Abstract
The association between inflammatory properties of diet and ovarian cancer risk has been investigated in some Western populations. However, little evidence is available from Asian women whose ovarian cancer incidence rates are low and dietary and lifestyle patterns are very different from their Western counterparts. We aimed to examine whether more pro-inflammatory diets, as indicated by higher dietary inflammatory index (DII®) scores, are associated with increased odds of epithelial ovarian cancer in southern China. A case-control study was conducted during 2006-2008 in Guangzhou, Guangdong Province. Energy-adjusted DII (E-DII) scores were calculated based on dietary intake assessed by a validated food frequency questionnaire administered to 500 incident epithelial ovarian cancer patients and 500 hospital-based controls. Logistic regression models were used to assess the relationship between E-DII scores and odds of ovarian cancer. Positive associations were observed between higher E-DII scores and ovarian cancer odds, using both continuous DII scores (odds ratio (OR) 1.87; 95% confidence interval (CI) 1.65, 2.13) and by DII tertiles (ORtertile3vs1 7.04, 95% CI: 4.70, 10.54, p for trend < 0.001). Likewise, a more pro-inflammatory diet was associated with a higher chance of serous and mucinous ovarian tumors. Our results suggest that a pro-inflammatory diet was associated with increased odds of developing epithelial ovarian cancer in southern Chinese women. The findings add to epidemiological evidence for the role of dietary inflammatory potential in ovarian cancer development.
Introduction
Ovarian cancer is the third most prevalent gynaecological malignancy (next to cervical and uterine cancer) and the eighth leading cause of cancer-related death in women worldwide. 1 According to global estimates nearly 300,000 new cases were diagnosed and more than 184,000 women died from the disease in 2018. 1 Ovarian cancer is usually detected at advanced clinical stages when the 5-year survival rate is only around 30%. 2 The incidence rate of ovarian cancer in China is relatively low, with an age-standardized rate of 5.32 per 100,000. 3 Despite relatively low incidence, due to its large population, China currently has more women diagnosed with ovarian cancer annually than any other country in the world. An estimated 52,100 new cases and 22,500 related deaths occurred in China in 2015. 4
Chronic inflammation has been hypothesized as a potential contributor to carcinogenesis and cancer progression, including ovarian tumors. 5,6 Diet, a modifiable lifestyle factor, has the ability to modulate inflammatory pathways and thus may be associated with the risk of ovarian cancer. 7 The intake of highly pro-inflammatory dietary components, such as simple carbohydrates and saturated fat, has been shown to be correlated with increased concentrations of inflammatory markers. 8,9 However, only a few studies in Western countries have evaluated the effects of an inflammatory diet on ovarian cancer risk, 10 -14 and more evidence, especially from low-incidence Asian populations whose dietary and lifestyle patterns are very different from Westerns populations, is needed before conclusions can be drawn.
Recently, a literature-derived, population-based dietary inflammatory index (DII®) was developed to evaluate the inflammatory potential of an individual’s diet. 8 A pro-inflammatory diet primarily consists of foods that are rich in saturated fat and refined carbohydrates and low in poly-unsaturated fatty acids, fiber, flavonoids and other antioxidant food components. A number of longitudinal and cross-sectional studies have validated the DII with various inflammatory markers, including C-reactive protein (CRP), 15 interleukin-6 (IL-6) 16 and tumor necrosis factor-α (TNF-α). 17 More pro-inflammatory diets, as defined by higher DII scores, have been linked to an increased risk of several cancers, including colorectal, 18,19 breast, 20,21 prostate, 22 endometrial, 23 pancreatic, 24 hepatocellular 25 and esophageal cancers. 26 Specifically related to ovarian cancer, the DII has been shown to be associated with ovarian cancer in the United States, 11 Italy 14 and Australia. 10
Although Chinese people have been shifting away from their traditional dietary pattern toward high-fat and low-fiber diets, considerable differences in nutrients intake between Chinese, Mediterranean (Italian) and Western diets remain. 27 The DII has been tested in the Chinese population in the past, to examine the association between inflammatory potential of diet and hip fracture, 28 breast, 29 hepatocellular 30 and esophageal cancers. 26 However, there have been no studies to our knowledge examining the relationship between DII scores and ovarian cancer in China. The present study aimed to investigate whether pro-inflammatory diets, as measured by the DII, are associated with increased odds of epithelial ovarian cancer in southern Chinese women.
Materials and Methods
Details of the study methods have been described previously, 31,32 and a summary is provided below.
Study Design and Participants
Between August 2006 and July 2008, a hospital-based case-control study of epithelial ovarian cancer was conducted at 4 public hospitals in Guangzhou, Guangdong Province of China. Guangzhou is an affluent and populous city in southern China with rapid economic growth. Eligibility criteria for participants were age ≤ 75 years and residence in metropolitan Guangzhou for at least the past 10 years. 31
Medical records and pathology reports were reviewed to identify patients with histopathologically confirmed and incident epithelial ovarian cancer (invasive and borderline malignancies) that had been diagnosed within the previous 12 months. Pathological diagnoses were based on the International Histological Classification of Ovarian Tumors. 33 Patients with self-reported memory problems affecting their recall of past events were excluded.
Malignancy-free, frequency age-matched (± 5 years) controls were recruited from inpatient departments of ophthalmology, orthopaedics, respiratory diseases, gastroenterology and physiotherapy at the same hospitals of cases. Exclusion criteria of controls were previous malignant disease, a history of bilateral oophorectomy, adherence to long-term medical diet, and self-reported memory problems. In both case and control groups, eligible participants were recruited and consented until the goal of 500 women were reached.
Based on previous research, the prevalence of consuming a pro-inflammatory diet was estimated at 33% in ovarian cancer patients and at 25% in control participants. 13 A sample size of 500 cases and 500 controls achieved a 79.7% power to detect a significant difference between these 2 groups at the significance level of 0.05.
The study was conducted in accordance with the Declaration of Helsinki, and the protocol was approved by the Human Research Ethics Committee of Curtin University (approval number HR 78/2006). Written informed consent was obtained from all participants before they were enrolled into the study. All participants were assured that the information provided would be kept confidential and they had the right to withdraw at any time.
Data Collection
All participants were interviewed face-to-face by trained interviewers in either Cantonese or Mandarin, usually in the presence of their next-of-kin to help the recall of dietary habits. 34 Information collected included data on demographic characteristics, anthropometric measures (current body mass index (BMI) and BMI 5 years before the interview), reproductive history, hormonal status, past and family medical histories, diet and lifestyle. 31 In addition, total physical activity energy expenditure was quantified in terms of metabolic equivalent tasks (MET)-hours per week. 32
A 125-item semi-quantitative food frequency questionnaire (FFQ), which was developed and had been validated for use among the southern Chinese population, 35,36 was administered to collect dietary information. Participants were asked to indicate their average frequency and amount of consumption for each item. The reference recall period for dietary and other lifestyle variables was 5 years prior to diagnosis for cases and 5 years prior to interview for controls. Each dietary item was transformed into daily intake, and nutrients and total energy (kcal) were estimated using the Chinese Food Composition Tables. 37
Calculation of DII Scores
The development 8 and validation 15 of the DII has been described elsewhere. A total of 1943 articles, published between 1950 and 2010, linking 45 food parameters (i.e., foods, nutrients, and other dietary components) with inflammatory biomarkers were first reviewed. Points were then assigned to each food parameter based on whether it increased (+1), decreased (−1) or had no (0) effect on 6 established inflammatory biomarkers (i.e. IL-1β, IL-6, TNF-α, CRP, IL-4, and IL-10). The score for each food parameter was weighted according to the study design. A food parameter-specific overall inflammatory effect score was next derived by deducting the anti-inflammatory fraction from the pro-inflammatory faction. This score was adjusted if the total weighted number of articles was less than the median number of 236 weighted articles. For the current study, data on 24 of the 45 food parameters could be derived from the FFQ and were used for calculating the DII scores. These include carbohydrate, protein, energy, cholesterol, fat, fiber, niacin, thiamin, riboflavin, vitamin A, vitamin C, vitamin E, vitamin B6, vitamin B12, β-carotene, isoflavones, selenium, zinc, iron, magnesium, tea, garlic, ginger, and onion. z-score and centered-percentiles for each of the 24 food parameters for each participant were calculated based on the average and standard deviation for each food parameter obtained from the global database which was created from the consumption of the original 45 food parameters. The centered percentiles for each food parameter was multiplied by the respective food parameter-specific overall inflammatory effect score to obtain the food parameter-specific DII score. All of these food parameter-specific DII scores were then summed to produce the overall DII score. To control for total energy intake, the DII was calculated per 1,000 calories of food consumed and used a calorie-adjusted standard global database.
Statistical Analysis
The energy-adjusted DII (E-DII) scores were analyzed both as a continuous variable and in tertiles (based on the distribution of controls). 38 Descriptive statistics were conducted to profile and compare the sample characteristics and E-DII scores between case and control groups. The χ2 test was used for categorical variables. Either the Student’s t test or the Mann-Whitney U test was used for continuous variables. The χ2 test and 1-way ANOVA were next performed to examine differences in participant characteristics across tertiles of E-DII among cases and controls.
Unconditional logistic regression analyses were then undertaken to estimate odds ratios (ORs) and 95% confidence intervals (95% CIs) for the association between E-DII score and epithelial ovarian cancer, using E-DII score as a continuous variable and with the DII expressed as tertiles (the lowest level being the reference category). Tests for linear trend were performed by assigning the median value of each tertile category of E-DII and modeling it as a continuous variable in the logistic regression analyses. Covariates included in the logistic regression models were age at interview (years), BMI (5 years ago, kg/m2), marital status (never married, married, widowed/divorced/separated), education level (none/primary, secondary, vocational/tertiary), parity, oral contraceptive use (never, ever), menopausal status (pre, post), tubal ligation (no, yes), physical activity (MET-hours/week), smoking status (never, ever), alcohol drinking (no, yes), and family history of ovarian or breast cancer (no, yes). These variables were either established or plausible risk factors from the literature. 39,40 Sensitivity of the analyses to histologic subtypes of epithelial ovarian tumors were also conducted. All statistical analyses were performed using the SPSS® package version 22 (IBM Corp.).
Results
Of the total 504 eligible patients and 512 eligible controls identified, 500 cases and 500 controls gave their consent to participate. No significant differences were found in educational level, marital status and age between participants and non-participants in both groups.
Half of the 500 epithelial ovarian cancer patients (n = 250) were histologically diagnosed as serous carcinoma, while 80 cases (16.0%) were mucinous tumors. Other histologic subtypes included borderline malignancy (n = 65, 13.0%), undifferentiated carcinoma (n = 59; 11.8%), endometrioid cystadenocarcinoma (n = 19; 3.8%), mixed epithelial cystadenocarcinoma (n = 13; 2.6%), clear cell carcinoma (n = 7; 1.4%), transitional cell carcinoma (n = 4; 0.8%) and malignant Brenner’s tumor (n = 3; 0.6%).
Participant characteristics and E-DII scores by case-control status are summarized in Table 1. The average ages of cases and controls were 59.1 (SD 5.7) and 59.7 (SD 6.5) years, respectively. The participants were predominantly postmenopausal (95.2%) and non-smokers (96.6%). More than half of the participants had attained secondary school education or above (59.9%), never had a tubal ligation (64.9%) and seldom drank alcoholic beverages on a regular basis (72.4%). Only a few women had a family history of breast or ovarian cancer (3.5%). However, women with ovarian cancer tended to have fewer pregnancies, less oral contraceptive use, and a higher mean BMI, and were less physically active than their control counterparts (p < 0.01). E-DII scores in this study ranged from −3.2 (most anti-inflammatory score) to 3.9 (most pro-inflammatory score). The median E-DII score among ovarian cancer patients (1.4, interquartile range (IQR) 1.2) was significantly higher than that of the controls (0.7, IQR 1.8). Characteristics of participants across tertiles of E-DII are provided for controls in Table 2. It shows that controls in the lowest tertile of E-DII were more likely to be nulliparous.
Characteristics of Ovarian Cancer Cases and Controls.

a Chi-square test, Student’s t test or Mann-Whitney U test for difference between cases and controls.
b Calculated using the density approach wherein nutrients are converted to per 1000 kcal.
Participant Characteristics Across Tertiles (T) of Energy Adjusted-Dietary Inflammatory Index (E-DII)a Among 500 Controls.
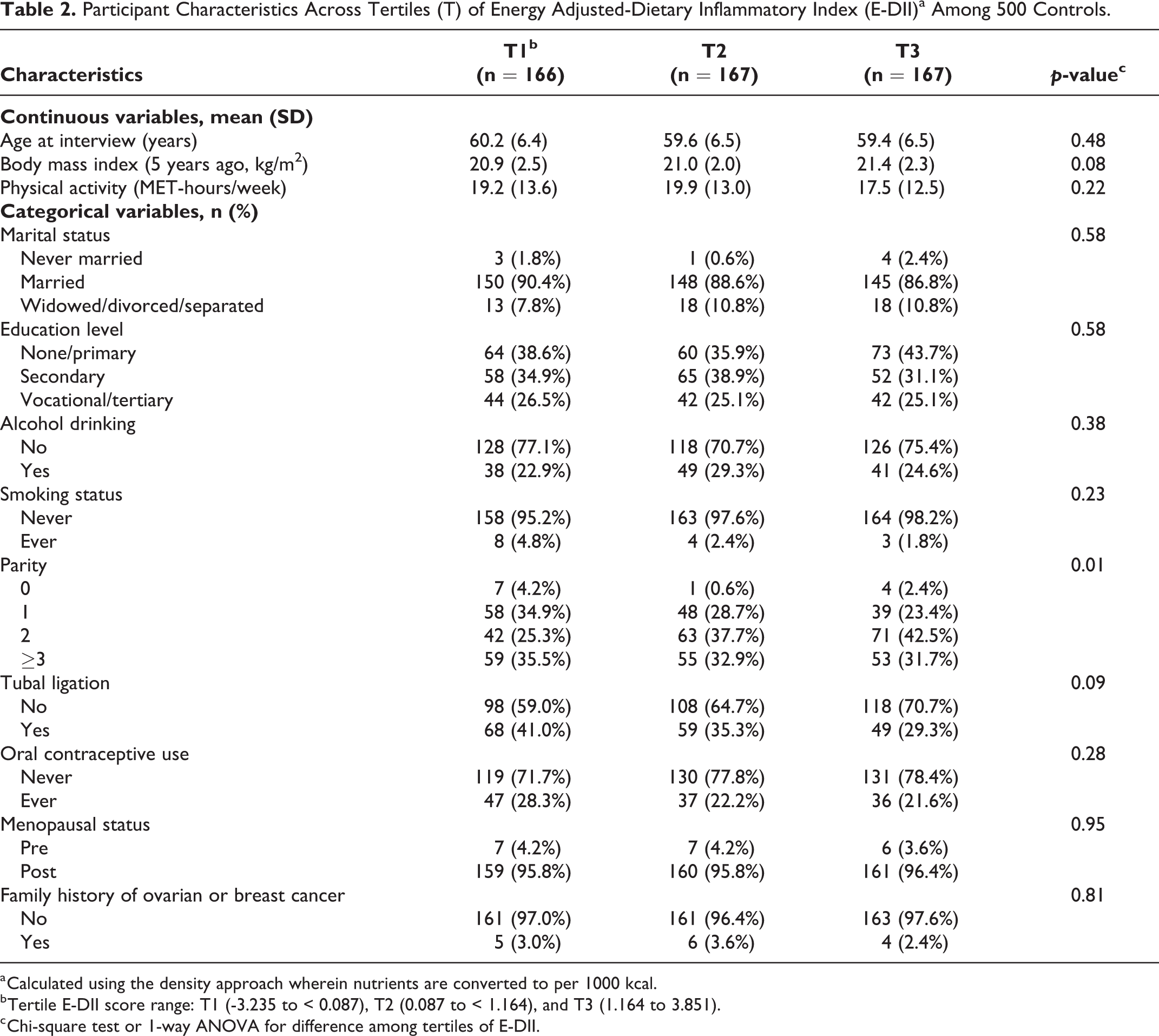
a Calculated using the density approach wherein nutrients are converted to per 1000 kcal.
b Tertile E-DII score range: T1 (-3.235 to < 0.087), T2 (0.087 to < 1.164), and T3 (1.164 to 3.851).
c Chi-square test or 1-way ANOVA for difference among tertiles of E-DII.
Table 3 presents the results of multivariable logistic regression analyses according to E-DII tertiles and continuous E-DII for all ovarian cancers and serous and mucinous ovarian tumors. Analyses were not conducted for other histologic subtypes due to the small number of cases available. The E-DII score (tertiles) was positively associated with the odds of ovarian cancer (adjusted ORtertile3vs1 = 7.04; 95% CI: 4.70, 10.54; p for trend < 0.001), and similarly for the continuous E-DII score (adjusted OR = 1.87; 95% CI: 1.65, 2.13). Also, a more pro-inflammatory diet, as indicated by a higher E-DII score, was associated with a higher chance of serous and mucinous ovarian tumors.
Adjusted Odds Ratios (95% Confidence Intervals) of Epithelial Ovarian Cancer for Energy Adjusted-Dietary Inflammatory Index (E-DII).

a Calculated using the density approach wherein nutrients are converted to per 1000 kcal.
b Based on logistic regression model adjusting for age at interview (years), body mass index (the previous 5 years, kg/m 2 ), marital status (never married, married, widowed/divorced/separated), education level (none/primary, secondary, vocational/tertiary), parity, oral contraceptive use (never, ever), menopausal status (pre, post), tubal ligation (no, yes), physical activity (MET-hours/week), smoking status (never, ever), alcohol drinking (no, yes), family history of ovarian or breast cancer (no, yes).
c Tertile E-DII score range: T1 (-3.235 to < 0.087), T2 (0.087 to < 1.164), and T3 (1.164 to 3.851).
d For 1 unit increment in E-DII score.
Discussion
This case-control study of southern Chinese women suggested a significant positive association between inflammatory potential of diet, as measured by increasing E-DII scores, and the odds of epithelial ovarian cancer. This is the first study in a low-incidence country, such as China, to examine the relationship between DII scores and ovarian cancer.
Our previous analyses in this population found a protective role of high intakes of fruit, vegetables, tea and isoflavones against epithelial ovarian cancer. 31,41,42 By contrast, an increased risk was observed with increased consumption preserved vegetables and meats. 43 Fresh fruit and vegetables are rich in anti-inflammatory components that are involved in DII calculation, such as fiber, beta carotene, isoflavones, niacin, folate and vitamins C and E. In contrast, preserved foods are usually high in saturated fat, which increases the pro-inflammatory capacity of the diet. 8 Our findings also are in line with the other limited studies available in China that explored dietary components contributing to the DII score and ovarian cancer. 44,45 For instance, in a hospital-based case-control study conducted in Hangzhou, eastern China, consumption of foods low in fat but high in fiber, carotene and vitamins C and E was inversely associated with ovarian cancer risk. 44
Results from the present study are generally consistent with the findings from a case-control study from New Jersey, in the United States, in which a positive association was observed between E-DII and ovarian cancer risk among postmenopausal women. 13 A population-based case-control study of African-American women also reported a statistically significant increased risk of epithelial ovarian cancer among those consuming the most pro-inflammatory diets (i.e., with the highest level of E-DII scores) in comparison to those consuming the most anti-inflammatory diet (i.e., with the lowest level of E-DII scores). 11 Similarly, higher DII scores have been linked with increased risk of ovarian cancer in Australia 10 and Italy. 14 Taken together, the results from our study support the previous findings showing that adhering to a pro-inflammatory diet may increase the odds of ovarian cancer.
One of the potential mechanisms behind the observed association between pro-inflammatory diet and increased ovarian cancer odds could be related to the tumor-promoting characteristics of chronic inflammation and its interaction with the bioactive components from diet. Although inflammation is commonly a healthy process needed to mount a competent immune response, chronic inflammation can be caused by the persistence of inflammatory triggers including lifestyle factors, such as poor dietary patterns, tobacco use, excessive alcohol drinking and physical inactivity. 46 Higher DII scores, which represent greater overall inflammatory properties of the dietary components, have been shown to be linked with increased levels of inflammatory markers such as CRP, IL-6 and TNF-α across different populations, 47 -49 including Chinese. 50 A chronic low-grade inflammatory status may lead to abnormal cellular proliferation, apoptosis inhibition and mutagenesis, which could contribute to tumorigenesis and may affect tumor progression. 51
Several strengths and limitations should be considered when interpreting the findings from this study. In order to avoid misclassification of the case-control status, we recruited only incident patients who had been histopathologically confirmed with epithelial ovarian cancer within the previous 12 months, and all controls were carefully screened. Hospital records were reviewed daily and a standardized identification procedure was implemented to ensure that ascertainment of patients was maximized and complete. Another strength was the high response rate (98%). Information on habitual food intake and dietary pattern was obtained using a validated and reliable questionnaire specifically developed for the southern Chinese population. Information bias and recall bias were unlikely given the lack of public awareness of any diet-related hypotheses on the pathogenesis of ovarian cancer in China at the time of interview. Nevertheless, it is possible that some ovarian cancer patients might have modified their dietary habits since the onset of the disease. To minimize reverse causation, the reference period for habitual food consumption was set at 5 years before diagnosis for cases and 5 years before interview for controls. No participant reported any changes in eating habits for medical reasons within the past 5 years.
A major limitation of this study was the retrospective nature of the case-control design. No cause-effect relationship between a pro-inflammatory diet and ovarian cancer could be established, even though participants were instructed to report on their dietary exposures in the past. Another possibility is that DII includes ovarian cancer-promoting dietary components exerted through other pathways such as oxidative stress, 52 adiposity, 53 and hormones. 54 Additionally, information on comorbid conditions of the participants, a potential confounder, was not collected and controlled for in the analyses. Only a small proportion of our patients had clear cell tumors. This histological subtype of epithelial ovarian cancer is known to be associated with excessive levels of endogenous estrogen. 55 Nulliparity and low parity have been reported to be more closely associated with clear cell tumors than other subtypes. 56 The high proportion of childbearing (98.6%) in our cases with 64% of them gave birth to at least 2 children might contribute to the small proportion of clear cell carcinoma in the present study. Moreover, as patients who died from ovarian cancer in the past 12 months were not included, selection bias cannot be eliminated. In addition, the use of hospital-based controls might also lead to selection bias if their characteristics are different from those of the general population; yet community-based controls are generally difficult to recruit in China due to the high rate of refusals. Nevertheless, the use of 4 hospitals that serve the entire catchment region reduced sampling bias, and our participants could still be considered as representative of the target population. In addition, the E-DII scores calculated from the 24 available food parameters in the study have not been validated with inflammatory markers. However, as noted there have been many construct validations conducted in other populations. 15 -17,57 Finally, the dietary habits were measured retrospectively based on self-report, and recall bias may have occurred. Face-to-face interviews instead of self-administered surveys were thus conducted to reduce recall bias. 58 Moreover, it has been suggested that dietary information collected from the spouses of elderly Chinese is quite reliable. 34 We therefore expected that the participants might provide more accurate dietary responses in the presence of their next-of-kin (mainly spouses).
Conclusions
Consumption of a pro-inflammatory diet, as identified by the E-DII, was associated with increased odds of epithelial ovarian cancer in southern Chinese women. Our findings add to epidemiological evidence on the association between dietary inflammatory potential and ovarian cancer.
Footnotes
Acknowledgments
The authors are indebted to the ovarian cancer patients and control participants who agreed to be interviewed. Thanks are also due to the medical and nursing staff of the participating hospitals for their assistance in patient recruitment.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Dr. James R. Hébert owns controlling interest in Connecting Health Innovations LLC (CHI), a company that has licensed the right to his invention of the dietary inflammatory index (DII®) from the University of South Carolina in order to develop computer and smart phone applications for patient counseling and dietary intervention in clinical settings. Dr. Nitin Shivappa is an employee of CHI.
Ethics Statement
The protocol of this study was approved by the Human Research Ethics Committee of Curtin University (approval number HR 78/2006). Written informed consent was obtained from all participants before they were enrolled into the study.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
