Abstract
Dendritic cells (DC) are professional antigen-presenting cells that activate T cells to kill cancer cells. The extracellular products of DCs have also been reported to perform the same function. In this study, we examined the in vitro differentiation of umbilical cord blood monocytes into DCs in the presence of GM-CSF, and interferon (IFN)-α. The resulting DC population (called IFN-DCs) were then matured in the presence of TNF-α, and pulsed with total protein extracted from A549 cancer cell line. The pulsed DCs and their conditioned medium were then used to stimulate allogeneic lymphocytes (alloLym). The proliferation and cytotoxicity of alloLym were then determined. The results showed that after 5 days of differentiation, the stimulated monocytes had the typical morphology and characteristic surface markers of DCs. Both unpulsed and pulsed IFN-DCs can induce the proliferation of alloLym, especially Vγ9γδ T cells. The conditioned medium from pulsed and unpulsed IFN-DCs culture also prompted the growth of Vγ9γδ T cells. Moreover, alloLym stimulated with pulsed DCs and their conditioned medium had a greater cytotoxic effect on A549 cells than the ones that were not stimulated. Our results indicated that IFN-DCs and their conditioned medium could induce the anti-tumor immunity in vitro, providing evidence for application of cord blood monocytes-derived, interferon-α- stimulated dendritic cells and their extracellular products in anti-cancer therapy.
Introduction
Dendritic cells (DCs) are professional antigen-presenting cells of the immune system. They are specialized in capturing, processing and presenting antigens on the cell surface along with co-stimulating molecules 1. Antigen-presenting DCs activate T cell mediated-immune response, and stimulate the proliferation and differentiation of naive T cells. In addition, mature DCs play an important role in maintaining the function of B lymphocytes and setting up immune memory. 1,2
DCs play an important role in activating host defense; thus it is promising candidate for vaccine therapy in treatment of infectious diseases and cancers. Autologous or allogeneic DCs have been used to treat some types of cancer such as melanoma, renal cell carcinoma, colorectal cancer, Non-Hodgkin lymphoma, multiple myeloma and prostate cancer in many clinical trials. 3 -9 Sipuleucel-T, which is a personalized immunotherapy developed by Dendreon Corporation and uses autologous DCs to treat prostate cancer, is the first therapeutic cancer vaccine approved by FDA in 2010. 10
DCs secrete extracellular products, including cytokines and exosomes, into its surroundings. These extracellular products of DCs have been reported to stimulate the immune system against infectious diseases and cancers. 2,11 The cytokines that are produced by DCs in response to inflammation can activate the adaptive immune response. 1,2,12 -14 For example, interleukin (IL)-12 and IL-18 are known as stimuli for the growth and activation of T cells. 1,2 IL-12 and IL-18 enhance the cytotoxicity of CD8+ T-cell in killing cancer cells and cells infected with bacteria and viruses. In addition, IL-12 inhibits angiogenesis (anti-angiogenic), preventing tumor formation. 1,2,11,14,15 DC-derived exosomes have the essential immune-stimulatory characteristics of DCs such as the ability to present antigens to T cells. 1,2,12,11,15,16 Products of lyophilized exosomes still fully retain their function after being kept at a temperature of 4 oC up to 36 months. 11,16
It has been shown that monocytes differentiate into DCs in vitro when incubated with stimulators such as granulocyte/macrophage colony stimulating factor (GM-CSF) and interleukin-4 (IL-4). 12 -14,17 -21 The maturation of the in vitro-generated DCs is achieved in the presence of tumor necrosis factor (TNF-α) and antigen stimulators. 17 -22 The other reported papers indicated that interferon (IFN)-α can induce CD14+ peripheral blood monocytes in vitro to differentiate into DCs 22,23 and these DCs can promote the proliferation of Vγ9γδ T cells. 22 Both in vitro 24 -26 and in vivo studies 27 -30 have shown that Vγ9γδ T cell sub-population express many antitumor characteristics, indicating that they could serve as anti-tumor factors. However, the Vγ9γδ T cell population only accounts for less than 5% of peripheral blood cells. 29,30 Thus, it is necessary to increase the number of Vγ9γδ T cells to develop them into an effective antitumor immunotherapy.
In this study, we determined the generation of DCs from human cord blood monocytes (CBMDCs) using IFN-α and GM-CSF stimuli. We also evaluated whether these CBMDCs and their extracellular products can be used to induce the proliferation of anti-tumor immunity of allogeneic peripheral blood lymphocytes. Particularly, we focused on the effects of CBMDCs induced by IFN-α on Vγ9γδ T cell proliferation. The results provided evidence for application of cord blood dendritic cells and their extracellular products in anti-cancer therapy.
Materials and Methods
Autologous Plasma Collection
Five cord blood units from 5 different donors were spun down at 4 oC for 10 minutes at 1710 × g .After centrifuge, autologous plasma was collected as much as possible without touching the red layer of blood cells. Autologous plasma was inactivated by heating at 58 oC for 60 minutes. The plasma was then centrifuged again at 1710 × g for 5 minutes at 4 oC. The supernatant was collected and stored at 4 oC. The red layer containing blood cells was reconstituted with phosphate buffer saline (PBS—Gibco, Grand Island, NY, USA) to the corresponded volume before collecting autologous plasma for mononuclear cell isolation.
Blood Mononuclear Cells Isolation
Purification of cord blood mononuclear cells (CBMCs) and PBMCs were performed using 1.077 ± 0.001 g/ml Ficoll-Paque (GE Healthcare, USA) and density-gradient centrifugation. In brief, the blood solution was centrifuged for 20 minutes at 840 × g with low acceleration and no brake at 4 °C. The buffy coat was washed twice with PBS containing 20% (v/v) fetal bovine serum (FBS—Gibco).
Cell Culture
Lung carcinoma cell line A549 was cultured in Dulbecco’s Modified Eagle Medium (DMEM—Gibco) supplemented with 10% (v/v) FBS. A549 cells provided tumor cell lysate and also served as target cells in cytotoxicity assay.
Preparation of tumor cell lysate
4 × 107 A549 cells were flash frozen in liquid nitrogen, followed by thawing in 37 ºC water bath. This process was repeated at least 10 times to obtain a crude lysate. A549 crude lysate was centrifuged at 2000 × g for 10 min at 4 ºC; and then filtered using 0.22 µm filter. The protein concentration was determined by the Bradford protein assay and the aliquots were stored at -80 °C until use.
Generation of IFN-DCs From Cord Blood Monocytes
CBMCs were seeded into T25 flask containing AIM-V medium (Gibco, Tokyo, Japan) supplemented with 10% autologous plasma for 2 hours at 37 ºC. After the removal of non-adherent cells, the adherent cells were cultured in AIM-V medium supplemented with 5% autologous plasma and DC cocktail I (Biotherapy Institute of Japan (BIJ), Tokyo, Japan) containing 1000 U/ml GM-CSF (Primmune Inc., Kobe, Japan) and 1000 U/ ml IFN-α (; INTRON, MSD K.K., Tokyo, Japan). At day 4 of culture, these cells were induced with DC cocktail II (BIJ, Tokyo, Japan) containing 10 ng/ml TNF-α (Peprotech Ltd., Hereford, UK), 1 µM Zoledronate (Novartis Pharmaceuticals, Switzerland), and 1000 U/ml IL-2 (Chiron Corp, Emeryville, CA, USA); and pulsed with the total cell lysate isolated from A549 lung cancer cells (100
Induction of Allogeneic Lymphocytes and Vγ9γδ T Cells by IFN-DCs and IFN-DC Conditioned Medium
IFN-DCs and their conditioned medium were analyzed for their ability to stimulate proliferation of allogeneic lymphocytes and Vγ9γδ T cells. AlloPBMCs were culture in AIM-V medium supplemented with 5% autologous plasma for 2 h, then non-adherent cells fraction was collected. Lymphocyte as non-adherent cell fraction (hereafter, alloLym) and DC/DC conditioned medium were mixed with ratio of 10:1, with the volume of DC conditioned medium equivalent to the number of DCs at this ratio. Cells were then cultured in AIM-V medium supplemented with 5% autologous plasma and IL-2 (100 IU/ml; Invitrogen, USA) for 7 days at 37 ºC and 5% CO2. After that, the non-adherent cell fraction was collected for determining the percentage of the induced Vγ9γδ T cells by flow cytometry, and the number of lymphocytes was counted using cell counting slide (Thermo Fisher, USA).
Collection of IFN-DCs and Their Conditioned Medium
On the day of harvest, the supernatant medium in each corresponding wells was gently collected by pipetting, and then centrifuged at 270 × g for 8 minutes. The supernatant was used as the corresponded conditioned medium. Since most of DCs could be detached by pipetting only, 5 mL of AIM-V medium was added onto the adherent cells in the flask and pipetting was carried out for dendritic cells collection.
Cytotoxic Assay
Effector cells (E) were prepared as followed: alloLyms were incubated with either IFN-DCs or IFN-DC conditioned medium at volume equivalent to 1 DCs: 4 alloLyms. After that, cells were cultured in AIM-V medium in the presence of 50 U/mL IL-2 for 7 days at 37 °C and 5% CO2. Target cells (T) A549 were labeled by Calcein-AM (Dojindo Molecular Technologies, Inc., Japan) for 30 min at 37 °C, followed by PBS wash. The labeled A549 cells were seeded at 3 × 103 cells/well to 96-well flat-bottomed plates. Effector cells were then incubated with labeled target cells at various E: T ratios. The plate was spun down at 50 × g for 2 min. Initial fluorescence intensity (FI) was measured by Terascan VPC2 counter (Minerva Tech, Japan). Cells were incubated for another 2 hours at 37 °C and 5% CO2. In maximum release control wells, cells were incubated with alkaline reagent DCN90 (Sigma, USA).
After incubation, the plate was centrifuged at 50 × g for 2 min. Then, 80 µl of the supernatant from each well was replaced by 80 µl fresh AIM-V. Centrifuge the plate again at 50 × g for 2 min and take the second FI measurement.
The cytotoxicity (%) was calculated as followed:
Experiment: FI value of E + T; Spontaneous: FI value of T + medium; Maximum: FI value of T + DCN90
Phenotypic Analysis
The cell phenotype was analyzed by flow cytometry using Navios Cytometer, Navios EX software, version 2.0 and Kaluza software (Beckman Coulter, USA). The following monoclonal antibodies (mAbs) and the corresponding isotypes were purchased from Beckman Coulter (USA) specific for: cluster of differentiation (CD)3, CD4, CD8, CD11c, CD14, CD40, CD80, CD86, CD123, Vγ9TCR, and major histocompatility complex molecule – HLA-DR (human leukocyte antigen D-related). The mAbs were conjugated with FITC (fluorescein isothiocyanate), Pacific Blue, Phycoerythrin Cyanin (PC)7, Allophycocyanin (APC) Vio, APC-Alexa Fluor 750.
Statistical Analysis
Statistical analyses were performed with Stata 12.0. p-Value less than 0.05 represented statistically significant difference.
Results
IFN-DCs Generation
At the first day of culture, monocytes were observed as relatively uniform size spheres with an average diameter of about 10 μm (Figure 1A). After stimulated with IFN-α for 4 days, the monocytes had characteristics of immature DCs, in which they were doubled in size with about 20 μm in diameter, and had a few short dendritic projections (data not shown). After incubating with DC cocktail II and pulsing with or without the total protein prepared from A549 lung cancer cells for 24 hours, no obvious change in the morphology of the unpulsed IFN-DCs (upIFN-DCs) was observed (Figure 1A). Meanwhile, the pulsed IFN-DCs (hereafter, pIFN-DCs) increased in cell size, number of dendritic projections and projection length. The average size of pIFN-DCs was bout 25-30 μm and the lengths of dendritic projections on the cell surface were between 5 and 15 μm (Figure 1A). These observations suggested that immature IFN-DCs were successfully maturated and pulsed with A549 cell lysate.

IFN-DCs characterization. (A) morphology of umbilical cord blood monocyte–derived IFN-DCs as representative from CB unit 1. After 4 days of culture, IFN-DCs were incubated with TNF-α and pulsed with (pIFN-DCs) or without (upIFN-DCs) A549 total cell lysate for another 24 hours. Left panel: cells at day 0 (the arrow indicated an example of adherent monocytes after removing the non-adherent cells). Middle and right panels: cells at day 5. (B) Flow cytometry results, which are representative from CB unit 1, showed the expression of DC markers on unpulsed and pulsed IFN-DCs. (C) Average expression of DC markers from 5 samples of unpulsed and pulsed DCs. (*p-value < 0.05).
To further confirm the maturation of DC, the upIFN-DCs and pIFN-DCs were stained for DC-specific markers including HLA-DR, CD40, CD80, CD86, CD14, CD56, CD11c and CD123. The flow cytometric results of DC surface staining were summarized in Figure 1B and C. Higher percentage of cells expressed these markers was observed in pIFN-DCs (> 90%) compared to upIFN-DCs (> 50%) (Figure 1C).
Interestingly, the average percentage of cells expressed CD14 and CD56 markers increased in pIFN-DCs compared to that in upIFN-DCs, with the average percentages of 50.3% and 38.7% in upIFN-DCs, and 93.6% and 89.6% in pIFN-DCs, respectively. These phenotypic characters of IFN-DCs were different from conventional mature monocyte-derived DCs induced by IL-4, which do not expressed CD56 and CD14. These results indicated that CBMDCs was successfully stimulated by IFN, and had a typical immune character of IFN-DCs.
IFN-DCs and IFN-DC Conditioned Medium Stimulated Proliferation of Allogeneic Lymphocytes
To test the allogeneic stimulatory capacity of DC, allogeneic lymphocytes (alloLym) as non-adherent cell fraction were cultured with IFN-DCs or IFN-DC conditioned medium. At day 4 of co-culture, the alloLym grew as clusters with various sizes and floated in the culture medium, indicating that these cells were at the log phase of growth (Figure 2A). The number of primed-alloLym was significantly induced in all primed conditions compared to the unprimed-control (p-value < 0.05), and significant difference to that at day 0 (p-value < 0.05), except the unprimed one (p-value>0.05) (Figure 2B). The number of alloLym primed with upIFN-DCs and pIFN-DCs increased 1.4 ± 0.2 and 2.2 ± 0.3 fold, respectively, compared to the control alloLym after 4 days of co-culture (Figure 2B). After 7 days of co-culture, alloLym primed with pIFN-DCs reached the highest fold increase in the number of cells (2.7 ± 0.2) compared to upIFN-DCs (1.7 ± 0.3), and pIFN-DCs conditioned medium (2.2 ± 0.4). The difference in cell number from day 4 to day 7 was not significant in these primed cells (p-value>0.05), except cell primed with pIFN-DCs (p-value < 0.05). Interestingly, pIFN-DCs conditioned medium showed better effect on alloLym proliferation than the upIFN-DCs at both day 4 and day 7 of the co-culture, but not as effective as with pIFN-DCs (Figure 2B).
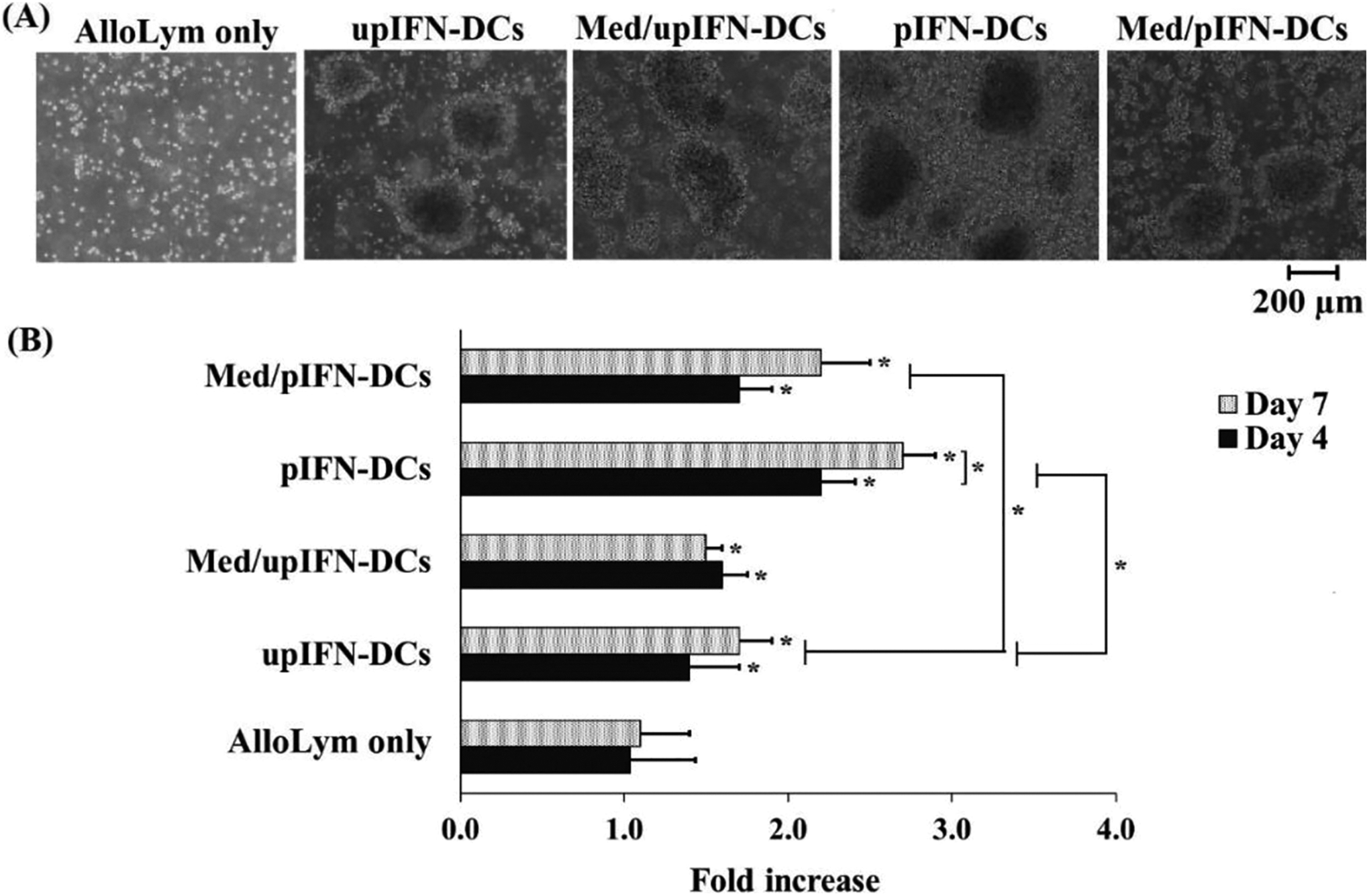
AlloLym proliferation induced by IFN-DCs and IFN-DC conditioned medium. (A) Stimulated allolym grew as clusters but not non-stimulated control (AlloLym only). (B) Average fold increase in the total cell number of alloLym incubated with IFN-DCs or IFN-DC conditioned medium. pIFN-DCs induced the highest proliferation of alloLym. Data was presented as mean ± SD in triplicate cultures (*p-value < 0.05).
IFN-DCs and IFN-DC Conditioned Medium Stimulated Proliferation of Allogeneic Vγ9γδ T Cells
We also checked the percentage of Vγ9γδ T cells (CD3+ Vγ9TCR+) in lymphocyte population obtained after stimulating alloLym with IFN-DCs or IFN-DC conditioned medium (Figure 3A). After 7 days of co-culture, the percentage of the induced Vγ9γδ T cells among alloLym population was determined by flow cytometry based on 2 markers CD3 and Vγ9TCR. Cell population, which was positive with both CD3 and Vγ9, was defined as Vγ9γδ T cells. All gated line was determined based on the isotype signal. The results showed that the percentage of Vγ9γδ T cells in lymphocyte population was induced sharply from less than 10% in the control to 85.4 ± 8.6% and 81.4 ± 9.2% in pIFN-DCs and upIFN-DCs treated cell populations, respectively (Table 1, Figure 3B). Additionally, med/pIFN-DCs and med/upIFN-DCs also were able to induce the percentage of Vγ9γδ T cells to 78.9 ± 7.9% and 74.5 ± 2.9% in lymphocyte population, respectively (Table 1, Figure 3B). The fold increase in the number of Vγ9γδ T cells was induced most with pIFN-DCs (46.1 ± 9.3), followed by med/pIFN-DCs (34.7 ± 11.3), upIFN-DCs (27.7 ± 10.4), and med/upIFN-DCs (22.4 ± 7.5) compared to the number of these cells at day 0 of co-culture (p-value <0.05) (Figure 3C). In the meantime, the percentage of CD4+ T cells and CD8+ T cells decreased after 7 days of induction (Table 1). These results demonstrated that IFN-DCs and their conditioned medium were able to induce the proliferation of Vγ9γδ T cells.

Vγ9γδ T cell proliferation induced by IFN-DCs and their conditioned medium. (A) The percentage of Vγ9γδ T cells population increased among lymphocyte population after incubating with IFN-DCs and their conditioned medium, presentative from CB unit 1. (B) Average percentages of Vγ9γδ T cells and (C) the fold increase of cell number were higher when inducing with pIFN-DCs compared to upIFN-DCs. Particularly, pIFN-DC conditioned medium had ability in inducing Vγ9γδ T cells proliferation similar to pIFN-DCs. Data was presented as mean ± SD in triplicate cultures (*p-value < 0.05).
Proportion (%) of T Cell Subpopulations in Allolym After Stimulated With IFN-DCs and Their Conditioned Medium.

* p < 0.05.
IFN-DCs and their Conditioned Medium Stimulated AlloLym to Kill A549 Cancer Cells
To test cytolytic activity of alloLym against target cells, stimulated alloLym were then incubated with Calcein AM labeled—A549 cells at 3 ratios of E: T: 12.5:1; 25:1; and 50:1. After 2 hours incubation with alloLym, fluorescent intensity of labeled-A549 cancer cells were reduced in co-culture wells, indicating that A549 cancer cells were lysed (Figure 4). Interestingly, more decayed fluorescent signal of labeled-A549 cancer cells was observed at higher ratio of E: T, underlining the efficiency of cytotoxicity of primed alloLym in killing A549 cancer cells (Figure 4). pIFN-DC stimulated alloLym efficiently killed A549 cancer cells with the highest cytotoxicity of 40%, 61.5% and 71%, respectively, at the 3 tested ratios. The cytotoxicity of alloLym stimulated by upIFN-DCs and their conditioned medium was significantly lower than the ones stimulated with pIFN-DCs (p-value < 0.05), with the cytotoxicity levels at the 50:1 ratio of effector/target were 23 ± 4.7% and 17.5 ± 7.4% compared to 71 ± 3.4%, respectively. Remarkably, the cytotoxicity of Med/pIFN-DC stimulated alloLym on A549 cells was higher than upIFN-DCs (p-value < 0.05), with 57 ± 9.7% cytotoxicity at E: T ratio of 50:1 (Figure 4).
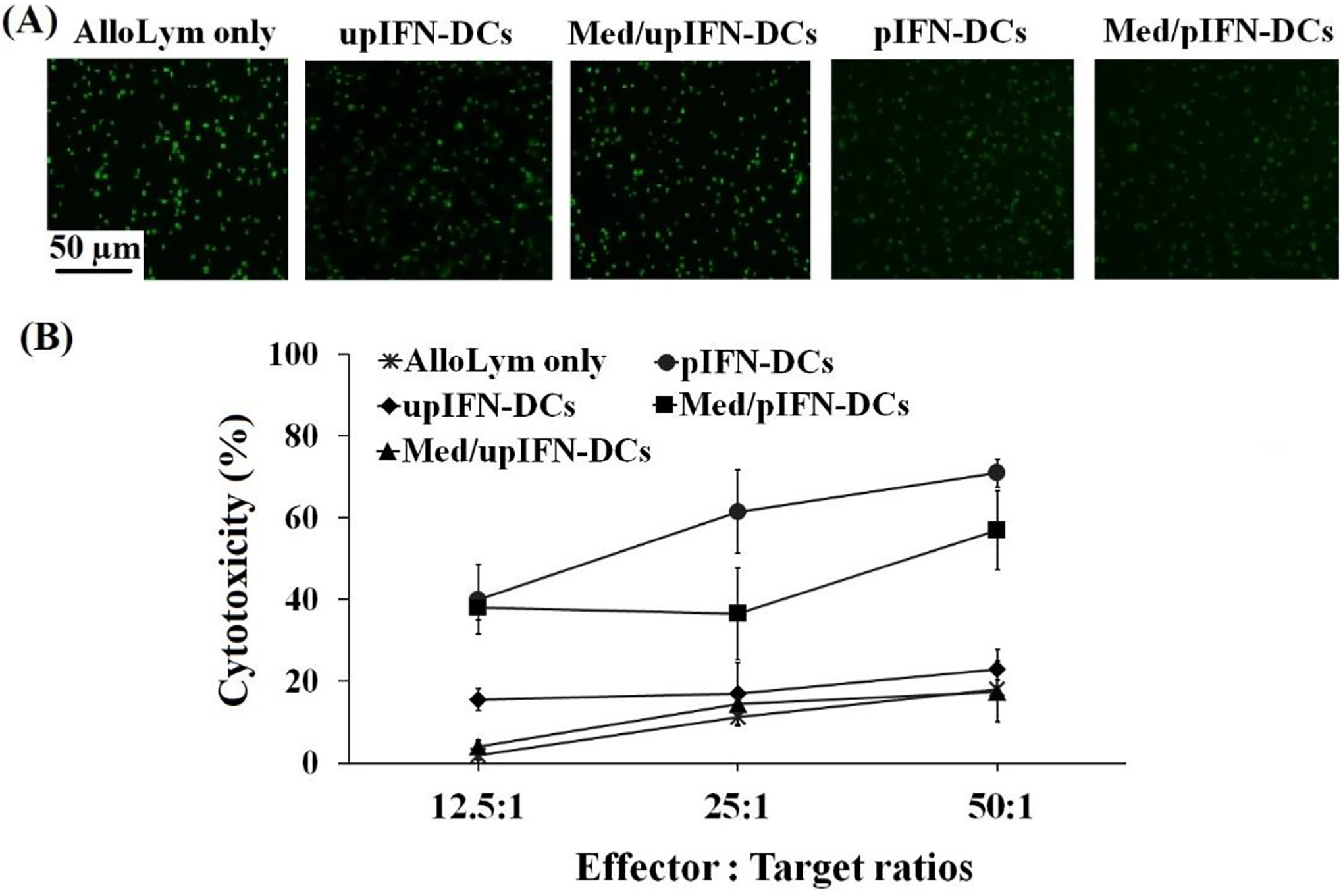
A549-cytotoxic activity of alloLym induced by pIFN-DCs and Med/pIFN-DCs. (A) Calcium AM labeled-A549 cancer cells were presented as green fluorescence dots in the pictures at E: T ratio of 50:1. The fluorescence signal was decreased in the samples incubated with alloLym stimulated by pIFN-DCs and pIFN-DC conditioned medium. (B) The statistical results showed that alloLym stimulated by pIFN-DCs and their conditioned medium had cytotoxic ability to A459 cells at all tested ratios. Data was presented as mean ± SD in triplicate cultures (*p-value < 0.05).
Discussion
Our study found that cord blood monocyte-derived DCs could be generated by IFN-α induction and that IFN-DCs were able to induce the proliferation and cytotoxicity of gamma delta T cells – a very promising weapon for fighting against cancer.
Autologous DC vaccination showed limited efficiency in treating cancer. 31 Moreover, using autoDCs may cause adverse effects to patients through peripheral blood collection. 6 -8,11,15,16 Compared to autoDCs, alloDCs have higher capability to induce a specific immune response because they can activate a wider range of CD8+ T cell immunity, including tumor responsive T cells and stimulate broad inflammatory response by polyclonal stimulation of allogeneic T cells. 31 However, alloDC using donor blood may be hindered due to ethical problems. 31 In our study, we showed that umbilical cord blood could be an alternative source for DC production. Previous studies have reported that the cord blood monocyte-derived DCs have the typical size and morphology of DCs, and express multiple DC makers. 32,33 We followed the protocol developed by Paquette et al. (1998) and Nidea et al. (2015), in which DCs were generated in vitro from monocyte of peripheral blood by inducing them with IFN-α. 22,23 Our DCs, which were generated from umbilical cord blood monocytes, had characteristics similar to the DCs that were generated from peripheral blood monocytes such as long dendrites, and high expression of DC surface marker profile including CD80high, CD86high, CD40high, HLA-DRhigh. 30 -34 In addition, DCs in this current study also expressed CD14high and CD56high, which are the 2 markers of monocytes. Nidea et al. (2015) also reported that DCs obtained from IFN-α-induced monocytes still kept these 2 markers on their surface. 22 These results demonstrated that we succeeded in inducing DCs from cord blood monocytes by IFN-α.
Beside the morphology and immune phenotypic characters, our results also showed that the obtained IFN-DCs could induce antitumor immunity. The number of alloLym increased more than 2 folds when incubated with pIFN-DCs and 1.7 times with upIFN-DCs. More importantly, the number of Vγ9γδ T cells was increased dramatically after alloLym was primed with IFN-DCs, accounting for more than 80% of the lymphocyte population. The number of these cells increased more than 45 times when inducing with pIFN-DCs, as compared to the control alloLym.
Recently, Vγ9TCR T cells have been shown to express many antitumor characteristics in both in vitro 24 -26 and in vivo studies. 27 -29 However, in natural conditions, Vγ9TCR T cell population only accounts for less than 5% in peripheral blood. 29,30 Thus, it is possible that increasing the population of Vγ9TCR T cells will improve antitumor immunity in patients. In our present study, IFN-DCs and their conditioned medium could stimulate the proliferation and activation of allogeneic Vγ9γδ T cells. It is known that Vγ9γδ T cells are activated and proliferated by bisphosphophonate in vitro. However, the method of cell expansion to obtain a sufficient number of Vγ9γδ T cells both in vitro and in vivo is still a problem. 35 -37 Recently, some research groups have developed a technique of using dendritic cells treated with bisphosphophonate in vitro to activate Vγ9γδ T cells and promote their proliferation. 38 -40 IFN-DCs in our study were also pulsed with bisphosphophonate prior to incubating with alloLym and showed similar effect on Vγ9γδ T cell expansion and activation. Moreover, DCs generated in our study highly expressed CD56 marker. These results were consistent with the study of Nidea et al. (2015), which reported that highly expressing CD56 IFN-DCs induced the proliferation and activation of Vγ9TCR T cells in vitro and in vivo better than conventional IL-4DCs. 22 In a recent published study of our research group, 34 we also generated IL-4DCs from cord blood monocytes and the proliferation of Vγ9γδ T cells stimulated with these IL-4DCs are smaller than that stimulated by IFN-DCs in this current study.
Moreover, our results indicate that alloLym incubated with IFN-DCs could killed A549 cells effectively. In particular, the cytotoxicity of alloLym stimulated by pIFN-DCs increased about 4 times, compared to the unstimulated control, and 2.6 times compared to upIFN-DC stimulated one. This result suggests that the alloLym was not only proliferated but also activated by IFN-DCs to recognize and destroy cancer cells. Our results also indicated that the IFN-DCs stimulated alloLym showed the much higher toxic to target cells compared to non-stimulated alloLym. This result may reflect the antigen-specifically killing activity of alloLym activated by IFN-DCs and their condition medium. Previous studies presented that the lymphocyte stimulated by IFN-DCs could product more Th1 cytokines such as IFNg and TNFa and then increase the antitumor activity of the lymphocyte. 22,23 However, to QG.18.09 prove the antigen-specific killing activity of stimulated lymphocytes, we should perform the experiment to analyze the cytokine profile of our IFN-DCs and allogeneic lymphocytes stimulated by these DCs in order to clearly support the anti-tumor immunity induction of these cells.
As shown in the results, Vγ9γδ T cells accounted for up to 85% of alloLym; thus, they could be a key player in providing A549 cytotoxicity activity of IFN-DCs stimulated alloLym. In this current study, both pulsed DCs and unpulsed DCs could stimulate T cells. However, the pIFN-DCs showed better effect than upIFN-DCs in Vγ9γδ T cells proliferation and cytotoxicity induction. The immune activity of tumor antigen-unpulsed DCs in inducing T cell growth and activation was reported. 41,42 It has also been shown that DCs can induce T cell proliferation as they interact with the target cells; and that immature DCs can stimulate antitumor activity. 41-42 Furthermore, adding antigen to DC therapy can greatly enhanced their efficiency. 43,44 Therefore, pulsed DCs stimulated T cell proliferation better than unpulsed DCs. 41 -44
Notably, our results revealed that pIFN-DC conditioned medium could also induce the proliferation and cytotoxicity of allogeneic Vγ9TCR T cells, similar to their parental cells. Remarkably, pINF-DC conditioned medium showed greater effects than upIFN-DCs in inducing alloLym proliferation and stimulating Vγ9γδ T cells to kill A549 cancer cells. DC conditioned medium has been reported to contain immunological cytokines and exosomes that can act like DCs. 11,15,16 By using the conditioned medium, we can take advantage of not only exosomes but also the other molecules that may play important functions in inducing the antitumor immunity of allogeneic lymphocytes. In perspective direction, we need to figure out these key molecules and the relationship with the other components in the conditioned medium that can be used in vaccination for cancer treatment.
Conclusion
In conclusion, we were successful to generate DCs from human umbilical cord blood monocytes by using GM-CSF and IFN-α. After maturating and pulsing with A549 total cell lysated, these IFN-DCs and their conditioned medium could induce the proliferation of allogeneic lymphocytes and Vγ9γδ T cells. In addition, alloLym stimulated by pIFN-DCs and pIFN-DC conditioned medium were able to kill A549 cancer cells efficiently. The data is very promising in developing an alternative therapy for cancer treatment in the future.
Footnotes
Acknowledgments
The authors would like to thank Vinmec Research Institute of Stem Cell and Gene Technology for supporting laboratory equipment. We are also grateful to Dr. Le Tra My, for English grammar reviewing.
Authors Contribution
Bui Viet Anh, Msc, and Chu Thi Thao, MSc are authors contribute equally to this paper.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Statement
The protocols were reviewed and approved by the Ethics Committee of the Dinh Tien Hoang Institute of Medicine (document no. IRB.009 on 20th June 2018), in compliance with the Helsinki Declaration. All methods were performed in accordance with the relevant guidelines and regulations. The mothers and the PBMC (peripheral blood mononuclear cell) donors were fully informed about the purposes of study, and their voluntary consent was obtained in written format. All the donors were healthy, and not carrying any of the following viruses: human immunodeficiency virus, hepatitis B virus, hepatitis C virus, cytomegalovirus, and syphilis.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article. This project was funded by Vietnam National University, Hanoi. Project code: QG.18.09. Bui VA and Chu TT were funded by Vingroup Inc, Hanoi. Project code: DT.01.

