Abstract
Microsecretory adenocarcinoma (MSA) is a rare, recently defined, low-grade salivary gland neoplasm characterized by a recurrent MEF2C::SS18 gene fusion and a generally indolent clinical course. Most tumors arise in the oral cavity, particularly the hard palate and buccal mucosa, and display microcystic and tubular architecture with bland cytological features and a consistent immunoprofile, including SOX10, S100, and p63 positivity, while p40 is negative. To date, fewer than 50 patients have been reported, with only 2 demonstrating high-grade histological features or distant metastasis. Herein, we report a 34-year-old woman with a tumor of the retromolar trigone, initially classified as polymorphous adenocarcinoma. Fifteen years later, the patient developed pulmonary metastases with distinct histomorphological features, including solid-cribriform differentiation and comedo-type necrosis. Molecular analysis identified an MEF2C::SS18 gene fusion and an immunohistochemical profile consistent with MSA. This report highlights the histological variation and the potential for late metastatic progression in MSA, emphasizing the need for accurate molecular diagnosis and long-term clinical surveillance, even in tumors that appear histologically low-grade at initial presentation.
Introduction
Microsecretory adenocarcinoma (MSA) is a rare and recently recognized salivary gland tumor defined by a recurrent MEF2C::SS18 gene fusion, typically involving exon 7 of MEF2C and exon 4 of SS18. 1 This tumor predominantly arises in the oral minor salivary gland, with a predilection for the hard palate and buccal mucosa. 1 Histologically, MSA displays a lobulated architecture composed of microcysts and tubules filled with basophilic secretions, along with cytologically bland epithelial cells. Immunophenotypically, MSA is consistently positive for SOX10, S100, and p63, while negative for p40, reflecting its intercalated duct-like phenotype. Despite its infiltrative growth pattern, the tumor generally behaves indolently.1,2 Interestingly, most of the tumors do not recur after surgery in the currently published follow-up periods. 3 To date, only 42 tumors have been reported in the literature, including an entity counterpart in skin. 4 Aggressive features have been identified in only 2 tumors: one demonstrated solid/cribriform architecture patterns, increased mitotic activity, and regional lymph node metastases, while the other, despite displaying low-grade histological features, was associated with both local recurrence and distant metastasis.5,6 Here, we report an MSA with aggressive features, in which the patient developed lung metastasis 15 years after the initial diagnosis. The metastatic lesion exhibits more solid-cribriform nests with fewer microcystic spaces, increased mitotic activity, a higher Ki-67 labeling index, with focal comedo-type necrosis features not typically seen in conventional MSA. This report highlights the potential for late metastatic progression in a tumor generally considered low grade, underscoring the need for long-term surveillance and further study to better define the full spectrum of its biological behavior.
Patient Presentation
A 34-year-old woman presented with a 1.5 cm exophytic mass of the right retromolar trigone. Gross examination revealed a firm, white lesion. Histologically, the tumor was unencapsulated, with lobular nests exhibiting a vague cribriform and microcystic architecture, infiltrative borders, and microcysts containing basophilic secretions (Figure 1A and B). Tumor cells were cuboidal, containing clear cytoplasm, monotonous nuclei with mild nuclear atypia, and rare mitoses (Figure 1C). The overlying mucosa exhibited pseudoepitheliomatous hyperplasia.
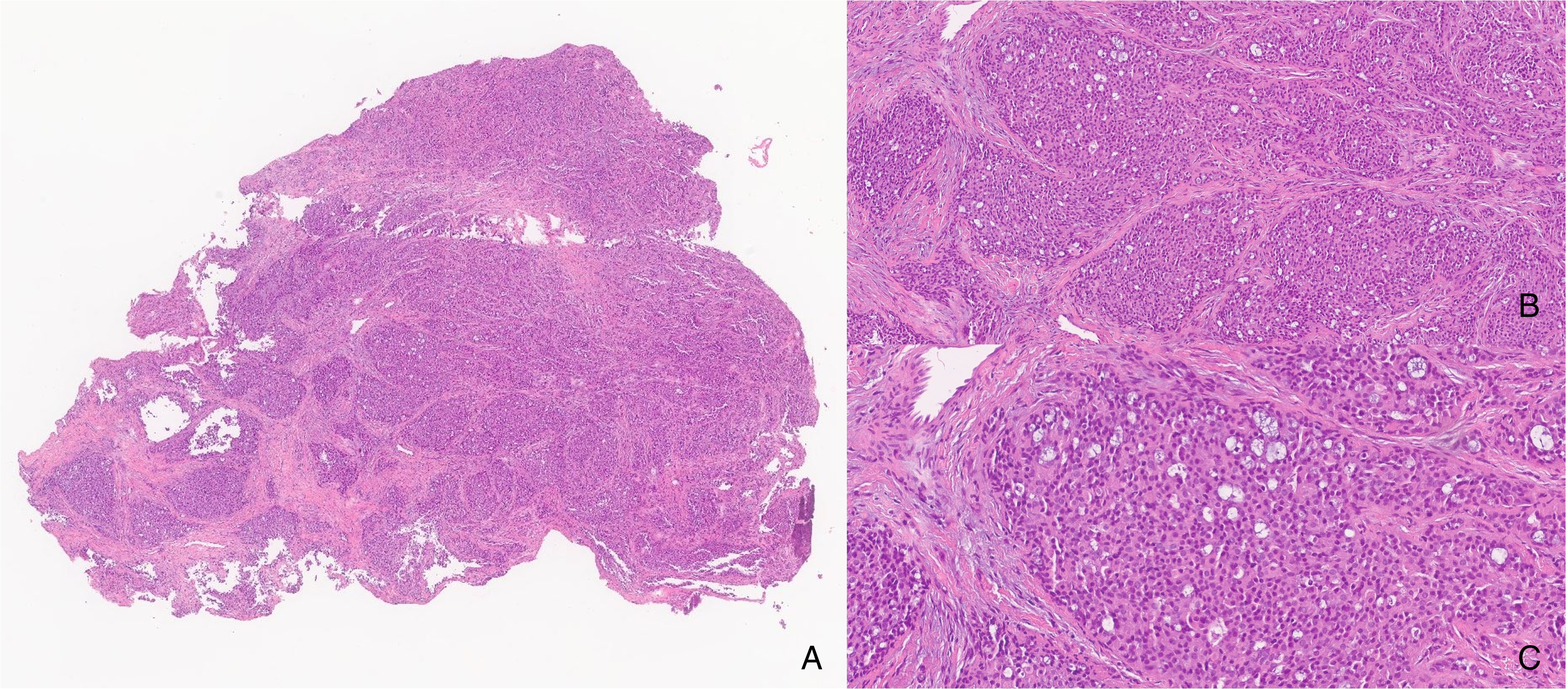
Histological Features of the Right Retromolar Trigone Tumor Resection. Histology showed lobular yet infiltrative architecture (A) with solid-cribriform microcystic spaces containing basophilic secretions (B). The tumor cells contain monotonous nuclei with mild nuclear atypia, eosinophilic to clear cytoplasm, and rare mitoses (C) (H&E, magnifications: A, 20×; B, 100×; C, 200×). Abbreviation: H&E, haematoxylin and eosin.
Immunohistochemical studies revealed diffuse positivity for keratin cocktail, keratin 7 (KRT7), S100, vimentin, and glial fibrillary acidic protein (GFAP). There was scattered and patchy positivity for p63 and E-cadherin. The tumor cells were negative for epithelial membrane antigen (EMA), p40, KIT, DOG1, smooth muscle actin (SMA), calponin, CD10, carcinoembryonic antigen (CEA), estrogen receptor (ER), and progesterone receptor (PR). The Ki-67 labeling index was approximately 8%. Due to its unusual morphology and immunophenotype, 2 pathologists rendered divergent initial diagnoses of polymorphous adenocarcinoma and myoepithelial carcinoma, respectively.
Fifteen years later, the patient was diagnosed with ER-positive, PR-positive, HER2-negative invasive ductal carcinoma of the breast (Figure 2). As part of cancer staging, positron emission tomography/computed tomography (PET/CT) revealed multiple pulmonary nodules with a maximum standardized uptake value (SUV) of 9.1. Wedge resection of a 1.6 cm solid nodule from the right upper lobe showed a well-circumscribed lesion that, on histological review, resembled the retromolar trigone tumor. However, compared to the retromolar trigone specimen, the lung lesion demonstrated more solid growth with fewer microcysts containing basophilic secretions. The tumor cells contained eosinophilic cytoplasm and round, uniform nuclei with dense chromatin (Figure 3A and B). Focal necrosis, increased mitoses (up to 8/mm²), and elevated Ki-67 labeling index (∼35%) were noted.
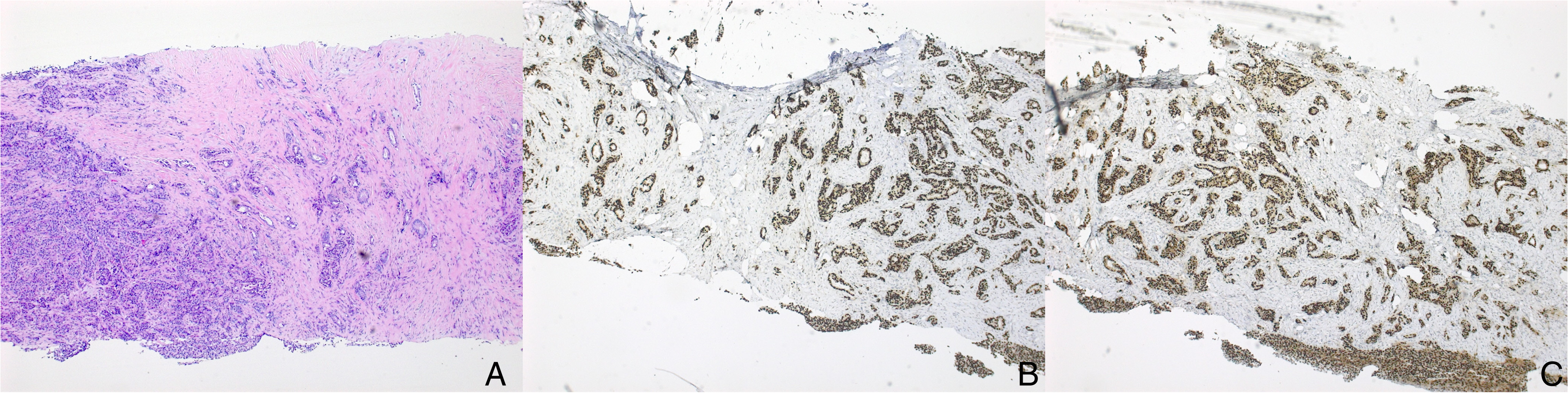
Histological Features of the Breast Core Biopsy. Histology showed invasive ductal carcinoma (A) that was positive for GATA3 (B) and estrogen receptor (C) (magnifications: A-C, 100×).
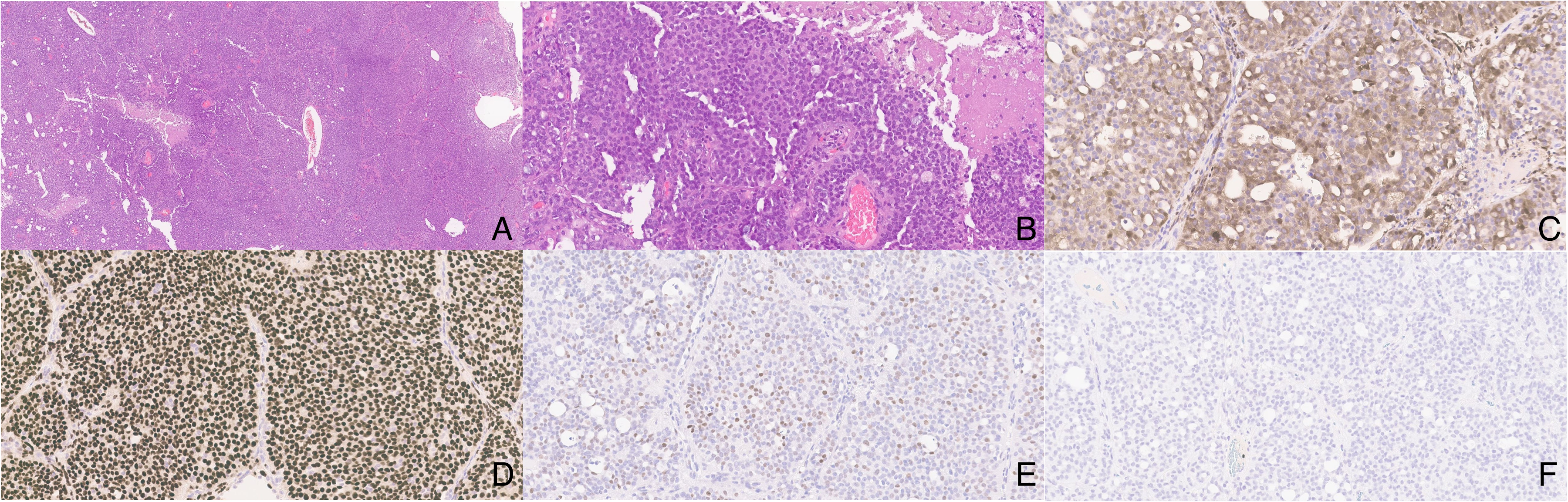
Histological Features of the Right Upper Lung Lobe Wedge Resection. Histology demonstrated solid architecture with fewer microcysts (A). The tumor cells contain uniform nuclei with dense chromatin surrounded by eosinophilic cytoplasm, with focal necrosis (B). Immunohistochemical stains showed that the tumor cells were positive for S100 (C), SOX10 (D), and p63 (E), while negative for p40 (F) (magnifications: A, 40×; B–F, 200×).
Immunohistochemical (IHC) studies demonstrated diffuse positivity for keratin cocktail, keratin 7, keratin 8, keratin 19, S100 (Figure 3C), SOX10 (Figure 3D), BCL2, and E-cadherin, supporting an epithelial phenotype with features suggestive of salivary gland origin. Patchy or focal positivity was observed for p63 (Figure 3E), keratin 14, and KIT. PanTRK demonstrated a heterogeneous membranous staining pattern. The tumor cells were negative for a broad range of lineage specimen markers, including SMA, h-caldesmon, calponin, myosin, mammaglobin, GATA3, ER, PR, HER2, DOG1, synaptophysin, chromogranin, TTF1, p40 (Figure 3F), EMA, CDX2, CD10, ALK, and GFAP, thereby excluding myoepithelial, neuroendocrine, breast, and other site-specific differentiation. p53 by IHC showed a wild-type staining pattern, and PD-L1 (clone 22c3) expression was considered negative with a combined positivity score of <1.
Molecular studies with next-generation sequencing revealed no mutations in EGFR exons 18 to 21. Alterations in the SWI/SNF complex were identified, specifically involving ARID1A and subclonal SMARCB1; however, ARID1A and SMARCB1 protein expressions by IHC were retained.
Given the absence of histological or immunophenotypic similarity to the patient's known breast carcinoma, along with the shared morphology, immunoprofile, and molecular findings between the current lung lesion and the prior retromolar trigone tumor, the lung lesion was best interpreted as metastatic salivary gland carcinoma. Subsequent expanded next-generation sequencing using a specific salivary gland tumor panel, “SalvGlandDx v2,” 7 identified a MEF2C::SS18 gene fusion, thereby pointing to the diagnosis of MSA, which had not yet been described at the time of the patient's initial presentation.
Discussion
MSA is a recently described salivary gland tumor with a characteristic molecular signature and a generally indolent clinical course. 8 Most reported tumors are low-grade, localized, and well-circumscribed, yet have a microscopically infiltrative tumor border. Histologically, MSA is composed of microcysts, tubules, and cords in a fibromyxoid stroma. The uniform epithelial cells exhibit clear cytoplasm and small nuclei with minimal atypia. The majority arise in the oral cavity and lack overtly malignant features such as necrosis, high mitotic rates, or metastasis. Perineural invasion is rare. 8 Given its architectural overlap with other low-grade salivary gland tumors, including the recently proposed entity microcribriform adenocarcinoma, careful histopathological evaluation and molecular studies are essential for accurate diagnosis. 9
To date, only 2 examples of MSA with aggressive histological and clinical features have been reported. The first, described by Gui et al, demonstrated overtly high-grade characteristics including solid architecture, extensive invasion, high mitotic activity (10/10 high-power field), necrosis, apoptosis, and lymph node metastases, with loss of p63 expression. Despite these features at the initial disease presentation, the patient remained disease-free for 20 months after being treated with surgery followed by adjuvant chemoradiation. 5 The second tumor, reported by Jurmeister et al, occurred in a patient who initially presented in 2009 and subsequently developed multiple pulmonary nodules within 1 year, followed by a local recurrence in 2016. The tumor was initially described as resembling mucoepidermoid carcinoma but was later reclassified as MSA upon the emergence of diagnostic criteria and molecular confirmation for this entity.1,6
Our report represents a further instance of MSA with aggressive clinical behavior, underscoring both the histological heterogeneity and the potential for late distant metastasis in the otherwise indolent neoplasm. The morphologic features in our report parallel those described by Gui et al, while the delayed pulmonary metastasis mirrors the clinical course seen in the report by Jurmeister et al.
Given its histological overlap with other salivary gland neoplasms, MSA should be carefully distinguished from entities such as polymorphous adenocarcinoma, microcribriform adenocarcinoma, 9 myoepithelial carcinoma, or adenocarcinoma not otherwise specified. Among these, polymorphous adenocarcinoma presents the greatest diagnostic challenge as a subset of these tumors, especially those harboring PRKD1, PRKD2, or PRKD3 fusions, may demonstrate cribriform pattern and an immunophenotype that is p63-positive and p40-negative features also seen in MSA. However, the presence of MEF2C::SS18 fusion in our report corroborates the diagnosis of MSA and helps to exclude other salivary gland neoplasms, including PRKD-rearranged polymorphous adenocarcinoma. Nevertheless, the solid-cribriform pattern observed in our report is usually seen in polymorphous adenocarcinoma/cribriform adenocarcinoma and poses some overlap. Other entities in the differential diagnosis include adenoid cystic carcinoma, secretory carcinoma, myoepithelial carcinoma, and sclerosing microcystic adenocarcinoma, which should also be considered in the differential diagnosis. Each can be distinguished based on a combination of histomorphology, immunophenotype, and molecular profiling.
This report adds to the growing recognition of MSA and expands the morphological and clinical spectrum of the tumor. While typically low-grade, MSA can rarely demonstrate aggressive behavior, including distant metastasis without prior recurrence. Our report illustrates that histological blandness at presentation does not preclude late metastasis, although further studies have to reveal if the unusual solid-cribriform morphology might be associated with the more aggressive clinical course. Molecular diagnostics, including fusion detection and sequencing, are critical for accurate diagnosis and prognostication. Long-term follow-up may be beneficial, even in tumors initially deemed indolent.
Footnotes
ORCID iDs
Ethics Approval
The authors confirm that all procedures performed in this case report were following the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Consent to Participate
Informed consent obtained, though for this type of study, it's not required.
Consent for Publication in the Manuscript
Consent for publication obtained, though for this type of study, it's not required.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
