Abstract
Background
Patients with cancer frequently reported sleep problems during their treatments which can affect their sleep quality have an impact on patients’ quality of life (QOL).
Objective
to assess the prevalence of sleep quality and associated factors in adult cancer patients on treatment in the Oncology unit of Tikur Anbessa Specialized Hospital, Addis Ababa, Ethiopia, 2021
Methods
institutional-based cross-sectional study design was used and data was collected by using face-to-face structured interview questionnaires from March 1 to April 1, 2021. Sleep Quality Index (PSQI) consisted of 19 items, the social support scale (OSS-3) consisted of 3 items, and the Hospital Anxiety and Depression Scale (HADS) consisted of 14 items were applied. Logistic regression including bivariate and multivariate analysis was done to examine the association between dependent and independent variables, and P< 0.05 was considered the level of significance for associations.
Results
A total of 264 sampled adult cancer patients on treatments were included in this study, with a response rate of 93.61%. About 26.5% of the participants’ age distribution was between 40 to 49 years, and 68.6% were female. 59.8% of the study participants were married. Concerning education, about 48.9% of participants attended primary and secondary school and 45% of participants were unemployed. Overall, 53.79% of individuals had poor sleep quality. Low income ((AOR=5.36 CI 95% (2.23, 12.90), fatigue (AOR=2.89 CI 95(1.32, 6.33), pain (AOR 3.82 C I95 % (1.84, 7.93), poor of social support (AOR =3.20 CI 95% (1.43, 6.74), anxiety (AOR=3.48 CI 95% (1.44, 8.38) and depression (AOR 2.87 CI 95 % (1.05-7.391) were all associated with poor sleep quality.
Conclusion
This study revealed a high prevalence of poor sleep quality, which was significantly associated with factors like low income, fatigue, pain, poor social support, anxiety, and depression among cancer patients on treatments.
Introduction
In 2018, 18.1 million cancer patients were reported globally, and almost around half of them died due to cancer. These figures are going to be doubled by 2040 and will increase in Low and Middle-Income Countries (LMIC), as World Health Organization (WHO) figures showed, two-thirds of the world’s cancer will happen at LMIC. The reason for 30% of premature death from Non-communicable Diseases (NCDs) among those aged 30-69 was cancer.1,2 Cancer is also the leading cause of death before age of 70 years in 91 of 172 countries; and in Eastern, Africa cancer was reported as the top incidence and cause of death in 2018. 3 Sleep is essentially important for cancer patients to achieve a good prognosis for treatment outcomes and to oppose the cancer disease.4,5 Cancer symptoms and cancer treatment side effects affect sleep quality.6,7
Among cancer symptoms pain is a common problem in cancer,8-10 however, less than 27.8% of cancer patients were taking analgesics. 11 As the stage of cancer increases, pain due to cancer also increases and disturbs the sleep quality of cancer patients.12,13
Patients with cancer are frequently reported sleep problems; especially during their treatments such as chemotherapy, surgically removal of the tumor, and radiation therapy. The problem is common and these treatments’ side effects lead cancer patients to physical, psychological, and economically disturbs, and the consequences of these problems affect patients’ sleep quality.10,14 Fatigue, nausea, and vomiting due to side effects of chemotherapy treatments were reported as a factor for poor sleep quality in the previous study among patients.11,15-17
Poor sleep quality has the following problem on the patient’s life, low quality of life (QOL), daytime dysfunction, recurrence of cancer, retrieval failure, fatigue, pain, irritability, and unhappiness; these problems are affecting the patients’ ability to confront the disease.18,19
The magnitude of poor sleep quality was different according to literature, but it’s common problem worldwide among patients with cancer on treatments; the previous study showed that the prevalence of poor sleep quality was between (30.5% to 99.5%) during patients were on their treatments19,20 and in other study conducted 98 breast cancer patients, 60% of them reported as their sleep quality was affected when they were undergoing chemotherapy treatments. 21
In a global, multi-center study among adult patients with cancer using opioids at 9 hospitals in 5 European countries, 78% of respondents reported poor sleep quality. 22 In the study conducted in New Zealand among patients with prostate cancer 30.5%, 23 and in another study conducted in Switzerland 42.8% of participants reported poor sleep quality during chemotherapy. 24
Based on different studies done in different countries like China 56.1%, Turkey 54.5%, and India 61.67% showed that patients with advanced cancer were reported to have sleep problems during their treatment.25-27
In Africa, there were limitations in research conducted related to this title, but an article conducted in Tunisia showed that 66% of breast cancer patients reported poor sleep quality during their treatments. 8
Generally, a different previous study revealed that poor sleep quality was found to be associated with pain, fatigue, depression, and anxiety.28-32 Poor social support and low education level,33-36 being a female and single, 37 poor hygiene, gastrointestinal, neck, and head cancer types were significantly associated with poor sleep quality.38,39
In a previous study, effectively managing cancer pain and fatigue, aerobic exercise, and providing comfort to patients, such as back massage, were recommended to enhance cancer patients’ sleep quality.9,40 Even though sleep quality in cancer patients is influenced by a variety of factors, currently in Ethiopia, no published research reflects on sleep quality among cancer patients. Therefore, this study will help to bridge the gap regarding sleep quality among cancer patients. This study aimed to assess sleep quality and associated factors in adult cancer patients on treatment at the Oncology unit of TASH Ethiopia, 2021.
Methods
Study Area, Period, and Design
The study was conducted at Tikur Anbessa Specialized Hospital (TASH) Oncology unit, in Addis Ababa Ethiopia. This hospital provides multi-dimensional aspects of cancer treatment services for the entire country. A cross-sectional institutional-based study design type was conducted from March 1/2021 to April 1/2021.
Population, Sample Size, and Sampling Methods’
The study population included all adult cancer patients aged above 18 years who were on treatment follow-up at TASH’s oncology unit; while patients previously diagnosed with mental health problems and had hearing impairments were excluded.
The single population proportion formula was used to determine the sample size. A margin of error of 5%, a confidence level of 95%, and a prevalence of 50% were taken. Since the population was <10 000, a correction formula was applied, by adding a 10% non-response rate, and then the final sample size was calculated to be 282. Finally, the study participants were approached by using a systematic random sampling process.
Data Collection Tools and Materials
The social support scale (OSS-3) consists of 3 items and arrays from 3-14 47 with current Cronbach’s alpha coefficients of .89; “Pittsburgh Sleep Quality Index (PSQI) questionnaire consists of 19 items with Cronbach’s alpha coefficients of .83 in previous and .81 Cronbach’s alpha coefficients in the current study; which clustered into 7 components (‘subjective sleep quality, sleep latency, sleep duration, habitual sleep efficiency, sleep disturbances, use of sleeping medication, and daytime dysfunction) with a possible score of (0-3) and each making score 0 to 21 were applied, 41 and the Hospital Anxiety and Depression Scale (HADS) consisting of 14 items with the reliability of Cronbach’s alpha coefficients of the total for both Anxiety and Depression of .91 in a previous study and .94 in the current study were applied. Seven of the items indicate anxiety and the remaining items indicate depression. 44 The other substance use Questionnaire on behavior/Substance use was adopted from Ethiopia Demographic Health Survey (2016 EDHS). 48
Three BSc Nurses collected data, and before data collection, half-day training was given to them. Data quality was ensured by correctly designing questionnaires, daily questionnaires were reviewed for completeness and necessary feedback was provided to data collectors. The data collection tool was translated into the local language Amharic version from the English version. The questionnaires were pre-tested on 5% of the total sample size participants who were not included in the study 1 week before data collection. Before data collection, an approval letter was obtained from the Ethical Committee of Addis Ababa University’s School of Nursing and Midwifery.
Operational Definitions
Ethical Statement
Ethical permission for this research was taken from the Institutional review board of Addis Ababa University, College of health sciences, School of Nursing and Midwifery with the reference number AAUMF 03-008 Protocol No. 67/21/SNM. A letter of cooperation was written to the hospital. Permission was obtained from the medical director and cancer treatment center focal person of the Hospital. Written informed consent was obtained from patients before the interview. Participants’ privacy and confidentiality of the information were maintained by the declaration of Helsinki.
Data Processing and Analysis
The data entering and cleaning were done by using Epi-data manager version 4.6 and analysis of data was done using SPSS version 25. The strength of an association between dependent and independent variables and their significance was computed by using an odds ratio with a 95% confidence interval. Binary and multivariate Logistic regression techniques were employed. At bivariate logistic regression analysis, variables with P < .25, were taken as a candidate for multivariate logistic analysis. In the multivariate logistic analysis pvalue <.05 was considered to be statistically significant.
Result
Socio-Demographic Characteristics of Respondent
Socio-demographic characteristics of adult cancer patients on treatment at Tikur Anbessa Specialized Hospital of Oncology Unit, Addis Ababa Ethiopia, 2021 (n = 264).

Clinical-Related Characteristics of Study Participants
Clinical related characteristics in adult cancer patients on treatment at Tikur Anbessa Specialized Hospital Oncology Unit, Addis Ababa, Ethiopia, 2021 (n = 264).
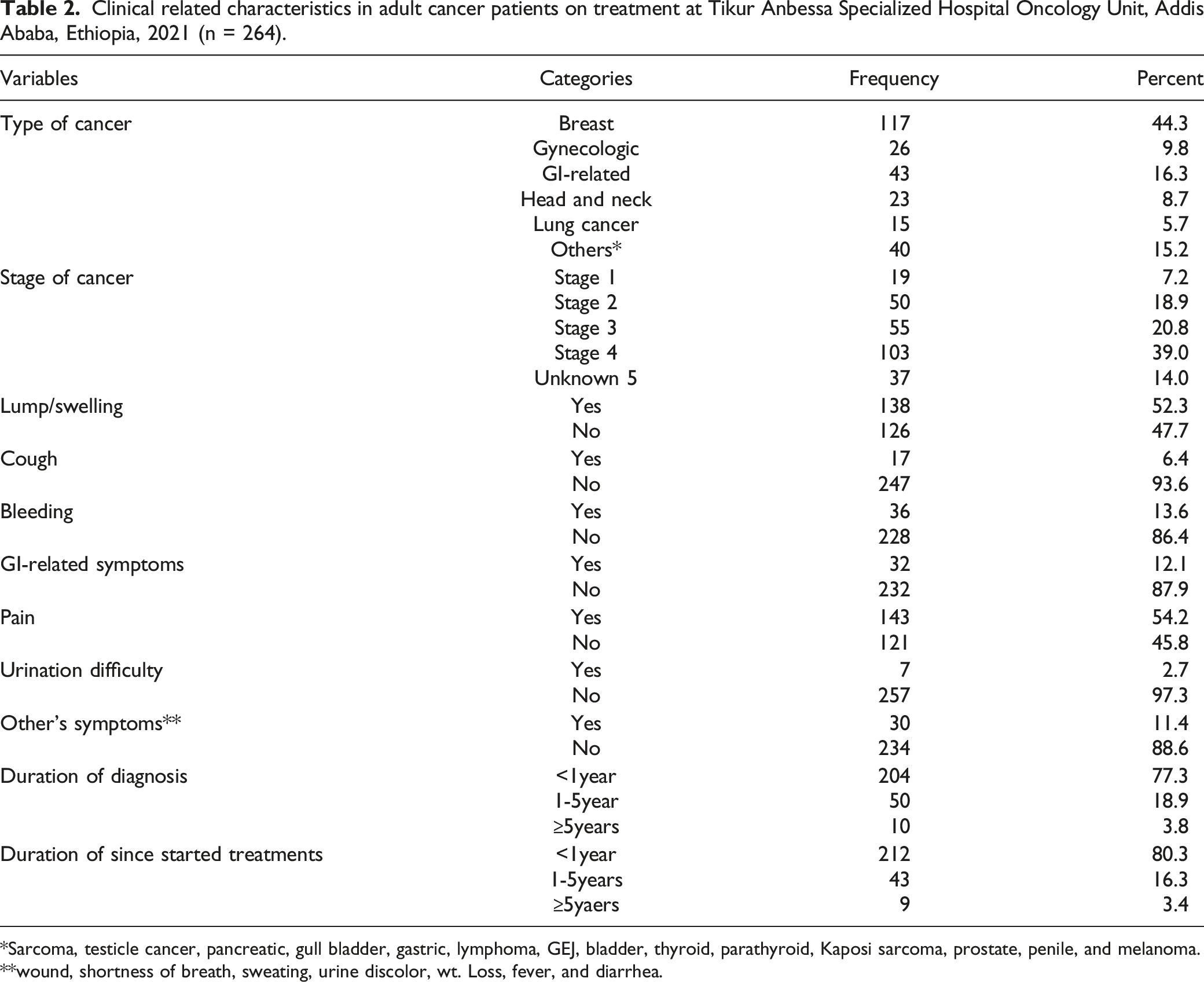
Sarcoma, testicle cancer, pancreatic, gull bladder, gastric, lymphoma, GEJ, bladder, thyroid, parathyroid, Kaposi sarcoma, prostate, penile, and melanoma.
wound, shortness of breath, sweating, urine discolor, wt. Loss, fever, and diarrhea.
Types of Treatments, Treatments Side Effects, and Environmental Factors
Type of treatments, side effects, and environmental factors among adult cancer patients on treatment at Tikur Anbessa Specialized Hospital Oncology Unit, Addis Ababa Ethiopia, 2021 (n = 264).

Psychosocial, Social Support, and Substance Use of Study Participants
Psychosocial, social support, and substance use of adult cancer patients on treatment at TASH Oncology Unit, Addis Ababa Ethiopia, 2021 (n = 264).

Sleep Quality of Study Participants
Sleep quality of adult cancer patients on treatment at Tikur Anbessa Specialized Hospital Oncology Unit, Addis Ababa, Ethiopia, 2021, (n = 264).

Factors Associated with Sleep Quality of Study Participants
factor association with sleep quality in adult cancer patients on treatment at Tikur Anbessa Specialized Hospital Oncology Unit, Addis Ababa Ethiopia, 2021, (n = 264).
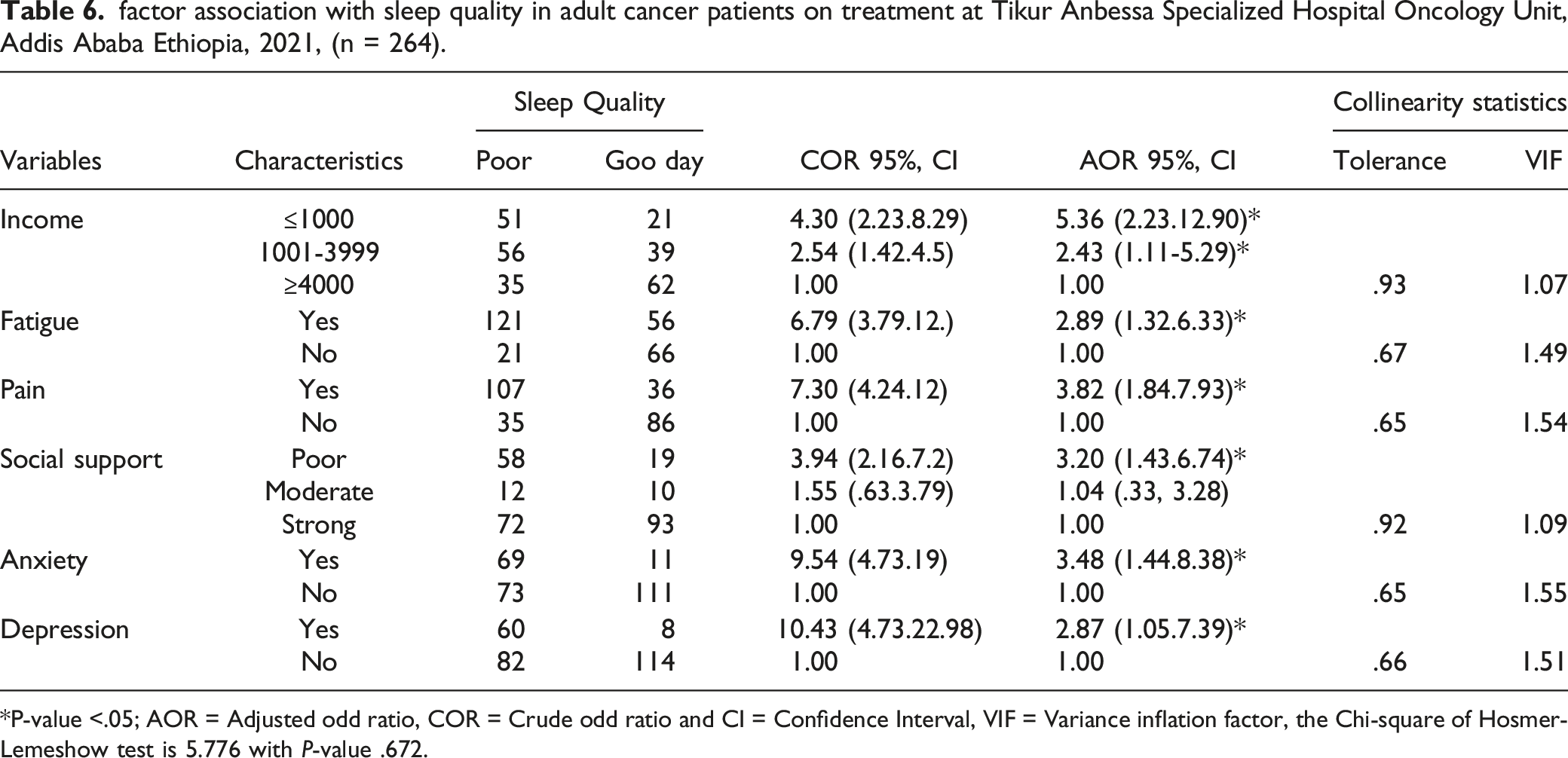
P-value <.05; AOR = Adjusted odd ratio, COR = Crude odd ratio and CI = Confidence Interval, VIF = Variance inflation factor, the Chi-square of Hosmer-Lemeshow test is 5.776 with P-value .672.
Discussion
The quality of sleep and associated factors in adult cancer patients undergoing treatment were investigated in this study. It showed that more than half of the participants had poor sleep quality. Accordingly, 53.8% of respondents reported having poor sleep quality.
But study enrolled 2371 participants from all types of cancer in the USA reported about 34% of sleep trouble as a common problem in cancer patients; while they were on treatment follow-up. 20 Which showed lower than the current study result, the disparity might be the fact that the study from the USA showed more participants found in high-income status, higher educational status, and about 4% also used sleep aid medication, but in the current study, only about 1.5% of participants used once or twice a week sleep aid medication.
As of the study conducted in New Zealand among patients in the treatment of cancer about 30.5% of individuals reported sleep difficulty during their treatments 23 which could be lower than the current study results. The inconsistency might be due to the previous study participants’ educational status; even the income and life status difference of the study participants might have an impact on the outcome of the result. However, in a study of 98 breast cancer patients from the United States, 60% of them reported poor sleep quality and another study among African Americans with breast cancer reported 61% deprived of sleep quality when they were treated.21,23
In a study from Greece, 58.2% of breast cancer reported poor sleep quality during undergoing chemotherapy treatments and the results were from specific breast cancer. 30 The disparity could be attributed to being a woman biologically and the burden of family responsibility as a result of stress might be high among females. An article from Switzerland 42.85%, 27 also reported a lower proportion than the current study, the difference might be about (31%) of participants were in stage 2, while about 39% of the current finding participants were in stage 4; when the stage of cancer increase, pain due to metastasis increasing and as the results sleep quality might decrease.
Findings from China 56.1%, 25 Turkey 54.4%, 26 and 2 studies found in India 57.6%, and 61.67%19,27 were nearly comparable with the current finding. The similarity might be participants from China, and India and the current were found in similar stage 4 (IV) of the disease. Research from Tunisia found that 66% of breast cancer patients indicated poor sleep quality after surgery. 8 This disparity might be attributed to the fact that the article was limited to breast cancer and all participants had undergone surgical treatments in addition to chemotherapy, while the current study included patients diagnosed with all cancer types and all types of treatments provided as well as the tool used in different setting contribute for the difference.
Income less than 1000 Ethiopian Birr was significantly associated with poor sleep quality. The previous study from Tunisian, 8 and Pakistan, 34 found that low financial status was significantly associated with poor sleep quality. The similarity might be more cancer patients terminated their job due to disease processes (characteristics).
Being fatigue was found to be a significant factor to be associated with poor sleep quality, accordingly, research conducted in the USA, China, Egypt, and Iran11,29,36,49,50; reported a consistent finding with the current study. The possible justification might be the fact that fatigue due to cancer treatment could result from hormonal disturbance, reduction of red blood cells (hemoglobin), and shortage of oxygen due to treatment side effects. 35
In a previous study from Ohio and Morocco having pain was statistically associated with poor sleep quality,28,39 which had similarities with the current study. The reason might be due disease pathophysiological process, especially during cancer metastasis in all cancer patients.
Poor social support was significantly associated with poor sleep quality which can be augmented with other studies conducted elsewhere.33,36 That might be justified as the chronicity of the disease process and fatality could minimize the patients’ ability to cope with crises they suffer from. During an early diagnosis, most clients need an emotional and psychological intervention to overcome with uncertainty related to the diagnosis. On the other hand, the information that the health personnel provides might play a significant role in the patient developing a coping mechanism.
Similar to the current finding, different studies that enrolled all types of cancer in the USA reported that anxiety and depression had significantly associated with sleep quality.28,29,36,38 The reason for that could be attributable to the psychological stress that emerges as anxiety and depression due to patients' fear of death, poor prognosis of treatment, and complications of cancer itself.
Study Limitations
• Being the study cross-sectional based design, does not allow infering causality. Prospective study is warranted. Further, this study does not include cancer patients from the region of the countrywhere treatment is recentely launched. • The lack of literature locally and the study that existed were specific to a single type of cancer. • The tool (PSQI) used to measure sleep quality in this study was not validated in Ethiopia.
Conclusion
This study revealed a high prevalence of poor sleep quality (53.79%) among cancer patients on treatment; the variables such as low income, fatigue, pain, poor social support, anxiety, and depression were found to be significantly associated with the outcome variable.
Footnotes
Acknowledgments
We would like forward our thanks to the School of Nursing and Midwifery, College of Health Sciences, Addis Ababa University, for sponsoring us to conduct this article. Our gratitude goes to Oncology Unit staffs for their cooperation during data collection and the study participants involved in this study; and also, to Jimma University Medical Center. We would also extend our heartfelt thanks to the authors for their invaluable professional input throughout the development of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
the sponsor for this paper was maintained from Addis Ababa University, College of Health Sciences School of Nursing and Midwifery.
Ethical Approval
Ethical permission for this research was taken from the Institutional review board of Addis Ababa University, College of health sciences, School of Nursing and Midwifery with the reference number AAUMF 03-008 Protocol No. 67/21/SNM. A letter of cooperation was written to the hospital. permission was obtained from the medical director and cancer treatment center focal person of the Hospital. written informed consent was obtained from patients before the interview. Participants’ privacy and confidentiality of the information were maintained by the declaration of Helsinki.
Authors’ Contribution
All authors had contributed equally for the development of this manuscript.
Data Availability
Data of this article can be available up on the request of legal body.
