Abstract
Transformative paradigms to proactively mitigate fatigue through noninvasive neurostimulation, such as transcranial direct current stimulation (tDCS), have been shown to be more effective than existing interventions. The study aimed to evaluate the effectiveness of HD-tDCS (High-Definition) as a fatigue countermeasure on vigilance and perceived fatigue on longer bouts of mental fatigue. Thirty-two participants were assigned to active stimulation and sham groups, and they took part in a 2 hr fatigue protocol of 2-back visuospatial working-memory tests. To evaluate the effect of active stimulation, separate linear mixed-effect models with the group (stimulation/sham), phase (pre/post), and sex (male/female) were analyzed. The results showed the effectiveness of HD-tDCS in slowing fatigue-related declines across both cognitive and perceptual channels, with fatigue reduction being more effective in women. These observations could inform sex-based considerations to improve the operational readiness of tDCS-based countermeasure systems to help mitigate fatigue among emergency and military personnel.
Objectives
Fatigue among emergency response (ER) workers has been associated with 3-fold increases in injury and depression risks and a 4-fold increase in health and safety-compromising behaviors. In particular, mental fatigue impacts situation awareness, cognition, and decision-making through persistent global network changes (Lim et al., 2010) and decreased brain coordination (Karthikeyan et al., 2021). Existing ER-based interventions, for example, breaks, education, caffeine, are largely reactive, resource-taxing, and, in some cases, accompanied by substantial health side effects. Transformative paradigms to proactively mitigate fatigue through noninvasive neurostimulation, such as transcranial direct current stimulation (tDCS), have proven more effective than existing interventions in mitigating fatigue in clinical populations.
tDCS has also been shown to boost cognition under fatigue. Active tDCS at ~2 mA for ~30 min has been shown to enhance vigilance to a greater extent and for a much longer time (>6 hr) when compared to traditional countermeasures, such as caffeine (McIntire et al., 2017). Studies have also found that anodal tDCS at 1 mA for 10 min enhanced vigilance and improved cerebral hemodynamics compared to non-stimulation and a sham placebo (Karthikeyan & Mehta, 2020). In a similar vein, another study reported that anodal tDCS at 1 mA for 10 min on the dorsolateral prefrontal cortex (DLPFC) enhanced performance on a working memory task (Karthikeyan et al., 2021). Collectively, these results highlight that a short 10 min dose of active tDCS stimulation mitigates fatigue outcomes, which prevails for at least 30 min post-stimulation. These findings highlight the benefits of tDCS on attention networks across different task demands. However, it should be noted that these time-on-task studies were conducted for ~60 min. Moreover, whether greater dosages of tDCS (e.g., ~2 mA for ~20–30 min) focally stimulation using high-definition tDCS (HD-tDCS) on such working memory-based tasks for longer durations show similar benefits remains unclear.
Mental fatigue is a neurobiological state related to prolonged exposure to mentally taxing activities (Van Cutsem et al., 2022), and thus, perceptions of fatigue remain a strong assessment of fatigue. Beyond its operational effectiveness in enhancing cognition, tDCS has been shown to preserve mood scores during extended wakefulness (McIntire et al., 2017), an outcome not seen with caffeine-related fatigue countermeasures (Azevedo et al., 2016). However, recent studies with shorter-duration fatiguing tasks have reported that perception of fatigue and efforts remained unchanged despite the delay seen in performance declines with tDCS intervention, questioning its benefits in reducing perceived fatigue (Karthikeyan et al., 2021).
The goal of this study was to evaluate the effectiveness of HD-tDCS as a fatigue countermeasure on vigilance and perceived fatigue on longer bouts of mental fatigue. The main hypothesis is that 20 min of ~2 mA HD-tDCS applied to the DLPFC would reduce fatigue-related declines in vigilance, collected using the Psychomotor Vigilance Task (PVT) after a 120 min working memory task. The Profile of Moods States (POMS) (Shahid et al., 2012) was utilized to assess mood pre/post fatiguing test, and perceptions of fatigue, motivation, and effort were collected intermittently during the fatiguing task. It was hypothesized that while the POMS scores will be preserved with stimulation, perceptions collected during the fatiguing tasks will not be influenced.
Approach
Thirty-two participants (16 F) with a mean age of 25 (6) years and mean sleep quality of 7.3 (1.4) out of 10, were recruited in a single-blind experiment and were randomly assigned to active stimulation and sham groups. Participants were provided training on the study tasks to minimize any learning effects during the experiment. After completing the demographic survey and POMS mood scores, participants were instrumented with the HD-tDCS electrodes (Soterix), with the anode at F3 and the four cathodes at F1, F5, FC3, and AF3 that surround F3.
Once completed, participants began a fatigue protocol (Figure 1) of twelve 10 min blocks of 2-back visuospatial working memory tests (Karthikeyan et al., 2021). Participants completed a 4-point questionnaire to intermittently assess their current levels of motivation, fatigue, effort, and mental demand. At the start of the third block, participants received either the active stimulation with HD-tDCS lasting 20 min with 2 mA amplitude and a 30 s ramp up and ramp down at the start and end of the stimulation duration, or sham stimulation with a 30 s ramp up followed by an immediate ramp down, to mimic the tDCS sensation (Karthikeyan et al., 2021). Before and after the fatigue protocol, participants completed a 10 min PVT task and 4-point questionnaire.
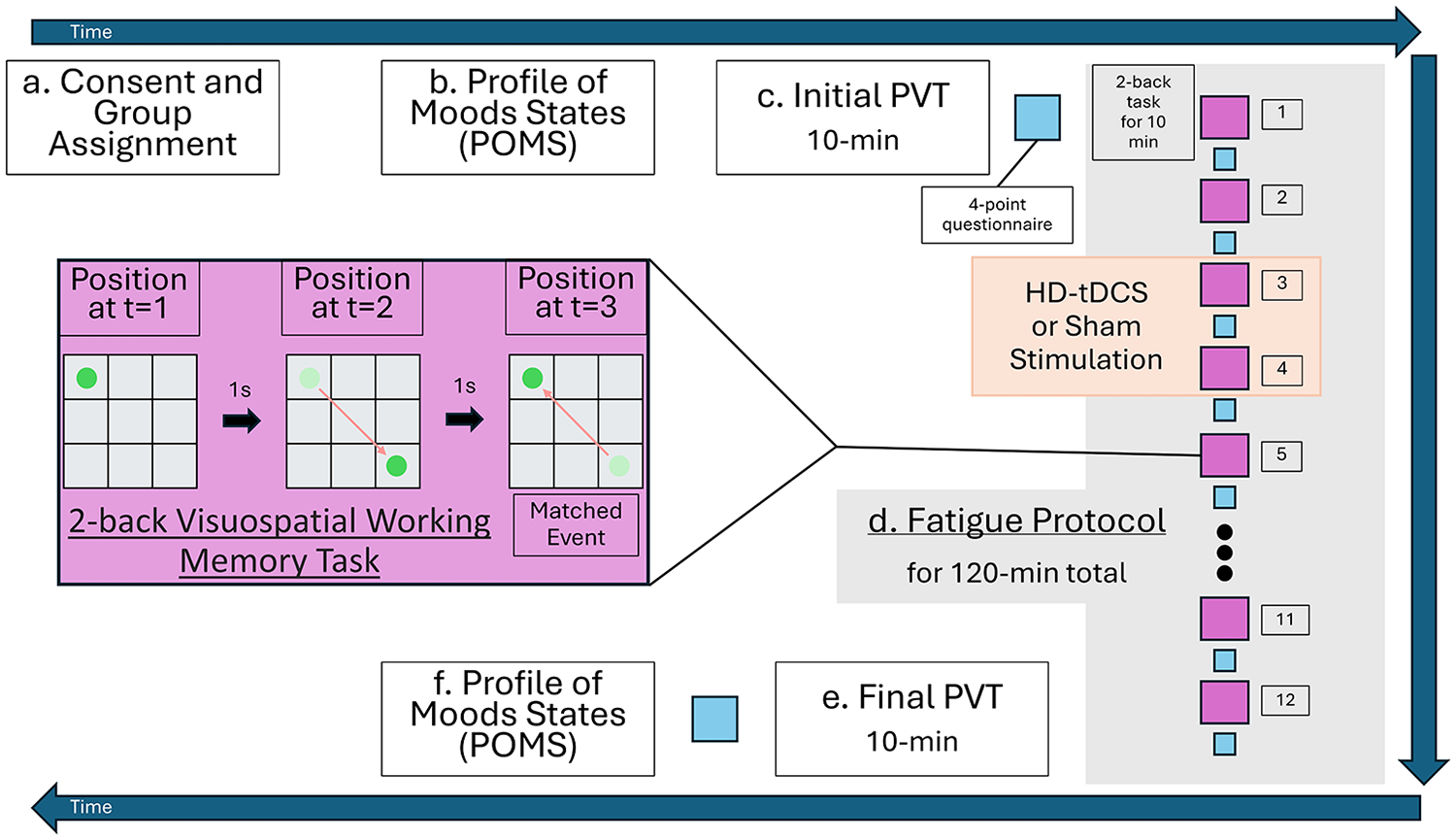
Experimental protocol moving through time in a clockwise direction starting at the top left and ending at the bottom left.
To evaluate the effect of active stimulation, separate linear mixed-effect models (LMMs) with the group (stimulation vs. sham), phase (pre vs. post), and sex (male vs. female), with participants as the random variable, were performed on the reaction times and lapse counts from the PVT, POMS scores, and perceived fatigue, motivation, and effort.
Findings
The stimulation group exhibited faster RTs (294.95 [31.41] ms) than the control group (317.39 [29.6] ms; p = .016) (Figure 2). Additionally, participants exhibited poorer PVT performance (response times [RT] and lapse counts) in the post-fatiguing tasks (both p’s < .001), and a phase x sex interaction found that women exhibited greater PVT lapses than men, however, this was only found post-fatigue (p = .05). A marginal group × phase × sex interaction (p = 0.06; Figure 2) was found on RT, where the poorer performance post fatigue in women was only observed in the sham group, while the stimulation group’s performance remained comparable with that of the men.
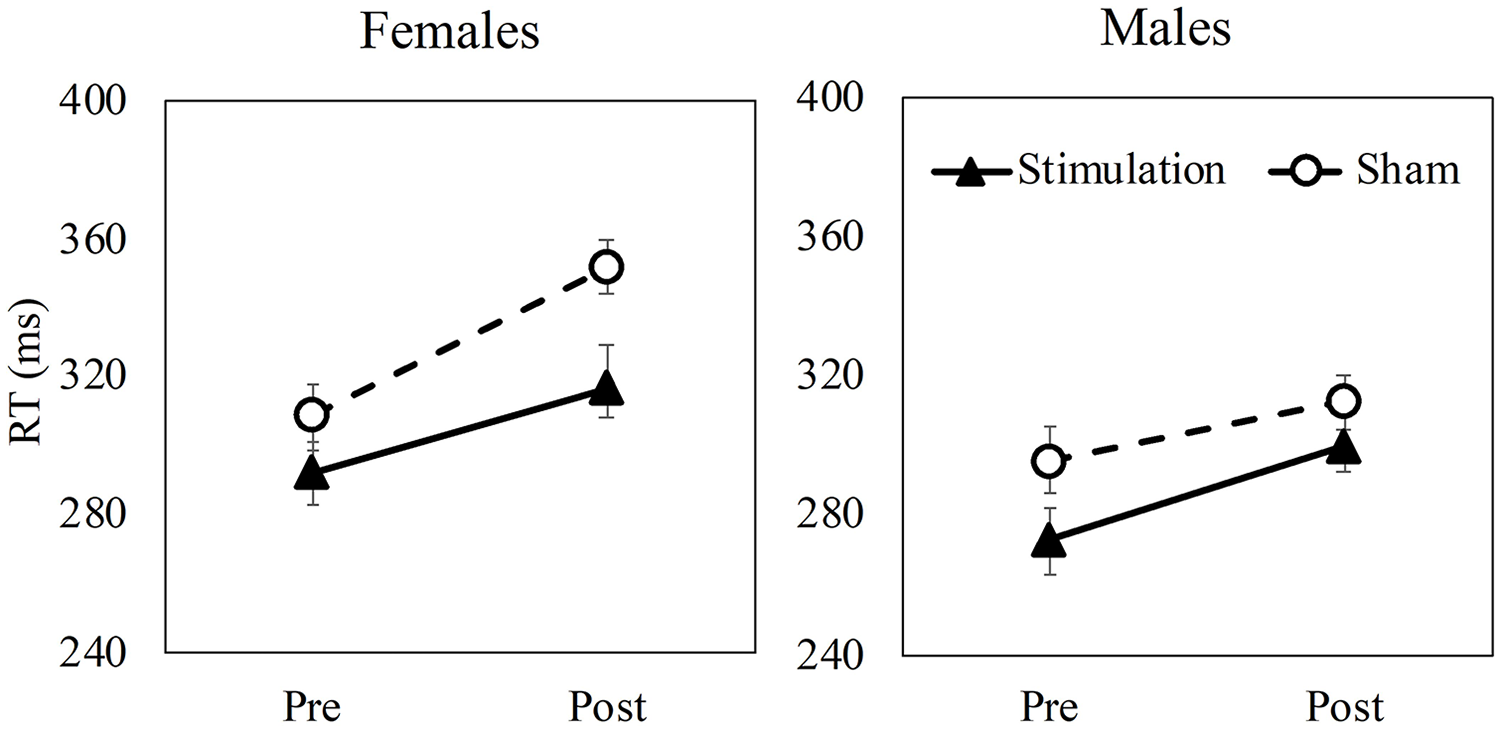
A significant three-way interaction effect of group (stimulation vs. sham), phase (pre vs. post), and sex (male vs. female) on PVT response times (RT). Error bars denote standard error.
Participants reported higher perceptions of fatigue, effort, and workload and lower motivation over time (all p’s < 0.001) (Figure 3). While the main effect of group was not found significant across these surveys (all p’s > 0.54), women reported higher perceived fatigue than men (p = .034). Additionally, a three-way interaction effect of group × phase × sex (p = .017) was observed. Among women, the stimulation group reported lower perceived fatigue than the sham group in the later fatigue phases (Figure 3), while no such group × phase effects were seen among men. Finally, POMS mood scores deteriorated post-fatigue (p < .001), however, no group or sex effects nor their interactions were found to influence POMS scores (all p’s > .168).
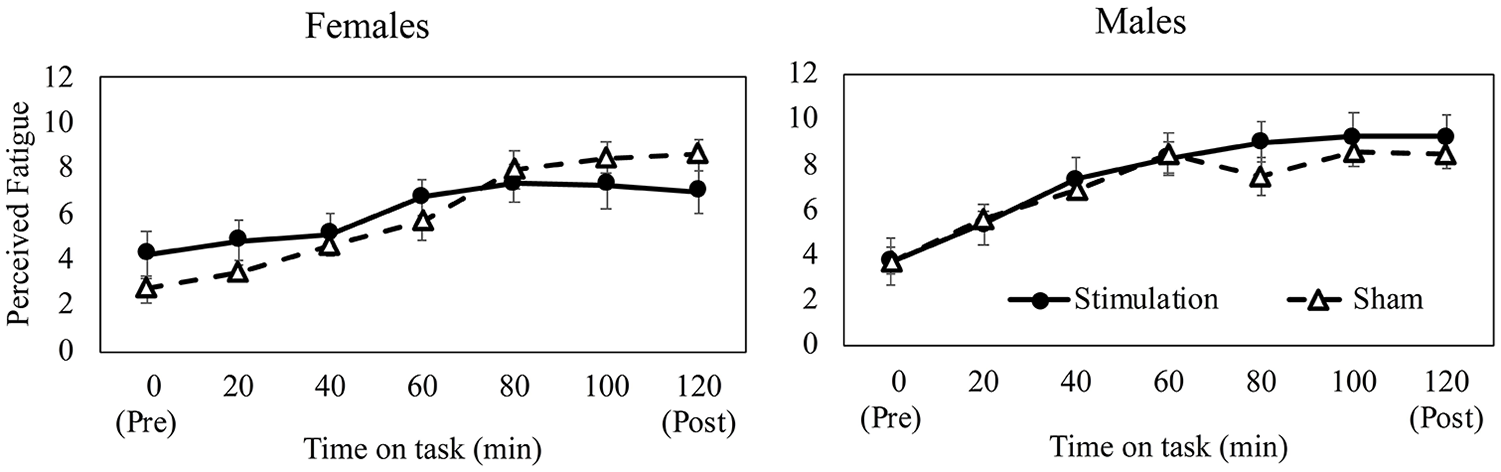
A significant three-way interaction effect of group (stimulation vs. sham), phase (every 20 min during time on task), and sex (male vs. female) on perceived fatigue. Error bars denote standard error.
Takeaways
Our study identifies two key takeaways. First, effective fatigue detection and countermeasure systems for performance in safety-critical areas like emergency response and defense sectors require an in-depth examination of their effectiveness on both the human cognitive and perceptual systems. Previous studies (Karthikeyan & Mehta, 2020; Karthikeyan et al., 2021) demonstrated that working memory, a key executive function for decision-making and situation awareness, improved with 1 × 1 tDCS, with no changes in perceptual channels. In contrast, the present work highlights the effectiveness of HD-tDCS, which offers a more focal neural stimulation, in slowing fatigue-related declines across both cognitive and perceptual channels for a prolonged working memory time-on-task.
Second, while the stimulation group exhibited faster response times than the sham group, fatigue reduction with HD-tDCS was more effective in women, while the sham group exhibited faster declines in RT-based performance. Indeed, the inherent sex-specific differences in PVT performance were leveled with stimulation. This cognitive augmentation in women was also accompanied by lower perceptions of fatigue. In contrast, perceived fatigue remained comparable between groups among men. It should be noted that beyond lower perceived fatigue and improved PVT performance, perceived motivation, effort, or POMS scores remained unchanged, thereby implicating distinct HD-tDCS gains through fatigue-specific neural pathways.
Future studies that incorporate neuroimaging and other bio-behavioral assessments to assess specific pathways influenced by focal HD-tDCS during a variety of fatiguing protocols (e.g., physical, sleep, mental) are needed. These observations could improve mechanistic understanding and operational readiness of tDCS-based countermeasure systems to help detect and predict fatigue among first responders and military personnel.
Footnotes
Acknowledgements
The authors thank the lab members of NeuroErgonomics Lab at Texas A&M University for collecting the data for this study.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
