Abstract
Background
25 hydroxyvitamin D [25(OH)D] and serum calcium have been associated with incident prostate cancer (PCa). However, there is limited data on whether these metabolites predict survival in men of African descent, a population disproportionately affected by PCa. We studied the relationship of 25(OH)D at PCa diagnosis with all-cause and cancer-specific mortality among Jamaican men and examined whether serum calcium modified any associations.
Methods
Serum 25(OH)D from 152 Jamaican men with incident PCa within the Prostate Cancer Risk Evaluation (PROSCARE) study were re-evaluated approximately 11 years after enrollment. 25(OH)D analyses were stratified using the using Holick criteria. PCa-specific and all-cause mortality were examined in Kaplan–Meier survival curves and Cox regression models adjusted for age, body mass index (BMI), smoking and Gleason score. Restricted cubic splines evaluated nonlinear associations. Serum calcium was assessed as an effect modifier of the association between 25(OH)D and mortality.
Results
Of cases with available 25(OH)D, 64 men with PCa survived, 38 deaths were PCa specific and 36 died of other causes. At baseline, 9.9% of cases were vitamin D deficient and 61.2% were vitamin D sufficient. Compared to 25(OH)D sufficient men, those with 25(OH)D <20.0 ng/mL concentrations were associated with higher PCa-specific mortality (adjusted HR, 4.95; 95% CI, 1.68, 14.63, P = .004) and all-cause mortality (adjusted HR, 2.40; 95%CI, 1.33, 4. 32, P = .003). Serum calcium was not associated with survival and did not modify any associations with 25(OH)D.
Conclusions
25(OH)D deficiency at PCa diagnosis predicted decreased survival for overall and PCa-specific cancer in Caribbean men of African ancestry.
Introduction
Globally prostate cancer (PCa) was the second most prevalent malignancy among men, with approximately 1.28 million men living with the disease and 360,000 dying from the condition in 2018. 1 PCa incidence among African-American men is 50% higher than in Caucasian men. Additionally African American men are twice as likely to die from PCa as Caucasian men. 2 In the South-east of England, African-Caribbean men had three times higher risk of the disease than their European counterparts. 3 The Prostate Cancer Ethnic Subgroups (PROCESS) study also found that the incidence of PCa in blacks was 3.1 times higher and occurred about 5.1 years earlier than among their Caucasian counterparts.4,5 In Jamaica, a country of predominantly African descent, PCa is the most frequently reported incident malignancy and registry data suggests that the incidence continues to rise.6,7 We have previously reported a mortality rate of approximately 7 deaths per 100 person years in Jamaican men with newly diagnosed PCa. 8
Meta-analysis and competing risk analyses show that high 25(OH)D concentrations significantly increase the incidence of PCa.9,10 Prospective studies provide conflicting findings of the relationship between low circulating vitamin D concentrations and PCa incidence.11-13 In contrast, Mendelian randomization studies which investigate genetic evidence of a causal role for 25(OH)D levels on PCa show no evidence of a relationship.14,15 In temperate countries, Black ethnicity has been associated with a greater likelihood of vitamin D deficiency. Studies have suggested that the high incidence of PCa in men of African ancestry with vitamin D deficiency may explain some of the ethnic disparities in PCa outcomes as the cutaneous synthesis of vitamin D from the vitamin D prohormone is ultra-violet induced, a process which is inhibited by melanin. 16
Despite the association with PCa incidence, Vitamin D may play a beneficial role when it comes to cancer survivorship. In temperate countries, lower serum 25(OH)D levels have been observed among men of African ancestry compared to Caucasians.17,18 The anticancer properties may operate through a range of molecular mechanisms that influence the progression of cancers, including PCa. Studies have demonstrated that physiological levels of vitamin D negatively affect the transduction pathways of different receptor tyrosine kinases by controlling transcription and modulate genomic functions19,20 reducing cell proliferation, inhibiting angiogenesis, activation of cell differentiation and promoting apoptosis in prostate cells. 21
While Vitamin D exerts control on calcium metabolism, the role of calcium in PCa development is uncertain. Investigations linking serum total calcium and PCa mortality have demonstrated increased likelihood of death, 22 a protective effect 23 or no association. 24 High concentrations of calcium reduce renal synthesis of calcitriol [1,25(OH)2D], suggesting that high calcium concentrations may moderate or reduce the useful effect of Vitamin D on PCa.25,26
In a previous analysis of the Prostate Cancer Risk Evaluation (PROSCARE) case control study of incident PCa, we found a positive relationship between serum 25(OH)D and the likelihood of PCa development, with a 23% increase in risk for each 10 ng/mL difference in 25(OH)D. Serum calcium also increased the likelihood of PCa. 27 There is however limited data on serum vitamin D and PCa survival in people of African ancestry, particularly those resident in the Caribbean. 28 We therefore studied the association of circulating serum 25(OH)D on PCa-specific and all-cause mortality in Jamaican men with PCa. We also investigated whether any association was modified by serum calcium levels.
Materials and Methods
Study Population and Follow-Up
The study re-enrolled men with incident PCa from the Prostate Cancer Risk Evaluation (PROSCARE) case control study approximately 11 years after initial participation. Survivors were invited to form part of a cohort study, the details of which have been previously published. 8 In summary, the PROSCARE study enrolled Jamaican men aged 40-80 years old attending urology clinics at two major hospitals and private urologists’ offices in the Kingston Metropolitan area in Jamaica were enrolled in a case-control study from March 2005 to July 2007, (cases, n = 242; controls, n = 275). Cases were incident PCa that were histologically confirmed and classified by histologic grade using the Gleason system. 29 Men with advanced metastatic cancer, previous prostate surgery, with recent severe weight loss, on hormonal treatment or taking finasteride were excluded from the study. Participants were followed up approximately 11 years later. Attempts were made to find these men using their last known contact information, hospital and practice outpatient records and with visits to the last known address to determine their Vital Status up to December 31, 2017. For participants who could not be contacted by telephone or email, the Registrar General’s Department (RGD) database was searched to determine if their death had been registered. The Cause of Death was gleaned from medical records, death certificates on file or obtained from the RGD.
All mortality data were reviewed by an end-points committee. Nonsense codes were removed and the primary cause of death determined according to the International Classification of Diseases and Injuries and coded using ICD-10 codes. Outcomes of interest included deaths from all-causes as well as PCa-specific mortality as documented by Jackson et al. 8
Data on risk factors were obtained by trained interviewers (nurses) at recruitment and included socio-demographic factors (age, education), smoking (non-smoker, former smoker, current smoker), physical activity (inactive/moderately inactive; moderately active/active), family history (yes, no) and medical history (diabetes mellitus: yes, no) and vitamin/mineral use (yes, no). Anthropometrics were measured (height and weight).
The study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Ethics Committee of The University of the West Indies Mona Campus (May 19, 2014/ECP 155, 13/14). Written informed consent was obtained from individual participants enrolled in the study.
Laboratory Assays
All biological samples were obtained at initial PROSCARE enrolment. Whole blood (5 mL) were taken from non-fasting participants, stored in ice-packed coolers and transported to the Clinical Laboratory, Chemical Pathology Sub-Department, The University of the West Indies where they were processed and analyzed.
Serum 25(OH)D
To determine serum 25(OH)D concentrations, ultra-performance liquid chromatography/tandem mass spectrometry was used as described by Schleicher. 30 Acceptable results were obtained with a coefficient of variation of11.0%, the limit of detection (.5 ng/mL) and limit of quantification (2.2 ng/mL).
Serum Total Calcium
Serum total calcium concentration was measured on the Abbott C8000 Clinical Chemistry Architect using the Aeroset Calcium assay. 31 The intra-assay coefficient of variation for the serum total calcium was 7.9%.
Statistical Analyses
Duration of follow-up was determined from the date of PCa diagnosis to death or our censor date (31/12/2017). Demographic, clinical and anthropometric characteristics of PCa men with available 25(OH)D were compared according to survivorship, and all-cause/cause-specific mortality.
Continuous variables that were normally distributed were presented as mean and standard deviation and compared with the Student t-test and ANOVA. For ANOVA, significantly different groups were examined using post hoc tests. Other continuous variables were expressed as median and interquartile range (IQR) and analyzed by the Mann-Whitney U test. Categorical variables were recorded as percentage and group differences examined using Fisher’s exact and Chi-square tests.
Serum 25(OH)D (ng/mL) levels were classified into three categories using cut-points suggested by Holick et al. 32 : “deficient” if < 20 ng/mL, “insufficient” 20 to 29 ng/mL and “sufficient” if ≥30 ng/mL.
In the analysis of all-cause and PCa-specific mortality, age-adjusted and multi-variable-adjusted hazard ratios (HRs) in addition to 95% confidence intervals (95% CI) were determined to measure the independent effects of 25(OH)D from Cox regression models. Kaplan-Meier cumulative survival curves were constructed and compared by the log-rank test. Multivariable adjusted restricted cubic splines analyses were used to explore the association between 25(OH)D and mortality. Five knots were placed at equal intervals across the distribution of 25(OH)D with a reference value of 40 ng/mL 25(OH)D. Models were adjusted for age and BMI (continuous), smoking (non-smoker, former smoker, current smoker) and Gleason score (2-6, 7, 8-10) as well as serum total calcium. Years since PCa diagnosis were used as the primary timescale. Serum calcium was investigated as a potential effect modifier of the relationship between 25(OH)D and PCa by adjusting for calcium and creating a cross-product term between serum calcium and 25(OH)D. Likelihood ratio tests were used to assess any indication of effect modification. To reduce potential reverse causality, we carried out secondary analyses after excluding men who died within two years of enrolment.
All statistical tests were two-sided and statistical significance was defined as P values <.05. Data were analyzed using Stata/MP (version 16.1, College Station, TX) and SPSS Version 20.
Results
Unavailable 25(OH)D
Cases with unavailable 25(OH) D (n = 90) were not significantly different in age (not available, 67.6 ± 8.2 vs available, 69.91 ± 7.5 years; P = .755), BMI (24.8 ± 3.8 kg/m2 vs 25.3 ± 4.7 kg/m2; P = .433), Gleason score ≥7 (19.3% vs 21.8%; P = .645), moderately active/active physical activity (64.0% vs 56.7%; P = .261) and current smoking (13.3% vs 15.6%; P = .848).
Characteristics of Prostate Cancer Cases
The study population comprised 152 men with histologically confirmed PCa who were followed up a mean of 10.9 ± 3.3 years (median, 11.3 years). Over the review period, 139 deaths were identified, yielding an all-cause mortality rate of 6.98 deaths per 100 person years. Most men with PCa (61.2%) had sufficient concentrations of 25(OH) D, however 9.9% of cases (n = 15) were vitamin D deficient (<20 ng/mL) at enrolment (data not shown).
Characteristics of the cohort of PCa cases at enrolment, deaths and PCa survivors.

BMI - Body mass index; sd represents the standard deviation. †Significant difference between PCa-specific deaths and those due to other causes, P < .05. ǂSignificant difference between PCa-specific deaths and PCa survivors, P < .05. ǂǂ Significant difference between deaths and PCa survivors, P < .05.
aUnknown causes of death (excluded): n = 14.
bDate of censor, 30/12/2017.
Missing cases.
Table 1 characterizes the PCa cohort by their vital status at follow up. Men who died from prostate cancer recorded lower survival time (median, 4.5 years) than those who died from other causes (median, 7.2 years). The observed 5-and 10-year PCa-specific mortality was 53.5% and 32.6% respectively. Survivors were similar in age to their counterparts who had died and were less likely to have high-grade (Gleason score 8 – 10). Fewer PCa survivors self-reported diabetes mellitus as a comorbid condition when compared with men who died from PCa or other causes. Serum 25(OH)D levels at baseline did not differ significantly among PCa-specific deaths (33.3 ± 14.9 ng/mL)], those who died from other conditions (32.3 ± 11.6 (ng/mL) and PCa survivors (35.4 ± 11.6 ng/mL). There were no statistical differences in the serum total calcium levels among the groups (Table 1).
Baseline Characteristics of PCa cases by Reference Categories of 25(OH)D a .

aCut-points based on Holick et al. (Ref. 24).
bDate of censor, 30/12/2017.
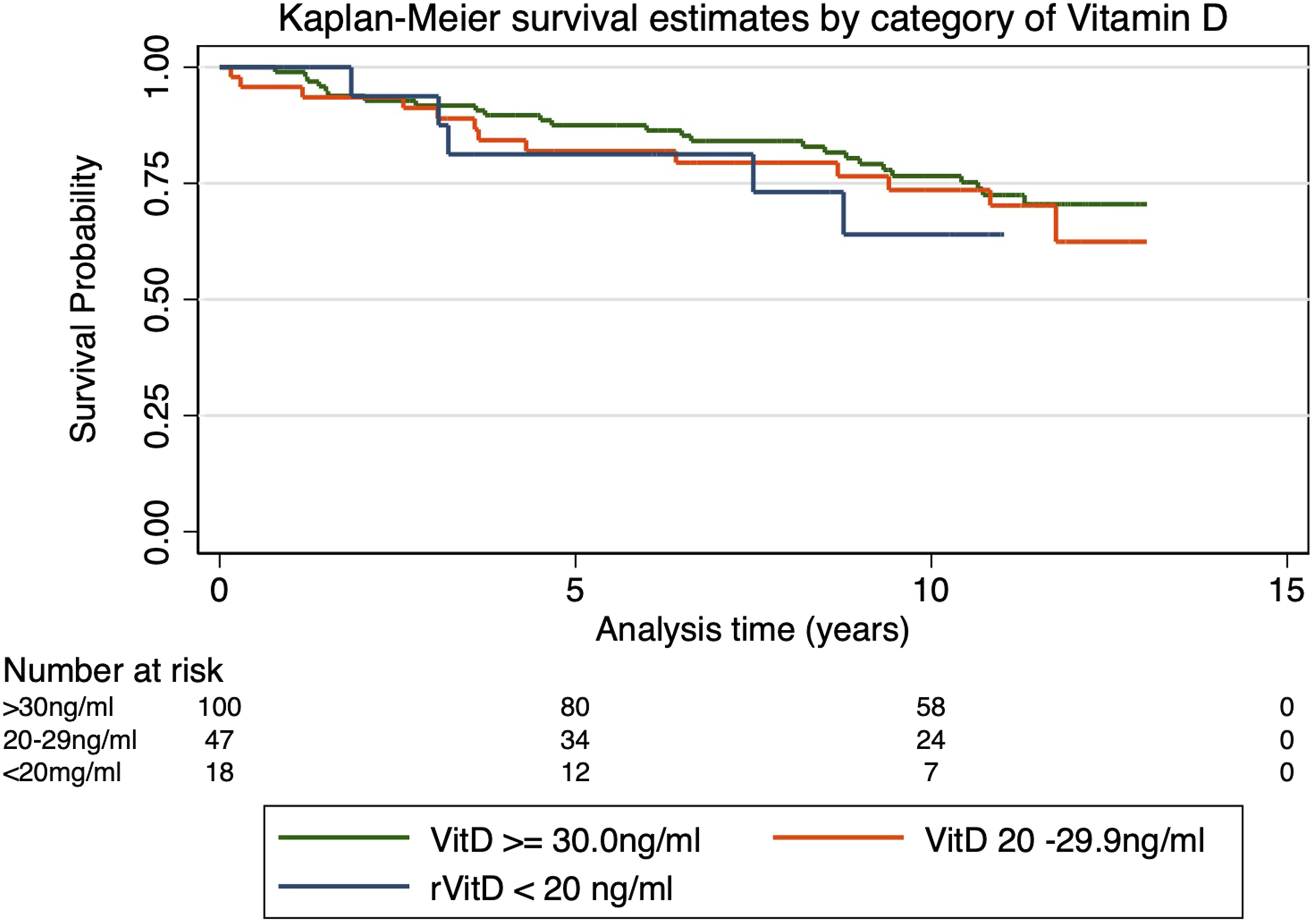
Kaplan–Meier curves for all-cause and PCa-specific- mortality according to 25(OH)D category at enrolment are presented in Figures 1 and 2, respectively. Significant differences were observed in all-cause but not PCa-specific mortality for the reference categories using the log-rank test in unadjusted models. Men with deficient 25(OH)D concentrations were associated with significantly higher risk of all-cause mortality than men with higher concentrations. Restricted cubic spline analysis suggested that at the lowest levels (<20 ng/mL) of vitamin D the adjusted effect on all-cause mortality is for an increased risk of death (greater than 200%) compared to normal concentrations (at 40 ng/mL) (Figure 3) but not PCa-specific deaths (Figure 4). Log-rank for Kaplan-Meier Curves for All-cause Mortality. Log-rank for Kaplan-Meier Curves for PCa-specific Mortality. Riskof all-cause mortality with increasing 2S(OH)D concentration usingrestricted cubic spline analysis with five knots. Risk of Pea-specific mortality with increasing 2S(OH)D concentration using restricted cubic spline analysis with five knots.


Serum 25(OH)D and Risk of All-Cause and Prostate Cancer-specific Death
HRs and 95% CIs for All-cause and PCa-specific Mortality by Reference Categories of 25(OH)D levelsa at Enrolment.

Abbreviations: HR – hazard ratio; CI – confidence interval.
aCut-points based on based on Holick et al. (Ref. 24).
bHR and 95% CI from Cox proportional hazards model adjusted for age (continuous) at diagnosis.
cModel adjusted for age and BMI (continuous), smoking (non-smoker, former smoker, current smoker), Gleason score (2-6, 7, 8-10).
dModel excluding deaths within the first two years of enrollment.
Multivariable Analysis of Serum 25(OH)D, Calcium and Mortality
Multivariable HRs and 95% CIs for All-cause and PCa-specific Mortality of Baseline 25(OH)D levels 2 with Additional Control for Calcium.

aCut-points based on based on Holick et al. (Ref. 24).
1Model adjusted for age and BMI (continuous), smoking (non-smoker, former smoker, current smoker), Gleason score (2-6, 7, 8-10).
2Model 1 excluding deaths within the first two years of enrollment.
Discussion
In this investigation of long-term PCa survival of predominantly African ancestry men, those with serum vitamin D deficiency at enrolment had higher mortality rates compared to men with sufficient vitamin D concentrations. Using clinical/reference values, men with 25(OH)D levels <20 ng/mL had significantly higher risk of PCa-specific mortality and all-cause mortality after adjustments were made for potential confounders, including serum total calcium, and excluding deaths within two years of enrolment.
PCa-specific Deaths
Our analysis indicated an increased risk of PCa-specific mortality for 25(OH)D concentration <20 ng/mL compared to those with sufficient concentrations (≥30 ng/mL). Although other studies have shown that low concentrations of 25(OH)D increase the risk of mortality 33 direct comparisons are limited due to differences in clinical cut-points used in the categorization of 25(OH)D. In a recent study of Danish men serum vitamin D deficiency (<25 nmol/L; 10 ng/mL) compared to vitamin D sufficiency (50 – 75 nmol/L; 20 - 30 ng/mL) were associated with higher risk of PCa-specific mortality (HR: 2.37, 95%CI: 1.45-3.89, P < .001). 34 A study of 5-year survival of US veterans reported that 25(OH)D <20 ng/mL was strongly related to death from PCa. 35 Other long-term epidemiological studies of pre-diagnosis circulating 25(OH)D levels on mortality observed higher 25(OH)D concentrations predicted lower PCa-specific mortality.36-38 The cohort assembled by Mondul et al. during 23 years of enrolment observed that higher pre-diagnostic serum 25(OH)D improved PCa survival. 37 Song et al.’s dose-response meta-analysis of cohort studies (n = 7,808) highlighted that higher pre-diagnosis of 25(OH)D concentration among men with more than ten years follow-up, at increments of 20 nmol/L of the metabolite, reduced PCa-specific mortality (HR: .92; 95% CI .89-.96). 39
All-Cause Mortality
Vitamin D deficiency was also associated poorer survival of men who died from all-cause mortality compared to men with sufficient 25(OH)D levels are consistent with other studies.34,35,40 Studies of US veterans with PCa (1999-2012), showed that vitamin D deficiency reduced the likelihood of survival. 35 Similar findings were documented among a Norwegian cohort where a 25(OH)D of <50 nmol/L increased the risk of all-cause mortality. 40 Meta-analyses of randomized trials of vitamin D revealed a twelve percentage decrease in total cancer deaths, in men who took vitamin D supplement. 41 In the meta-analysis by Song et al. 39 increments of 20 nmol/L levels of vitamin D were inversely related to all-cause mortality among men with PCa. 39
Serum 25(OH)D demonstrated no direct association with total calcium (r, .13; P = .133) nor was there any indication of any relations between vitamin D levels and serum total calcium, as well as PCa risk. The relationship between 25(OH)D levels and death may be a result of the metabolite, however 25(OH)D could be a marker of lower socioeconomic status or diet quality which may decrease survival. While we adjusted for socioeconomic status (SES) in our analysis the indicators used to measure SES may not be precise enough to account for this effect.
Interestingly in this study, men with deficient 25(OH)D concentration were associated with a significantly higher risk of all-cause and PCa-specific deaths compared to men with sufficient vitamin D levels. Similar findings were observed by Stroomberg et al.’s investigation of Danish men. 34 Other studies highlighted that the similarity in findings appear to be related to a higher a priori risk of dying caused by other characteristics of deficient vitamin D men such as increased comorbidities, Black ethnicity or the relationship of vitamin D with immune system functions.42,43 It is suggested that after PCa diagnosis, serum vitamin D is unlikely to become a reference biomarker for mortality. 34
In our analysis controlling for confounding factors which influence PCa prognosis, 25(OH)D deficiency predicted PCa-specific mortality and all-cause mortality. In contrast to the inverse relationship between sufficient concentrations of 25(OH)D and mortality39,44 suboptimal levels have been associated with increased risk of death due in part to declines in anti-inflammatory and immune modulating effects of vitamin D.45,46 Low 25(OH)D which has the potential to lower 1,25(OH)2D, may increase PCa progression. Normal prostate cells possess 1-α-hydroxylase activity, which has the capacity to convert 25(OH)D to 1,25(OH)2D. PCa cells show markedly reduced 1-α-hydroxylase activity resulting in declines in 1,25(OH)2D, a metabolite with anti-proliferative, pro-differentiating, and anti-metastatic effects on prostatic epithelial cells. Notably, due to loss of 1-α-hydroxylase activity, PCa cells may become insensitive to circulating 25(OH)D.47,48 Although the underlying molecular mechanisms has not been clearly elucidated, it is suggested that genetic variants in the vitamin D pathway on PCa are associated with risk of PCa mortality. 49 Shui et al. demonstrated that men with an allele associated with higher circulating 25(OH)D, but had low circulating 25(OH)D concentrations, were at higher risk for fatal PCa. They further reported that other SNPs were mainly located in CYP24A1, an enzyme critical for the catabolism of vitamin D, and VDR, the key nuclear receptor that mediates the genomic effects of vitamin D. 50 Other studies show that overexpression of CYP24A1 are associated with worse outcomes in solid tumors such as the prostate. 51 The increased risk of all-cause and PCa specific mortality observed in this study may also be related to other a priori risk of dying among men with low circulating 25(OH)D. Factors such as increased comorbidities, or deficiencies in immune system functions 52 may partly explain mortality findings.
Strengths and Limitations
This study of the effect of baseline serum 25(OH)D and calcium on mortality approximately 11 years after PCa diagnosis has strengths and limitations. While this is a relatively small sample, we are not aware of other data examining this issue in Black men residing in a middle-income country.
Serum 25(OH)D levels were measured at diagnosis in men with histologically confirmed incident PCa and is in contrast to cancer mortality studies that collected serum 25(OH)D after the cancer diagnosis, which increases the risk of reverse causality. 53 Follow-up of participants 11 years after diagnosis reflect long-term outcomes. We conducted sensitivity analyses by excluding men who died within the first two years of enrolment to reduce the risk of reverse causality as a probable bias and found the results were similar. Adjustments were made for BMI which has been shown to be associated with risk of PCa-specific mortality. 39 Seasonal variability is unlikely to influence the availability of 25(OH)D as on average, the country records approximately 8.2 hours of sunlight daily. While we considered other significant confounders, residual confounding cannot be excluded.
Study limitations include the absence of data on cancer recurrence, molecular profile of tumor and other predictive factors subsequent to PCa diagnosis. The presence of other chronic illnesses was based on participant self-report. Our study population includes men of primarily African ancestry, and may limit generalizability to other ethnic or racial groups with lower rates of survival from PCa. 1 A single blood specimen was used to determine 25(OH)D levels, which may not provide a true indication of the metabolite’s concentration during PCa development or progression. However, other studies report that one measurable quantity of plasma 25(OH)D is characteristic of the metabolite over extended periods.54-56 In addition, we did not measure the active hormone 1,25(OH)2D which mediates the effects of vitamin D. While the International Society of Uro-pathologists grade groups which more accurately assign grade and is more strongly related to prognosis, only Gleason scores were available for this cohort. 57
Conclusion
In this long-term follow-up of men diagnosed with PCa, circulating 25(OH)D levels <20 ng/mL concentrations identified in few patients, predicted increased risk of PCa-specific mortality and all-cause mortality in a predominantly African descent population. Serum calcium did not appear to modify the effect of 25(OH)D on survival.
Footnotes
Author Contributions
Conceptualization: Donovan McGROWDER, Marshall TULLOCH-REID and Maria JACKSON Methodology: Marshall TULLOCH-REID and Maria JACKSON Formal analysis: Maria JACKSON and Marshall TULLOCH-REID Acquisition of data: Maria JACKSON, Marshall TULLOCH-REID, William AIKEN, Leroy HARRISON, Affette McCAW-BINNS, Trevor FERGUSON and Kathleen COARD Writing— original draft preparation: Donovan McGROWDER, Marshall TULLOCH-REID, Simon ANDERSON and Maria JACKSON Supervision, critical revision of the manuscript and important intellectual input: Donovan McGROWDER, Marshall TULLOCH-REID, Simon ANDERSON, Affette McCAW-BINNS, Trevor FERGUSON, Kathleen COARD, William AIKEN, Leroy HARRISON and Maria JACKSON.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Research Awards for Cancer in the Caribbean, a collaborative small grants program administered by Caribbean Public Health Agency (CARPHA) utilizing funds from the U.S. National Cancer Institute (NCI).
Ethical Approval
The study received approval from The University of the West Indies/University Hospital of the West Indies (UHWI/UWI) Faculty of Medical Sciences Ethics Committee.
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with The University of the West Indies/University Hospital of the West Indies (UHWI/UWI) Faculty of Medical Sciences Ethics Committee approved protocols.
