Abstract
Background
Our purpose is to develop a model combining radiomic features of radiotherapy localisation computed tomography and clinical characteristics that can be used to estimate overall survival in patients with nasopharyngeal carcinoma treated with intensity-modulated radiotherapy following induction chemotherapy.
Methods
We recruited 145 patients with pathologically confirmed nasopharyngeal carcinoma between February 2012 and April 2015. In total, 851 radiomic features were extracted from radiotherapy localisation computed tomography images for the gross tumour volume of the nasopharynx and the gross tumour volume of neck metastatic lymph nodes. The least absolute shrinkage and selection operator algorithm was applied to select radiomics features, build the model and calculate the Rad-score. The patients were divided into high- and low-risk groups based on their Rad-scores. A nomogram for estimating overall survival based on both radiomic and clinical features was generated using multivariate Cox regression hazard models. Prediction reliability was evaluated using Harrell’s concordance index.
Results
In total, seven radiomic features and one clinical characteristic were extracted for survival analysis, and the combination of radiomic and clinical features was a better predictor of overall survival (concordance index = .849 [confidence interval: .782-.916]) than radiomic features (concordance index = .793 [confidence interval: .697-.890]) or clinical characteristics (concordance index = .661 [confidence interval: .673-.849]) alone.
Conclusion
Our results show that a nomogram combining radiomic features of radiotherapy localisation computed tomography and clinical characteristics can predict overall survival in patients with nasopharyngeal carcinoma treated with intensity-modulated radiotherapy following induction chemotherapy more effectively than radiomic features or clinical characteristics alone.
Keywords
Introduction
Nasopharyngeal carcinoma (NPC) is a unique form of head and neck cancer characterised by its distinct geographical distribution, with an average incidence of approximately 1.1% per 100,000 people worldwide.1-3 NPC is also distinct from other head and neck cancers owing to its association with the Epstein–Barr virus, naturally aggressive behaviour and high risk of distant metastases.4-6 Although improvements in living conditions and quality of life have helped to decrease the incidence of NPC, it is relatively more prevalent in East and Southeast Asia than in other regions.
Treatment options for NPC include radiotherapy alone, concurrent chemoradiotherapy and induction chemotherapy plus concurrent chemoradiotherapy. 7 Radiotherapy is the primary treatment for patients with NPC. The current standard treatment for locoregionally advanced NPC is concurrent chemoradiotherapy with or without induction chemotherapy. Advancements in imaging technology, such as the development of intensity-modulated radiotherapy (IMRT), have substantially improved survival rates among patients with advanced NPC. 8 However, there is no effective method for predicting prognosis in patients with NPC.
Several recent studies have demonstrated that radiomic-based methods can be used to predict the prognosis of patients and guide clinical decision-making.9-11 Radiomic analysis, in which several high-throughput quantitative features are extracted from radiological images, provides valuable information associated with tumoural heterogeneity. 12 To date, radiomic analyses have been used to predict survival in patients with various types of tumours, such as hepatocellular carcinoma, gastric carcinoma and breast cancer.13-15 Some scholars have reported that a nomogram combining radiomic and clinical features can be used as a convenient and reliable tool for predicting prognosis in patients with NPC.16-18 Radiomic features are extracted from radiotherapy localisation computed tomography (CT) images obtained prior to radiotherapy, which differ from conventional CT images, in that they allow clinicians to directly sketch regions of interest (ROIs), saving time for both clinicians and patients. Although radiotherapy localisation CT is widely used in hospitals, radiomic analysis based on radiotherapy localisation CT is rare. Therefore, in this study, we aimed to develop and validate a novel radiotherapy localisation CT-based radiomic model for predicting survival among patients with NPC treated with IMRT following induction chemotherapy.
Material and Methods
Patients
Patients were considered eligible if they met the following inclusion criteria: age ≥18 years; pathologically confirmed NPC; Karnofsky functional status score of at least 70 points; at least one measurable lesion that has not received local treatment, such as radiotherapy, according to the Response Evaluation Criteria in Solid Tumours 1.1; and expected survival ≥3 months. Treatments and data analyses were conducted in accordance with the Declaration of Helsinki. All patients were treated at the Affiliated Hospital of Southwest Medical University in Luzhou City, Sichuan Province, China, from February 2012 to April 2015. Ethical approval was obtained from the Medicine Ethics Committee of the Affiliated Hospital of Southwest Medical University. Diagnoses and treatments for NPC were based on the guidelines of the Chinese Society of Clinical Oncology. All patients underwent magnetic resonance imaging (MRI) initially and were subsequently diagnosed with NPC based on the results of a pathological biopsy.
First, all patients received induction chemotherapy with either paclitaxel plus cisplatin or 5-fluorouracil plus cisplatin every 3 weeks for a total of two cycles. Concurrent chemoradiotherapy with cisplatin plus IMRT was performed at week 7 to 14 of the total treatment cycle
IMRT
In recent years, IMRT has been widely used in the treatment of various head and neck tumours, including NPC and other rare cancers.19,20 In addition, a clinical study reported that IMRT not only significantly improved treatment outcomes but also reduced the incidence of some side effects. 21 Intensity-modulated radiation allows the clinician to adjust the intensity of each radiation according to the actual situation, so each beam field can have one or more high-intensity radiation areas and any number of low-intensity areas within the same field, thus allowing greater control over the dose distribution with the target. 22 IMRT uses linear accelerators to safely deliver a precise dose of radiation to the tumour while minimising the dose delivered to the surrounding normal tissue.
Clinical Endpoints and Follow-Up
After the completion of treatment, all patients were monitored every 3 months during the first 2 years, every 6 months for the next 3 years and annually thereafter. The follow-up rate was 93.3%. All patients were followed-up for more than 2 years, and the median follow-up duration was 27 months. Overall survival (OS) was calculated from the start of IMRT to the date of death or the last follow-up visit.
ROI Segmentation
ROIs included the gross tumour volume of the nasopharynx (GTVnx) and gross tumour volume of neck metastatic lymph nodes (GTVnd). They were sketched by a specialist with more than 15 years of experience in head and neck cancer research using a Philips Pinnacle 9.10 planning system (Philips, Cleveland, Ohio, USA).
Radiomic Analysis
Image Acquisition
The images for radiomic analysis were obtained from radiotherapy localisation CT performed prior to IMRT. Radiotherapy localisation CT was performed using a LightSpeed RT 4 scanner (GE Healthcare, Chicago, IL, USA). The scanning parameters used in this study were as follows: tube voltage, 120 kVp; field-of-view, 250-400 mm; pixel size, 512 × 512; slice thickness, .25 cm; and average number of slices, 116. Before feature extraction, all images were resampled according to a voxel size of 1×1×1 mm3. Wavelet-based methods were used to process the CT images from which the radiomics features of the GTVnx and GTVnd were extracted.
Feature extraction
The radiomic features of GTVnx and GTVnd were extracted via the three-dimensional (3 D) slicer platform using the ‘pyradiomics’ package: (available at: http://PyRadiomics.readthedocs.io/en/latest/) (accessed on June 10, 2020). 23
Statistical Analyses
All statistical analyses were performed using R software (version 3.6.3; R Foundation for Statistical Computing, Vienna, Austria) and X-tile software (version 3.6.1; Yale University School of Medicine, New Haven, Conn). Data dimension reduction, feature selection and radiomics feature building were achieved through least absolute shrinkage and selection operator (LASSO) Cox regression modelling to select the most valuable predictive radiomic features from the gross tumour volume. Multivariate Cox regression hazard models were used to assess survival outcomes, radiomic features and clinical characteristics. Radiomic features and clinical characteristics were presented using a nomogram, which graphically displayed the prediction model using numerical scale lines based on traditional statistical methods. Radiomic features were selected via LASSO regression to fit the Cox proportional model using the ‘glmnet’ package in the R software. The ‘survival’ and ‘rms’ packages in R software were used for the multivariate Cox regression hazard models, nomogram and calibration curve, respectively. Harrell’s concordance index (C-index) was used to evaluate the nomogram model. The linear combination of selected radiomic features was used to calculate the radiomic scores (Rad-scores) for each patient, and the Rad-scores were weighted by their respective coefficients. X-tile software was used to calculate the cut-off value of the Rad-score, which was used to divide patients into high-risk and low-risk groups. Associations between radiomic features and clinical parameters were investigated using heat maps, and heatmap analysis was performed using the ‘pheatmap’ package in R (version 3.6.3).
Results
Patient Characteristics.

Abbreviations: KPS

LASSO regression reduced the high-throughput radiation signature. LASSO coefficient profiles of the 851 texture features (A). Tuning parameter (λ) selection in the LASSO model using 10-fold cross-validation with minimum criteria (B). LASSO: least absolute shrinkage and selection operator.
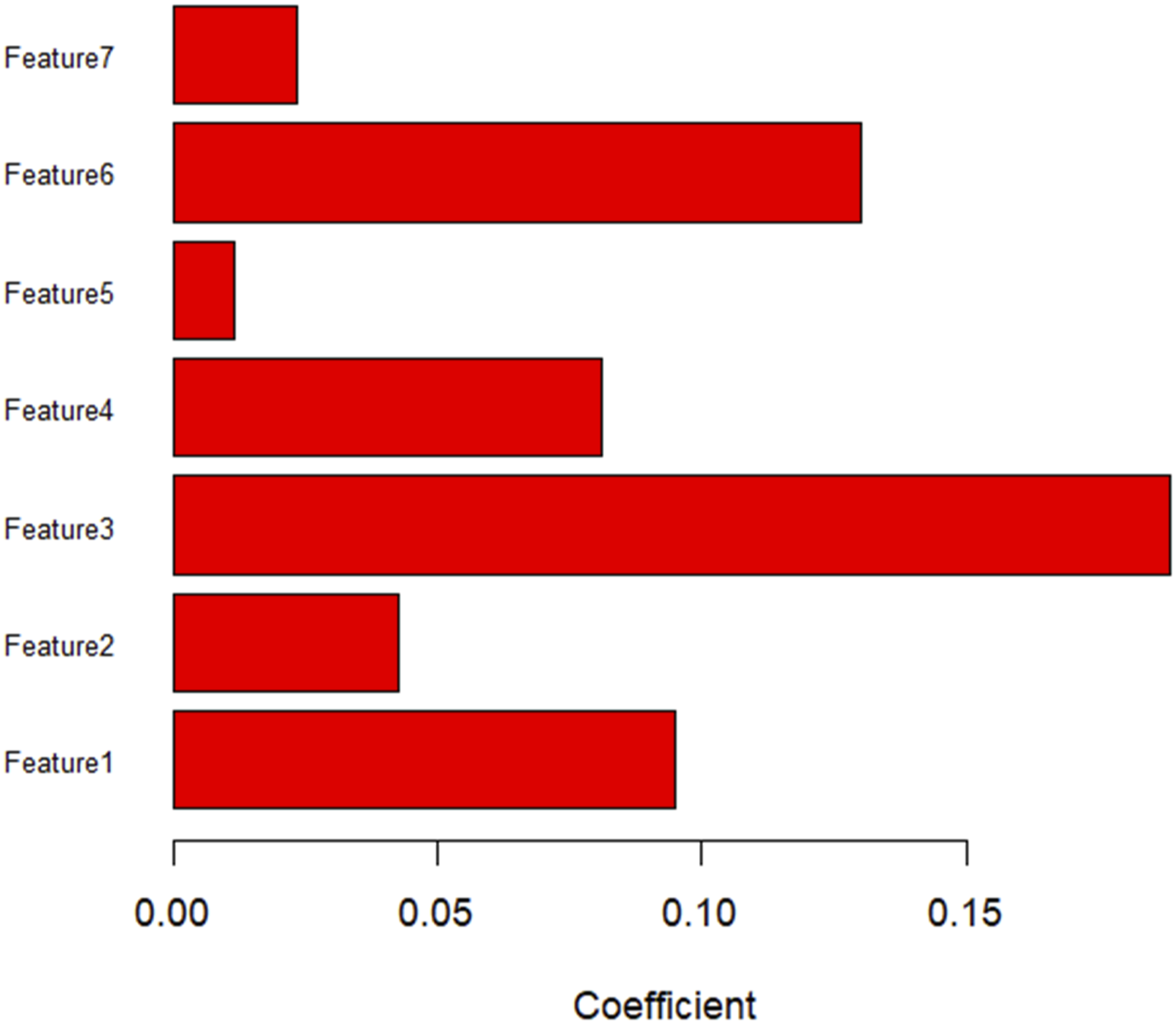
Histograms showing the roles of individual features contributing to the radiomic features. Contributing features are plotted on the y-axis, and their LASSO coefficients are plotted on the x-axis.
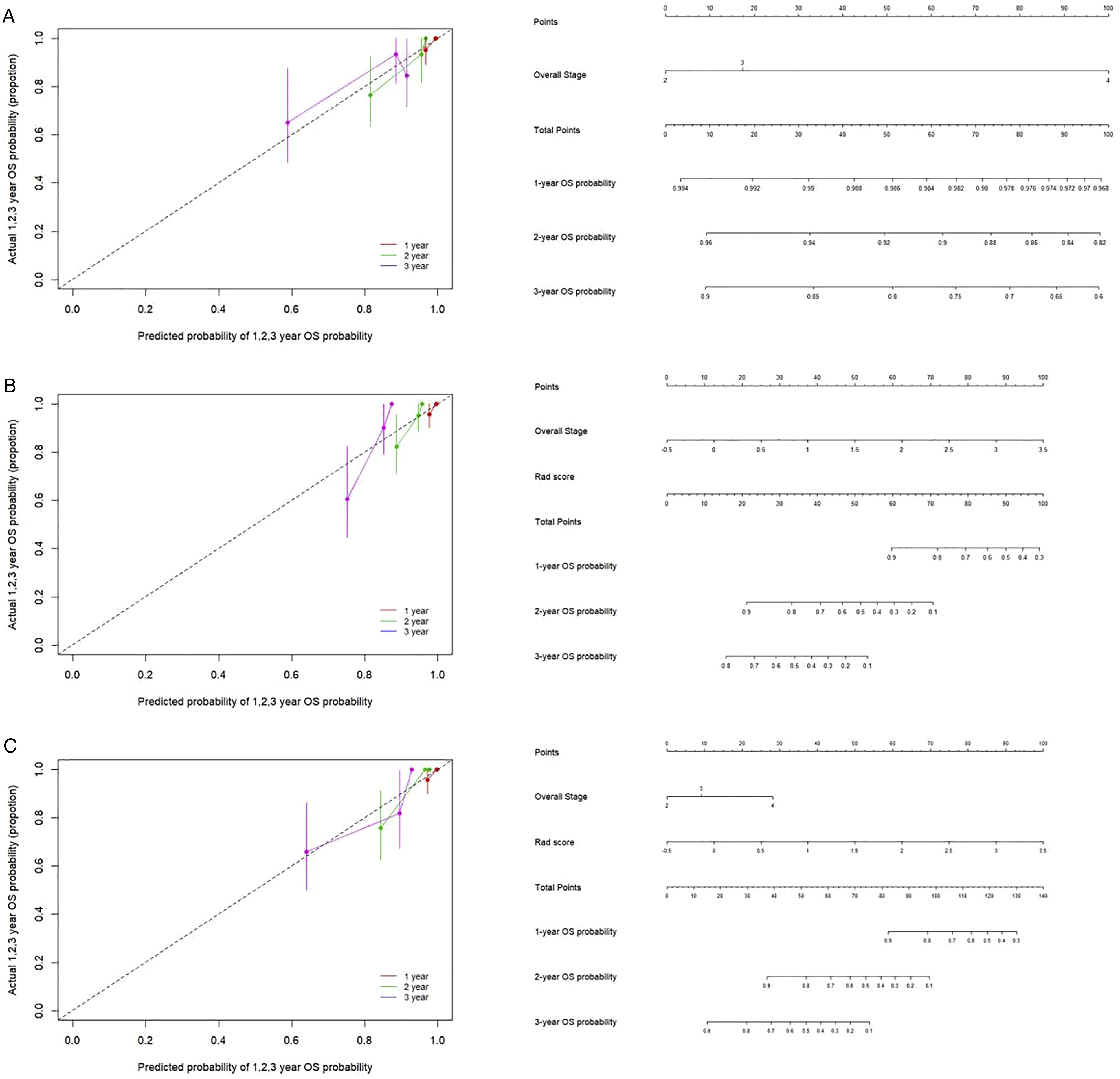
The cut-off Rad-score was −.06, which was used to classify patients into a high-risk group (Rad-score ≥ −.06) and a low-risk group (Rad-score < −.06). The survival curves for both groups are shown in Figure 3. The high-risk group exhibited shorter OS than the low-risk group. The nomograms for radiomic and clinical features, as well as the nomogram for the combination of the two, and the corresponding calibration curves are shown in Figure 4. Heat maps revealed associations between the radiomic signature selected by LASSO regression and clinical parameters (Figure 5). The results of the heat map analysis indicated that features 1-7 were correlated with T stage (all P <.05). Our results also indicated that the combination of radiomic and clinical features was a better predictor of OS (C-index = .849 [confidence interval {CI}: .782-.916]) than radiomic features (C-index = .793 [CI: .697-.890]) or clinical features (C-index = .661 [CI: .673-.849]) alone. Kaplan–Meier curves for overall survival in the low-risk and high-risk groups. The nomogram and the associated calibration curve. The nomogram (left) and the associated calibration curve (right) developed when clinical characteristics alone were used (A). The nomogram (left) and the associated calibration curve (right) when radiomic features alone were used (B). The nomogram (left) and the associated calibration curve (right) for the combination of radiomic features and clinical characteristics (C). Heat map for radiomic features and clinical characteristics.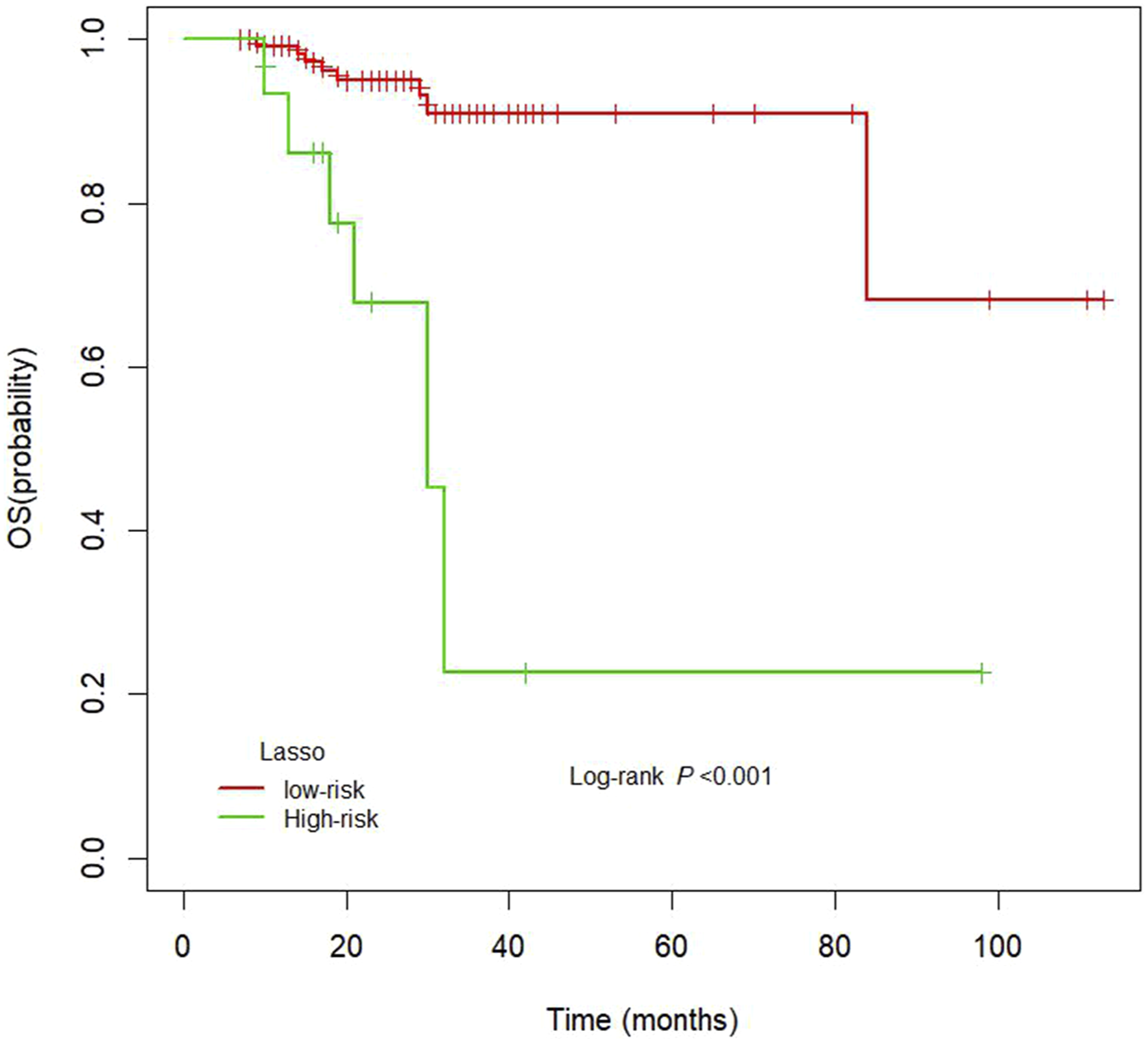

Discussion
In this study, we used radiotherapy localisation CT images to develop a novel radiomic model for predicting survival among patients with NPC treated with IMRT following induction chemotherapy. A total of 145 patients with NPC underwent radiotherapy localisation CT, and the images obtained were directly used by clinicians to outline the ROIs. Seven radiomic features and one clinical feature were selected for model construction. Our findings indicated that a nomogram combining these features can predict OS in patients with NPC treated with IMRT following induction chemotherapy more effectively than models based on radiomic features or clinical characteristics alone.
Radiomic analysis has been applied to many types of cancer, such as colorectal cancer, breast cancer, pancreatic ductal adenocarcinoma and hepatocellular carcinoma,24-27 and previous studies have suggested its advantages in predicting treatment efficacy and prognosis among patients. Some studies have demonstrated that LASSO regression can be used for the accurate analysis of radiological characteristics in relatively small sample sizes and can also prevent overfitting of the model.28,29 Radiomics combines radiomic features with clinical features to better predict OS, progression-free survival and disease-free survival, which effectively compensates for the lack of single clinical or radiomic predictions. In this study, we developed and validated a nomogram based on clinical and radiomic features obtained from radiotherapy localisation CT scans acquired before IMRT, which helped predict OS in patients with NPC. The method used in this study is simple and repeatable because these features are generated by a validated package, which is freely available from the 3 D slicer. 23 Furthermore, the information provided by this method may aid doctors in making clinical decisions for patients.
Several previous studies have reported that MRI-based radiomic models can be used to predict survival in patients with NPC.30-32 At the same time, some scholars have reported the value of positron emission tomography-CT-based radiomic analysis in predicting NPC prognosis. 33 However, unlike these studies, we used radiotherapy localisation CT-based radiomic models to predict patient survival. Therefore, the biggest innovation of this study is that we used radiotherapy localisation CT as the basis for model construction to predict the prognosis of patients with NPC. Prior to radiotherapy, each patient underwent radiotherapy location CT. The images obtained from radiotherapy localisation CT can be directly used by clinicians to outline ROIs, which saves clinicians more time and improves clinical work efficiency compared with the use of images obtained from ordinary CT and MRI. All images used for radiomics analysis were acquired via radiotherapy localisation CT before IMRT. Ultimately, our results indicated that the combination of radiomic and clinical features was a better predictor of OS than radiomic features or clinical features alone.
This study had some limitations. Owing to the small sample size, a validation cohort was lacking. To overcome these limitations, our research group intends to recruit more patient groups to conduct a multicentre study. In addition, previous research has shown that OS should be assessed by following patients up for more than 5 years 34 ; however, the median follow-up duration in our study was only 27 months.
In summary, our findings indicated that radiotherapy localisation CT-based radiomic models can effectively predict the survival of patients with NPC. Recent studies have reported that MRI-based radiomic models can be used to diagnose NPC stage and as adjunct tools to evaluate local recurrence, thus providing personalised treatment for different patients.31,35 Therefore, future studies should aim to develop and validate radiotherapy localisation CT-based radiomic models for staging and the prediction of local recurrence in patients with NPC. Furthermore, additional studies are required to determine whether radiotherapy localisation CT-based radiomic analyses can be applied to other cancers, such as lung cancer, liver cancer and other head and neck tumours. Such studies will not only provide diversified and more effective means for clinical diagnosis and treatment but will also provide patients with more personalised and effective treatments.
Conclusion
The combination of radiotherapy localisation CT-based radiomic features and clinical characteristics may serve as a reliable and powerful tool for predicting prognosis in patients with NPC treated with IMRT following induction chemotherapy.
Footnotes
Declarations of Conflicting Interest
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved by the Approved by Luzhou Medical College Affiliated Hospital drug clinical trial Committee.The approval number is K2012001.
