Abstract
Introduction
The association between the expression of HIF-1α in the laryngeal carcinoma and the prognosis of disease is quite well documented, but the significance of HIF-1α C1772T polymorphism and its relation to disease phenotype have to be clarified. The aim of this study was to investigate the influence of C1772T polymorphism on the clinical-pathological characteristics and disease-free survival after initial surgical treatment of patients with laryngeal carcinoma.
Materials and methods
The prospective cohort study included 65 patients with laryngeal carcinoma. Two representative tumor tissue specimens were taken in each patient during surgery; 1 specimen was used to asses HIF-1α C1772T polymorphism and the other 1 to determine the immunohistochemical expression of HIF-1α, VEGF, as well as CD 34 proteins. The comparison of polymorphism frequency between study and control population was conducted by collecting a 5 mL of peripheral venous blood samples in each subject.
Results
Clinicopathological characteristics of laryngeal carcinoma didn’t affect the expression of hypoxia-related biomarkers, such as HIF-1α, VEGF or MVD. The statistically significant association between HIF-1α and VEGF expression was found (P = .034), but not between HIF-1α expression and MVD value (P = .696). The expression of HIF-1α was significantly higher among CT heterozygotes (P = .029). We found a significantly more recurrence among CT heterozygotes compared with patients with CC homozygous alleles (57.10% and 24.30%, respectively; P = .007). Patients with C1772T polymorphic variants had significantly worse disease-free survival compared with patients without polymorphism (Log-rank test, P = .007).
Conclusion
HIF-1α C1772T polymorphism was significantly associated with worse disease-free survival which nominates it as a predictor of laryngeal carcinoma relapse. The preoperative assessment of hypoxia-related biomarkers should be used in everyday practice in order to determine the treatment modalities for laryngeal carcinoma.
Keywords
Introduction
The incidence and prevalence of laryngeal squamous cell carcinoma (LSCC) have both increased during the past 30 years (12% and 23.8%, respectively). 1 With nearly 200.000 of newly discovered cases per year, these tumors are the most common head and neck carcinomas. 2 They are more frequent in males than in females (5:1) with peak incidence in the sixth or seventh decade of life. Tobacco and alcohol consumption increase the risk for these tumors. Treatment options for laryngeal carcinoma are limited and the 5-year survival rate is about 60%. 3
Despite their moderate frequency, biology of these tumors is not well understood. However, it is known that hypoxia plays a significant role in the pathogenesis of LSCC 4 ; therefore, the effect of hypoxia-related biomarkers remains to be elucidated. The key transcription factor that mediates cellular adaptive response to hypoxia is Hypoxia-inducible factor-1 (HIF-1). HIF-1α induced genes provide dual protection to the growing tumor, promoting the development of blood vessels and helping tumor tissue to metabolically accommodate to reduced oxygen level. Vascular endothelial growth factor (VEGF) seems to be the critical factor responsible for blood vessel development and represents a downstream target of HIF-1α. The relation between HIF-1α and VEGF is interfered with various proangiogenic and antiangiogenic factors and it is more likely that HIF-1α affects the angiogenesis by mechanisms that are much complex than simple VEGF induction, possibly by recruiting additional target genes involved in vessel maturation. Therefore, the correlation of HIF-1α with VEGF expression and microvessel density (MVD), as measurement of angiogenesis in laryngeal carcinoma need further analysis.
It has been demonstrated that overexpression of HIF-1α in the tumor tissue of patients with LSCC is associated with poor locoregional control. 5 Additionally, some known polymorphisms within the HIF-1α gene could affect the cancer phenotype by increasing tumor aggressiveness. Recently, Wu et al demonstrated that HIF-1α C1772T polymorphism is significantly associated with increased head and neck carcinoma risk. 6 This single nucleotide polymorphism (SNP) is the most frequently analysed polymorphism of the HIF-1α gene in the literature. It involves substitution that results in replacement of the amino acids, proline 582 with serine. These alleles appear to be associated with enhanced transcription activity and the expression of HIF-1α in upper aerodigestive tract carcinomas (UADTC). 7 While the association between the expression of HIF-1α in the tumor tissue and the prognosis of disease is quite well documented, the significance of the SNP and its relation to disease phenotype have to be clarified. In general, current data in literature on HIF-1α gene variants and their potential role on development and progression of laryngeal carcinoma are scanty.
The main objective of this study was to investigate the influence of C1772T SNP in the HIF-1α gene on the clinical-pathological characteristics and disease-free survival (DFS) after initial surgical treatment of patients with LSCC. Further, the study investigated the interrelations between the expression of HIF-1α, VEGF and MVD in surgically treated patients with LSCC.
Material and Methods
This prospective cohort study was conducted from 1st March 2013 to 1st March 2019 and was approved by Institutional Ethics Committee (Approval Number 128/2). Study has been carried out in accordance with the Helsinki declaration and the investigators have obtained written consent from each patient and healthy controls for the participation in the study. All patients and healthy controls gave written consent for their anonymized information to be published in this article. Additionally, we de-identified all patient details. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines. 8
Patients were consecutively included in the study if they fulfilled inclusion criteria: (1) The diagnosis of LSCC confirmed histologically by the 2 independent pathologists; (2) Initial surgical treatment carried out due to LSCC; (3) Initial surgical treatment approved by the Institutional Oncological Council; (4) Signed written consent for the participation in the study. Exclusion criteria were: (1) patients who underwent previous surgery due to laryngeal malignancy; (2) patients diagnosed with other type of laryngeal malignancy than LSCC; (3) patients who underwent previous chemotherapy or radiation therapy; (4) patients with distant metastasis; (5) patients with synchronous malignant tumors and (6) patients that were followed inconsistently due to lack of their motivation.
Two representative tumor tissue specimens were taken in each patient during surgery; 1 specimen was used to asses C1772 T SNP of HIF-1α gene and the other 1 to determine the immunohistochemical expression of HIF-1α, VEGF, as well as CD 34 proteins. The comparison of SNP frequency between study and control population was conducted by collecting a 5 mL of peripheral venous blood samples in each subject.
The tumors were stratified according to the eighth Edition of the TNM classification, UICC. According to previous studies, laryngeal carcinomas were stratified in 2 groups considering the size of primary tumor (early stage, T1-T2; advanced stage, T3-T4) and cervical lymph node metastasis was evaluated as present or absent. 4 Surgical and postoperative oncological treatment were carried out according to the standard protocol approved by the Institutional Oncological Council. Each patient was followed for a period of 5 years, assessing the frequency of disease recurrence.Fresh representative tumor tissue samples from each patient were immediately frozen at −800C. The DNA was extracted using standard methods, as described previously. 7 The DNA from peripheral blood was isolated using the QIAamp DNA Mini Kit from Qiagen (Hilden, Germany) according to the manufacturer`s instructions. To assess HIF-1α we focused on the polymorphisms found in human HIF-1α that resulted in an amino acid substitution within exon 12. Consequently, PCR was performed to amplify the 178-bp fragment of human HIF-1α gene Using a primer set, HIF-1α (forward 5′-CAT GTA TTT GCT GTT TTA AAG-3′) and HIF-1α (reverse 5′-GAG TCT GCT GGA ATA CTG TAA CTG-3′) under the following conditions: 30 cycles of denaturing at 95°C for 30 s, annealing at 61°C for 30 s and extension at 72°C for 30 s.1,2 PCR products were bidirectionally sequenced in capillary electrophoresis (ABI PRISM Genetic Analyzer 3130; Applied Biosystems, USA) using the aforementioned primers.
CATGTATTTGCTGTTTTAAAGGACACAGATTTAGACTTGGAGATGTTAGCTCCCTATATCCCAATGGATGATGACTTCCAGTTACGTTCCTTCGATCAGTTGTCACCATTAGAAAGCAGTTCCGCAAGCCCTGAAAGCGCAAGTCCTCAAAGCACAGTTACAGTATTCCAGCAGACTC)
Tumor tissue was fixed in 10% neutral buffered formalin for 10 hours and embedded in paraffin. Hematoxylin and eosin-stained histological sections were used to confirm the diagnosis of LSCC. Immunostaining was performed on 4-μm sections. The expression of HIF-1α, VEGF, as well as CD34 molecules was demonstrated by immunohistochemical methods and intensity of expression in tumor cells was assessed by semiquantitative analysis.
Mouse monoclonal anti-human HIF-1α antibody (Abcam, Cambridge, UK) und rabbit polyclonal anti-human VEGF antibody (Santa Cruz Biotechnology, Santa Cruz, California, USA; sc-152) were used to detect HIF-1α and VEGF expression. A specific and sensitive immunohistochemical method EnVisionTM+Dual Link System-HRP (DakoCytomation, Carpinteria, California, USA; code K4065) was applied to assess the expression of individual markers HIF-1α and VEGF. Also, double immunohistochemical staining was applied, which enabled simultaneous expression of HIF-1α and VEGF on the same tissue section, using EnVisionTM G/2 Doublestain System, Rabbit/Mouse (DakoCytomation, Carpinteria, California, USA; Code K5361). During the immunohistochemical staining procedures, the principles of quality control and specificity of staining were followed by applying positive and negative control procedures, according to the propositions of UK NEQAS (UK National External Quality Assessment for Immunohistochemistry).
All morphometric and stereological analysis were performed on Olympus BX-51 microscope networked with a CCD video camera (PixeLink) connected to a 19″ PC monitor, using a computer software for microscopic image analysis (new CAST stereological software package, VIS - Visiopharm Integrator System, version 2.12.1.0; Visiopharm; Denmark).
The level of HIF-1α expression was scored according to the system used by Silva et al. 9 The scoring system was as follows: 0, no nuclear staining; 1, nuclear staining less than 10%; 2, nuclear staining 10-29% and 3, nuclear staining 30% or higher. Scores 0 or 1 were considered low and scores 2 or 3 were considered high HIF-1α expression. On the other hand, a cut-off of 20% staining was used to differentiate low and high VEGF expression, according to Kyzas et al. 10 The assessment was independently carried out by 2 investigators.
We used CD34 for microvessel staining and determination of MVD. Three areas with the highest numbers of stained microvessel were identified as “hot spots” on light microscopy and were used for microvessel counting on high-magnification. MVD was expressed as the number of stained microvessel in the optical field over each hot spot.
Statistical Analysis
Data were presented as n (percentage) and mean ± standard deviation. Pearson’s chi-squared test, Fisher’s exact test and Mann-Whitney U test were used to test statistical hypotheses. The analysis of time elapsed between surgical treatment and recurrence of malignant disease was performed using Kaplan-Meier method and Log-rank test was used to compare survival distributions of patients with and without polymorphisms. Potential predictors of laryngeal carcinoma relapse were determined by Cox regression analysis. A P value of ≤.05 was considered to be statistically significant. Statistical data analysis was performed using IBM SPSS Statistics 22 (IBM Corporation, Armonk, NY, USA).
Results
Age, Gender and C1772T Genotype Distribution of Study and Control Group of Patients.

Correlation of Clinicopathological Characteristics with MVD, HIF-1α and VEGF Expression.

Genotypic distribution of C1772 T polymorphism of HIF-1α gene in both patients and controls (Table 1) exhibited no deviations from the Hardy-Weinberg equilibrium (respectively, χ2 value = 2.697 and χ2 value = 2.742; P = .358). The presence of C1772 T SNP of HIF-1α gene was not associated with the size of the primary tumor (P = .969) or the presence of cervical lymph node metastasis (P = .950).
Correlation of HIF-1α and Recurrence with MVD, VEGF Expression and C1772T Polymorphism.

Bold values were considered to be statistically significant (P value of ≤.05 ).

Immunihistochemical analysis in patient with HIF-1α C1772T polymorphism. A) High expression of HIF-1α protein in tumor tissue (magnification ×20), and B) High expression of VEGF protein in tumor tissue (magnification ×40).
All patients were followed up for 5 years after surgery and 38% of them developed recurrent disease. The mean period or recurrence was 21.04 ± 5.29 months, with a range of 5-56 months. Out of 25 patients with recurrent disease, 12 patients developed local recurrence, 5 patients had cervical lymph node metastasis and locoregional recurrence was found in 8 patients. Distant metastasis was found in 4 patients and disease-related mortality of patients with recurrent disease within 5-year follow-up was 28%.
Recurrence of malignant disease wasn’t affected by the size of the primary tumor or the initial presentation of patients with cervical lymph node metastasis (P = .746 and P = .495, respectively). On the other hand, we found a significantly more recurrence of LSCC among patients with CT heterozygotes of HIF-1α C1772T compared with patients with CC homozygous alleles (57.10% and 24.30%, respectively; P = .007). Further analysis considering the size of primary tumor revealed that the correlation between recurrence and C1772T SNP was significant in patients with early stage of primary tumor, but not in patients with advanced stage (P = .004 and P = .313, respectively). DFS was estimated using Kaplan-Meier curve (Figure 2) and compared according to the HIF-1α genotype. Patients with C1772T polymorphic variants had significantly worse DFS compared with patients without polymorphism (Log-rank test, P = .007). According to the results of Cox regression the only potential predictor of LSCC relapsing was the presence of CT heterozygote of HIF-1α C1772T (HR = 2.931, 95%CI HR = 1.29-6.65, P = .010). Recurrence wasn’t affected by the level of VEGF expression or MVD (respectively, P = .170 and P = .326). Disease-free survival in patients with CC homozygous and CT heterozygous alleles of HIF-1α C1772T.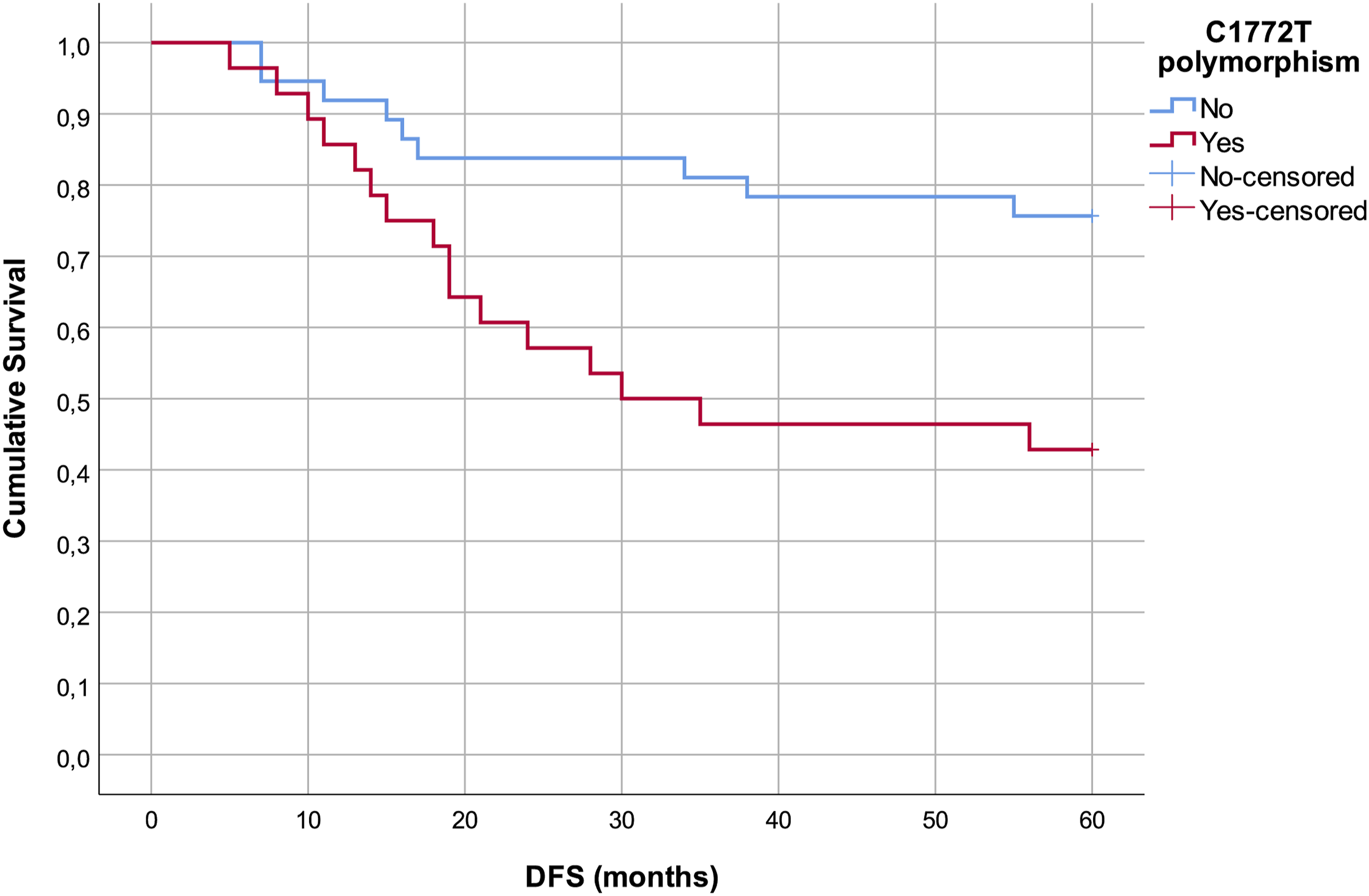
Discussion
This prospective clinical study on patients with laryngeal carcinoma revealed: (i) the statistically significant association between HIF-1α and VEGF expression in LSCC, but not MVD, (ii) a significant correlation between C1772T SNP in the HIF-1α gene and overexpression of HIF-1α protein in tumor tissue and (iii) significantly worse DFS in patients with CT heterozygotes of HIF-1α C1772T.
Hypoxia occurs during the development of solid tumors and is considered to be a key feature of tumor microenvironment. Under conditions with low oxygen level in tumor tissue, a wide array of adaptive behaviors is triggered, leading to acquisition of a more aggressive tumor phenotype. Evidence has shown that hypoxia affects the prognosis and outcome of different types of solid tumors, including LSCC by promoting proliferation, differentiation, invasion and epithelial-mesenchymal transition.11,12 Considering the crucial role of hypoxia during the LSCC progression, hypoxia-related biomarkers are expected to provide substantial information about recurrence and survival of LSCC patients. Targeting the transcriptional factors involved in hypoxic response might provide fresh perspectives in oncological treatment. 13
Phenomenon that HIF-1α drives initial response to hypoxia and HIF-2α subsequently takes over the main role during abiding hypoxic condition is described in the literature as the HIF switch. Befani and Liakos confirmed distinct functions of HIF-α isoforms by reporting that HIF-1α is involved in cell proliferation and migration in early angiogenesis, whereas HIF-2α promotes remodeling and the maturation of the microvasculature. 14 While the HIF switch is quite evident and documented during the evolution of renal cell carcinoma, literature data regarding laryngeal carcinoma are inconclusive.15,16
Study on oral carcinomas showed that the expression of HIF-1α is an early event in carcinogenesis. 17 The explanation behind this theory is that HIF-1α expression reflects the level of oxygen reduction in tumor tissue, which is a characteristic of fast-growing tumors, particularly early forms of carcinomas. Kwon et al found various radioresistance among patients with early stages of LSCC, depending on the different levels of HIF-1α expression and tumor hypoxia. 5 Furthermore, Rzepakowska et al reported HIF-1α overexpression in intraepithelial vocal fold lesions with early stage of dysplasia, affecting invasiveness and development of cellular atypia. 18 The relationship between hypoxia and the size of primary tumor is not as linear as previously thought. Actually, the lower limit of hypoxia tolerance and tumor response to hypoxia depend on the type of healthy tissue from which the tumor originated, the pathohistological type of tumor and localization. Our data showed no relation between HIF-1α expression on 1 side and tumor size or the presence of cervical lymph node metastasis on the other side (P = .515 and P = .172, respectively), indicating that clinical characteristics of a tumor don’t necessarily reflect its aggressiveness. Thus, the clinical stage of the disease is greatly influenced by the time that has passed from the onset of symptoms to the diagnosis of the malignant disease. It is more likely that hypoxia and HIF-1 expression are be more pronounced in fast-growing stages than in advanced stages of the malignant disease.
On the other hand, we found direct correlation between HIF-1α and VEGF expression in tumor tissue. Proangiogenic effects of HIF-1α in cancer cells are achieved under hypoxic conditions through the synthesis of angiogenesis-related-proteins such as VEGF. The mechanism which explains mentioned relationship is that HIF-1α activate the transcription of hypoxia-responsive genes by binding to specific five-nucleotide DNA sequence, known as the hypoxia response elements (HRE). The presence of HREs has been demonstrated in the promotor regions of numerous genes, including VEGF. 19 The downstream regulation of VEGF by HIF-1α is well exploited for the development of new antiangiogenic agents that targets HRE sequence and inhibits VEGF expression. 20
Popov et al demonstrated significantly higher VEGF levels in patients with LSCC who had upregulated HIF-1α expression. 15 Despite some controversies regarding HIF-1α and VEGF correlation in head and neck squamous cell carcinoma (HNSCC), these signaling molecules exert a coordinated effort to overcome hypoxic conditions. 21 Concurrent upregulation of HIF-1α and VEGF expression in LSCC found in our study confirms hypoxia and neovascularization as certain hallmarks of cancer.
Literature data are debatable whether VEGF overexpression is an early event in LSCC development or whether it is associated with an advanced tumor stage.22,23 Despite some reports pointing out a significant correlation between VEGF expression and the presence of LSCC cervical lymph node metastasis, most authors agree that there is a lack of relation of VEGF and T or N stage of laryngeal carcinoma. Tumors with high VEGF expression are characterized by disturbed process of angiogenesis and impaired vascular integrity which allows easier intravasation and dissemination of malignant cells. On the other hand, tumors with lower VEGF expression and less pronounced angiogenesis have a larger intercapillary distance and are therefore more susceptible to hypoxia, which can affect tumor aggressiveness in different ways, interfering in the relationship between VEGF and clinical characteristics of LSCC.
Our results showed that 49% of patients with advanced stage of primary tumor and 50% of patients with cervical lymph nodes metastasis had low level of VEGF expression, confirming a lack of correlation between stage of the disease and VEGF expression.
The results of our study showed no significant association between MVD on 1 side and HIF-1α or VEGF on the other side. By definition, MVD corresponds to the number of blood vessels stained on a given surface and accordingly reflects the intercapillary distance. It varies among different types of tumors and depicts the characteristics of healthy tissue from which the tumor originated, as well as newly formed tumor tissue. Vascularization of the tumor should reflect its metabolic demands; but unlike normal tissue, tumors very often develop blood vessels more extensively than is needed to settle energy requirements. Also, there is a difference in the ability of the tumor blood vessels to carry oxygen and nutrients. Therefore, it could be suggested that, despite the common opinion, MVD is not a true measure of tumor angiogenic activity, nor does it reflect functional status of the neovasculature. Furthermore, the total angiogenic potential of tumor microenvironment should be considered as the sum of the activities of positive and negative angiogenesis regulators. It is certain that a correlation between MVD and VEGF or any other single angiogenic regulator can’t be established in laryngeal cancer due to insufficiency of true angiogenic expression by MVD.
Zvrko et al reported MVD as a prognostic factor for the outcome of patients with LSCC that could help to identify patients with increased risk of malignant recurrence. 24 Previous meta-analyses tried to identify the correlation between MVD and 5-year overall survival in patients with HNSCC25,26; however, no precise conclusion was reached. Results from our study showed no impact of MVD on recurrence of LSCC. Deficiency of MVD to assess the angiogenesis quantitatively may be 1 of the reasons for the lack of this correlation.
Meta-analysis conducted by Yang and associates supported the hypothesis that C1772T SNP in the HIF-1α gene is a susceptibility marker of cancer. 27 True nature of the relationship between HIF-1α SNPs and carcinogenesis is still unclear. Malignant disease may arise after activation of oncogenes or after deactivation of tumor suppressor genes and it requires cell transformation which is almost certain that C1772 polymorphism in the HIF-1α gene is not able to be accomplished. It is more likely that the polymorphisms may affect some properties of previously existing tumors, such as growth potential or invasiveness. The single nucleotide polymorphism in HIF-1α gene C1772T is associated with increased expression of HIF-1α protein in UADTC. 7 Therefore, it emphasizes the HIF-1α pathway by up-regulation of HIF-1α expression and promotes aggressive malignant phenotype. Tanimoto et al reported higher MVD values and more advanced stages of primary tumor in patients with CT heterozygotes of HIF-1α C1772T. 28 Furthermore, some literature data indicate a predisposition of TT homozygous variant to the development of more voluminous LSCC and neck lymph node metastasis. 4 Contrary to the previous authors, our results showed the lack of correlation between the presence of HIF-1α variant form and clinical-pathological characteristics of LSCC, although patients with C1772 polymorphism in the HIF-1α gene had an increased expression of HIF-1α protein in tumor tissue.
Also, this SNP has an important part in the early stages of UADTC metastasis and has a negative influence on survival and prognosis. This is the first study that investigated the relationship between C1772 polymorphism in the HIF-1α gene and 5-year survival of patients with laryngeal carcinoma. However, Prasad et al. reported poor survival rate in oral carcinoma patients with CT genotype compared to CC genotype. 29 Our study patients with C1772 polymorphism expressed a higher frequency of local and regional relapse of malignant disease, especially those with early-stage of laryngeal carcinoma. These results support the hypothesis of HIF-1α pathway up-regulation during early stages of LSCC development.
A cancer recurrence is 1 of the major issues in surgical oncology. About 16% of patients with early-stage laryngeal carcinoma fail to respond to treatment, experiencing residual or recurrent disease. 30 A significant number of those patients will require salvage laryngectomy that entails high rate of postoperative morbidity. Tsetsos et al. reported advance-stage of recurrent disease in 84% of patients, confirming that recurrent laryngeal carcinoma is highly aggressive disease. 31 Biomarkers are the molecular signatures and hallmarks of pathological process, therefore may provide essential information about disease prognosis or recurrence. The potential of specific hypoxia-related biomarkers to define aggressive phenotype of LSCC with ominous prognosis is what demonstrate the necessity of their evaluation at the time of initial diagnosis or surgery. The identification of such LSCC patients through evaluation of specific hypoxia-related biomarkers may provide benefit by tailoring the treatment to the individual demands of the patient.
The main limitation of the study is limited number of included patients. Although preferred treatment of LSCC in our referral tertiary center is surgery, numerous patients are initially treated with radiation therapy or chemotherapy. Larger cohorts of patients involving randomized clinical trials would be a step further in determining true usefulness of hypoxia-related biomarkers in diagnostics, treatment and prognosis of LSCC.
Conclusion
The results of this study suggest that C1772T polymorphism in the HIF-1α gene promotes overexpression of HIF-1α protein in tumor tissue of patients with LSCC. This polymorphism was significantly associated with worse DFS, which nominates it as a predictor of laryngeal carcinoma relapse. It is up to future investigations to determine the specific pathway in which HIF-1α C1772T polymorphism affects the survival of patients with LSCC. The preoperative assessment of hypoxia-related biomarkers may provide further insight into malignant potential and aggressiveness of laryngeal carcinoma and it should be used in everyday practice in order to determine the treatment modalities.
Footnotes
Acknowledgments
We are grateful to Svetozar Damjanovic (Clinic for Endocrinology, Diabetes and Metabolic disease, Faculty of Medicine, University of Belgrade, Serbia) for inspiration, suggestions and insightful discussion.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved by the Ethical committee of the Clinical Center of Serbia (Approval Number 128/2). Study has been carried out in accordance with the Helsinki declaration and the investigators have obtained written consent from each patient and healthy controls for the participation in the study. All patients and healthy controls gave written consent for their anonymized information to be published in this article.
