Abstract
Introduction
Plexiform neurofibromas (PN) represent the main cause of morbidity in patients affected by Neurofibromatosis Type 1 (NF1). Until recently, surgery has been the main treatment option in these patients, but it is burdened with a low efficacy rate and a high incidence of side effects as well as recurrence. In recent years, MEK inhibitors (MEKi) such as selumetinib and trametinib have shown great promise.
Methods
We retrospectively describe a single center cohort of NF1 patients affected by PN1 and treated with MEKi since 2019 to 2021. Patients recruited in the study were affected by PN that were not eligible to complete surgical excision, symptomatic or with major cosmetic deformation or functional neurological deficits.
Results
Most patients experienced improvement in clinical symptoms and quality of life, with reduction or stabilization of lesions. However, no complete response was achieved. The most common adverse effects involved the skin, affecting every patient. Importantly, no life-threatening adverse effects occurred.
Conclusions
In our experience, MEKi treatment has been shown to be both safe and effective in improving symptomatology and quality of life.
Keywords
Introduction
Plexiform neurofibromas (PNs) represent the most common benign tumor occurring in patients affected by Neurofibromatosis Type 1 (NF1), arising in 30-50% of cases.1,2 They originate from the sheaths of peripheral nerve trunks and show rapid growth during early childhood 3 ; histologically, they are composed of Schwann cells, in which the loss of neurofibromin function determines the dysregulation of the Ras pathway that underlies the neoplastic transformation.4,5 The tendency to be widespread and infiltrative, their visual impact, as well as the often very important dimensions and the potential capacity for malignant transformation, make PNs the pathological manifestation determining the greater comorbidity in NF1, therefore also representing the main therapeutic challenge in these patients. 6
Magnetic resonance imaging (MRI) is the gold-standard radiological technique for the diagnosis and follow-up of PN. 7 However, there are no current guidelines on radiological surveillance in patients with NF18,9; radiological studies are performed at baseline and repeated based on the appearance of symptoms or changes in physical examination. 7
Until recently, surgery has been the main treatment option in these patients, although it is burdened with a low efficacy rate and a high incidence of side effects as well as recurrence. 5 In fact, the close relationship between PNs and the nerve fibers, from which they originate, makes it very difficult to achieve complete resection without iatrogenic nerve deficits. 1 Thus, in most cases, the surgical approach does not represent a wise treatment option, especially in bulky lesions that infiltrate internal organs. 10
For the aforementioned reasons, numerous efforts have been achieved to search for alternative medical therapy, exploring the efficacy of several drugs in the last decades: tipifarnib, pirfenidone, sirolimus, imatinib, and interferon (IFN) alfa-2b.11-15
Among these, MEK inhibitors (MEKi), which were initially developed for other malignancies such as melanoma and lung cancer, are currently used for the treatment of PNs. 16 Due to the recent approval by regulatory agencies, relatively few cases have been published regarding the use of MEKi in the treatment of PNs.17-23
Here we report a single center’s experience of treating NF1-associated PNs, with the aim of providing further evidence regarding the efficacy and tolerability of these drugs. The primary aim of this study was to evaluate the radiological and clinical response to two MEK inhibitors (selumetinib and trametinib) in NF1 patients with inoperable or symptomatic PN. Secondary aims were the assessment of treatment toxicity and the impact of therapy on the patients' quality of life (QoL).
Materials and Methods
This is a single-center retrospective clinical study. Patients recruited in the study were affected by PN that were not eligible to complete surgical excision, symptomatic or with major cosmetic deformation or functional neurological deficits, and treated with MEKi were included.
MEK inhibitors have been used as part of an expanded access program from July 2019 to May 2022.
NF1 clinical diagnosis was based on the diagnostic criteria established by the 1987 NIH Consensus Development Conference 24 and, in specific cases of non-satisfaction of all criteria, the genetic diagnosis was performed by next-generation sequencing (NGS) using a custom panel including the NF1 gene (Twist Bioscience, South San Francisco, CA). In case of a negative sequencing result, a Multiplex Ligation-dependent Probe Amplification (MLPA) analysis (MRC‐Holland, Amsterdam, The Netherlands) was performed to detect exon deletions/duplications of NF1. It should be recalled that a negative NF1 molecular testing does not rule out a diagnosis of NF1: some individuals (∼3-5%) diagnosed with NF1 based on clinical criteria do not have a pathogenic variant detectable by current technology. 25
The histological diagnosis of the PN was obtained by a surgical approach with biopsy or partial resection in all patients.
Informed and written consent for the processing by the legal representative or the person of legal age was obtained. The study was conducted according to the guidelines of the Declaration of Helsinki and approved by the Institutional Review Board of Bambino Gesù Children’s Hospital of Rome, Italy (RAP-2022-0008, 23th June 2022).
The reporting of this study conforms to STROBE guidelines. 26
Analysis of the Effectiveness of the Therapy
Radiological Evaluation
Radiological evaluation was performed with magnetic resonance imaging with 1.5 T magnet (Aera, Siemens, Erlangen, Germany) including axial and coronal STIR sequences. MRIs were performed at baseline and then every six months, identifying a target lesion (the most significant PN in size, or clinically most significant).
Images were saved in the Digital Imaging and Communications in Medicine format and target lesion segmented by using software available on the MR workstation (Siemens Syngo.via, Siemens Medical Solutions). The measurements were completed in consesus by two experienced pediatric radiologist (G.S.C. and P.T., 24 and 45 years of experience respectively).
The evolution of PN was evaluated with the Response Evaluation in Neurofibromatosis and Schwannomatosis (REiNS) consensus criteria. Following these criteria, the target lesion had to be measurable in at least three cross-sections and have a minimum volume of 3 cm3, with defined margins. 27
Clinical Evaluations
During the treatment period, periodic visits were carried out with a specific physical examination based on the location of the target lesion identified. Clinical assessments were performed at baseline, 1 month after the start of treatment, and then every three months or according to the needs of the individual patient.
Pain was coded according to the Visual Analog Scale, a psychometric tool that classifies the intensity of pain according to a score in a continuous range of values. Each value is associated with a figure that represents the mimicry correlated to that given intensity and the patient can express the intensity of their pain within this range, making a numerical and objective measure of pain. 28
Motor neurological deficits were analyzed using the British Medical Research Council muscle power scale. 29 A single impaired muscle was evalueted at baseline and was tested over time by two clinicians (A.C and G.M.).
Toxicity Evaluation
Toxicities secondary to treatment with MEK inhibitors were coded and graded according to the Common Terminology Criteria for Adverse Events (CTCAE) version 5.0, 30 assessed during periodic clinical visits according to the timings indicated above.
For the evaluation of toxicity, the following tests were regularly performed: blood analyzes (complete blood count, biochemistry, creatine phosphokinase (CPK) assay at baseline and then quarterly), cardiological visits and echocardiogram at the baseline and then every six months, dermatological visits at the baseline and then on a quarterly basis, eye examinations at the baseline and then on a quarterly basis.
Quality of Life
In order to evaluate the outcome in terms of clinical benefit from treatment, the patients’ perception of QoL (understood as the impact that the disease and treatment have on a child or adolescent self-perception and in the various social, school, and family settings) was assessed through the Italian version of the PedsQL TM 4.0 Generic Core Scale 31 before the start of the drug (T0), 3 months after the start of the therapy (T1) and then continued the analysis at 6 months (T2) and 12 months (T3). The questionnaire includes a section for the parents (2-18 years) and a section for the child (5-18 years), and is composed of 23 items that evaluate the main areas of health (as outlined by the World Health Organization): physical, emotional, social, and academic states. The items provide for an evaluation of the response according to a 5-point Likert scale ranging from 0 (ie it considers the problem absent - “never”) to 4 (which considers the problem “almost always”). The raw scores are then converted into a scale ranging from 0 to 100 in which a higher score therefore corresponds to a better QoL.
Statistical Analysis
Statistical analysis was conducted using Excel software. A descriptive analysis of the sample was performed with multiclass and multivariate distributions. For the primary objectives, the Student test was conducted to reject the null hypothesis (P-value); the significance threshold was conventionally chosen at .05.
Results
Characteristics of Patients Included. CALMs = cafè au lait Macules. NF= Neurofibromatosis. PN= Plexiform Neurofibroma. NGS= Next Generation Sequencing. MLPA = Multiplex Ligation-Dependent Probe Amplification. IFN = Interferon.

Patients’ median age was 3 years (age range: 3 months-17 years).
NF1 had been initially diagnosed primarily by clinical criteria in 5 cases, while genetic testing enabled diagnosis in 8 patients. Regarding clinical criteria, café au lait macules (CALMs) were present in all patients, while axillary or inguinal freckles were found in 6 out 13 (46%) and Lisch nodules in 3 out 13 patients (23%), respectively, characteristic bone changes were detected in 6 children (46%), and 4 out of 13 patients (31%) were affected by optic pathway glioma. Only 1 patient (8%) had a first-degree relative with NF1. Twelve of thirteen patients underwent genetic testing: a pathogenic variant in the NF1 gene was identified by NGS in 9 cases, while MLPA analysis resulted positive for NF1 deletions in 2 patients. One child resulted negative for both tests, and the diagnosis was carried out only by clinical criteria.
Among our cohort, 8 out of 13 patients (61%) were affected by more than 1 PN. The head and neck represented the most frequent location (31%, 4 out of 13), followed by limbs (3 patients, 23%), abdomen (2 patients, 15%), and the thoraco-mediastinal region (one patient, 8%). Spinal PNs occurred in 3 patients (23%).
Partial surgical resection was performed in 8 patients, whereas no complete excision was possible. Four children had previously undergone medical therapy with IFN.
Overall, twelve patients were given selumetinib, while 1 was treated with trametinib because unable to swallow tablets formulation. The two drugs were requested and obtained on a compassionate use program; selumetinib was administered daily, at a dose of 25 mg/m2 (maximum single dose 50 mg) twice daily (BID) in cycles of 28 days. The pharmaceutical formulation available is in 10 mg (white) and 25 mg (blue) capsules. Trametinib was administered as an oral suspension to a patient unable to take capsules at a dose of .032 mg/kg once daily in 28-day cycles.
The start of treatment was driven in all 13 cases by the onset of clinical signs/symptoms (motor or functional) and/or worsening of symptoms (e.g. pain) related to the presence of PN; in 6/13 cases a progression of radiological disease coexisted according to the RECIST guidelines and REiNS consensus. The most frequently referred signs/symptoms were pain (unresponsive to analgesic therapy) in 9 out of 13 patients (69%) and neurological motor deficits in 11/13 (84%) patients. A patient with PN, extended from the cervical area to the thorax, presented with total paresis of the right upper limb. Of note, a patient with a massive abdominal-pelvic PN experienced an obstructive renal failure, caused by urinary tract obstruction. A PN in the orbital region caused a severe ptosis in a patient, which led to a complete obliteration of the palpebral fissure. Overall, the symptomatology showed a distribution consistent with the localization of PN.
Outcomes
Summary of Patients’ Outcomes. BID = bis in die. PD = Progressive Disease. SD = Stable Disease. PR = Partial Response.

Radiological evaluation
Characteristics of the Target Lesions and Their Evolution Over Time During Treatment.

A radiological partial response (PR) was achieved in three of 13 patients (23%). Of note, in 2 patients the disease volume was reduced by almost a third (from 938.6 cm3 at baseline to 287.1 cm3 and from 11.9 cm3 to 4.4 cm3, respectively) at 12 months from the start of therapy (Figure 1). Stable disease (SD) was found in 9/13 patients (69%). Only 1 patient of 13 experienced a case of progressive disease (PD), with a considerable growth at the first control (+245%). A biopsy was performed and a malignant peripheral nerve sheath tumor (MPNST) was diagnosed at the histological examination, so MEKi treatment was stopped. The treatment is still ongoing for the other patients. MRI images showing impressive volume reduction of a head-neck PN from T0 (baseline) [left] to T2 (after 12 months) [right].
The median time to onset of response to treatment was 12 cycles (6-30 range). At T2, the volume of neurofibromas showed a median change of .74% in reduction, with a range from - 69.4% to +245.9% (SD = .74).
Clinical Evaluation
British Medical Research Council Muscle Power Scale Results at Different Timepoints.

Pain Valuations at Different Timepoints According to Visual Analog Scale.
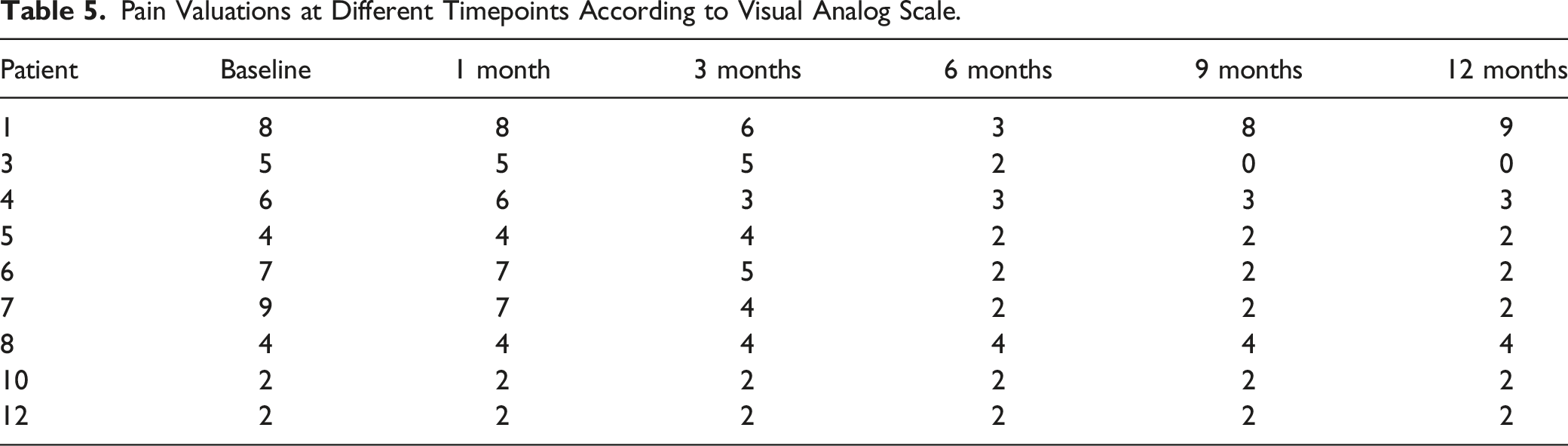
Among four patients with disfigurement, 1 experienced an improvement.
Quality of Life
Of 13 patients described in our cohort, two were excluded from the QoL assignment due to lack of compliance, whereas 1 was too young to respond adequately.
From our data, it emerged that the patients’ QoL, assessed at baseline (65.6 [SD = 19.8]), improved as early as the third month (81.5 [SD = 16.5]), with a continuation of the improvement in the following months, although tending towards a stabilization compared to the first period, reaching a value of 82.5 (SD = 19.1) at the third assessment (9 months) and a value of 83.9 at 12 months (SD = 17.9), as shown in Figure 2. In Table 6 the mean values of the questionnaire at the baseline and at the final evaluation (12 months) are reported. Significative improvement was achieved in three areas of the questionnaire: health perception (P = .02), emotionality (P = .01) and a high significance of the total value (P = .007) QoL evaluation at different timepoints. Mean Values of the QoL Questionnaire at the Baseline and at the Final Evaluation.

It can also be noted that the areas that obtain a greater increase in QoL are related to Health (T0 = 53.4 [SD = 33.4] - T1 = 70.0 [SD = 24.1]) and School Activities (T0 = 66.0 [DS = 28.9] - T1 = 82.8 [DS = 18.2]), as shown in Figure 2; this evidence confirms the perception of clinical improvement (reduction of symptoms, better physical and routine well-being, decrease in admissions) resulting in greater social and educational participation and learning.
Figure 3 shows the average values of the questionnaire administered to the caregivers, in which only four patients were able to be evaluated. Although there is evidence of an improvement in the quality of life after the start of the drug both in the self-assessment report and the Proxy evaluations, aimed at parents, it is possible to detect, even if the sample is small, how the scores are higher in the quality of life perceived by patients compared to that experienced by the caregiver (ΔPedsTOT proxy = 14.9; ΔPedsTOT patients = 18.3). Mean values of QoL administered to caregivers.
Toxicity
Toxicities Observed According to CTCAE 5.0.
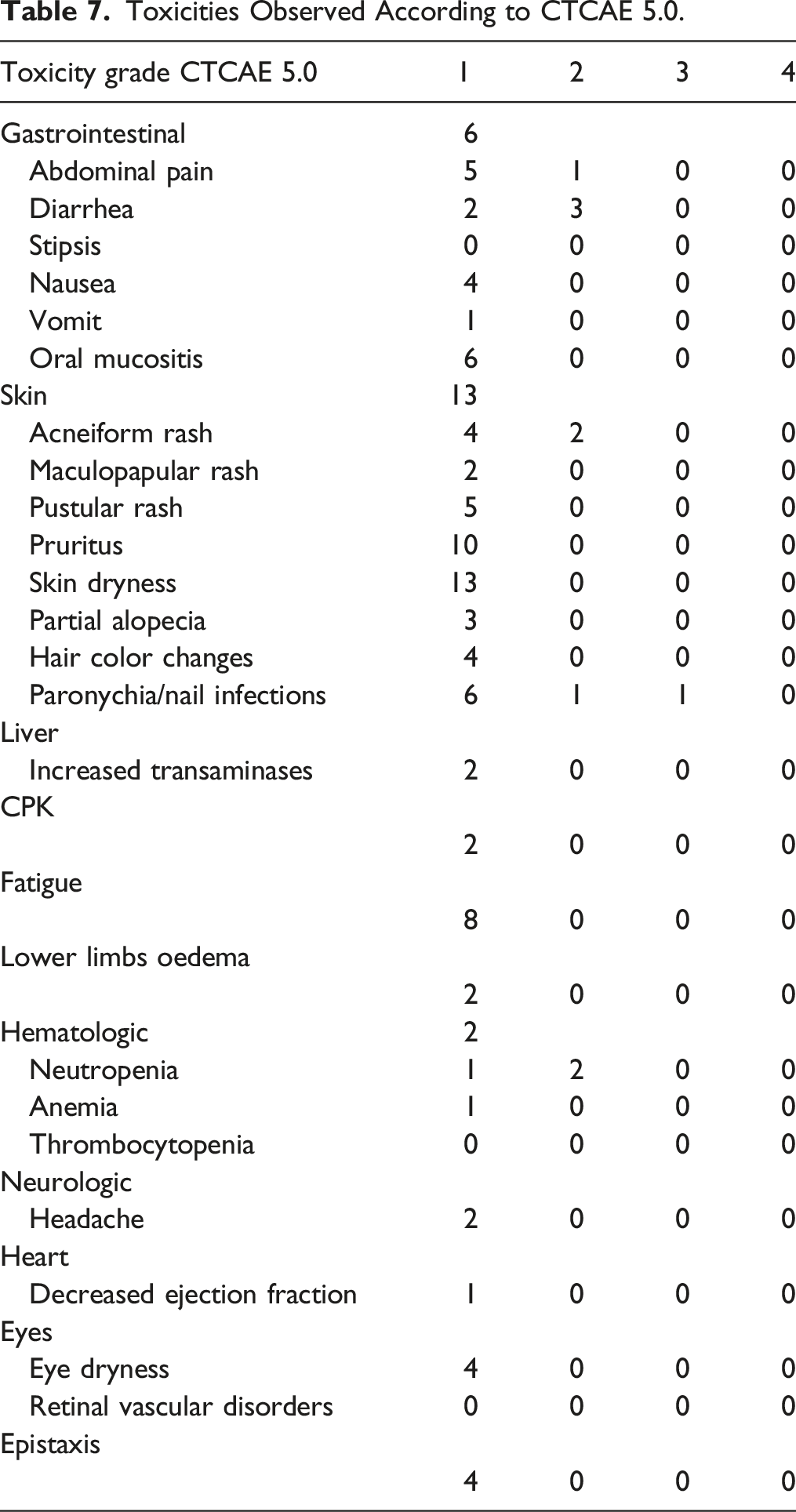
All patients experienced dermatological toxicity. Of note, 13/13 (100%) reported dry skin and 10/13 (78%) pruritus. Skin dryness involved less than 10% of body surface area (BSA) in all patients; there was no associated erythema, and pruritus was also modest in all patients, without leading therapy discontinuation. Two skin toxicities were of higher severity, namely acneiform rash and paronychia/nail infection. The former presented in the form of papules or pustules on a skin surface between 10 and 30% of the BSA in two patients (grade 2); paronychia required surgery with both local and systemic antibiotic therapy in 1 patient (grade 2) and surgery and antibiotics in another patient (grade 3). Gastrointestinal toxicities were the second most frequent: 6/13 patients (46%) developed grade 1 oral mucositis; 6/13 (46%) experienced abdominal pain, that only in 1 case interfered with activities of daily living (ADL); the most severe gastrointestinal toxicity was diarrhea, found in 5/13 patients (38%). An asymptomatic reduction in the left ventricular ejection fraction (LVEF) occurred in only 1 patient (8%). The other toxicities were: asthenia, headache, dry eye, edema in the lower limbs, and alteration of transaminases and creatine phosphokinases.
Two patients had to discontinue selumetinib during the first three months, due to grade 2 gastrointestinal toxicity (diarrhea) and grade 3 paronychia, respectively. After 10 days of suspension and supportive therapy, treatment was resumed at full dose in both cases. No patient required dose reduction.
Discussion
Until recently, therapeutic options were limited to surgical resection of symptomatic PNs only. Recent guidelines on the management of PN in NF1 6 have highlighted the need for specific management for these conditions. 32 Although benign, PNs are associated with significant comorbidity due to the compression of the surrounding areas and the infiltration of the nerve branches from which they originate and for which they cause pain, motor deficits, and disfigurement. 33 PNs also have the potential for malignant transformation into malignant peripheral nerve sheath tumors (MPNST), the leading cause of death in individuals with NF1. 34
Due to the typical infiltrating growth pattern along the nerve course, resection is always partial and burdened by numerous severe perioperative complications. 1 For these reasons, the need for additional treatments for PNs is widely recognized and a priority in the field of research in NF1. During past decades, several attempts have been made with different classes of drugs, such as antihistamines (ketotifen) as well as anti-angiogenetics (thalidomide), in the medical treatment of PNs.35,36 However, no satisfactory results were obtained.
Continuous advances in knowledge of the biology and molecular pathogenesis of PNs have driven interest in targeted treatment approaches. Sirolimus and everolimus were considered promising candidate compounds because they inhibit the mTOR pathway, which is constitutively activated in NF1 and neoplastic cells. 13 However, the data collected in clinical trials failed to provide any demonstration of response and radiological or clinical efficacy.13,37
Antifibrotic activity of imatinib and pirfenidone has generated several expectations. They do not act directly on neoplastic Schwann cells, but on the tumor microenvironment and on the additional cellular components typical of neurofibromas. In particular, imatinib acts on the stem cell factor (Kit-ligand), responsible for the interaction between the neoplastic Schwann cells and the mast cells, which are 1 of the major components of neurofibroma microenviroment, resulting in the mast cell activation. 12 Pirfenidone, on the other hand, acts on various cytokines and growth factors involved in the development of fibrosis such as TGF-β, secreted by the mast cells. 14 However, only modest results have been achieved.12,14
More recently, the drug that has shown the best results in increasing the time to progression (TTP) of PN in NF1 was pegylated IFN, which compared to the placebo showed more prolonged radiological stability over time in the PNs of treated patients; its usefulness has been limited however by the often intolerable toxicity. 11 Furthermore, with a rebound effect upon the suspension of the agent, the PNs showed an increase in size.
Ras pathway represents another interesting therapeutic target: in NF1 the loss of neurofibromin expression leads to constitutive activation of this pathway (responsible for tumorigenesis) and thus of the MAPK signal transduction cascades. 38 Tipifarnib blocks this pathway inhibiting Ras farnesylation but did not demonstrate clinical efficacy in treating PNs.15,39
Importantly, the MAPK signal transduction cascades work through the sequential activation of protein kinases (ERK1/2, c-Jun N-terminal kinase, p38 MAPK, and ERK) which regulate different cellular processes such as proliferation, differentiation, apoptosis, and migration. 40 Mutations in the ERK1/2 transduction cascade (extracellular signal-regulated kinase) are the most common oncogenic factors. 41 In this signaling pathway (Ras-Raf-MEK-ERK), MEK is a kinase characterized by a double specificity: it activates ERK by phosphorylating both tyrosine and threonine residues. Through this mechanism, the accuracy in signal transduction greatly increases, preventing errors in ERK activation. 40 The fundamental role that the ERK/MAPK pathway plays in carcinogenesis is widely recognized. 42
All these reasons made MEK inhibitors an appealing approach in treating PNs.
A phase I study, published in 2016, reported the evidence of the volumetric reduction of PN in pediatric patients who were treated with MEKi. 43 This study was followed by the Phase II trial, published in 2020, which confirmed the previous radiological response of a 20% reduction in tumor volume. In particular, after an average treatment period of 3 years, 74% of patients treated with selumetinib showed a partial radiological response with a reduction in PN volume greater than or equal to 20% and a reduction in pain; 48% reported an improvement in QoL and 56% and 38% achieved an increase in strength and in range of motion of the limbs, respectively. The median time to the best response to selumetinib was 16 treatment cycles (4-36 range). The incidence of adverse effects was confirmed compared to the phase I trial: 38% of patients showed dose-limiting toxicities and 10% had to stop therapy. 44 Therapy with MEKi has demonstrated a tolerable toxicity profile with the most common adverse events coinciding with gastrointestinal (nausea, diarrhea, vomiting), dermatological (acneiform rash, mucositis), increased creatine kinase, and paronychia. 45
The present work analyzes a monocentric series of 13 patients with inoperable and/or symptomatic NF1 and PNs treated with MEKi (selumetinib or trametinib).
The primary objective of the study was to evaluate the outcome in terms of radiological and clinical response to treatment. Secondary objectives were the assessment of treatment toxicity and the impact of therapy on the quality of life. The therapy was used following an expanded access plan, that is, for nominal compassionate use. As already mentioned, in NF1, the loss of functional neurofibromin causes the activation of the classic RAS-MAPK-MEK signaling cascade. MEK inhibitors (MEKi), initially used for cancers such as melanoma and lung cancer, 16 are selumetinib (AZD6244; ARRY-142886), second-generation allosteric MEKi and trametinib (GSK-1120212). The primary objective of our study was the evaluation of the efficacy of MEKi in radiological terms, evaluating the periodic volumetric magnetic resonance imaging (vMRI) and determining the volume of each PN, at each re-evaluation. One-dimensional (1-D) and two-dimensional (2-D) tumor measurements 1 have limited value in determining the treatment outcome of PNs, which are often large, have a complex (non-spherical) shape, and have a slow growth pattern. The Short TI Inversion Recovery (STIR) sequence is the sequence that allows the best characterization; the international commission REiNS standardized the use of vMRI in a consensus, thus standardizing radiological investigations in patients with NF1 and PN. 27 In order to reproducibly quantify the size of the PNs, following the REiNS indications, three diameters and the volume calculation are used. Differently from what was seen in phases I and II of the trials,43,44 in our study only 3/13 patients (23%) had a partial response (PR), with a reduction of the volume of the mass in the percentage of 69%, 42% and 63% from baseline, respectively. The rate observed in those studies was 68%. This difference may arise from the limited follow-up period and the small number of patients enrolled in this study. The most frequently observed response was disease stability (DS), found in 9/13 (69%) patients at vRM assessments. In 1 patient, a disease progression (DP) was evident and justified a new biopsy, which eventually led to the diagnosis of malignant transformation. In addition to the volumetric reduction of the lesion due to resonance re-evaluations, 8 patients (61%) had a clear clinical improvement characterized by the disappearance of pain and functional limitation, comparable to what has already been demonstrated by Gross et al. 44 Clinical assessments used objective assessment tools such as the British Medical Research Council Scale for Muscle Strength for motor neurological deficits and the Visual Analog Scale for pain. Muscle strength increased in 5/11 patients (45%), with a mean increase of .8 points (SD = 1.0; P = .03). Pain decreased in 5/9 patients (55%), with a mean decrease of 2.33 points (5.22 at baseline and 2.89 at 12 months [SD = 2.8; P = .03]). The discrepancy between the overall rate of symptom improvement (8/13 patients, 61%) and the tumor volume reduction rate (3/13 patients, 23%) was also observed in the phase II SPRINT study. 44 Another parameter of response to treatment was the determination of QoL in a multidimensional test which considers the impact that the disease and the treatment have on the child/adolescent’s self-perception. For the pediatric population, the PedsQL TM 4.0 Generic Core Scale [96] is used. In our study, quality of life improved in 8/10 patients, with an average score increase of 18.2 from baseline. Although there is an improvement in the quality of life after the start of treatment both in the self-report and in the Proxy evaluations, addressed to parents, it is noted that the scores are higher in the quality of life perceived by patients than that felt by the caregiver (ΔPedsTOT proxy = 14.9; ΔPedsTOT patients = 18.3). The data would suggest that the decrease in hospitalizations, due to the implementation of alternative therapies compared to the standards, is an important factor that would benefit the QoL perceived by patients and their families. In the Gross et al. study, 44 the majority of pediatric patients and parents (72% and 86%, respectively) reported improvements from baseline on the Global Impression of Change scale, and worsening toxic effects related to selumetinib, all of which were reversible on discontinuation of treatment.
MEK inhibitors in pediatric patients appear to be well tolerated. Gastrointestinal toxicities, increased creatine kinase and skin toxicity (according to CTCAE 5.0 grade) are more frequently observed. 43
MEK inhibitors demonstrated the highest rate of both tumor volume reduction (23% patients with PD vs 5% with Peg-IFNα2b and 17% with imatinib) and symptom reduction (61% of patients vs 30% with imatinib) and of the improvement of the QoL (80% of patients vs a slight effect on the cognitive and emotional spheres of tipifarnib).
In the two pediatric studies of selumetinib,43,44 elevation of creatine kinase and a rash were the most common grade 3 toxicities. Fatigue was also commonly reported but generally did not require dose modifications. In our study, the toxic effects of selumetinib were similar to those reported in the literature. No serious and irreversible critical events were reported. No patient presented symptomatic cardiotoxicity. The main reported toxicity was at the cutaneous level (13/13, 100%). Two patients had to discontinue treatment in the first 3 months due to grade 2 gastrointestinal toxicity (diarrhea) and grade 3 paronychia, respectively. For both, after 10 days of suspension and supportive therapy, selumetinib was resumed at 100% of the dose. To date, it is unclear whether treatment with the MEK inhibitor can be stopped after a certain period of time or whether this leads to a resumption of disease progression. Dombi et al. reported slow tumor regrowth after selumetinib dose reduction due to toxic side effects. 43 This indicates a dose-dependent effect of MEK inhibition and suggests that neurofibromas may progress after the conclusion of therapy.
Another question is knowing when to start treatment with MEK inhibitors. It is worth asking whether early initiation of MEK inhibitor treatment could have prevented neurofibroma growth and subsequent neurological symptoms or, at best, prevented malignant transformation in a patient enrolled in this study.
In our study, selumetinib and trametinib resulted in radiological stabilization in most of the patients analyzed and provided clinically significant benefits in terms of both function and quality of life. The level of toxicity and the absence of cumulative toxic effects allowed for long-term treatment.
Limitations to the use of MEKi are represented by the lack of complete radiological response in 100% of cases and the need to prolong treatment for the progression of rebound disease upon discontinuation; for these reasons we are currently evaluating the association with tyrosine kinase inhibitors (cabozantinib) that could have a crucial role in the action on the tumor environment in order to reach an era of pharmacological treatment for PNs. 6
Our study presents several limitations: first, the retrospective design and the small cohort limited the generalization of the results. In addition, the disfigurement was evaluated only by clinicians without using standardized tools as well as the muscle streght was not always tested by the same physician at each visit, making these measurements susceptible to inter-observer bias.
Conclusions
NF1 is a genetic condition predisposing to the development of neoplastic diseases; PNs are the most common clinical manifestation of NF1. Historically, the surgical approach has always been the first therapeutic option in the treatment of symptomatic plexiform neurofibromas, in the face of their natural tendency of progression and morbidity related to the resection itself. Recently, the range of choices has been enriched with new target therapies, among which MEKi drugs, such as selumetinib and trametinib, have shown great promise.
In our single-center experience, MEKi treatment has been shown to be both safe and effective in improving symptomatology, representing nowadays the best card to play against PNs. However, some issues such as the rebound effect upon discontinuation and the lack of complete response to treatment still persist, calling for further studies in search of more effective treatments.
Footnotes
Acknowledgments
We are grateful to all patients and their families.
Authors’ Contributions
Conceptualization, A.C. and A.M.; Data curation, M.D., F.F., S.R, G.M. and G.M.M.; Formal analysis, M.A.D.I., F.F. and R.A.; Supervision, L.B, G.D.B., A.C., C.I., G.M.M. and A.S.; Writing—original draft, M.A.D.I., F.F. and M.L.; Writing—review and editing, M.A.D.I. and A.M. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Institutional Review Board of the Bambino Gesù Children’s Hospital of Rome, Italy (RAP-2022-0008, 23th June 2022).
Informed Consent Statement
Informed consent was obtained from all subjects involved in the study.
