Abstract
Background:
This study aimed to mimic the changes from Charcot neuropathic arthropathy in humans by examining the effects of exposing diet-induced obese (DIO) mice to neurotrauma through a regimented running protocol.
Methods:
Forty-eight male wild-type C57BL/6J mice were obtained at age 6 weeks and separated into 2 groups for diet assignment. After a 1-week acclimation period, half of the mice consumed a high-fat diet (60% fat by kcal) ad libitum to facilitate neuropathic diet-induced obesity whereas the other half were control mice and consumed an age-matched standard low-fat control diet (10% fat by kcal). At age 12 weeks, half of the animals from each group were subjected to a high-intensity inclined treadmill running protocol, which has been previously demonstrated to induce neurotrauma. Sensory testing and radiographic analyses were periodically performed. Histopathologic analyses were performed post killing.
Results:
DIO mice had significantly higher bodyweights, higher body fat percentages, and lower bone mineral density than wildtype control mice that were fed a normal diet throughout the experiment (P < .001 for each). DIO mice displayed significantly reduced sensory function in week 1 (P = .005) and this worsened over time, requiring 20.6% more force for paw withdrawal by week 10 (P < .001). DIO mice that ran demonstrated greater midfoot subluxation and tarsal instability over all time points compared with normal-diet mice that ran (P < .001). Histopathologic analyses revealed that DIO mice that ran demonstrated significant changes compared with controls that ran (P < .001 for each parameter).
Conclusion:
Changes akin to the earliest changes observed in or before joint destruction identified in diabetic Charcot neuropathic arthropathy in humans were observed.
Clinical Relevance:
There is currently no standard of treatment for patients with Charcot neuropathic arthropathy. This study establishes a protocol for an animal model that can be used to study and compare interventions to treat this disease.
Introduction
Charcot neuroarthropathy (CNA) is a debilitating, neurodegenerative arthropathy that predominantly affects the lower leg in humans. 21 The development of CNA is dependent on a combination of neuropathy and trauma, yet the genesis and disease progression are poorly understood. Although neuropathy has many etiologies, in the United States, diabetic neuropathy is a common instigating factor in CNA. Furthermore, the morbidity and mortality associated with the disease is comparable to some malignancies with a 5-year mortality as high as 50%.7,9,18,21,35,40 It is hypothesized that a loss of peripheral sensation combined with repetitive microtrauma activates pro-inflammatory cytokines, thus promoting osteoclastogenesis with eventual bone and joint destruction.18,21,35,40 Other theories for Charcot pathophysiology involve local increased blood flow, increasing compartment pressure, and monocytes in the joint space, causing osteoclast enrichment and bone resorption.12-20
Current treatments for CNA include immobilization, bracing, and surgical intervention.9,14,35 With an average 12.7-year survival after the first diagnosis, a 44% 3-year postoperative mortality rate, and 10% of patients requiring a major amputation following their initial surgery, it is imperative that models to study and evaluate CNA be developed.2,21,33
Although previous studies have described animal models for diabetic neuropathy and musculoskeletal degeneration using treadmill exercise, none have combined the 2 factors shown to produce CNA. However, a recently published pilot study by Stauch et al exposed a murine, neuropathic model with microtrauma and produced Charcot-like neuroarthropathic changes mimicking humans.3,4,10,12,20,22,26,32,34
The purpose of this study was to further investigate a mouse model for CNA through a neurotraumatic mechanism. This is the first multivariable designed study used to evaluate an animal model for CNA and expands on previous work in terms of cohort size and controls. The present work hypothesizes that mice with peripheral neuropathy exposed to a regimented running protocol will experience pathophysiologic and radiographic changes similar to CNA in humans.17,26,27,36,37,39
Methods
Animal Model
This study was approved by the Institutional Animal Care and Use Committee (protocol no. 01100). Forty-eight 6-week-old male C57BL/6J mice (Jackson Labs, Bar Harbor, ME) were used, with 12 per cohort. The cohorts consisted of the following 4 groups: (1) diet-induced obese mice (DIO), known to demonstrate peripheral neuropathy; (2) diet-induced obese mice that underwent a running protocol (DIO+R); (3) wild-type control mice fed a standard diet (WTC); and (4) wild-type control mice fed a standard diet that underwent a running protocol (WTC+R).
DIO and DIO+R mice were fed a high-fat that contained 60% kcal from fat starting at the age of 6 weeks to facilitate diet-induced obesity. The WTC and WTC+R mice were fed a standard control diet containing 10% kcal from fat. Diets (respectively, D12492 and D12450B, Research Diets Inc, New Brunswick, NJ) were maintained ad libitum with access to water throughout the entirety of the study. Three mice were housed in each cage with a 12-hour light-dark cycle. Veterinary staff monitored the animals throughout the study.
Exercise Protocol
Following 6 weeks of being fed their respective diets (12 weeks old), the DIO+R and WTC+R mice began a 1-week acclimation to an inclined treadmill running protocol designed to induce hind-paw microtrauma. The rodent treadmill (Exer 3/6, Columbus Instruments, Columbus, OH) was set to a 10-degree incline to increase the force placed on the hind paws. During this acclimation period, the mice began running for 5 minutes, and then 5 minutes were added each following day, amounting to 25 minutes of running by the end of the week. The protocol continued with treadmill running for 25 minutes per day 4 days each week for 10 weeks. The speed was set at a constant 5 m/min for 5 minutes, followed by the incorporation of a 0.017 m/s² acceleration providing an increase in speed of 1 m/min each minute up to a maximum speed of 25 m/min.16,28
The mice were observed over the duration of each running period. If mice ceased running at any point, a soft pipette nudge was applied to the hindquarters. 34
Body Composition
The bodyweights, body-fat percentage (BFP), and bone mineral density (BMD) were recorded on nonrunning days. Bodyweights were obtained using a standard precision balance and were recorded on weeks 1, 3, 5, 7, and 10 of the running protocol. Body fat percentage and bone mineral density were obtained for each mouse using an UltraFocusDXA dual-energy X-ray absorptiometry (DEXA) scanner (Faxitron, Tuscon, AZ). BFP and BMD were obtained at weeks 1, 5, and 10 of the running protocol while the mice were under anesthesia using isoflurane.
Sensory Function
Sensory function testing was performed before running at weeks 1, 3, 5, 7, and 10. Baseline measurements were obtained at week 1 before the start of the running protocol. Two tests were performed to evaluate the sensory function: electronic digital von Frey filament testing and hot plate testing. von Frey filament testing employed a 3-g force to the paw to inflict a nociceptive withdrawal response.11,20 This was performed on both hind paws twice with approximately 10 minutes between each test.
Radiographic Analysis
Plain radiographic images were obtained in the lateral plane using DEXA imaging at weeks 1, 5, and 10 of the running protocol. Radiographs were assessed by a single board-certified foot and ankle orthopaedic surgeon (M.C.A.) who was masked to the groups to evaluate the collapse of the hind-paw longitudinal arch and mid-paw joint subluxation using Meary angle, calcaneal–fifth metatarsal (C5MT) angle, as well as the percent uncoverage of cuneiform over the first metatarsal (%T/MT).8,38 The %T/MT was used as a measurement of mid-paw subluxation and was performed by measuring the height of the medial cuneiform and the length between the dorsal surface of the second metatarsal and the second cuneiform. The %T/MT was the percentage of the second cuneiform that was above the joint space.
Endpoints and Histopathology
Following the 10-week running protocol, the animals were humanely killed as stated in our approved protocol using CO2 euthanasia. Subsequently, the proximal tibia to distal metatarsals of each animal were harvested with care to avoid disruption of bones or joints for histologic analysis. Histologic specimen preparation consisted of immediately fixing specimens in 10% formalin for 72 hours, followed by decalcification in Formical (10% formic acid in formaldehyde for 10 days, per established mouse protocol). Tissues were processed and embedded in paraffin for 5-µm slices in the sagittal plane. The specimens were stained with hematoxylin-eosin.
The histopathologic analysis was performed by a masked board-certified, experienced musculoskeletal pathologist (J.F.S.) and included observations of mid-paw pathology of the bone and soft tissues similar to previous work. 34 The following measures were scored: (1) perichondral ossification of the articular surface; (2) endochondral ossification of the subchondral location, interpreted as perichondral endochondral ossification causing osteosclerosis (the earliest changes observed in human CNA); (3) distance of the articular surface to the closest marrow space measured in microscopic arrow lengths; and (4) cortical endochondral ossification. Scoring ranges included the following: 0 = healthy/normal, 1 = minor changes within normal range or below level of detection, 2 = pathologic (moderate) changes, 3 = pathologic (severe) changes. Standardized microscopy arrow lengths were used for articular to marrow distance (measuring the degree of subchondral osteosclerosis as occurs in early phase Charcot): less than or equal to 1 arrow length = 1, more than 1 up to 2 arrow lengths = 2, and 3 = greater than 2 arrow lengths. The soft tissue analysis of nerves and small capillary vessels included the involvement by arteriolosclerosis, and the severity of intraneural vacuolization and intraneural myxoid change (edema) as observed in diabetic neuropathy. This involvement was graded on a 0-2 scale, with a higher score correlating to the severity of the pathologic change; 0 = healthy, 1 = normal/minimal or focal changes, 2 = pathologic (abnormal).
Statistics
Repeated measures analysis of covariance was used for longitudinally collected data including body composition, sensory function testing, and radiographic analyses. For each outcome, a model was fit that could estimate within-group change over time as well as between-group differences. Each model contained factors for diet, exercise, diet by exercise interaction, and a nested effect for the week within the interaction term. This parameterization also allowed for the estimation of overall group comparisons across time points. Results were reported in terms of model-adjusted means and 95% CIs, and a Bonferroni adjustment was applied for multiple comparisons when results were reported for the time points separately. Wilcoxon rank-sum tests were used for the categorical histopathology ratings measured after killing, and median scores were generated. Significance was defined as P < .05, and statistical analyses were performed using SAS statistical software, version 9.4 (SAS Institute, Inc, Cary, NC).
Results
Mouse Neuroarthropathy Regime
All 48 mice completed the 16-week protocol, including the 10-week running protocol, without complications. All mice, with n = 12 per group, were considered for all histopathologic and statistical analyses.
Body Composition Analyses
Bodyweight changes
The bodyweight results indicated a significant increase in bodyweight for all 4 groups (Table 1). The average increases for the nonrun groups were not significantly larger than the run groups for both the high-fat diet and the normal diet. However, the DIO+R group had significantly higher bodyweights at week 3 when compared to the DIO group (P = .02). Average bodyweight increases for the high-fat vs normal diets were greater for both the nonrun group (P < .001) and the run group (P < .001). Both the DIO and DIO+R groups had significantly higher bodyweights than the WTC and WTC+R, respectively, over all time points (P < .001 for both) (Figure 1A).
Changes in Each Outcome Measure From Beginning of Running Protocol (Week 1) to the End of the Protocol (Week 10) With Statistical Significance in Adjacent Columns.
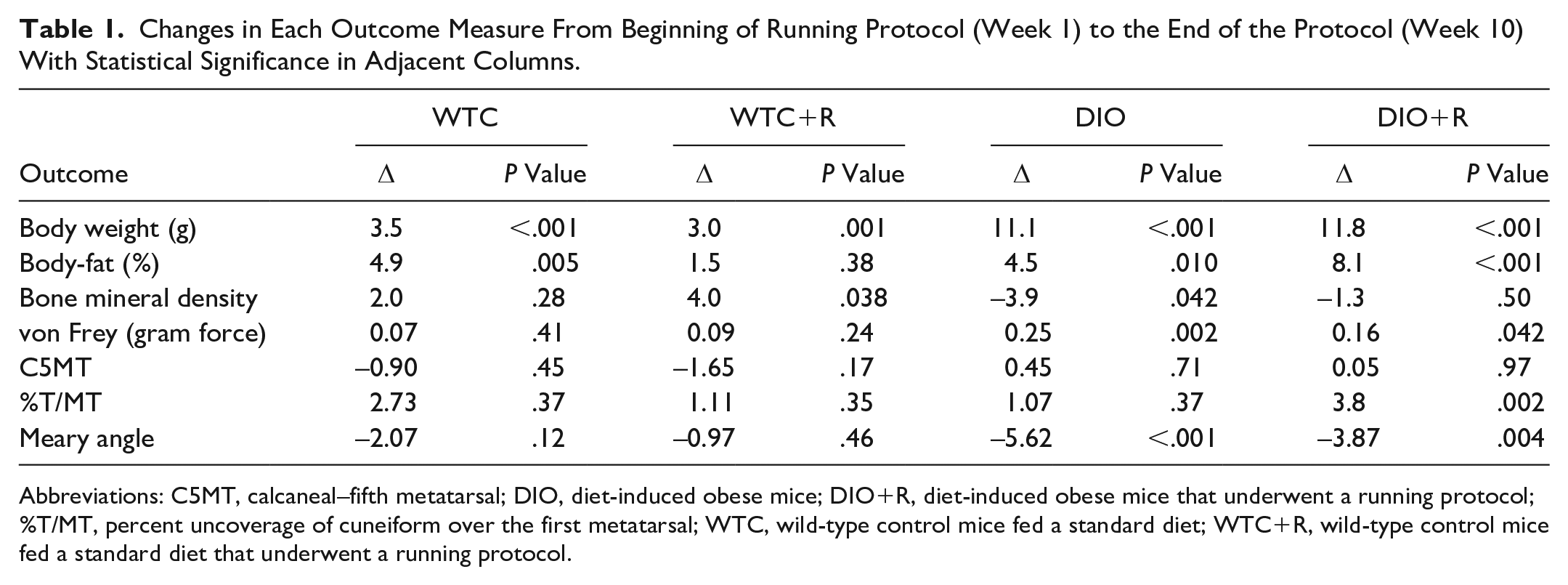
Abbreviations: C5MT, calcaneal–fifth metatarsal; DIO, diet-induced obese mice; DIO+R, diet-induced obese mice that underwent a running protocol; %T/MT, percent uncoverage of cuneiform over the first metatarsal; WTC, wild-type control mice fed a standard diet; WTC+R, wild-type control mice fed a standard diet that underwent a running protocol.
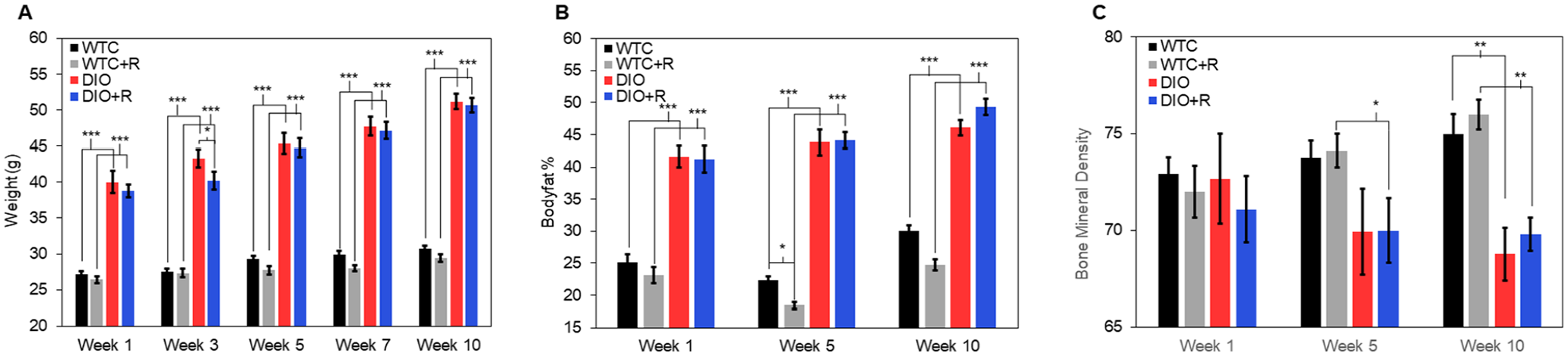
Comparison of (A) average weight, (B) bodyfat percentage, and (C) bone mineral density throughout the experiment (n = 12 per group). Error bars indicate standard error of the mean.
Body-fat percentage changes
The results from BFP measurements demonstrate significant increases for all but the WTC+R group (Table 1). Significant differences in total BFP change between the WTC+R and the DIO+R (P = .008) were detected. However, significant differences were not detected in average increases between nonrun vs run for either the high-fat or the normal diet. Additionally, the nonrun group did not show a significant difference between high fat vs normal diets (P = .86). Both the DIO and DIO+R had significantly higher BFPs than WTC and WTC+R over all time points (P < .001 for both) (Figure 1B).
Bone mineral density changes
The BMD measurements revealed a significant decrease in the DIO group (P = .042) and a significant increase in the WTC+R group (P = .038) (Table 1). Significant and marginally significant differences were detected in DIO vs WTC (P = .029) and DIO+R vs WTC+R (P = .052). At week 5, the DIO+R group had significantly lower BMD than the WTC+R group (P = .04). At week 10, the DIO and DIO+R groups both had significantly lower BMD at week 10, when compared to the WTC+R groups (P < .005 for both).
Sensory function changes
A significant increase in the force required for a response during von Frey testing was observed for the DIO and DIO+R groups over time (P = .002 and P = .042, respectively) (Table 1). The changes over the course of the running protocol did not produce significant differences between nonrun vs run groups or high-fat vs standard diets. However, the DIO+R group required significantly more force than the WTC+R at all weeks (week 1: P < .001, weeks 5 and 10: P < .001). The DIO group required significantly more force than the WTC at weeks 5 and 10 (P < .001). (Figure 2).
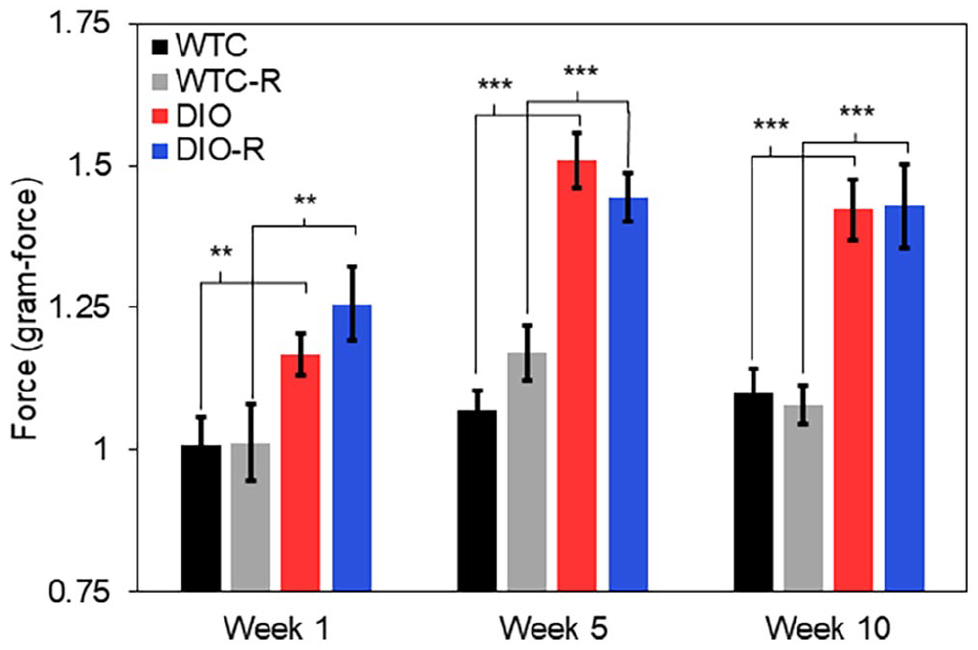
Comparison of the force required to elicit a positive response in mouse hind paw using von Frey filament over the 10-week time period (n = 12 per group). Error bars indicate standard error of the mean.
Radiographic changes
The midfoot subluxation, described by the %T/MT measurements, was significantly greater for the DIO group when compared to WTC over all time points (week 1: P = .001, week 5: P = .004, week 10: P = .030). The DIO+R group also had significantly greater subluxation than the WTC+R group at all time points (P < .0001).
Meary angle and C5MT angles did not demonstrate any clear trends in the data. Significant differences detected within these measurements are in Figure 3.
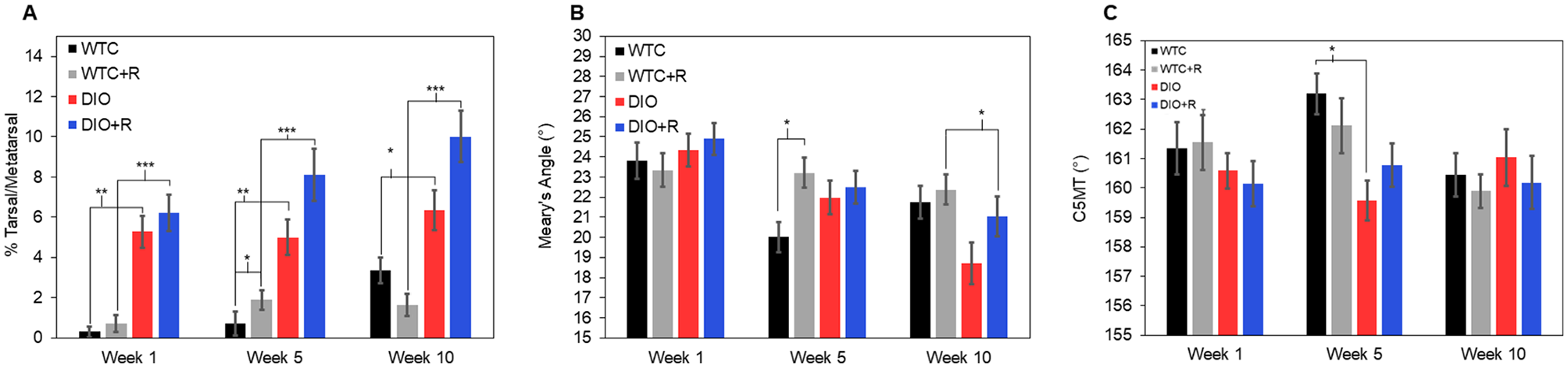
Comparisons of (A) subluxation percentage of the tarsal over the metatarsal bones (B) Meary angle, and (C) calcaneus–fifth metatarsal angle between each group over the duration of the study (n = 12 per group). Error bars indicate standard error of the mean.
Histopathology analysis
The median scores for all regions of analysis are shown in Table 2 and Figure 4.
Median Values for Masked Histopathologic Grading for Each Group.
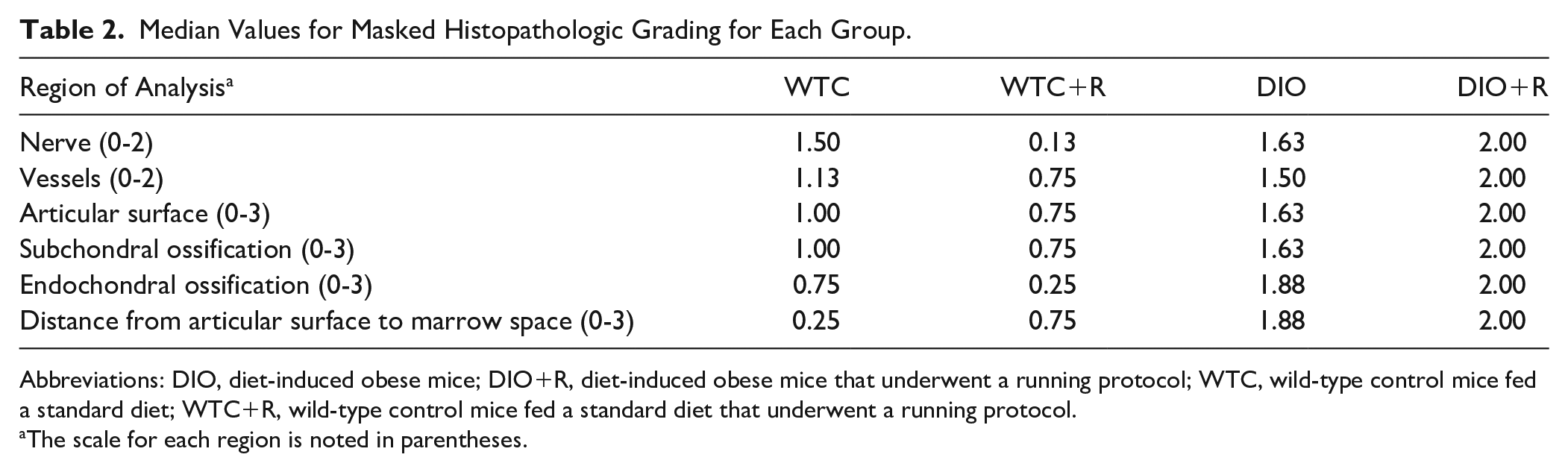
Abbreviations: DIO, diet-induced obese mice; DIO+R, diet-induced obese mice that underwent a running protocol; WTC, wild-type control mice fed a standard diet; WTC+R, wild-type control mice fed a standard diet that underwent a running protocol.
The scale for each region is noted in parentheses.
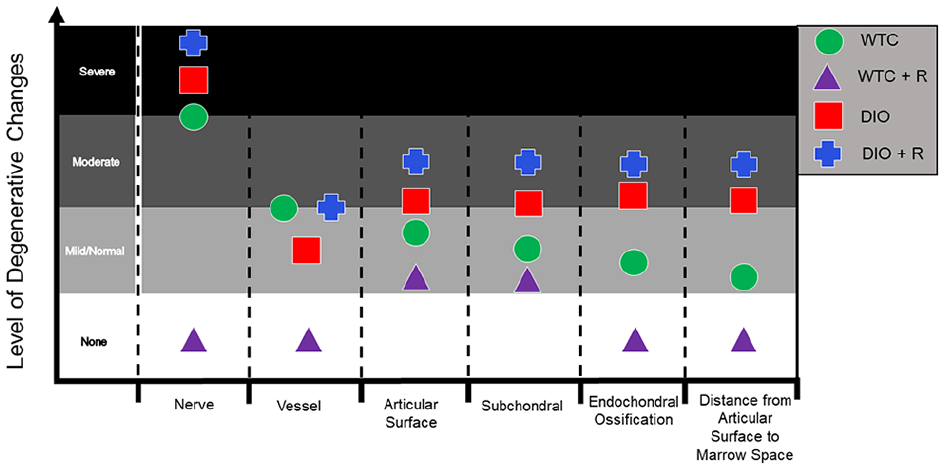
Graphic representation of histopathologic changes of Charcot neuropathic arthropathy model (n = 12 per group). This includes assessments of nerve intraneural myxoid (edema) change and fibrosis (column 1), small vessel arteriolosclerosis (column 2), articular peculiar perichondral ossification (column 3), subchondral osteosclerosis (microfracture endochondral ossification) (column 4), increased distance from the articular surface to closest marrow space due to osteosclerosis (column 6), and cortex endochondral ossification remodeling (column 5). The level of degenerative changes on this plot correlates to the scores for the respective grading scale for each measurement.
Articular surface perichondral ossification
Results indicated significant degenerative, peculiar perichondral concentric ossification in the articular surface of the DIO+R (P < .001) and the WTC (P = .003) groups when compared with the WTC+NR group (Supplemental Figure 1). No significant differences were detected between the DIO group and the DIO+R group, yet the median rating was higher in the former when compared to the WTC group (P = .015). The mean values are plotted with respect to their assigned level of degenerative changes in Figure 4.
Subchondral evidence for microfracture endochondral ossification
The subchondral space of the DIO+R and the WTC had significantly greater degenerative change ratings when compared to the WTC+R group (P < .001 and P = .013, respectively) (Supplemental Figure 1). Additionally, the median rating was significantly higher in the DIO group when compared to the WTC group (P = .005) (Figure 4)
Subchondral Bone Osteosclerosis Causing Bone Thickening
The median rating of the distance from the articular surface to the closest marrow space (abnormal 2 and 3) was significantly lower in the WTC+R group when compared to the WTC and the DIO+R groups (P = .041 and P < .001, respectively) (Supplemental Figure 1). Furthermore, the median rating was significantly higher in the DIO group when compared to the WTC group (P < .001).
Cortical bone remodeling
Cortical endochondral ossification rating (abnormal 2 and 3) was significantly lower in the WTC+R group when compared to the DIO+R group (P < .001) (Supplemental Figure 1). Additionally, the DIO group had significantly greater changes compared to the WTC group (P < .001).
Neuropathic changes
Comparisons of neuropathic change revealed significantly healthier nerves in the WTC when compared to the WTC+R group (P = .005) and the DIO group (P < .001) (Supplemental Figure 1). The DIO+R group displayed the highest median grade among all groups for neuropathic change (Figure 4).
Vasculopathic changes
Comparisons of vasculopathic change indicated that the vessels of the WTC group were significantly healthier than both the WTC+R group (P < .001) and the DIO group (P < .001) (Supplemental Figure 1). The DIO+R group had the highest median vessel grades indicating the highest degree of vasculopathic changes.
Discussion
This study is the first to evaluate a CNA mouse model with a large cohort for extended treadmill protocol running while controlling for both running and diet. This model confirms the combination of neuropathy in the C57BL/6J diet–induced obese mouse combined with microtrauma from treadmill running produces neuroarthropathic changes similar to those seen in humans.
Leading theories of CNA development suggest a combination of neuropathy and trauma are required to develop the condition, which is consistent with our findings.1,19 This is supported by the observation that DIO mice developed neuropathy, but only DIO+R mice had significant radiologic and pathologic changes; thus, the combination of neuropathy and trauma is essential to CNA development and progression in a murine model.
This model builds on a previous pilot model by Stauch et al 34 that investigated a viable animal model for idiopathic neuroarthropathy. The present study builds on the previous model by including the appropriate controls to establish the combined effect of obesity and microtrauma in the described mouse model for CNA. Additional improvements include a higher number of subjects to support statistical power, quantifiable sensory data, and increased study duration to observe more dramatic effects. This study reveals similar but more developed changes surrounding all outcome measures with this increased running duration, supporting a proportional agreement, and the reproducibility of the model. These changes, however, still reflect the early changes before the complete joint fragmentation of CNA.
Further supporting the neurotraumatic theory, only the DIO+R group revealed significant degeneration through neurologic, histopathologic, and radiographic analyses. Both DIO and DIO+R had increased weight gain and body fat percentage with decreased bone mineral density; although studies suggest obesity to be protective from osteoporosis, newer data demonstrate that increased adipose tissue causes harm to bony structures.5,6,15 Specifically in CNA, bone mineral density is decreased, which increases the susceptibility for fracture and dislocation.30,31
Various studies have described von Frey filament testing as a method of quantifying nociception with clinical implications.11,24 The DIO and DIO+R mice in this study demonstrated increased force for nociception on von Frey testing, indicating neuropathic changes in both groups and were significant throughout the study.
Furthermore, DIO and DIO+R groups both experienced a decrease in BMD over time, suggesting that repeatedly loading the hindfoot through a regimented exercise protocol will not result in BMD improvements for these mice. This finding seems to be due to the combination of diet and exercise, as exercise alone has been found to increase bone density in previous mouse models. 25 Furthermore, although the DIO+R mice ran 4 times per week, their body weight and body fat percentages were comparable to the DIO mice that did not run throughout the entirety of the study.
Charcot arthropathy results in characteristic radiologic collapse of the longitudinal arch, resulting in changes in Meary angle, C5MT, and %T/MT; DIO+R mice had significantly greater %T/MT than DIO mice at week 10 (Figure 5). Meary angle and C5MT did not have significant changes in the DIO+R mice.1,29,38 We theorize that the characteristic changes to Meary angle in Charcot foot did not occur because the mice ran on an angled treadmill that increased stress on the hindfoot, which may have emphasized hindfoot joint subluxation but not the arch. Furthermore, the radiographs were nonweightbearing, limiting the ability to observe the collapse of the arch. Moreover, the lack of significant differences in these measurements, used for clinical assessment of bipedal humans, may be attributed to the fundamental differences in locomotion in the quadrupedal mice, causing an inherent difference in stresses within their hind paws.
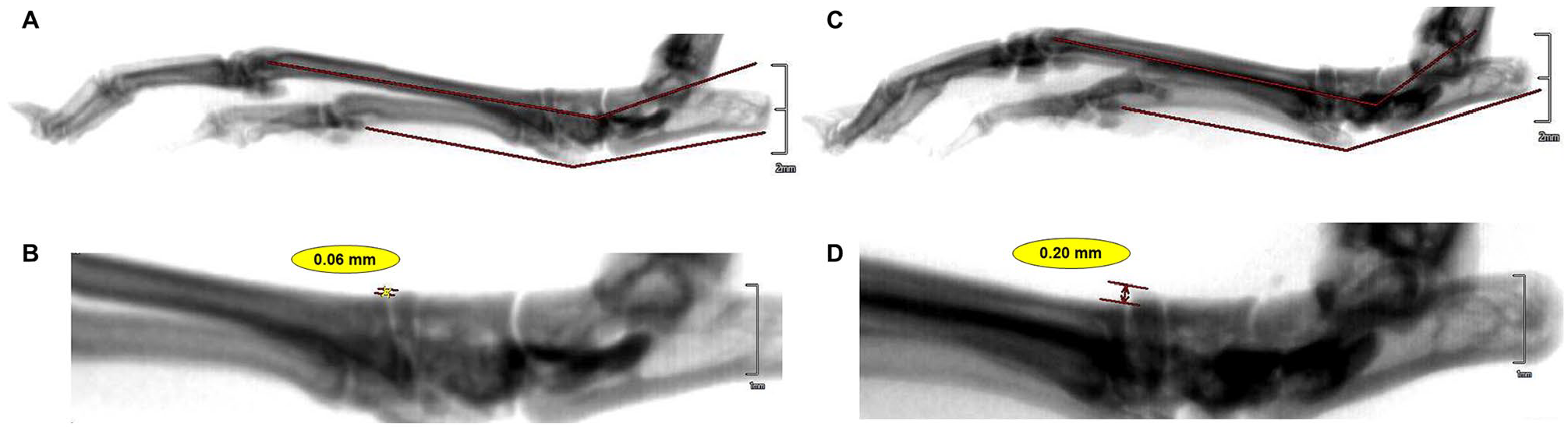
Representative radiographic images in the sagittal plane of (A) diet-induced obese runner (DIO+R) with Meary angle and calcaneal–fifth metatarsal angle and (B) midfoot subluxation. As a comparison, wildtype control nonrunner (WTC) mouse (C) Meary angle and calcaneal–fifth metatarsal angles, and (D) measured midfoot subluxation.
Pathologic grading systems, correlating to Eichenholtz staging, have also been developed to predict outcomes based on joint soft tissue and bone etiologies. 23 For this study, pathology was graded similar to the FEMASK grading methodology, as previously described by King et al 23 in humans. The histopathologic changes indicated the earliest findings of CNA in the DIO+R mice. The first osteosclerotic phase of proposed repeated microfracture and peculiar (concentric cortical-like, and possibly unstable) endochondral ossification prior to the secondary phase of complete joint destruction was observed. These changes involving peculiar perichondral ossification and subchondral and cortical endochondral ossification (microcallus-like) osteosclerosis may be early reparative changes after microfracture that cannot be sustained with persistent microtrauma and repeated microfracture. In human patients, this would ultimately lead to final joint subluxation and destruction.
Limitations of this study include the limited duration of observation and a lack of available HbA1c or blood glucose levels in the mice. An additional limitation regarding studying the effect of running duration with respect to number of weeks was the lack of gene expression data to observe worsening at the molecular level. Furthermore, this model only uses male mice, and it is unknown whether these changes are translatable to females. Additionally, because of survivorship considerations during experimental planning involving the use of older mice, the mice were initially younger than the age considered to be skeletally mature. This mouse model allows for future focus on gene expression, prevention, slowed progression, and treatment, using both pharmacologic and surgical methods. It provides a framework to further evaluate the mechanical traumatic and obese-induced vasculopathic neuropathic degenerative features of CNA.
Conclusion
This multivariable designed study used to evaluate an animal model for CNA expands on previous work in the establishment of an animal model. Changes akin to Charcot foot in humans were observed through the combination of neuropathy in the C57BL/6J diet–induced obese mouse and microtrauma from treadmill running. This model may serve future studies surrounding treatments and intervention protocols for Charcot foot.
Supplemental Material
sj-jpg-2-fai-10.1177_10711007231198822 – Supplemental material for Establishment of a Neurodegenerative Charcot Mouse Model
Supplemental material, sj-jpg-2-fai-10.1177_10711007231198822 for Establishment of a Neurodegenerative Charcot Mouse Model by Zachary A. Koroneos, Anna Ptasinski, Christopher Stauch, Tonya S. King, Julie C. Fanburg-Smith and Michael Aynardi in Foot & Ankle International
Supplemental Material
sj-pdf-1-fai-10.1177_10711007231198822 – Supplemental material for Establishment of a Neurodegenerative Charcot Mouse Model
Supplemental material, sj-pdf-1-fai-10.1177_10711007231198822 for Establishment of a Neurodegenerative Charcot Mouse Model by Zachary A. Koroneos, Anna Ptasinski, Christopher Stauch, Tonya S. King, Julie C. Fanburg-Smith and Michael Aynardi in Foot & Ankle International
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. ICMJE forms for all authors are available online.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplementary material is available online with this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
