Abstract
Oncoplastic surgery is an evolving field in breast surgery combining the strengths of breast surgical oncology with plastic surgery. It provides the surgeon the ability to excise large areas of the breast in the oncologic resection without compromising, and possibly improving, its aesthetic appearance. The purpose of this review is to provide a guide that could help a breast surgeon excise breast cancer in most areas of the breast using 5 oncoplastic techniques. These techniques would be used depending primarily on the location of the cancer in the breast and also on the size of the tumor.
Background
In 1907, Halsted published his results for radical mastectomy in the treatment of breast cancer. 1 Depending on the type of cancer, his cure rate results ranged between 25% and 75%. The surgery itself was morbid but remained the standard for many years to come. Since the advent of the radical mastectomy, an effort has been made to treat breast cancer effectively not only oncologically but also aesthetically. The radical mastectomy gave way to the modified radical mastectomy that followed with the simple mastectomy. Today, if a surgeon can maintain the breast form, the standard has become the partial mastectomy operation when operating for breast cancer. Large volume displacement oncoplastic surgery allows the surgeon the ability to resect large volumes of breast tissue and achieve the advantages in large volume resection which include fewer positive margins, 2,3 while maintaining and possibly improving the aesthetics of the breast form using mastopexy and breast reduction designs. Patients who would have been previously thought to be mastectomy candidates can now have breast-sparing procedures with improved aesthetics.
The oncoplastic surgical team typically consists of a breast surgeon and a plastic surgeon who work together in designing mastectomy skin incision patterns to best access the region of the breast with cancer and subsequent pedicles that have anatomically established blood supply patterns to tissue areas that can be displaced to fill in the void created by the breast tissue partial mastectomy. It is possible to have 1 surgeon with appropriate training in breast surgical oncology and breast plastic surgery who can do oncoplastic surgery by himself or herself as long as the training is complete with regard to performing the surgery safely and effectively based on clinical outcomes and with regard to having the knowledge to deal with postoperative complications in both the breast surgery oncologic and plastic surgery realms.
Oncoplastic surgery in the broadest definition involves the oncologic resection of breast cancer with subsequent reconstruction. To be more practical, it has been divided into volume replacement and volume displacement oncoplastic surgery. 4 Volume displacement oncoplastic surgery, which involves the techniques described in this article, is the removal of breast cancer followed by the manipulation of local ipsilateral breast tissue to recreate the breast form. This is often done with mastopexy or breast reduction techniques among others. Volume displacement oncoplastic surgery can remove up to a third of the breast, with the use of the remaining breast tissue for reconstruction, assuming the patient has moderate- to large-sized breasts. 4,5 Volume replacement oncoplastic surgery involves large volume removal of breast cancer and the use of regional flaps or implants to reconstruct the breast. An example of this could include thoracodorsal perforator flaps rotated into large upper outer quadrant defects following oncologic resection. 6,7
Purpose
Our goal is to describe 5 techniques that would allow breast surgeons access to cancers in most parts of the breast with the ability to excise a large volume of breast in an effort to achieve negative margins with the reliance on mastopexy and breast reduction techniques to close the breast while achieving good aesthetics.
Achieving any of these techniques is dependent on the understanding of the blood supply of the breast (Figure 1A). The blood supply medially is off the internal thoracic artery with strong complex of perforating vessels. There is additional bloody supply from the lateral thoracic artery and thoracoacromial branches. Inferiorly, there are intercostal branches. 8

A, Blood supply of the breast. B and C, Standard incision patterns for oncoplastic surgery (B) and blood supply for 3 major oncoplastic breast flaps (C). D, Oncoplastic skin markings 1 through 9 for vertical reduction mastopexy. Markings: (1) preoperative view, (2) midline, breast meridian and Pitanguy’s point markings, (3) breast shifted laterally for marking vertical limbs, (4) breast shifted medially for marking vertical limbs, (5) symmetry of the markings checked by shifting both breasts medially, (6) symmetry of the markings checked by using a paper tape absorb the ink, (7) symmetry of the markings checked by using a paper tape on the opposite breast, (8) sternal notch to nipple distance measured by measuring tape to 21 cm (alternatively, new nipple position should ideally be at the inframammary crease level), and (9) nipple to inframammary fold measured by measuring tape to 7 cm. (Authors had −21 and −7—confirm they mean measured “to” 21 cm and 7 cm.)
Each technique builds upon 2 surgical principles that are the basis of any breast mastopexy or reduction technique: the skin incision type and the pedicle design. The skin incision types involve a circumvertical, periareolar, 4 crescentric, 9 L-shaped incision, 10,11 or an inverted T (Wise) pattern incision that allows the breast surgeon access to most regions of the breast for cancer resection. Figure 1B shows the 2 basic skin incision types. The pedicle design type provides blood supply to the nipple areola complex and any other additional tissue that can be used to fill in the partial mastectomy defect. Figure 1C shows 3 key pedicle designs involving the superior pedicle, the superomedial pedicle, and the inferior pedicle. Mixing and matching the skin incision type and the pedicle design type allows the breast surgeon access and the ability to resect large regions of breast tissue in most areas of the breast. The general principles for marking the breast skin with patient standing upright are illustrated in Figure 1D for the vertical oncoplastic breast reduction (technique 2). Skin markings in general are similar for all 5 techniques. To align the new site for the superior areola, a rough guide is to use the middle finger slid behind the breast from inframammary fold (IMF) and the nipple areola complex is generally in correct position (Figure 1D, markings 2). Vertical limbs are marked by shifting the breast laterally and medially (Figure 1D, markings 3 and 4). Symmetry between the breasts should be assured and can be checked by several simple methods shown such as the use of tape (Figure 1D, markings 5-7) or a ruler can be also used. The proper nipple placement can be verified with sternal notch to nipple distance of about 21 cm (Figure 1D, markings 8). It is critical to mark the IMF and maintain nipple to mammary fold measure of about 7 cm (Figure 1D, markings 9).
Technique 1
Double doughnut mastopexy 4,12 (Figure 2):
Addresses small to medium cancers centrally located in any quadrant.
Appropriate for grade I ptosis. Especially useful in patients with enlarged nipple areola complexes.

Double doughnut mastopexy incisions for small central tumors.
Operative Markings
Patient should be standing while marking is made. Initial markings should mark the IMFs on each side, the sternal midline, and the paramedian line (that splits the breast in half; Figure 1D, marking 2). The difference is that the nipple areola complex (NAC) complex will not be relocated because it is already in good position. If the cancer is palpable, mark the region of the cancer or use the wire localization to mark the tumor area.
Intraoperative Steps
Position the patient supine with arms out on an arm board at 90°.
Complete sentinel or complete axillary lymph node dissection first as appropriate through separate axillary incision or alternatively can be performed through same breast incision.
Use assistant to place the nipple areola complex in full stretch. Use a 42- or 38-mm cookie cutter and a marking pen to mark the new size of the nipple areola complex centered over the nipple. If there is no nipple areola complex enlargement, in full stretch, draw a concentric circle outside the nipple areola complex using a width of approximately 2 cm between the outer and inner concentric circles. The patient should be warned in the informed consent that there may be long-term postoperative nipple areola enlargement.
Start by de-epithelialization of the skin region between the 2 concentric circles.
Make up to a 50% full-thickness incision in the outer circle to allow maximal access to the quadrant or region where the cancer is present.
Raise appropriate skin flaps not only over the tumor area but at least 2 cm additionally around the tumor area that will help facilitate the closure of the defect later.
Remove the cancer and orient the specimen accordingly. If the cancer is central, consider taking the skin that would have been de-epithelialized en bloc with the cancer deep to this tissue.
Ask pathology to weigh the specimen that has been removed. It is critical to leave approximately 10% extra volume on the side to undergo radiation. Undermine the breast tissue surrounding the cavity in a plane superficial to the pectoralis fascia. Consider taking the pectoralis fascia as a deep margin if the cancer is close to the fascia.
Place medium clips at the level of the deep fascia, superiorly, medially, laterally, and inferiorly to help with radiation guidance.
Place a drain in the cavity if necessary.
The breast tissue should be able to be approximated over the drain collapsing the defect using 2-0 or 3-0 sutures. While absorbable sutures can be used for smaller breasts, it is important to use a nonabsorbable suture around the areolar incision in order to prevent distortion of areola and chronic scar widening.
Approximate the skin with deep dermal sutures followed by a running subcuticular stitch.
The contralateral breast can be operated on in a similar manner with judicious resection of breast tissue with patient being sit up to evaluate symmetry after incisions are either sewn or stapled.
Dressings and bra should follow.
Technique 2
Wise or circumvertical incision pattern, superior pedicle 13 -15 (Figure 3A):
Small, moderate, large inferior pole or lower outer/inner quadrant cancers.
Appropriate for grade II to III ptosis.
Should involve a plastic surgeon or a surgeon trained in plastic surgery techniques.
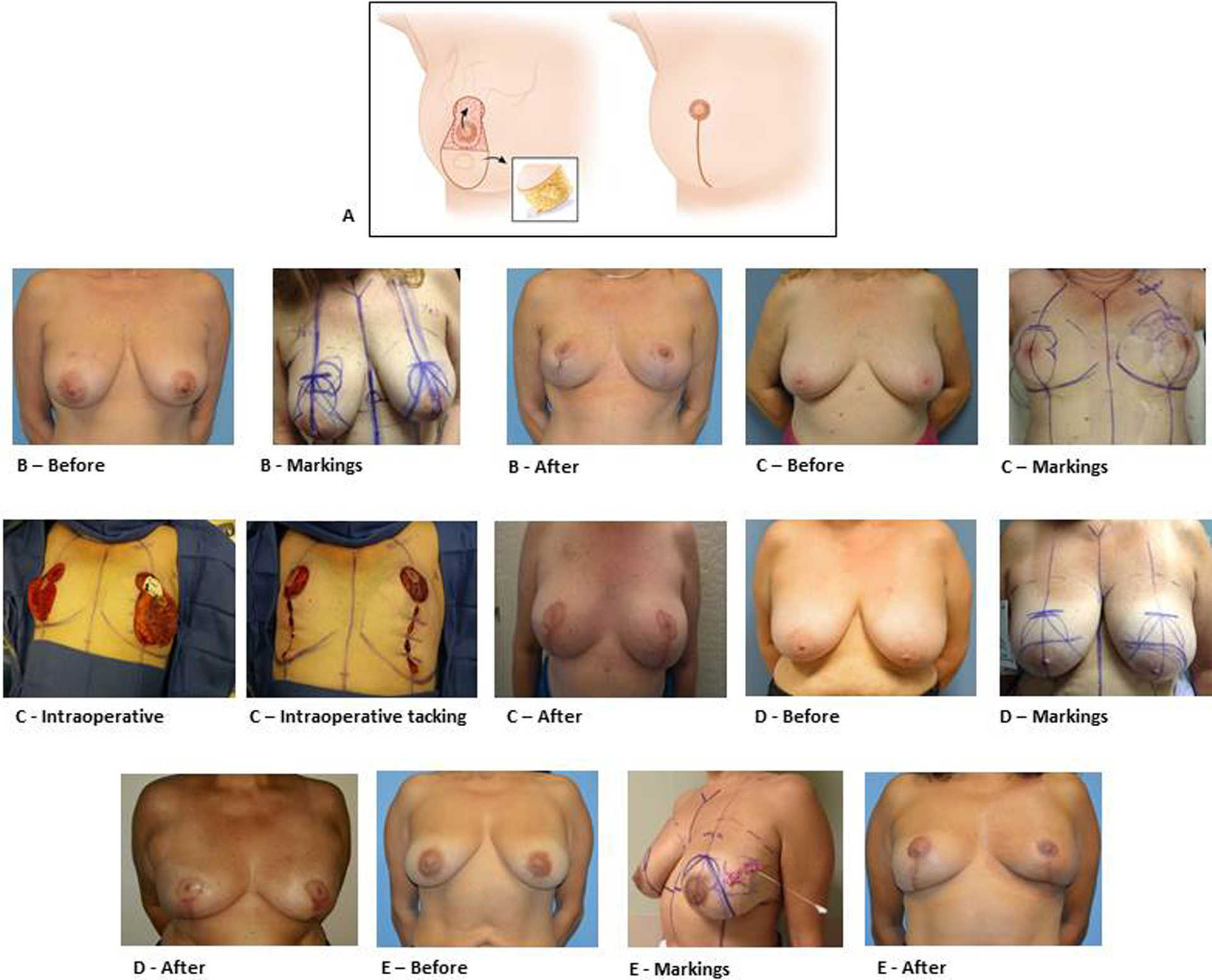
A, Inverted T incision for inferior pole breast cancers with before and after results showing mastopexy effect. B, A 47-year-old female with pathological stage T1N0M0 21 mm ER+, PR+, Her2 invasive ductal carcinoma at right breast at the 1-o’clock position. Total of 275 g on the right and 144 g on the left breast were excised using superior pedicle vertical scar technique. B, A 50-year-old female with pathological stage T2N0M0 28 mm ER+, PR+, Her2 invasive ductal carcinoma at left breast at the 1-o’clock position. Total of 189 on the right and 218 on the left breast removed using medial pedicle vertical scar technique. See intraoperative pictures of the procedure depicting the location of the tumor and vertical pattern. C, A 60-year-old female with pathological stage TISN0M0 5 mm DCIS at left breast at the 1-o’clock position. Total of 374 g on the right and 374 g on the left removed using vertical pattern medial pedicle technique followed by radiation. D, A 52-year-old female with pathological stage T2N1AMO 35 mm ER+, PR+, Her2 lobular carcinoma at left breast at the 2-o’clock position. Total of 273 g removed from each breast using vertical pattern superomedial pedicle technique followed by radiation (E).
Operative Markings
A discussion between the plastic surgeon and breast surgeon determines what type of skin incision is ideal. From an oncologic standpoint, inferior midline cancers (at the 5- to 7-o’clock position) can be excised with a circumvertical incision design. Inferior outer lateral or outer medial cancers are more appropriately treated with a Wise pattern incision. With this, the skin superficial to the cancer can be excised assuring a negative anterior margin without compromising cosmesis. Additionally, tissue including the pectoralis fascia in this region is taken to increase the likelihood of a posterior negative margin. Both breasts are marked while standing. If the cancer is palpable, the cancer is also outlined during the marking process. The markings for this step are outlined in Figure 1D.
Intraoperative Steps
Position the patient supine with arms out on an arm board at 90°.
The plastic surgeon can begin the contralateral mastopexy at the same time the breast surgeon operates on the breast with cancer, or lumpectomy can be performed with the assistance of the plastic surgeon.
Complete sentinel or complete axillary lymph node dissection first as appropriate through separate axillary incision or alternatively through same breast incision.
Use assistant to place the nipple areola complex in full stretch. Use a 42-mm cookie cutter and a marking pen to mark the new size of the nipple areola complex centered over the nipple.
De-epithelialize around the region outside the cookie cutter marking defining the new nipple areola complex and in the region of the superior pedicle (including the superior keyhole).
Perform the partial mastectomy by making full-thickness skin incisions down and including the pectoralis fascia in the region of the inferiorly based cancer. This may include just the midline inferior pole if the cancer is at the 5- to 7-o’clock position (and the skin incision design is a circumvertical design) or also the medial and lateral wing if the cancer is in the lower outer or lower inner quadrants (and the skin incision design is a Wise pattern).
Mark the partial mastectomy for orientation for pathology margin assessment.
Ask pathology to weigh the specimen that has been removed. It is critical to leave approximately 10% extra volume on the side to undergo radiation. Complete the elevation in the superior pedicle and approximation of the lateral and medial pillars in a standard mastopexy design fashion (completed by the plastic surgeon).
There are 4 patient examples shown using this technique in Figure 3B to E. The preoperative markings and some intraoperative images are included to highlight the steps involved with this technique. The specific details of the location and type of breast cancer are included in the caption of Figure 3.
Technique 3
Wise incision pattern inferior pedicle 5,16 -18 (Figure 4A):
Small, moderate, large superior pole or upper inner and upper outer quadrant lesions.
Appropriate for grade II or III ptosis.
It has an added advantage (compared to mastectomy) for use in younger patients with regard to maintaining sensation to the nipple areola complex, even in larger resections.
Should involve surgeon trained in plastic surgery techniques.
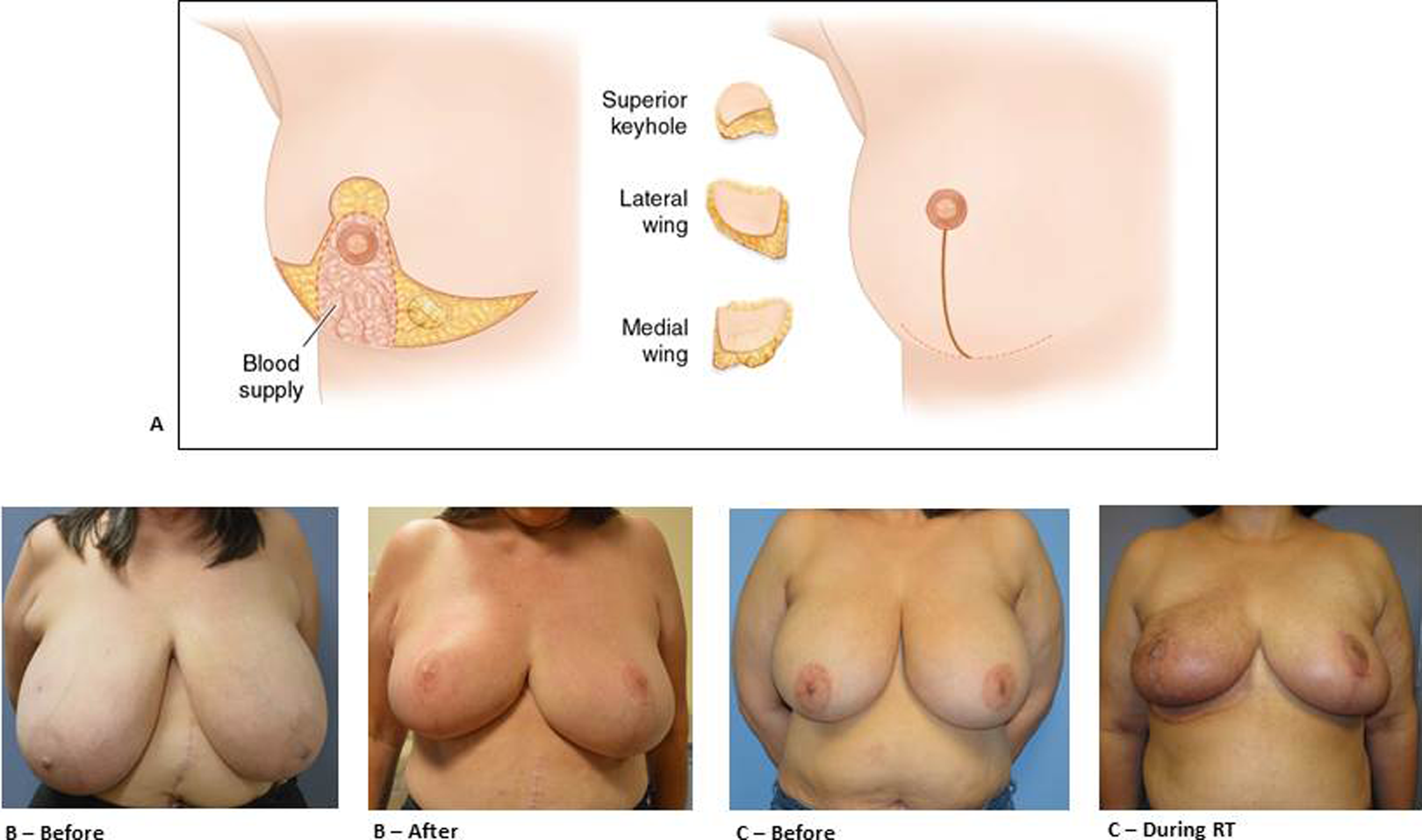
A, Wise pattern incision inferior pedicle and various lower pole tumor excision sites. B, A 63-year-old female with right pathological stage T1N0M0 invasive lobular carcinoma at the 11-o’clock position. Total of 1687 g on the right and 1300 g on the left breast removed using Wise pattern inferior pedicle. C, A 49-year-old female with pathological stage T1N0M0 15 mm invasive cribriform carcinoma ER+, PR+, Her2 at the 7-o’clock position. Total of 1245 g on the right breast and 1313 g on the left breast removed using Wise pattern inferior pedicle technique followed by radiation (C).
Operative Markings
A discussion between the plastic surgeon and breast surgeon determines what type of skin incision is ideal. From an oncologic standpoint, large superior quadrant cancers can be accessed easily and excised without much trouble. Additionally, tissue including the pectoralis fascia in this region is taken to increase the likelihood of a posterior negative margin. Both breasts are marked while standing. If the cancer is palpable, the cancer is also outlined during the marking process.
Intraoperative Steps
Position the patient supine with arms out on an arm board at 90°.
The plastic surgeon can begin the contralateral mastopexy at the same time the breast surgeon operates on the breast with cancer.
Complete sentinel or complete axillary lymph node dissection first as appropriate through separate axillary incision.
Use assistant to place the nipple areola complex in full stretch. Use a 42-mm cookie cutter and a marking pen to mark the new size of the nipple areola complex centered over the nipple.
De-epithelialize around the region outside the cookie cutter marking defining the new nipple areola complex and in the region of the inferior pedicle.
Perform the partial mastectomy by making full-thickness skin incisions down and including the pectoralis fascia in the region of the superiorly based cancer. Cancer access should be optimized by elevating the superior mastectomy skin envelope in the process.
Mark the partial mastectomy for orientation for pathology margin assessment. Ask pathology to weigh the specimen that has been removed. It is critical to leave approximately 10% extra volume on the side to undergo radiation. Complete the elevation of the inferior pedicle and approximation of the lateral and medial pillars in a standard Wise design fashion (completed by the plastic surgeon). Special care needs to be given to avoid an asymmetrical IMF appearance, especially if the medial and lateral pillars cannot be fixed to the midpoint of IMF. This underscores the importance of preoperative markings.
Complete closure.
There are 2 examples shown using this technique in Figure 4B and C. The specific details of the location and type of breast cancer are included in the caption of Figure 4. There is a completion photo following radiotherapy.
Technique 4
Wise or circumvertical incision pattern, superomedial pedicle 17 -19 (Figure 5A):
Small, moderate, large inferior pole, superior pole, lower outer quadrant, upper outer quadrant lesions.
Differs from the superior pedicle technique, as cancers just superior to the nipple areola complex can be excised with the superior keyhole excision.
Not appropriate for inner, medial cancers as the pedicle arises from this point.
Good for grade II or III ptosis.

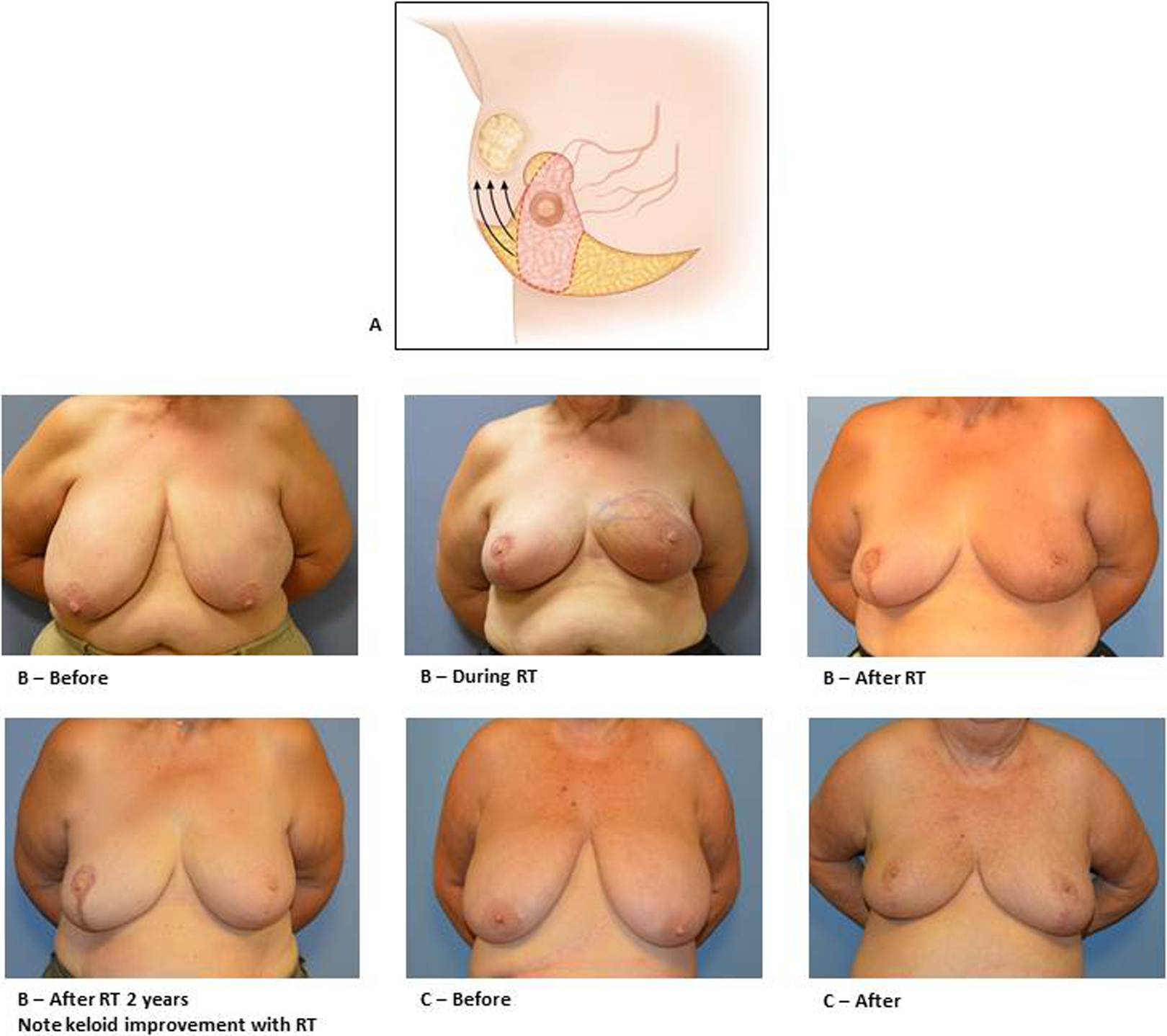
A, Superior medial pedicle flap used for lower outer quadrant tumors, inferior pole, upper outer quadrant, and superior quadrant tumors. B, A 36-year-old female with pathological stage T1N1MO 25 mm ER+, PR+, Her2 invasive ductal carcinoma at right breast at the 6-o’clock position. Total of 561 g on the right and 613 g on the left removed using Wise pattern superomedial pedicle technique with axillary node dissection followed by radiation.
Operative Markings
A discussion between the plastic surgeon and breast surgeon determines what type of skin incision is ideal. Inferior outer lateral cancers are more appropriately treated with a Wise pattern incision. With this, the skin superficial to the cancer can be excised assuring a negative anterior margin without compromising cosmesis. Additionally, tissue including the pectoralis fascia in this region is taken to increase the likelihood of a posterior negative margin. Both breasts are marked while standing. If the cancer is palpable, the cancer is also outlined during the marking process.
Intraoperative Steps
Position the patient supine with arms out on an arm board at 90°.
The plastic surgeon can begin the contralateral mastopexy at the same time the breast surgeon operates on the breast with cancer.
Complete sentinel or complete axillary lymph node dissection first as appropriate through separate axillary incision.
Use assistant to place the nipple areola complex in full stretch. Use a 42-mm cookie cutter and a marking pen to mark the new size of the nipple areola complex centered over the nipple.
De-epithelialize around the region outside the cookie cutter marking defining the new nipple areola complex and in the region of the superior medial pedicle.
Perform the partial mastectomy by making full-thickness skin incisions down and including the pectoralis fascia in the region of the inferiorly based cancer. This may include just the midline inferior pole if the cancer is at the 5- to 7-o’clock position (and the skin incision design is a circumvertical design) or also the medial and lateral wing if the cancer is in the lower outer or lower inner quadrants (and the skin incision design is a Wise pattern) or a cancer located in the superior keyhole.
Mark the partial mastectomy for orientation for pathology margin assessment.
Ask pathology to weigh the specimen that has been removed. It is critical to leave approximately 10% extra volume on the side to undergo radiation.
Complete the elevation of the superomedial pedicle and approximation of the lateral and medial pillars in a standard mastopexy design fashion (completed by the plastic surgeon).
There is an example shown using this technique in Figure 5B. The specific details of the location and type of breast cancer are included in the caption of Figure 5. There is completion picture following radiotherapy.
Technique 5
Wise pattern extended superomedial pedicle 20 (Figure 6A) and hemi-wing pedicle (Figure 7):
Large to very large upper outer quadrant defects.
Steps for this are similar for technique 4. Exposure to the upper outer quadrant is identical to technique 4, and the breast surgeon can remove up to a quadrant worth of breast tissue in the upper outer quadrant. To fill this large defect, the plastic surgeon can use the inferior pole (making this the extended superomedial pedicle flap) to fill the defect. If more tissue is required, the plastic surgeon can use both the inferior pole and the lateral wing tissue (making this the hemi-wing pedicle flap) to fill in the much larger defect. There are 2 examples shown using this technique in Figure 6B and C. The specific details of the location and type of breast cancer are included in the caption of Figure 6. There is completion picture following radiotherapy.
A, Extended superior medial pedicle flap for upper larger outer quadrant tumors. B, A 59-year-old female with pathological stage T1N1M0 5 mm ER−, PR−, Her-2+ invasive ductal carcinoma at left breast at the 2-o’clock position. Total of 1057 g on the right and 827 g on the left removed using Wise pattern medial pedicle technique with axillary node dissection followed by radiation. C, A 70-year-old female with pathological stage T2N0MO ER+, PR+, Her2 invasive ductal carcinoma at left breast at the 1-o’clock position. She underwent Wise pattern medial pedicle technique followed by radiation.
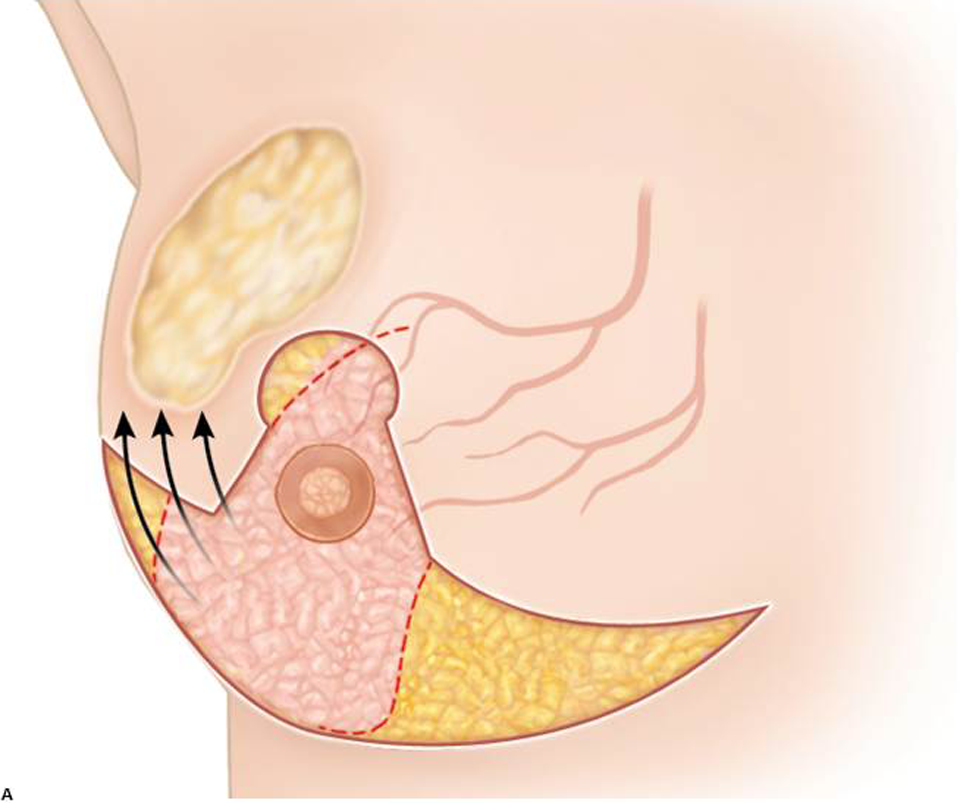
Illustration of incision for hemi-wing superior medial pedicle flap for very large upper outer quadrant tumors.
Discussion
The surgical goal in breast cancer treatment is primarily the safe removal of cancerous tissue with adequate margins while maintaining acceptable aesthetic breast form. Volume displacement oncoplastic surgery expands the role of breast conservation surgery by allowing greater amounts of breast tissue to be removed by employing the strengths of plastic surgery in maintaining and sometimes improving upon aesthetic breast form. Patients who would have only been considered for mastectomy now can have breast preservation surgery using oncoplastic techniques. Clough et al 14 divided volume displacement oncoplastic surgery into 2 subtypes, with the first involving smaller excisions of breast volume involving moderate undermining of breast tissue and local tissue rearrangement and the second involving removal of large volumes of breast tissue followed by the use of Wise pattern and circumvertical incision patterns and varying pedicle designs with the necessity of performing a symmetry operation on the contralateral breast. In a similar fashion, Silverstein et al 4,21 described oncoplastic surgeries adding hemi-batwing and batwing to facilitate the removal of central, upper outer, and inner quadrant lesions to the armamentarium of oncoplastic operations. While past papers have given us many reasonable options, the goal in this review was to provide no more than 5 surgical oncoplastic options that could address the oncologic resection in the vast majority of cancers regardless of their location in the breast, while using only incision patterns that minimize aesthetic deformity and visualization of incision lines on the breast. Each of the 5 techniques described earlier use traditional breast mastopexy and mammoplasty incision pattern designs commonly used in plastic surgery for typical cosmetic operations and have been very well accepted aesthetically based on cosmetic outcome assessments. All the techniques presented here should include a contralateral breast symmetry discussion with the patient, given that the nipple areola complex is often reduced in size and is moved to a different location with the exception being a small cancer removal using technique 1 doughnut mastopexy.
Oncologic Safety of Oncoplastic Surgery
The safety of oncoplastic breast conservation surgery has been well established by several studies. 2,3,22 -25 Local regional recurrence rates and survival rates have shown no difference between oncoplastic breast conservation surgery and standard lumpectomy. This should be of no surprise, given that oncoplastic surgery allows for larger lumpectomy specimens to be removed if necessary, which should remove more of the involved tissue than a standard lumpectomy allows.
Benefits of Oncoplastic Surgery Versus Partial Mastectomy
There are several advantages to oncoplastic surgery versus standard partial mastectomy. Oncologically, the primary advantage is the significantly lower positive margin rate found after an oncoplastic resection compared to a standard partial mastectomy. Positive margin rates have typically ranged from 19% to 40% for standard partial mastectomy. 2,26 Furthermore, only after employing the strengths of additional shaved margins after completion of a partial mastectomy are rates as low as 19% reported. 26 The ability to perform a large partial mastectomy thanks to the volume displacement plastic techniques in reconstructing the breast allows for low positive margin rates reported to be in 10% to 12% range in several large review studies. 2,3 This halving of positive margin rates when using oncoplastic surgical techniques is a major advantage to patients, especially when counseling them about options for breast conservation surgery. Furthermore, in the rarer subset of patients who have insidious extensions of tumor that imaging did not preoperatively identify (eg, invasive lobular carcinoma, ductal carcinoma in situ), the initial removal of a large section of breast tissue provides for the best chance for negative margins with one operation versus multiple re-excisions that would be needed when performing a traditional standard partial mastectomy. If there still are multiple focuses of positive margins even after a large oncoplastic resection, then a mastectomy is warranted and this quicker realization for the need for mastectomy, compared to multiple re-excision surgeries historically needed, using standard partial mastectomy techniques is another advantage for oncoplastic surgery. Other advantages to oncoplastic surgery involve the aesthetic advantages created when providing the patient with a breast lift or reduction using inherited plastic surgery incision markings. Patients with ptotic or symptomatically large breasts enjoy the aesthetic results that have historically been favored and reported in the plastic surgery literature. Furthermore, the obliteration of dead space secondary to the displacement and rearrangement of tissue decreases the theoretical likelihood of seroma formation and nipple retraction seen in standard partial mastectomy operations, especially in regions like the inferior pole that scar and sink in after radiation therapy is performed resulting in poor aesthetic outcomes known as “swan beak deformities.” 4 The lower positive margin rates and generally improved aesthetic outcomes found in oncoplastic surgery compared to standard partial mastectomy make it a more cost-effective option in the appropriate patient with breast cancer.
Benefits of Oncoplastic Surgery Versus Mastectomy
Breast conservation surgery and mastectomy as surgical treatment options have no difference in overall survival. 27,28 In breast conservation surgery, when comparing standard partial mastectomy, complete mastectomy does have an advantage in its lower locoregional recurrence disease rate. 27 However, when oncoplastic surgery has been compared to complete mastectomy, there has been no statistically significant difference in long-term locoregional recurrence. 29 Additionally, a complete mastectomy compared to oncoplastic surgery is an operation with higher short- and long-term potential morbidity in addition to psychological stress of losing a breast. 30 A mastectomy without reconstruction has the disadvantage of poor cosmesis and gross breast asymmetry that oncoplastic surgery clearly does not. With reconstruction, be it implant based or flap based, the inherent disadvantages of a foreign body with implant-based reconstruction or possible flap complications that include donor site morbidities when using autologous tissue reconstruction are avoided in oncoplastic surgery. Oncoplastic surgery does provide the aesthetic advantages that are inherent with breast lift and reduction surgeries, but direct comparisons using patient outcomes data against implant and flap reconstructions with regard to aesthetics have not been performed and are needed. While such research is needed, less surgery often requires less revision and a presumptive advantage with a 1-stage oncoplastic resection of tumor and reconstruction that typically has a patient returning home the same or the next day is that it does not require the longer postoperative recovery needed in implant and flap-based reconstructions. Postoperatively, implant based and flap based reconstructions require multiple days in the hospital and possibly multiple stages for revision surgery. In such, the patient with breast cancer can more reliably and efficiently move on from the surgical treatment phase and continue with any adjuvant medical or radiation treatment that is appropriate for them.
Disadvantages of Oncoplastic Surgery
There are few disadvantages to oncoplastic surgery. First, it often requires a contralateral symmetry operation if the patient wants to have a symmetric outcome to facilitate bra wearing and obtain a good to excellent aesthetic outcome. While this may be viewed by some as an advantage in that the breasts may have a final, better aesthetic appearance, most would view more surgery as not necessarily an advantage, especially when the primary goal is the treatment of cancer. Second, oncoplastic surgery still falls under the wing of breast conservation surgery and as such requires that at least whole breast radiation be given to the cancer-affected breast. Third, there have been reports of fat necrosis, especially after smaller tissue rearrangement oncoplastic surgeries described by Clough et al. 5 These were mostly in fatty and less glandular breasts. Using plastic surgery techniques with established pedicles and blood supplies (inferior, superior, and superomedial pedicles) with historical incision patterns described by the 5 techniques mentioned earlier, fat necrosis has not been a major problem with regard to screening mammography and clinically in general as noted in the plastic surgery breast mastopexy and reduction mammoplasty outcomes literature. Newer technology applications such as using indocyanine green with intraoperative angiograph to assess oncoplastic flap perfusion have further helped as adjunctive tools in addition to clinical judgment in assessing and resecting compromised tissue to avoid future fat necrosis during the oncoplastic operation. 31,32
Conclusion
These 5 large volume displacement oncoplastic surgical techniques allow the breast surgeon to have excellent access to most regions of the breast and the ability to excise large volumes of tissue with the advantage of excellent aesthetic closure. The use of judicious patient selection and education has improved oncological and aesthetic outcomes of breast cancer operations using oncoplastic techniques.
Footnotes
Authors’ Note
Dr. Chatterjee is a consultant for Allergan and Novadaq.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
