs. 42
To collect the required cost data, a researcher-made data collection form was used. It had been designed based on the research objectives, the review of the literature, and consultation with oncologists and health economists. The first part of the form dealt with demographic information of the patients (including age, gender, place of residence, marital status, being the head of the family or not, education level, etc.), and the cost data were recorded in the second part of the form.
The direct medical costs in this study included the costs of physician’s visits, drugs and medicines, laboratory tests, radiography, consumables, port insertion, hospital hotelling, etc. which were obtained from interviewing the patients or their families and referring to the bills they had paid and their medical records. In order to find the exact price of the drugs, the researchers referred to the Deputy of Food and Drug of Shiraz University of Medical Sciences.
The direct non-medical costs included the mean cost of accommodation, transportation within and outside the city, food, purchase of auxiliary tools and equipment such as wheelchairs, walkers, crutches, abdominal braces, etc., childcare and kindergarten, telephone and internet contact with the family, and the cost of changes in the environment and living space for the patient’s adaptation to the disease, such as making toilets, elevators, etc. These costs were determined through interviews with the patients or their families.
The indirect costs of the disease also included absenteeism and potential productivity lost due to the disease or patient care, which were collected through direct questions from the patients and their companions. The indirect costs based on productivity lost were calculated using the human capital method, and the individuals’ wages were used to calculate lost income. According to the Labor Department’s law of 2021, the minimum wage was considered to be equivalent to 18.12 USD per day and every 8 working hours was equivalent to 1 working day. 43
It is worth noting that in the present study, there was no patient death due to the use of the investigated drug combinations. Therefore, there was no potential productivity lost due to the premature death of the patient.
To convert the costs from Iranian Rials to USD, one US dollar was considered equal to 42,000 Rials. 44 Given that the time period of the study was one year, the discount rate was not considered.
Health Outcomes (Effectiveness and QALY)
The effectiveness index in the present study was the rate of major response to the drug combination used. Measuring and evaluating the effectiveness index was based on the tumor shrinkage. The effectiveness data were collected 2 weeks after the completion of drug therapy.
Another part of the data was related to measuring the utility of the patients in order to calculate their QALYs. Once the utility was determined, the QALY was obtained by multiplying the time spent in a certain condition by the utility associated with that condition. 45 In the present study, the utility values needed for QALY calculation were extracted from the EuroQol-5 Dimension (EQ-5D). The weights of this scale had been estimated by Gudarzi et al. for Iran. 46 The utility data were collected immediately after the completion of the drug treatment period.
Model Structure
In this study, the Treeage 2011 software was used to draw the following decision tree (Figure 1): Decision tree in the treatment of studied stage IV colorectal cancer patients.
In the above decision tree, the patients were first divided into 2 groups based on the type of drug regimens, and finally, they were classified into the major response (>20%) and no response (<20%) groups based on their responses to the drug combinations. 47
After the decision tree was drawn, the incremental cost-effectiveness ratio (ICER) and the incremental cost-utility ratio (ICUR) were calculated by dividing the difference in costs by the difference in effectiveness (the tumor shrinkage) and by dividing the difference in costs by the difference in QALYs of the 2 drug combinations, respectively. If the value of either ICER or ICUR was negative, one of the regimens would be more cost-effective than the other, and if positive, the ICER or ICUR had to be compared with the threshold in order to make a decision.
Willingness-To-Pay
To calculate the willingness-to-pay (the threshold), the World Health Organization’s method, that is, one to three times the GDP per capita, was used. 48 It should be noted that one-time GDP per capita was used in the present study. Iran’s GDP per capita (the threshold) in 2021 which was equal to $16,783 49 was considered as the amount of willingness to pay.
Sensitivity Analysis
In order to increase the accuracy and deal with uncertainty in this study, the one-way definitive sensitivity analysis (tornado diagram) and probabilistic sensitivity analysis were performed. In addition, the cost-effectiveness acceptability curve was drawn to show the probability of being cost-effective in different amounts of willingness to pay. The mean, standard deviation, and distribution of the input parameters used in the decision tree model of the study have been provided in Appendix 1.
The collected data were analyzed using Treeage 2011 and Excel 2016 software.
Ethical Considerations
This study was approved by the Ethics Committee of Shiraz University of Medical Sciences on 2021.05.01(Code: IR.SUMS.REC.1400.073). The patients were free to choose to participate in the study, and once the objectives of the research were explained to them, their written informed consent was obtained. The questionnaires and checklists were completed anonymously and the patients were assured of the confidentiality of their answers to the questions. It should be noted that in order to comply with ethical considerations and the confidentiality of patient information, the patients were distinguished by the codes at the top of the cost information forms and the effectiveness and utility checklists.
Results
Demographic Characteristics of the Studied Patients (N = 88).

Mean Annual Direct Medical, Direct Non-Medical, and Indirect Costs per studied Colorectal Cancer Patients in 2021 (USD).

According to Table 2, the mean direct medical costs in the study year were 14,673.23 USD for the patients treated with FOLFOX6+Bevacizumab and 13,412.22 USD for the ones treated with FOLFOX6+Cetuximab. The consumed drugs accounted for the highest direct medical costs, with 8673.05 USD (59.1%) in the FOLFOX6+Bevacizumab group and 7253.89 USD (54.08%) in the FOLFOX6+Cetuximab.
As presented in Table 2, the mean annual cost of using direct non-medical services per patient treated with the FOLFOX6+Bevacizumab regimen was 769.91 USD, and 560.21 USD per patient treated with the FOLFOX6+Cetuximab. The highest portion of direct non-medical costs in the group treated with FOLFOX6+Bevacizumab was 331.44 USD (43.05%) for the accommodation of the patients and their companions, and it was 171.56 USD (30.61%) in the group treated with FOLFOX6+Cetuximab for the transportation of the patients and their companions.
According to the results presented in Table 2, the mean annual indirect costs per patient treated with FOLFOX6+Bevacizumab and FOLFOX6+Cetuximab regimens were 1302.42 USD and 1225.37 USD, respectively, most of which were related to the cost of absenteeism of the patient’s companions for accompanying and caring for the patient (832.55 USD (63.92%) and 914.63 USD (74.64%), respectively).
The Results Related to the Studied Drug Regimens’ Response Rates, QALYs, and Costs.
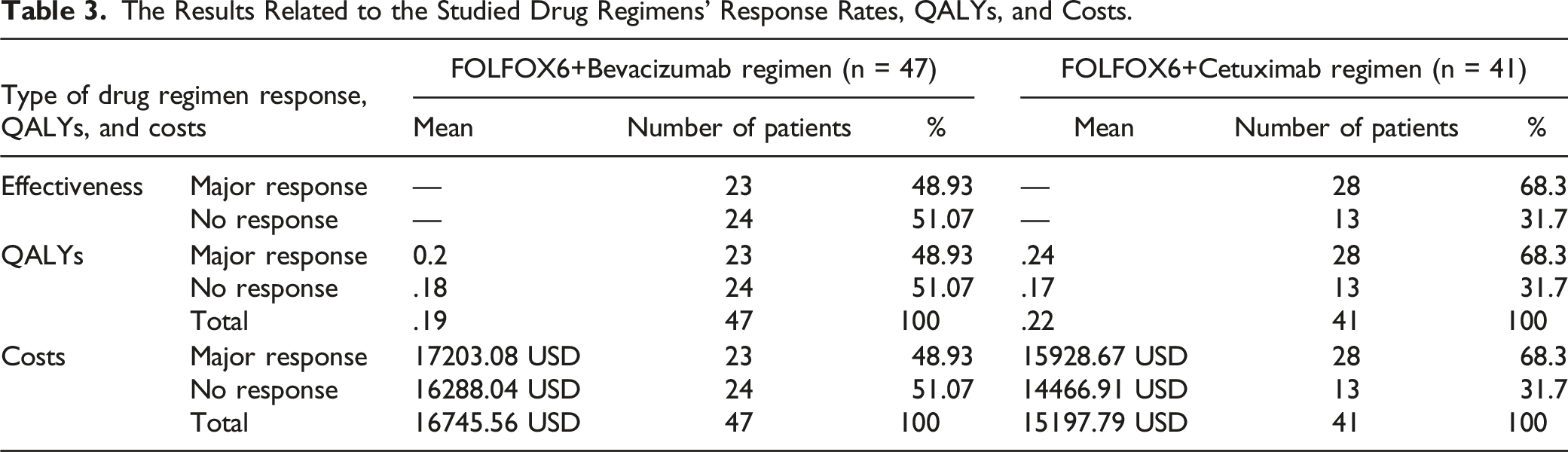
Table 3 also shows the QALY values based on the equation (5D) questionnaire for the 2 regimens. As can be seen, the mean QALY value in the FOLFOX6+Cetuximab regimen (.22) was higher than that of the FOLFOX6+Bevacizumab regimen (.19).
The Results of Cost-Effectiveness and Cost-Utility Analyses.

aThe confidence interval.

Results of the cost-effectiveness and cost-utility analyses. (a): Results of the cost-effectiveness analysis. (b): Results of the cost-utility analysis.
The results of the cost-utility analysis showed that the expected costs in the FOLFOX6+Bevacizumab and FOLFOX6+Cetuximab groups were 16,746.13 USD and 15,191.05 USD, respectively, and the expected QALYs calculated were 0.19 and .22, respectively. The results obtained from the decision tree have been presented briefly in Table 4. Therefore, it could be said that compared to the FOLFOX6+Bevacizumab, the FOLFOX6+Cetuximab saved 1555.08 USD and increased the QALYs by .03; As a result, FOLFOX6+Cetuximab was considered as the dominant option (reducing costs and increasing QALYs). According to Figure 2(b), FOLFOX6+Cetuximab had lower costs and higher QALYs (dominant option) than the other combination.
Results of the Sensitivity Analyses
In the one-way sensitivity analysis, each variable increased by 20%, based on which the Tornado diagram was depicted (Figure 3 (a)). According to the Tornado diagram, the results of the study were most sensitive to the increase in the costs of FOLFOX6+Bevacizumab and FOLFOX6+Cetuximab and least sensitive to the effectiveness of FOLFOX6+Cetuximab. Then, each variable decreased by 20% and the Tornado diagram was drawn again (Figure 3 (b)). As can be seen, the uncertainty in the results was high and the results were sensitive to the change of parameters in such a way that they were most sensitive to the decrease in the effectiveness of FOLFOX6+Cetuximab and the costs of FOLFOX6+Bevacizumab. Results of the one-way and probabilistic sensitivity analyses for comparing FOLFOX6+Cetuximab with FOLFOX6+Bevacizumab regimens in the studied patients.
As far as the probabilistic sensitivity analysis dealing with cost-effectiveness was concerned (Figure 3 (c)), the horizontal axis represented the incremental effectiveness (the difference in the effectiveness of the 2 drug regimens) and the vertical axis represented the incremental costs (the difference in the costs of the 2 drug regimens). The diagram was drawn using Monte Carlo simulation for a sample size of 1000 by assigning a beta distribution for the probabilities and effectiveness and a gamma distribution for the costs. The results showed that with the threshold (willingness-to-pay) of 16,783 USD, FOLFOX6+Cetuximab was more cost-effective than the other combination (the dots were mostly scattered in the acceptable area and below the threshold).
Figure 3 (d) shows the one-way sensitivity analysis of the cost-utility of the 2 drug regimens after increasing each variable by 20% According to the Tornado diagram, the results of the present study had the highest and the lowest sensitivities to the increase of the QALY of FOLFOX6+Bevacizumab in the major response arm and the QALY of FOLFOX6+Cetuximab in the no-response arm, respectively. Then, each variable decreased by 20% and the Tornado diagram was drawn again (Figure 3 (e)). As can be seen, the results of the study did not change much and had the highest sensitivity to the decrease in the QALY of FOLFOX6+Cetuximab in the major response arm.
In the probabilistic sensitivity analysis diagram dealing with cost-utility (Figure 3 (f)) where the horizontal axis represents the incremental QALYs (the difference in the QALYs of the 2 drug regimens) and the vertical axis represents the incremental costs (the difference in the costs of the 2 drug regimens), it was found out that with a threshold (willingness-to-pay) of 16,783 USD, FOLFOX6+Cetuximab had higher cost-utility than the other combination (the dots were mostly scattered in the acceptable area and below the threshold).
Overall, the results of the sensitivity analyses showed that there was a bit of uncertainty.
Cost-Effectiveness Acceptability Curves
The cost-effectiveness acceptability curve for the effectiveness outcome has been plotted in Figure 4(a), in which the horizontal axis shows the willingness-to-pay (USD) and the vertical one shows the probability that FOLFOX6+Cetuximab, compared to FOLFOX6+Bevacizumab, become cost-effective. According to the figure, at the willingness-to-pay amount of 16,783 USD, this probability was about 70%. Cost-effectiveness acceptability curves for effectiveness and QALY outcomes. (A): Cost-effectiveness acceptability curve for the effectiveness outcome. (B): Cost-effectiveness acceptability curve for the QALY outcome.
Figure 4 (b) indicates the cost-effectiveness acceptability curve for the QALY outcome. In this figure, the horizontal axis shows the amount of willingness-to-pay (USD) and the vertical one shows the probability that FOLFOX6+Cetuximab, compared to FOLFOX6+Bevacizumab, becomes cost-effective. According to the figure, at the willingness-to-pay amount of 16,783 USD, this probability was about 60–65%.
Discussion
In colorectal cancer, chemotherapy is used as an adjuvant treatment to surgery for some high-risk patients at stages Ⅱ, III, and even resectable stage IV. It is also used as the palliative treatment at the stage of metastatic disease. 50 Chemotherapy is recommended for 6 months, and no greater effect has been seen in its longer treatments of 9 and 12 months. 39 The present study aimed to compare the cost-effectiveness of FOLFOX6+Bevacizumab with FOLFOX6+Cetuximab in patients with stage IV colorectal cancer referred to medical centers in Shiraz, Iran, in 2021.
According to the results of the present study, the mean total cost of stage IV colorectal cancer patients who used FOLFOX6+Bevacizumabin 2021 was higher than that of patients who took FOLFOX6+Cetuximab. The results of the study by Lee et al. (2021) suggested that from the societal perspective, the total cost (direct and indirect costs) of chemotherapy with the anti-EGFR drug was lower than that of chemotherapy with Bevacizumab. 51 However, the results of the study by Bai et al. (2019) from the societal perspective indicated the higher total cost of FOLFOX4+Cetuximab compared to FOLFOX4 alone. 52 The drug combinations used in their study and the present one were different, though. In addition, the Markov model had been used in their study, while in the present study, the decision tree was used. Another difference is that in their study, Bai et al. divided the costs into 2 groups of direct and social costs, with the latter including time and travel costs. In the present study, however, direct non-medical costs included the costs of food, accommodation, auxiliary tools, etc., and indirect costs included absenteeism of the patients and their companions.
Among the different types of costs investigated in both groups of patients, direct costs accounted for the highest ones, and they were higher in the FOLFOX6+Bevacizumab group than in the FOLFOX6+Cetuximab, possibly due to the higher cost of the drugs used in the former group, since drugs accounted for the major costs of the patients. In their study, Bai et al. (2019) examined the cost-effectiveness of the FOLFOX4+Cetuximab compared to that of FOLFOX4 from the societal perspective and suggested that the main reason for the difference in the costs of the patients treated with the drugs studied was direct medical costs, especially chemotherapy costs. They indicated that the treatment method that had a higher cost of chemotherapy was more expensive and had higher direct costs. 52 However, Vahdatimenesh et al. (2017) conducted a study from the societal perspective and stated that the highest mean total cost was related to the indirect costs due to the mortality cost. 53 It should be noted that in the present study, there was no mortality cost due to the lack of mortality from the consumption of the studied drug regimens.
In the current study, the mean direct medical costs in the FOLFOX6+Bevacizumab group were higher than in the other group. Such a difference was mainly due to the higher cost of drugs in the FOLFOX6+Bevacizumab regimen group. The results of the studies by Sehnalova et al. from the perspective of the healthcare providers (2020) 54 and Pathak et al. (2018) 55 showed that the direct medical costs of the group taking Bevacizumab was higher than the one taking Cetuximab, mostly due to the costs of chemotherapy drugs. However, the results of Shida et al.'s study (2018) 38 from the public medical care viewpoint showed that the mFOLFOX6+Cetuximab treatment arm was more expensive than the other arms, which is contrary to the results of the present study. One of the reasons for this difference could be the number of chemotherapy courses. In other words, in their study, Cetuximab was administered once a week, but Bevacizumab and Panitumumab were administered once a fortnight. However, in the present study, both studied drug regimens were administered once a fortnight. In addition, the observed difference could be due to the difference in the study perspectives.
In the present study, the highest direct medical costs were attributed to consumed drugs, accounting for 59.1% and 54.08% of the direct medical costs in the FOLFOX6+Bevacizumab and FOLFOX6+Cetuximab groups, respectively, the reason for which seemed to be the high cost of Bevacizumab in some countries, including Iran. The results of the study by Sehnalovaet al. (2020) conducted from the perspective of healthcare providers from 2007 to 2017 showed that Bevacizumab was more expensive. 54 In their study from the perspective of the health system, Najafi et al. (2018) showed that the costs in the group of patients receiving Bevacizumab were estimated to be lower per patient than in the Cetuximab group over the lifetime horizon. 56 One of the differences could be in the time horizons of the studies; the time horizon in the present study was one year and thus, the discount rate was not taken into account. Also, the results of Giuliani and Bonetti’s study (2016 and 2018) showed that the cost of FOLFOX+Bevacizumab was lower than FOLFOX+Cetuximab.27,28 The reason for this difference could be that the costs studied were related to only drug costs of one treatment cycle; while in the present study, the direct medical, direct non-medical, and indirect costs of treatment in 6 to 12 cycles were considered.
The results of the current study also showed that direct non-medical costs and indirect costs were higher in the group of patients under treatment with FOLFOX6+Bevacizumab than in the FOLFOX6+Cetuximab group. In the former group, the percentage of patients referred to the hospital from outside Shiraz was higher, and consequently, they needed to stay longer in Shiraz. As a result, the absenteeism of the patients and their companions from work in this group was more than that in the group under treatment with FOLFOX6+Cetuximab. Furthermore, due to the fact that the side effects of FOLFOX6+Bevacizumab are usually greater and more severe than FOLFOX6+Cetuximab, and since chemotherapy by Bevacizumab increases the risk of cardiovascular diseases such as increased blood pressure and irregular heartbeat, 57 the cost of changes in the home environment for the adaptation of these patients was higher (approximately 2.5 times). These could have caused an increase in direct non-medical and indirect costs in the group receiving FOLFOX6+Bevacizumab.
The mean direct non-medical costs per patient in the year of the study were higher in the group under treatment with FOLFOX6+Bevacizumab than in the FOLFOX6+Cetuximab group, the reason for which could be the fact that most of the patients in the former group referred to the medical center from outside Shiraz, that is, from other cities of Fars and other provinces and stayed in inns and hotels.
The mean annual indirect costs per patient in the group receiving FOLFOX6+Bevacizumab were higher than in the other group. This could be due to the differences in the patients’ employment status, types of jobs, and daily wages received by the patients in the 2 groups.
In the present study, the index of major response to drug combination and tumor shrinkage was used to measure effectiveness. According to the results, FOLFOX6+Cetuximab had higher expected effectiveness than the other regimen (.68 vs. .49). One of the reasons could be that metastatic tumors may be sensitive to Cetuximab and Cetuximab has more tumor shrinkage than Bevacizumab. 58 Lee et al. (2021) concluded in their study that the effectiveness of Cetuximab-based chemotherapy was greater than that of Bevacizumab-based chemotherapy. 51 However, Nishizawa et al. (2021) showed that the drug regimen containing Cetuximab was less effective. In their study, the overall response rate in terms of tumor shrinkage, the median overall survival, and the median progression-free survival were respectively 59.1%, 25.3 months, and 11.7 months in the SOX+B-mab group, and 43.5%, 15.5 months, and 5.5 months in the SOX+C-mab group. 59 A systematic review by da Silva et al. (2018) indicated that treatment with Bevacizumab improved overall survival, progression-free survival, and post-progression survival compared to treatment without this medicine, but in terms of disease control, there was no difference between chemotherapy with and without Bevacizumab. Besides, Bevacizumab had severe side effects such as high blood pressure, perforation of the gastrointestinal tract, arterial and venous thromboembolism, diarrhea, and neutropenia. 60
The results of the current study showed that FOLFOX6+Cetuximab and FOLFOX6+Bevacizumab had the QALYs of .22 and .19, respectively, and the utility was higher (11 extra days of full health over a year) in the former group. This difference could be due to the longer hospitalization period of the patients receiving FOLFOX6+Bevacizumab, which had more negative effects on the quality of life. This finding is consistent with the results obtained by Su et al. (2022) 29 and Lee et al. (2021)51 who showed that the drug combinations containing Cetuximab caused more QALYs. However, Houtset al. (2019) compared the drug regimens and the outcomes of the regimens containing Bevacizumab or Cetuximab and found no significant difference in progression-free survival and overall survival between the patients treated with Bevacizumab compared to those receiving Cetuximab. However, the risk of death was higher among Cetuximab-treated patients with right-sided tumors compared to those with left-sided tumors, while this was not the case for Bevacizumab-treated patients. 31
According to the results of this study, FOLFOX6+Cetuximab was dominant due to its lower costs and higher effectiveness compared to FOLFOX6+Bevacizumab. The reason could be that since chemotherapy with FOLFOX6+Bevacizumab was more expensive than with FOLFOX6+Cetuximab in Iran, the patients who received the former combination had higher direct costs because the costs of drugs are considered as direct costs. The results of the studies by Lee et al. (2021) with the societal perspective 51 and Koilakou et al. (2021) 61 showed that the drug regimen containing Cetuximab was more cost-effective. However, the results of the studies by Najafi et al. (2018) from the perspective of the health system, 56 Ezatet al. (2013) from the societal perspective, 62 and Natrah et al. (2012) 63 from the societal perspective showed that Bevacizumab was more cost-effective in the management of last-stage colorectal cancer. The reasons for the difference between the results of this study and other studies could be attributed to the types of drugs compared and the investigation of only direct medical costs in some other studies.
In the present study, the results of the cost-utility analysis showed that FOLFOX6+Cetuximab with lower costs and greater QALYs compared to FOLFOX6+Bevacizumab had higher cost-utility and dominated the latter drug regimen. It could also be concluded that changing the treatment regimen from FOLFOX6+Bevacizumab to FOLFOX6+Cetuximab increased the patients’ quality of life at a lower cost. The results of the studies by Elsamanyet al. (2021) 64 and Lange et al. (2014) 33 also indicated that the drug regimen containing Cetuximab had higher cost-utility. However, the results of the studies by Ezatet al. (2013) 62 and Natrah et al. (2012) from the societal perspective 63 showed that Bevacizumab was cheaper and with higher utility. The reason for the difference between the results of these 2 and other studies could be attributed to the type of drugs and drug combinations compared.
The results of the one-way and probabilistic sensitivity analyses dealing with cost-effectiveness showed that the level of uncertainty about the results was high, and the study results were most sensitive to the increase in the costs of FOLFOX6+Bevacizumab and FOLFOX6+Cetuximab, as well the decrease in the effectiveness of FOLFOX6+Cetuximab and the costs of FOLFOX6+Bevacizumab. Therefore, adopting policies that prevented the change of costs of these 2 drug regimens could help increase the robustness of the results and reduce uncertainty. In other words, adopting such policies could possibly increase the stability and robustness of the results and reduce their sensitivity to parameter changes, and thus, the study results would be robust for the following years.
In the one-way sensitivity analysis dealing with cost-utility, the investigated variables were the costs, effectiveness, and QALYs of the 2 drug regimens including FOLFOX6+Bevacizumab and FOLFOX6+Cetuximab. The results of the sensitivity analysis showed that the ICUR was sensitive to changes in the incremental QALYs. According to the results obtained from the probabilistic sensitivity analysis dealing with cost-utility, FOLFOX6+Cetuximab had higher cost-utility than FOLFOX6+Bevacizumab.
Moreover, according to the cost-effectiveness acceptability curve for the effectiveness outcome, at the willingness-to-pay amount of 16,783 USD, the probability that FOLFOX6+Cetuximab became cost-effective was approximately 35% higher than that of FOLFOX6+Bevacizumab. As shown in Figure 4(a), by increasing the resources to over 16,783 USD (the threshold), the maximum cost-effectiveness probability of FOLFOX6+Cetuximab would reach 75%. In addition, it could be stated that if the willingness-to-pay increased up to 3 times the threshold, the probability of being cost-effective for this drug regimen would increase by only 5%, which was not economical for allocating more resources. The study by Stintzing et al. (2020) 34 from the perspective of German social health insurance showed that at the willingness-to-pay amount of 50,000 euros, the probability of being cost-effective for the 2 drug regimens was equal (approximately 50%), but above this amount, the probability of being cost-effective for FOLFIRI+Cetuximab versus FOLFIRI+Bevacizumab was 96%. Thus, the FOLFIRI+Cetuximab curve was above the FOLFIRI+Bevacizumab curve and it was more cost-effective at different willingness-to-pay amounts.
The cost-effectiveness acceptability curve for the QALY outcome at the willingness-to-pay amount of 16,783 USD showed that the probability that FOLFOX6+Cetuximab became cost-effective was approximately 25% higher than that of FOLFOX6+Bevacizumab. According to Figure 4(b), increasing the allocation of resources to over 16,783 USD (the threshold) would not lead to an increase in the cost-effectiveness probability of FOLFOX6+Cetuximab by over 65%–70%. Also, it could be said that if the willingness-to-pay increased up to 3 times the threshold, the probability of being cost-effective for FOLFOX6+Cetuximab would increase by a maximum of 5%, which was not economical. In the study by Stintzing et al. (2020), 34 the results showed that the FOLFIRI+Cetuximab was more cost-effective than the FOLFIRI+Bevacizumab at different willingness-to-pay amounts.
Regarding the generalizability of the results of this study, it could be stated that since FOLFOX6+Cetuximab and FOLFOX6+Bevacizumab regimens are used in other Iranian hospitals to treat stage IV colorectal cancer patients, the results can be generalized to them. However, given that the cost coverage by insurance organizations, the patients’ ability to pay the costs, and the incidence and prevalence of stage IV colorectal cancer vary in different countries, the results of this study may not be generalized to other countries with certainty.
Some limitations of the present study were as follows: the small population and sample size; the non-cooperation of some patients and their families in answering the questions of the questioners and checklists; forgetting some of the costs under study by some patients or their families and the existence of recall bias; incompleteness of some cost accounts in the patients' medical records; the impossibility of using the Markov model due to the lack of studies in this field and consequently, the impossibility of extracting transition probabilities; and changing the drug regimens of some patients by the specialists during the study and therefore, the exclusion of a number of patients from the study.
Conclusion
The results of the current study showed that FOLFOX6+Cetuximab compared to FOLFOX6+Bevacizumab in stage IV colorectal cancer patients in Shiraz, Iran, in 2021 was the dominant option in terms of cost-effectiveness and cost-utility. The findings of this study can be helpful in preparing clinical guidelines for colorectal cancer patients. It is suggested that basic and supplementary health insurance coverage for paying for FOLFOX6+Cetuximab increase. Due to the high cost of accommodation for the patients and their companions, free or low-cost places are needed to be considered near specialized medical centers for the accommodation of cancer patients and their companions during the period of receiving medicine and undergoing chemotherapy in order to reduce their such costs. The use of remote technology to guide patients by oncologists can effectively reduce direct non-medical costs (including travel costs) and indirect costs. Providing specialized services related to colorectal cancer in smaller cities can reduce direct non-medical costs (including travel and accommodation costs) and indirect costs as well. Finally, due to the fact that FOLFOX6+Cetuximab was found to have higher cost-effectiveness and cost-utility than FOLFOX6+Bevacizumab, it is suggested that it be prioritized as a standard treatment method for stage IV colorectal cancer in Iran.
Footnotes
Appendix
Acknowledgments
The present article was extracted from the thesis written by Sedighe Sadat Tabatabaei Far and was financially supported by Shiraz University of Medical Sciences grant No. 22955. The researchers would like to thank the studied patients for their kind cooperation with the researchers in collecting and analyzing the data.
Authors’ Contributions
RR, AJ, and MD contributed to the idea and design. SSTF contributed to the data collection. RR, AJ, and SSTF contributed to the data analysis. All authors contributed to the manuscript writing and revision. All authors approved the final version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shiraz University of Medical Sciences (22955).
Data Availability
The data used and analyzed in the study are available from the corresponding author on reasonable request.
Ethical Approval
This study was approved by the Ethics Committee of Shiraz University of Medical Sciences (Code: IR.SUMS.REC.1400.073). The patients were free to choose to participate in the study, and once the objectives of the research were explained to them, their written informed consent was obtained. The questionnaires and checklists were completed anonymously and the patients were assured of the confidentiality of their answers to the questions. It should be noted that in order to comply with ethical considerations and the confidentiality of patient information, the patients were distinguished by the codes at the top of the cost information forms and the effectiveness and utility checklists.
