Abstract
Objective
The objective is to investigate the factors that can predict early treatment response in patients receiving concurrent chemoradiotherapy (CCRT) for cervical cancer.
Methods
We assessed clinical factors and treatment response in patients who underwent CCRT for cervical cancer at four time points: initial, 2.5 weeks, 6 weeks after starting CCRT, and 3 months after completing CCRT. The final treatment response was determined by positron emission tomography-computed tomography (PET-CT) 3 months after completion of CCRT. Patients were divided into two groups according to the final treatment response: complete response (CR) group or non-CR group. And the early CCRT response prediction model was developed using stepwise multivariate logistic regression analysis.
Results
Of the 62 patients who underwent CCRT for cervical cancer, 57 patients who completed all 4 time points examinations were included in the analyses and classified as CR (n = 32) and non-CR (n = 25) group. Tumor volume and serum squamous cell carcinoma antigen (SCC Ag) of the initial, 2.5 weeks, and 6 weeks after CCRT were significantly associated with the final treatment response. For the early treatment response prediction model, we selected patient age, tumor volume, and SCC Ag measured at initial and 2.5 weeks of CCRT as variables, and the equation of the final model was yielded. Using a cutoff of 0.433, this model had a sensitivity of 72.0%, a specificity of 84.4%, and a probability of 0.8225 (P < .0001).
Conclusion
Short-term (at 2.5 weeks after starting CCRT) measurements of tumor volume and serum SCC Ag were significant predictors of response to CCRT in patients with cervical cancer.
Introduction
Concurrent chemoradiotherapy (CCRT) is regarded as the definitive standard treatment for patients with locally advanced cervical cancer.1,2 Overall response rates to cisplatin-based CCRT are over 90%,3,4 and 3-year disease-free survival (DFS) rates have been reported to range from 65% to 75%.5,6 However, 30% to 50% of cervical cancer patients who receive CCRT experience treatment failure, 7 with locoregional recurrence being the main cause of failure.7,8 Large tumor size and lymph node involvement are factors contributing to recurrence. 7
Salvage treatment options are generally ineffective in patients who experience treatment failure after definitive CCRT.9,10 Therefore, early prediction of treatment failure can provide a window to escalate treatment, such as intensification of radiation dose, addition of other chemotherapeutic agents, or enrollment in clinical trials.9-11 Moreover, discontinuation of ineffective treatment can reduce associated toxicity and morbidity. 12
Magnetic resonance imaging (MRI) was found to be optimal in the radiologic assessment of cervical cancer, playing significant roles in initial staging, as well as guiding primary treatment and evaluating treatment response. 13 Moreover, the serum concentration of squamous cell carcinoma antigen (SCC Ag), a biomarker of cervical cancer, has been used to predict outcomes and responses to therapy, as well as to detect early recurrence during posttreatment surveillance.11,14
The present study was designed to identify potential markers predicting the failure of CCRT in patients with cervical cancer and to determine whether a combination of MRI and SCC Ag measurement could predict early treatment failure in cervical cancer patients treated with CCRT.
Materials and Methods
The study protocol was approved by the institutional review board of the Catholic University of Korea St. Vincent’s Hospital, which waived the requirement for informed consent due to the retrospective nature of the study (VC17RESI0154). The reporting of this study conforms to the reporting recommendations for tumor marker prognostic studies (REMARK) guidelines. 15
Patients and Tumor Characteristics
This retrospective study enrolled patients with histologically confirmed squamous cell carcinoma of the uterine cervix who were scheduled to receive definitive CCRT between July 2012 and December 2016 at our hospital. Clinical data were collected from patients’ electronic medical records, with all patient data anonymized to maintain patient confidentiality. Each patient was staged according to the criteria of the International Federation of Gynecology and Obstetrics (FIGO). 16 Patients were included if they were aged ≥21 years, had FIGO stage IB2 to IVA tumors, and had no history of prior chemotherapy or radiotherapy.
CCRT Treatment
All patients were treated with a combination of concurrent radiotherapy and chemotherapy. Radiotherapy consisted of computed tomography (CT)-guided external beam radiotherapy (EBRT) and intracavitary high-dose-rate brachytherapy (ICR). The total dose of EBRT delivered to the whole pelvis was 50 Gy, in doses of 2 Gy/day, five days per week for 5 weeks. Chemotherapy consisted of 50 mg/m2 cisplatin once weekly for four or five weeks. At the end of EBRT, ICR was initiated at a dose of 5 Gy delivered to point A, defined according to the guidelines of the American Brachytherapy Society, 17 twice weekly for 3 weeks, for a total dose of 30 Gy. Patients completed CCRT within 8 weeks.
Evaluation of Treatment Response
All patients underwent positron emission tomography-computed tomography (PET-CT) at two time points: before initiation of CCRT to evaluate nodal and/or extrapelvic tumors, and 3 months after the completion of CCRT to evaluate the final response to treatment. Patients underwent MRI, and the concentrations of tumor markers (SCC Ag and CEA) were measured, at four time points: before initiation of CCRT (baseline), 2.5 weeks and 6 weeks after starting treatment, and 3 months after completing CCRT. Response to CCRT was determined by assessing tumor volumes measured by MRI 3D volumetry at these time points. All MRIs were performed using a 3.0-T MRI scanner (Siemens Magnetom Verio, Achieva 3.0 T, Philips Healthcare, The Netherlands), with a 16-channel torso phased-array body coil. Two radiologists independently evaluated the longest tumor diameter based on T2-weighted images at maximal and, after comparing their results, reached consensus. Tumor volume was calculated by multiplying the longest horizontal, vertical, and height diameters, and π/6. 18 Serum SCC Ag was measured by immunoradiometric assay using the ARCHITECT SCC Reagent Kit (Abbott Diagnostics, Chicago, IL, USA). Clinical responses based on PET-CT scans performed 3 months after the end of CCRT were determined using the Response Evaluation Criteria in Solid Tumors (RECIST) 1.1 criteria. 19 Responses were classified as complete response (CR), partial response (PR), stable disease (SD) or progressive disease (PD). Patients were divided into two groups, those who achieved CR and those who did not, with the non-CR group including patients with PR, PD, and SD.
Statistical Analysis
Continuous variables were expressed as mean and standard deviation (SD) and compared using unpaired two-sample Student’s t-tests, whereas categorical variables were expressed as numbers and percentages and compared using chi-square tests. Variables associated with CR were evaluated by logistic regression analysis, with the resulting predictive model validated by receiver operating characteristics (ROC) curve analysis.
Interobserver and intra-observer agreements were assessed by calculating intra-class correlation coefficients (ICCs) with 95% confidence internals (CI). Statistical analyses were performed using SAS version 9.4 (SAS Institute Inc., Cary, NC, USA.). A two-sided P-value < .05 was defined as statistically significant.
Results
Patients and Tumor Characteristics
Demographic and Clinical Characteristics of Patients in the CR and Non-CR Groups.

Footnote: All values are expressed as the mean (SD) or number (%).
CR, complete response; BMI, body mass index; FIGO, International Federation of Gynecology and Obstetrics; SCC Ag, squamous cell carcinoma antigen; CEA, carcinoembryonic antigen; CCRT, concurrent chemoradiotherapy.
aPositive on radiologic workup at the time of initial diagnosis.
b2.5 wks and 6 wks indicate the time points from the initiation of CCRT.
Prior to treatment, the mean tumor volume was 32.8 cm3 in the CR group and 73.1 cm3 in the non-CR group (P = .035). The percentage of patients with more advanced FIGO stage was significantly higher in the non-CR group (P = .024). Histological grade (P > .999) and the incidence of lymphatic metastasis (P = .183) did not differ significantly in these two groups.
Early CCRT Response Prediction Model: The CCRT Complete Response Index
Univariate Analysis of the Clinical Variables.

Footnote: OR, odds ratio; SCC Ag, squamous cell carcinoma antigen; CCRT, concurrent chemoradiotherapy; FIGO, International Federation of Gynecology and Obstetrics.
a2.5 wks and 6 wks indicate the time points from the initiation of CCRT.
The AUC Values of ROC Curves for Prediction Models for Complete Response Determined 3 Months after Completing CCRT.

Footnote: CCRT, concurrent chemoradiotherapy; TV, tumor volume; SCC, squamous cell carcinoma antigen. The AUC values were presentd with the 95% CI.
a2.5 wks and 6 wks indicate the time points from the initiation of CCRT.
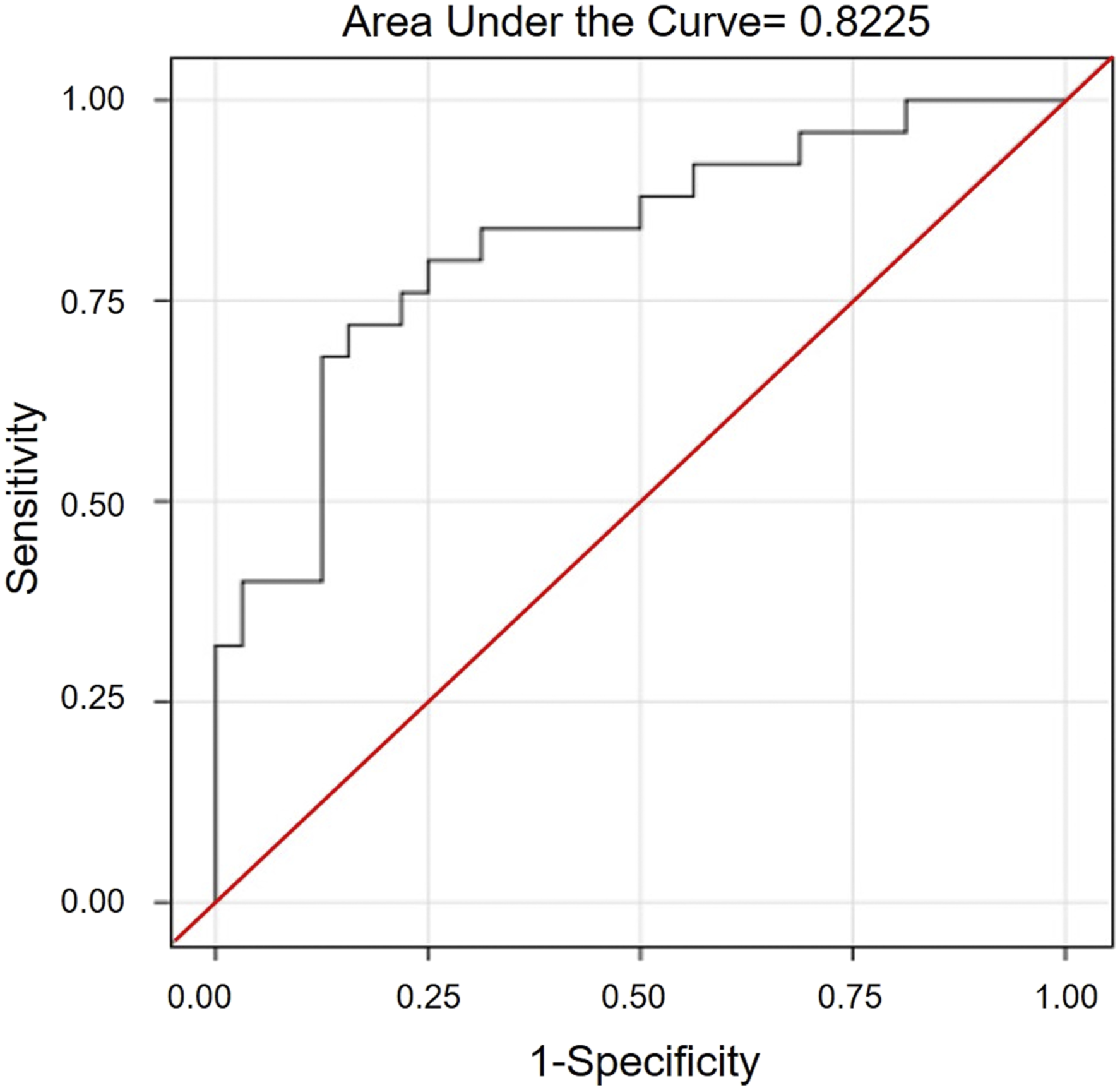
Receiver operating characteristics (ROC) curve analysis of CCRT complete response index (CCRT CRI).
Sensitivity, Specificity, Positive Predictive Value, Negative Predictive Value, and Accuracy at Various Cutoff Points for CCRT CRI.
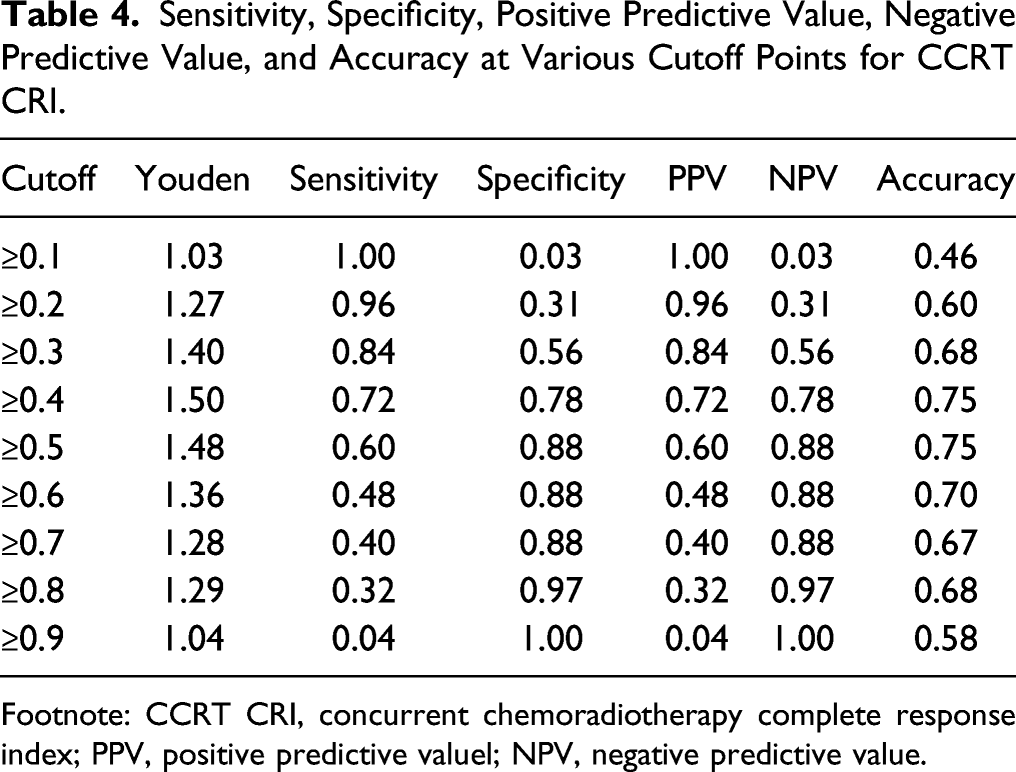
Footnote: CCRT CRI, concurrent chemoradiotherapy complete response index; PPV, positive predictive valuel; NPV, negative predictive value.
Discussion
The standard treatment for locally advanced cervical cancer is CCRT followed by brachytherapy. It has been recommended that the entire treatment course be completed within 8 weeks to avoid treatment failure.1,2 Response to treatment is evaluated 3 months after the end of CCRT 20 or 19–20 weeks after the start of treatment. 2 During this time, however, patients who do not respond well to CCRT may miss the opportunity to receive an effective alternative treatment and may be exposed to toxicity and morbidity from unnecessary treatment. Therefore, early prediction of response to CCRT is important for optimizing treatment of poor responders.9,10,21 Completion hysterectomy after chemoradiation, although not widely performed, may enhance survival in patients with residual disease after CCRT.22-24 The OUTBACK trial (NCT01414608), an ongoing international randomized phase III trial, is expected to determine the efficacy and toxicity of adjuvant chemotherapy following CCRT.
The present study evaluated the ability of a combination of MRI 3D volumetry and serum SCC Ag concentration to predict response to CCRT. Several studies have assessed the optimal timing of MRI to measure tumor volume or tumor regression rate during CCRT as a predictor of treatment outcome. For example, tumor regression rate, as determined by sequential MRI measurements of tumor volumetry 4–5 weeks after the start of radiation treatment (40–50 Gy), was shown to be a sensitive method of assessing response to radiation therapy.21,25-27 Although earlier prediction can improve the ability to choose an appropriate alternative treatment, measuring the tumor regression rate at an earlier time point, 2–2.5 weeks after the start of treatment (20–24 Gy), showed inconsistent results. Tumor regression rate measured at 2–2.5 weeks did not correlate with treatment response or local recurrence rate, perhaps because morphologic changes in tumors in response to CCRT occur later than changes at the cellular/molecular level.25,27 In contrast, another study found that both tumor volume and tumor regression rate after 2–2.5 weeks of radiotherapy could predict local control and disease specific survival rate, although results after 4–5 weeks of treatment were better predictors of these outcomes. 21
Inclusion of serum SCC Ag concentration in the algorithm improved the accuracy of predicting CR after 2–2.5 weeks of radiotherapy. SCC Ag is an important biomarker of cervical cancer, 28 with elevated serum SCC Ag level being associated with advanced tumor stage, larger tumor size, lymphovascular and regional nodal involvement, and deep stromal involvement. 29 Pre- and posttreatment SCC Ag concentrations, as well as its rate of reduction during CCRT, are useful predictors of treatment failure or survival outcomes in patients with cervical cancer.30-34 For example, normalization of serum SCC levels at the end of RT or a >70% reduction in concentration at 4 weeks correlated significantly with CR in patients with elevated pretreatment levels of serum SCC. 35 Moreover, failure of serum SCC Ag to normalize to < 2.2 ng/mL, after 4 weeks of CCRT was shown to be an early indicator of positive posttreatment FDG-PET. 11 However, the ability of SCC Ag after 2–2.5 weeks of CCRT to predict CR has not yet been determined. The present study found that serum SCC Ag concentrations measured before treatment and after 2.5 weeks of CCRT correlated with CR rate 3 months after the end of treatment. A review of 304 patients with cervical SCC treated with CCRT found that the recurrence-free survival rates in patients with pretreatment SCC Ag levels <4 ng/mL and ≥4 ng/mL were 80.2% and 56.6%, respectively (P < .001). 36 In addition, overall survival rates were lower and rates of local, regional, and distant metastases were higher in patients with elevated SCC Ag levels. 36 Higher pretreatment SCC Ag levels were also found to be associated with para-aortic lymph node relapse after CCRT. 37 Pretreatment SCC Ag levels have also been used to stratify risk in patients with cervical cancer.38,39 For example, an SCC Ag concentration > 2 ng/mL was found to be an independent risk factor for distant failure. Patients with SCC Ag concentration <2 ng/mL, stage IB-IIB disease, and negative lymph nodes were found to have a 5-year distant relapse-free survival rate of 83%, whereas those with SCC Ag > 2 ng/mL, stage III disease, and positive lymph nodes had a 5-year distant relapse-free survival rate of 43%. 39 Moreover, pretreatment SCC Ag level was shown to be independently prognostic of distant recurrence and was incorporated into a nomogram predicting the probability of distant recurrence within 5 years. 38
The present study found that tumor volume and tumor marker concentrations were factors associated with outcomes after initiation of CCRT. Treatment response 3 months after the end of CCRT correlated with tumor volume and serum SCC Ag at initial and 2.5 weeks of CCRT. According to our Logit model, a cutoff value of 0.4339 yielded a sensitivity of 72.0% and a specificity of 84.4%. ROC curve analysis confirmed an AUC of 0.8225. These results were very close to the predicted value at the post-CCRT time point.
This Logit model may predict early treatment failure in patients treated with CCRT for cervical cancer. The ability to predict treatment response before the end of treatment has several advantages, as well as suggesting another potential marker for prediction of CCRT failure in patients with locally advanced cervical cancer.
This study had several limitations. First, the number of included patients was relatively small, suggesting the need for additional studies and target groups to improve the ability of the model to predict CR. Second, this study included only patients with the squamous cell type of cervical cancer, suggesting the need for studies on patients with other types of cervical cancer. Third, this predictive model was unable to clearly delineate a treatment modality for patients resistant to radiation therapy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korea government (NRF 2017R1D1A1B3030021, NRF 2019R1A2C1086807).
Ethics Approval and Consent to Participate
All Subjects gave their informed consent for inclusion before they participated in the study. The study was conducted in accordance with Declaration of Helsinki, and the protocol was approved by the Ethics Committee of St Vincent’s Hospital (approval number: VC17RESI0154).
