Abstract
The purpose of this article is to review closure options for complex chest wounds in patients with locally advanced breast cancer. Experiences of the plastic and oncologic surgery teams at Moffitt Cancer Center were reviewed, and the literature researched for various surgical options of complex chest wound closure. Multiple treatment modalities exist for reconstruction of complex chest wall wounds with the external oblique and V-Y latissimus dorsi musculocutaneous advancement flaps serving as workhorses in reconstruction. Treatment of cancer has moved from simply a surgical solution to include other modalities such as hormonal therapy, chemotherapy, and radiation—the latter 2 having serious consequences for wound healing. A team approach and knowledge of available flap options are vital for closure of complex wounds in a timely manner. Appropriate planning can optimize the primary goal of the oncologic surgeon to remove the cancer and the plastic surgeon’s objective to reconstruct the defect and achieve a closed, durable wound prior to chemotherapy and radiation. We present the experience at the Moffitt Cancer Center in reconstructing challenging chest defects and review the reconstructive ladder.
Introduction
Despite advances in breast cancer outcomes, patients still present with locally advanced tumors due to a delayed presentation or unresponsiveness to systemic therapy. The Moffitt Cancer Center is privy to many complex wounds involving the chest as breast cancer serves as the highest cancer risk in women. In 2017, approximately 1291 patients were operated on for breast cancer with over 150 of these patients having at least stage 3 or 4 disease at the Moffitt Cancer Center. Even in situations of stage 4 disease, the role of the surgeon is to maintain local control. Locally advanced breast tumors can affect the patient’s quality of life once they start fungating through the skin, causing bleeding and infections. Excision of the primary tumor creates issues for the breast surgical oncologist in that primary closure can be difficult. Plastic surgeons must work with breast surgeons to achieve closure of wounds that heal quickly so as not to interfere with subsequent postoperative treatments such as radiation and chemotherapy. Nonsurgical interventions themselves can negatively affect wound healing and need to be considered in operative planning especially in regard to timing manner. Additionally, it is imperative the patient is aware of the potential necessity for wound care, osteoradionecrosis, and new cancer secondary to radiation. A review of the reconstructive options for the chest wall following cancer extirpation follows.
Complex Closure With Undermining
Nonfascial flaps involving skin and subcutaneous tissue can be considered on defects that can be closed with undermining via bilateral advancement flaps on the chest (Figure 1). The vascular supply is entirely dependent on the subdermal plexus of the flap and without a wide base the vascular supply to this flap can be compromised. Undermining is performed superiorly toward the clavicle and inferiorly toward the abdomen and closed in a straight horizontal line. Defects up to 15 cm can be closed vertically with larger defects warranting thoracoepigastric/thoracoabdominal flaps versus external oblique flaps as described below. 1 As long as the defect size is small and does not result in significant tension, this technique is simple, fast, and effective. At our institution, we sometimes use duoderm followed by taping to assist with tension offloading by holding the skin in place. 2 The quality of the periwound tissue and base of the wound must be considered with this technique. Radiation can cause significant changes to the soft tissue and impede wound healing, resulting in dehiscence. The risks and benefits of this technique as well as potential backup plans should failure result must be considered and discussed with the patient preoperatively.

Undermining via bilateral advancement and complex closure of a large chest wall skin defect (left) after bilateral mastectomy due to left advanced breast cancer. Due to left side closure being tighter than right, tape was applied over hydrocolloid dressing to reduce tension over the wound (middle). Post-op result (right).
Skin Graft
The simplest solution to achieve closure in wounds that are too large to close by primary intent or undermining is to simply transfer healthy skin from a separate donor site to the wound bed via a split thickness skin graft (Figure 2) relying on the wound bed to serve as a lifeboat to the graft by imbibition, inosculation, and neovascularization. 3 Skin grafting dates back as far as 3000 years ago in India with attempts to reconstruct nasal defects with the buttocks serving as the donor site resulting from nasal amputation as a form of punishment for thievery. 4 One major advantage of a skin graft is achieving immediate coverage of the underlying tissue. Further, meshing the skin allows for increased surface area coverage of the exposed wound that can subsequently heal independently in a planned manner. Usually, split thickness grafts are used, but there have been reported cases where full-thickness skin grafts were used in chest reconstruction. 5 Unfortunately, not only does a skin graft cause a defect at a secondary donor site but it also is completely infeasible when exposed bone and tendons inhibit adherence and necessitate dermal substitutes and multiple procedures. Additionally, the frequent need for early postoperative chemo/radiation therapy often precludes the use of skin grafts as these therapies can impair wound healing resulting in complete graft loss (for an example of radiation-induced tissue changes, see Figure 3). Another disadvantage is the increased time to healing with skin grafts, which can also delay radiation versus other closure techniques. In these instances, a flap or more complex closure is indicated.

Split thickness skin graft to chest wall defect before (left) and after (right).

External oblique flap advancing tissue from the abdomen over large distances. Before (left), after (right). Note the radiation changes to the skin causing hyper and hypopigmentation as well as contracture of tissue to the left breast in the photo on the left.
Fasciocutaneous Flaps
Thoracoepigastric/thoracoabdominal
The thoracoepigastric and thoracoabdominal flaps are 2 different fasciocutaneous flaps based off of a segmental blood supply with much confusion regarding their nomenclature. The medial version (thoracoepigastric flap) is based off of the superficial epigastric system and the lateral version (thoracoabdominal flap) is based off of the lateral intercostals and lumbar arteries. A significant difference is that the thoracoabdominal flap donor site can usually be closed while the thoracoepigastric flap may require backgrafting. 1 The very advantage that makes dissection of this flap simple is also the disadvantage of not having the robust blood supply of the musculocutaneous flaps. 6 Thus, they are reserved for inferior thoracic defects. 7 Yet, some studies claim the superiority of these flaps over musculocutaneous flaps as they present comparable results with tolerance of radiation but with decreased operating time, blood loss, and mean hospital stay. 8
Musculocutaneous Flaps
Mathes–Nahai
Any discussion of musculocutaneous flaps requires an understanding of the classification of the arterial supply. Table 1 shows the muscles can be categorized into 5 types. 9 Understanding the blood supply of the flap is key to optimizing its viability by ensuring dissection, including the pertinent vessels. Preparation involving a thorough study of the involved anatomy sets the surgeon up for the greatest chance of success. Please refer to this table as needed throughout this discourse.
Mathes–Nahai Classification of Flaps Based on Arterial Supply of Muscles.

External oblique
The external oblique flap serves as a workhorse flap in chest wall reconstruction for several important reasons. First, it is a reliable flap with both a dominant and segmental blood supply. The deep circumflex iliac artery serves as the dominant pedicle in the flap and is located laterally along with a segmental supply from the intercostal arteries. Though, as previously mentioned, debate does exist regarding the superiority of this flap versus the fasciocutaneous flaps of the abdomen and this is likely fueled by confusion regarding its classification as a type IV versus type V muscle. 10 Second, the flap accommodates a large-sized defect reported up to around 400 cm2 (and even up to 800 cm2 in an obese individual) with simple closure of the donor site (Figure 3). 7,11 Most importantly, for patients requiring a “one-and-done” surgery in order to pursue further treatment without concerns of wound healing delays, this flap is an essential component in the reconstructive toolbox.
Pectoralis advancement/turnover
For small chest wall defects over the middle portion of the chest, the pectoralis muscle is an excellent choice especially in cases of osteoradionecrosis. This type V muscle receives its dominant supply from the thoracoacromial artery with segmental supply from arteries off the lateral thoracic, internal mammary, and intercostals. 12 There are 2 main techniques of flap use with variations of each. The pectoralis major can be dissected off the pectoralis minor and advanced, medially retaining its major blood supply from the thoracoacromial artery. Alternatively, the flap can be turned over requiring loss of the thoracoacromial artery and receiving its blood supply from the internal mammary artery (IMA). When neither of these options achieves sufficient closure of the defect, backup plans include separating the sternal and clavicular heads of the muscle, releasing the humeral insertion, and using the contralateral pectoralis major muscle. In designing coverage using the pectoralis major flap, it is essential to consider the patient’s history and to plan accordingly. For example, patients with an extensive cardiac history often require the use of the IMA for bypass procedures and knowledge of previous harvesting of vessels prevents vascular embarrassment. Although dissection can be straightforward, understanding the anatomy allows for optimization of length and vascular supply.
Pedicled transverse rectus abdominis flap
The rectus abdominis muscle extends from the xiphoid to the pubic symphysis, lending it as a versatile flap that can be used in multiple regions on the body due to its length and vascular supply. Deemed a type III muscle with dual major supply from the deep superior epigastric artery (DSEA) and deep inferior epigastric artery (DIEA), the flap can be based either superiorly or inferiorly. The DIEA is superior to the DSEA in most individuals, and as such performing a delay procedure where the DIEA is clipped prior to reconstruction can allow for the DSEA to compensate, thus increasing the flap success when based off of the DSEA. 13 Superiorly based flaps are excellent for chest reconstruction, such as in sternal defects with turnover flaps and breast reconstruction via pedicled flaps. These pedicled rectus flaps must be tunneled underneath tissue to allow the donor muscle to fill the defect. The resulting abdominal defect and potential for bulging and hernias must be discussed with the patient preoperatively. Free flaps (discussed below) based on the DIEA can also be used for breast and chest wall reconstruction with sparing of the rectus muscle. As most plastic surgeons are comfortable with the dissection of this flap, it has become a popular option for reconstruction of chest wall defects. 14,15
Latissimus dorsi and VY-advancement
The latissimus dorsi, Latin for “broadest of the back,” debuted as a flap in 1896 with Dr Igninio Tansini using it for breast reconstruction following a mastectomy. It would take another 80 years before becoming a popular form of reconstruction once the work of Mathes and Nahai on the arterial classification of flaps prompted an era of musculocutaneous reconstruction. 16 The latissimus dorsi flap is versatile for a variety of defects, and its long pedicle length allows for wide rotation. The blood supply is type V with the main vessel being the thoracodorsal artery with secondary branches from the lumbar and intercostal arteries. Due to its large size, the muscle can be used in intrathoracic defects to fill a volume void as well as extrathoracic defects to provide durable coverage. 14 The flap can be tunneled under the axilla as in breast reconstruction, but it can also be used for large chest wall defects as a V-Y advancement flap with a skin paddle (Figure 4). The resulting defect can be closed primarily up to 13 cm in some reports. 17 Even further advancement can be obtained by releasing the muscle from the humorous and the thoracodorsal artery branch to the serratus anterior. The large amount of undermining with this technique makes seromas at the donor site a common complication and multiple drains are placed intraoperatively in anticipation. The ability to use large skin paddles, the reliability, and the ease of dissection with the V-Y advancement flap understandably make it an essential tool in which all plastic surgeons should be familiar. One reason this form of reconstruction has been preferred in our practice is because, in the rare instance of complete control or cure of cancer, total autologous abdominal-based reconstruction of the breast is still left as a viable option.

Latissimus dorsi V-Y advancement flap with primary closure of donor defect to treat large mass of left chest wall. Before (left), after (right).
Other
Posterior arm flap
For unique defects near the axilla and adjacent to the thoracic chest wall, a posterior arm flap can be considered (Figure 5). This flap is a fasciocutaneous flap based off of an unnamed vessel from the brachial or deep brachial artery. In one study of 35 flaps, the average flap dimension was 8 cm × 12 cm. 18 An obvious downside of this reconstruction is the ensuing scar and deformity that results at the donor site on the arm, which is visible with short sleeve shirts as opposed to other types of reconstruction. Although this flap is suitable in a limited number of situations, it is nonetheless helpful to bear in mind.

Posterior arm flap for coverage after removal of axillary mass. Before (left), after (right).
Omentum
When other reconstructive options have been exhausted, the omentum can be used for chest wall defects. Understandably, other flaps are preferable, especially in the case where a watertight seal is desired as there is no cutaneous paddle. The omentum is a Mathes–Nahai type III flap supplied by the left and right gastroepiploic arteries with preference given to using the right artery that typically presents as the larger vessel. The pliability and dimensions up to 25 cm × 34 cm are advantages, allowing the flap to reach deep crevices in wounds unreachable by musculocutaneous flaps. 7 Yet, there are many disadvantages that preclude its use as a first-line flap such as having to enter the abdominal cavity, potential for herniation through the break in fascia that allows externalization of the flap to fill the defect void, and the requirement of skin grafting over the flap with further delayed healing. Further, it is a flap whose dissection may require the assistance of the general surgeon. 14
Contralateral breast
For defects of the chest wall that are not too far off-midline, the contralateral breast can serve as a good source of soft tissue for coverage. The breast receives its blood supply from multiple sources, including the lateral thoracic artery, IMA, thoracoacromial artery, and intercostal arteries. 12 An internal mammary artery perforator (IMAP) propeller flap has been described for contralateral mastectomy defects. The flap is based off of the second and third IMAP with dissection occurring in the subcutaneous plane. The flap is especially beneficial in women with large breasts with a flap size reported of up to 26 cm × 11 cm, which can be saved from a breast undergoing a prophylactic mastectomy. 19 Unfortunately, lack of excess skin tissue in women with smaller breasts may necessitate the need for backgrafting of the donor site. The poor cosmesis of this flap lends it as a backup solution for coverage as the donor nipple can end in an odd location and because the pivot point of the flap can be bulky over the sternum. Finally, for patients with advanced cancer, completely removing the unaffected breast may be preferable to transplanting tissue that has potential for malignancy.
Free flaps
Free flaps were pioneered by surgeons in the fields of head and neck and hand where vessel anastomosis allowed for transfer of tissue from one area of the body to another by connecting donor vessels to recipient vessels in the defect site. 20 The microvascular technique was further refined by considering variables such as vessel diameter, pedicle length, and donor site deformity. Despite a wide variety of surgical options for closing defects of the chest wall, it is important the surgeon goes up the reconstructive ladder and considers the simplest yet still efficacious method first. 21 Free flaps are good options where all other options are not appropriate or for patient comorbidities such as smoking history. Popular free flaps include the deep inferior epigastric artery perforator flap (Figure 6) and anterolateral thigh flap.
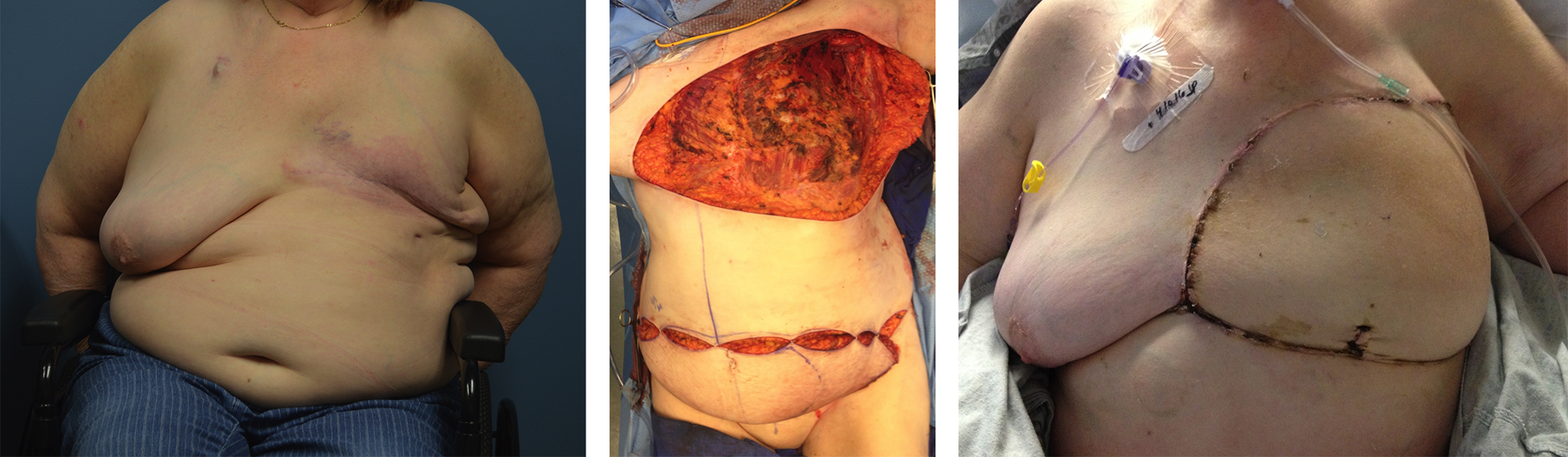
Deep inferior epigastric artery perforator flap (DIEP) using abdominal tissue to reconstruct large defect of chest wall using microvascular techniques. Before (left), intraoperative (middle), after (right).
Perforator Flaps
A new frontier in reconstruction involves the use of perforator flaps. These flaps are often based off of previously well-described muscle flaps but involve dissection through these muscles to free the perforator, thus serving as a muscle-sparing technique. An example is the thoracodorsal artery perforator flap that allows preservation of the latissimus dorsi muscle. While the idea of muscle preservation is appealing, it needs to be weighed against the technical challenge of perforator dissection and anatomic variability.
Guidelines and Pitfalls
When faced with a reconstructive challenge in the chest wall, we recommend asking the following questions: What is the size of the defect? Where is the location of the defect on the chest wall? What are all the options available for reconstruction? What is the easiest option? Is the patient a good candidate for a given reconstruction option based on medical factors? What is the backup plan if the first option fails?
The goal is to pick the option that gives the most durable, safest, easily executable result with the highest likelihood of success.
The most common pitfall in chest wall reconstruction is poor planning. The best option for one patient may be a completely impossible solution for another patient based on the previously listed questions. The most important consideration for coverage is the vascularity of the tissue needed. Creating templates of the defect and rotating these templates along the pedicle trajectory can help determine whether the proposed flap will fully cover the defect in a tension-free manner. If there is question of vascularity intraoperatively, modalities such as the external and implantable Doppler, needle pricking, and SPY technology can be used to assess flow in the flap that can help guide further refinement such as release of sutures, flap delay, trimming of nonviable tissue, or anastomotic revision. When designing flaps, it is always best to air on the side of excess as it is simple to trim tissue but deficiencies of even a few centimeters can result in reconstructive failure. Finally, inadequate communication with the general surgery team both preoperatively and intraoperatively can result in unnecessary challenges. We recommend the plastic surgeon be available for intraoperative assessment during the tumor extirpation in the event that unexpected findings occur so that both teams can discuss any alteration of previous plans that could affect reconstructive outcomes.
Conclusion
Despite improved treatment of cancer, surgeons continue to face complex requirements for tissue coverage of wounds. At the Moffitt Cancer Center, some of the most advanced cases are seen from around the world. Detailed here are modalities used for chest wall reconstruction that have proven safe and durable (Figure 7). 22 In our experience, the latissimus V-Y advancement flap and external oblique flap are the workhorses of choice. However, it is important to assess each patient individually and to think outside the box when go-to solutions aren’t feasible. Of utmost importance is communication between the patient, onocologic surgery team, and reconstructive surgery team to prevent ligation of essential vessels, thoracotomy tubes through potential flaps, unacceptable deformities, and so on. Before any scalpel is lifted to remove the tumor by the general surgeon, the plastic surgeon should first pick up a marking pen in the preoperative area and the surgeons should work together to ensure tumor resection and defect closure for the best patient outcomes.

Review of various chest wall reconstruction modalities. CONTRA BREAST indicates contralateral breast; EO, external oblique; LD adv, latissimus dorsi advancement; PEC, pectoralis major; POST ARM, posterior arm; TA/TE, thoracoabdominal/thoracoepigastric; VRAM, vertical recutus abdominis myocutaneous.
Footnotes
Acknowledgments
The authors would like to thank the thousands of patients who have allowed us to care for them and who have served as our greatest teachers.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
