Abstract
Adamantinoma-like Ewing sarcoma (ALES) is a newly described rare entity, which shows EWSR1::FLI1 rearrangement characteristic of Ewing sarcoma. This can be diagnostically challenging as it manifests histologically with epithelial differentiation and has diffuse keratin expression as well as p40 and p60 positivity. We hereby report a case of ALES in a 33-year-old woman with a past medical history of breast carcinoma who presented with a right-sided parotid mass. CT scan of the neck showed a heterogenous mass within the superficial lobe, measuring 17 mm in diameter for which the patient underwent superficial parotidectomy. Histopathology of the mass revealed a malignant neoplasm formed of solid nests, cords and sheets of cells with minimal cytoplasm and monomorphic nuclei with granular chromatin and indistinct nucleoli. Brisk mitotic activity and tumor necrosis were also present. The tumor showed strong and diffuse reactivity for pankeratin (clone AE1/AE3) and keratin 20, both in a dot-like pattern, raising the suspicion of metastatic Merkel cell carcinoma; however, molecular studies showed EWSR1::FLI1 rearrangement, supporting the diagnosis of ALES. In summary, it is prudent to have knowledge about this entity to avoid its misdiagnosis as other malignancies of the head and neck region which exhibit a different clinical course, prognosis and hence treatment modalities.
Keywords
Introduction
Adamantinoma-like Ewing sarcoma is a novel entity which was first described by Folpe et al., in the bone and soft tissue region in a study of 66 genetically confirmed cases. It belongs to the Ewing sarcoma family of tumors and demonstrates the EWSR1::FLI1 translocation. 1 Till date, 12 cases have been reported in the head and neck region, mainly in the salivary glands. This tumor displays complex epithelial differentiation characterized by cohesive growth, keratin pearl formation, peripheral nuclear palisading and diffuse keratin and p40 staining. 2 This morphology creates a diagnostic challenge because of the overlapping histopathologic features with undifferentiated small round blue cell tumors, such as alveolar rhabdomyosarcoma, olfactory neuroblastoma, NUT midline carcinoma, lymphoma, melanoma, and many others, that occur in the head and neck region. 3 However, contrasting with aforementioned tumors, ALES harbor the characteristic EWSR::ETS gene fusions similar to its counterpart in the long bones and elsewhere. Herein, we report a case of ALES in the parotid gland presenting a diagnostic challenge with an immunomorphologic featured that mimicked metastatic Merkel cell carcinoma.
Case Presentation
A 35-year-old woman with a history of stage IV oligometastatic breast cancer who presented with a gradually enlarging right-sided parotid mass, noticed 3 months prior to presentation. No associated fever, drainage, or other symptoms. CT of the neck revealed a heterogeneous mass within the superficial lobe of the parotid gland measuring up to 17 mm in maximum dimension. Findings were suspicious for metastatic lymphadenopathy in view of history of breast cancer or a primary salivary gland neoplasm. FNA of the mass revealed crushed lymphoid cells, admixed with rare benign epithelial cells and rare groups of atypical cells, however, no definitive diagnosis was made, therefore, a follow up superficial parotidectomy was performed. Grossly, the tumor showed a well-circumscribed lobulated mass with white-tan cut surface. Microscopically, the tumor was formed of small round blue cells arranged in solid nests, sheets, and cords separated by fibrocollagenous stroma. The cells were monomorphic with indistinct cytoplasm, containing nuclei with finely granular chromatin and rare prominent nucleoli. Brisk mitotic activity and tumor-type coagulative necrosis were present. Prior breast tumor morphology was compared with the parotid tumor and showed distinct histologic features of ER, PR, and Her2/Neu (ERBB2) positive invasive ductal carcinoma with focal apocrine features and high-grade ductal carcinoma in-situ in the former and malignant small round cell tumor in the latter. Immunohistochemistry showed positive reaction for pankeratin with clone AE1/AE3 and keratin 20, both were strong and diffuse in dot-like pattern. Reactions for synaptophysin, p40 and p63 were also strongly and diffusely positive, raising the suspicion of metastatic Merkel cell carcinoma; however, CD99 immunohistochemistry showed a strong and diffuse reaction, which raised the suspicion of a possible sarcoma of the Ewing family of tumors (Figure 1). Additional immunohistochemical reactions for smooth muscle/ myoepithelial markers (SMA, SMMHC, and calponin), melanoma markers (SOX10 and S100), breast carcinoma markers (ER, PR, GATA3, and keratin 7), hematolymphoid (LCA, CD3, and PAX5), desmin, SV40, and TTF1 were all negative. A clinically validated RNA solid tumor fusion panel testing using next generation sequencing showed EWSR1::FLI1 gene rearrangement. The fusion transcript originated from der(22) chromosome and contained the N terminal domain of the EWSR1 (exons 1–7, amino acids 1–265) that was fused in-frame with C-terminal domains of FLI1 (exons 7–9, amino acids 241–452) gene, thereby linking the transactivation domain of EWSR1 with the DNA binding ETS type domain of FLI1 to form an oncogenic chimeric transcription factor (Figure 2), confirming the diagnosis of Ewing sarcoma, adamantinoma-like.
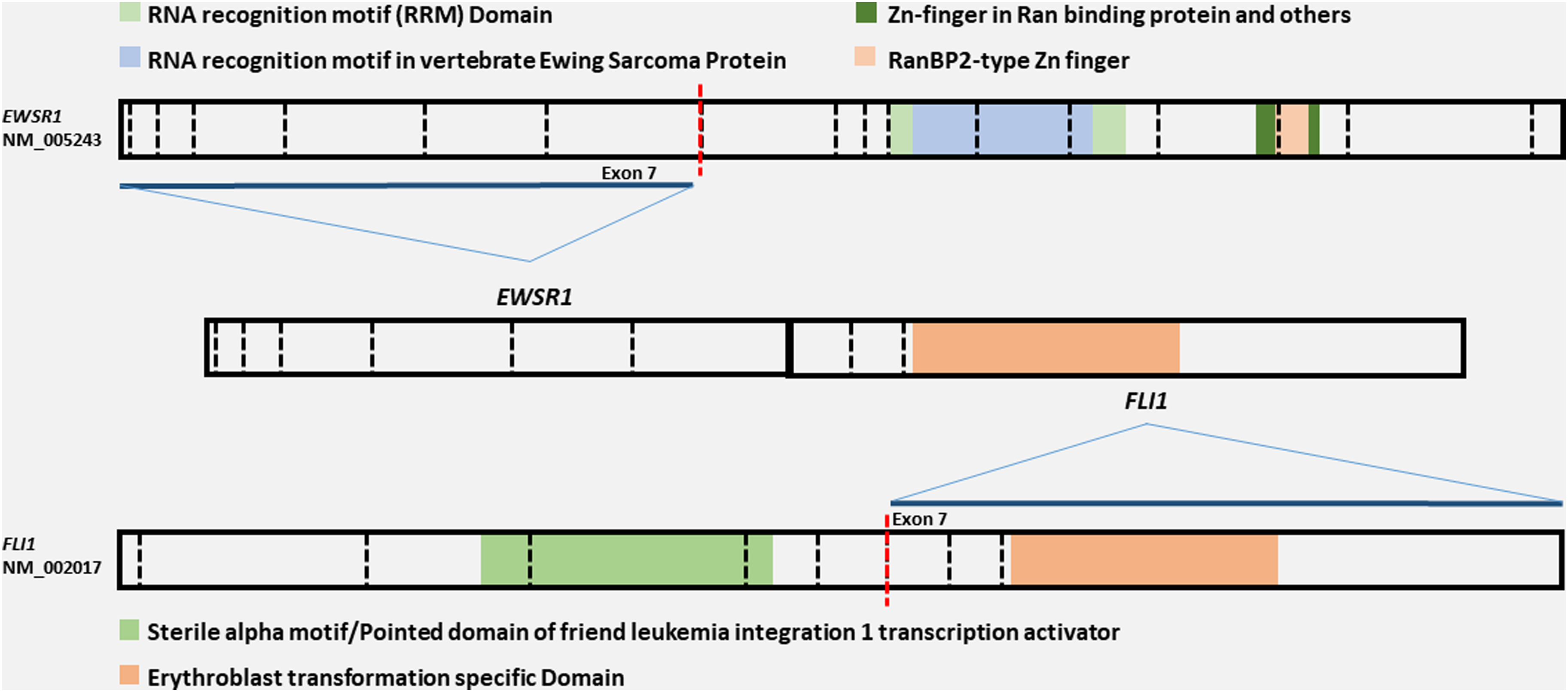
A schematic representation showing the EWSR1::FLI1 fusion, with the fusion transcript containing the N terminal domain of the EWSR1 (exons 1–7) and the C-terminal domains of FLI1 (exons 7–9) genes.
Discussion
Fusion of the Ewing sarcoma breakpoint region 1 (EWSR1) gene on chromosome 22 with the Friend leukemia virus integration 1 (FLI1) gene on chromosome 11 resulting in the t(11;22)(q24;q12) translocation is a recurrent fusion described in Ewing sarcoma/peripheral primitive neuroectodermal tumors (ES/PNET). The t(11;22)(q24;q12) EWSR1::FLI1 rearrangement is found in 85% of cases of Ewing sarcoma tumors where EWSR1::FLI1 expression has been shown to lead to transformation in cultured cells and altered transcriptional regulatory activity, increased binding levels and affinity for the SWI/SNF complex, and increased H3K27 trimethylation.4–6
Adamantinoma-like Ewing sarcoma is a recently recognized tumor that belongs to the Ewing Sarcoma family of tumors. It has a predilection for the head and neck region, especially salivary glands, where it is prone to misdiagnosis as basal cell adenocarcinoma or poorly differentiated adenocarcinoma with basaloid features according to the report by Rooper et al. 2 They occur over a wide age range (32–77 years), with a mean age of 52 years. Histologically, it has propensity to differentiate along the epithelial lines where monotonous cells show squamous differentiation with keratin pearl formation as well as basaloid morphology, rosette formation, and frequent association with necrosis and increased mitotic activity. 2 To add to the complexity, ALES also demonstrates immunophenotypic positivity for keratins, p40, CD99, NKX2.2, and neuroendocrine markers such as synaptophysin as seen in our case. 2 Therefore, this diagnosis carries a high potential for a diagnostic pitfall.
An important example to consider is Merkel cell carcinoma (MCC), an aggressive small blue round cell tumor with neuroendocrine differentiation, which usually occurs in the sun-damaged skin of the head/neck region. Microscopically, MCC tumors are formed of sheets and trabeculae of cells with scant cytoplasm, round monomorphic nuclei containing finely granular and speckled chromatin, and inconspicuous nucleoli. Mitotic activity is usually brisk and necrosis, vascular, and perineural invasions are usually seen. 7 All these attributes resemble ALES and make diagnosis challenging. Immunophenotypically, MCC shows positive expression of pankeratin with clone AE1/AE3, chromogranin, and synaptophysin, again resembling ALES. Additionally, the characteristic paranuclear dot like positivity for keratin 20 present in cases of MCC, is thought to be negative in ALES, although it was not performed/ reported in the vast majority of the reported cases. However, as seen in our case, even the characteristic pattern of keratin 20 reactivity turned out to be not specific to MCC. Interestingly, one of the cases reported by Rooper et al., was in fact diagnosed as Merkel cell carcinoma, although no details on the immunohistochemical characteristics were reported and one case reported by Alnuaim et al. showed focal keratin 20 positivity with no further characterization of the staining pattern reported.2, 8 Tumors with basaloid morphology like basal cell adenocarcinoma and adenoid cystic carcinoma are other examples. While basal cell adenocarcinoma is a low-grade malignancy with infiltrative growth pattern. 3 It shows a uniform morphology with small cells containing scant cytoplasm and ovoid hyperchromatic nuclei. Nuclear palisading is usually present and squamous differentiation can be seen. By immunohistochemistry, both basal cell adenocarcinoma and adenoid cystic carcinoma show evidence of epithelial/myoepithelial differentiation where the p63, p40, and calponin highlight the myoepithelial cells while keratin 5/6 and keratin 7 highlights the luminal (epithelial) cells. ALES, on the other hand, shows a single population of tumor cells with diffuse reaction for keratin 5/6 and p40. NUT midline carcinoma is also diagnostic pitfall, especially in the presence of squamous pearls, however, all the aforementioned differential diagnoses are negative for CD99. 8 Lastly, neuroendocrine differentiation as evident by the positive reactions for neuroendocrine markers, such as synaptophysin, is seen more frequently in salivary gland ALES (80% cases) as in our case. Therefore, tumors with neuroendocrine differentiation, such as large cell neuroendocrine carcinoma are of diagnostic consideration. However, these are again negative for CD99 immunohistochemistry.
In summary, the complex morphologic and immunophenotypic features of ALES with a broad differential diagnostic list create a diagnostic challenge. One needs to be alert and cognizant of this tumor for proper characterization and classification that requires ancillary molecular techniques for a definitive diagnosis and subsequently proper management.
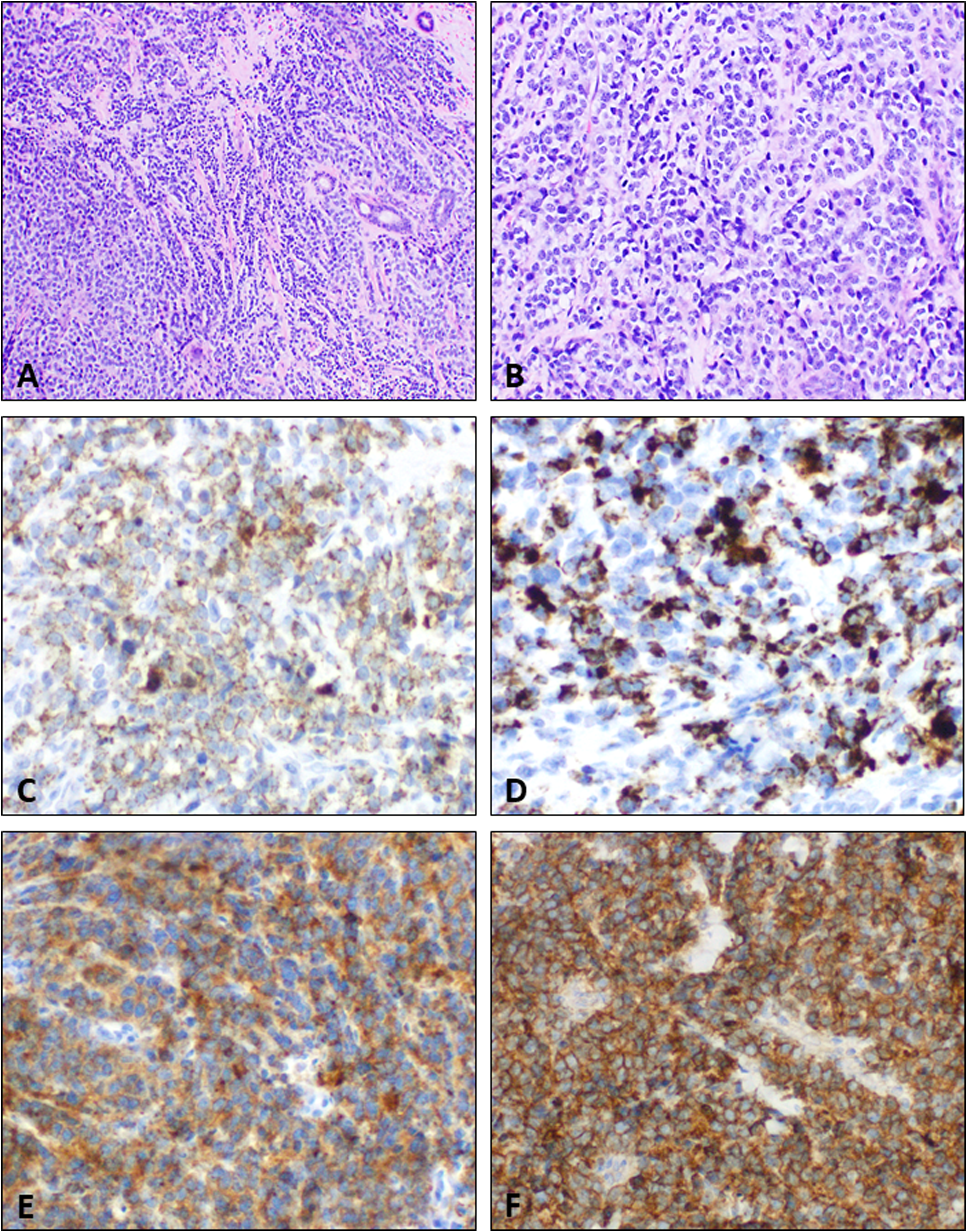
Adamantinoma-like Ewing sarcoma. Representative images show tumor cells arranged in sheets, nests and cords with intervening fibrocollagenous stroma and entrapped salivary gland ducts (A). The tumor cells have scant amount of cytoplasm, with monomorphic, round nuclei containing fine granular chromatin (B). On immunohistochemistry, the tumor cells show diffuse staining for pankeratin with clone AE1/AE3 (C) and keratin 20 (D), both in a stippling dot-like pattern (reminiscent to that seen in cases of Merkel cell carcinoma). Diffuse immunohistochemical staining for synaptophysin (E) and CD99 are also present (F).
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
