Abstract

Brief Report
Accurate grading of prostate cancer (PCa) is essential for prognostication and clinical decision-making. Risk stratification tools, such as the National Comprehensive Cancer Network (NCCN) system, incorporate both pathological and clinical parameters, including the Gleason score (GS)/Grade Group (GrG), tumour extent, serum prostate-specific antigen (PSA) levels, and clinical stage. 1 These risk groups inform decisions regarding additional investigations and optimal management.
Intraductal carcinoma of the prostate (IDCP) is a neoplastic proliferation within pre-existing ducts, typically representing retrograde spread of invasive cancer, though a subset may arise as a precursor lesion. 2 IDCP is recognized as an adverse prognostic finding, yet its incorporation into the GS/GrG remains controversial. The Genitourinary Pathology Society (GUPS) recommends grading only the invasive component, whereas the International Society of Urological Pathology (ISUP) advocates grading both the invasive and intraductal components.3,4 Of note, both organizations recommend commenting on the presence of IDCP.
Previous studies have demonstrated that incorporating IDCP into grading modifies GS/GrG in a subset of patients.5–7 However, as NCCN risk stratification considers multiple factors, the extent to which these grading changes affect overall risk categorization remains uncertain. A prior study found that different grading approaches altered NCCN risk classification in a minority of patients, but its findings were based on a small cohort. 5 In this study, we evaluate the impact of IDCP grading methods on NCCN risk categorization at diagnostic biopsy in a larger series of patients.
Consecutive prostate biopsies with GrG1-GrG4 invasive carcinoma and IDCP diagnosed between 2015 and 2021 at a single institution were identified. Part of this dataset formed the basis of a previous publication evaluating the impact of grading methods on GS/GrG alone but was expanded for the current study. 6 For each biopsy set, three haematoxylin and eosin (H&E)-stained level sections were evaluated from each biopsy site. To distinguish IDCP from invasive carcinoma, the presence of clearly visible basal cells on H&E-stained sections was the only acceptable morphological criterion; in all other specimens, immunohistochemical cocktail staining (p63/high molecular weight keratin/amethylacyl-CoA racemase) was performed (151/177, 85.3%)
All specimens were reviewed by a genitourinary pathologist (S.P.) and graded using two different methods. For the first method, only invasive carcinoma was graded, without incorporation of the IDCP component (invasive only). For the second method, both invasive carcinoma and IDCP components were graded (invasive + IDCP). The GS/GrG comparison between the two scoring methods was based on the highest GS/GrG at any site. Individual sites with IDCP only (without invasive carcinoma) were not graded.
The proportion of positive cores, serum PSA level, and clinical T-stage were recorded. The NCCN risk group was calculated twice based on the highest GrG using both grading methods.
Institutional ethical approval was obtained (ECM4(I)27/05/19).177 patients with GrG1-GrG4 invasive carcinoma and IDCP were included. Of these, 31 patients (17.5%) had a change in the highest GS/GrG after inclusion of IDCP in grading. The GrG increased by one grade in 21 patients (67.7%), by two grades in 9 patients (29.0%) and by three grades in 1 patient (3.2%) (Figure 1). Upgrading was due to comedonecrosis in IDCP (13/31, 41.9%), solid growth in IDCP (1/31, 3.2%) or increased proportion of Gleason pattern 4 (17/31, 54.8%).
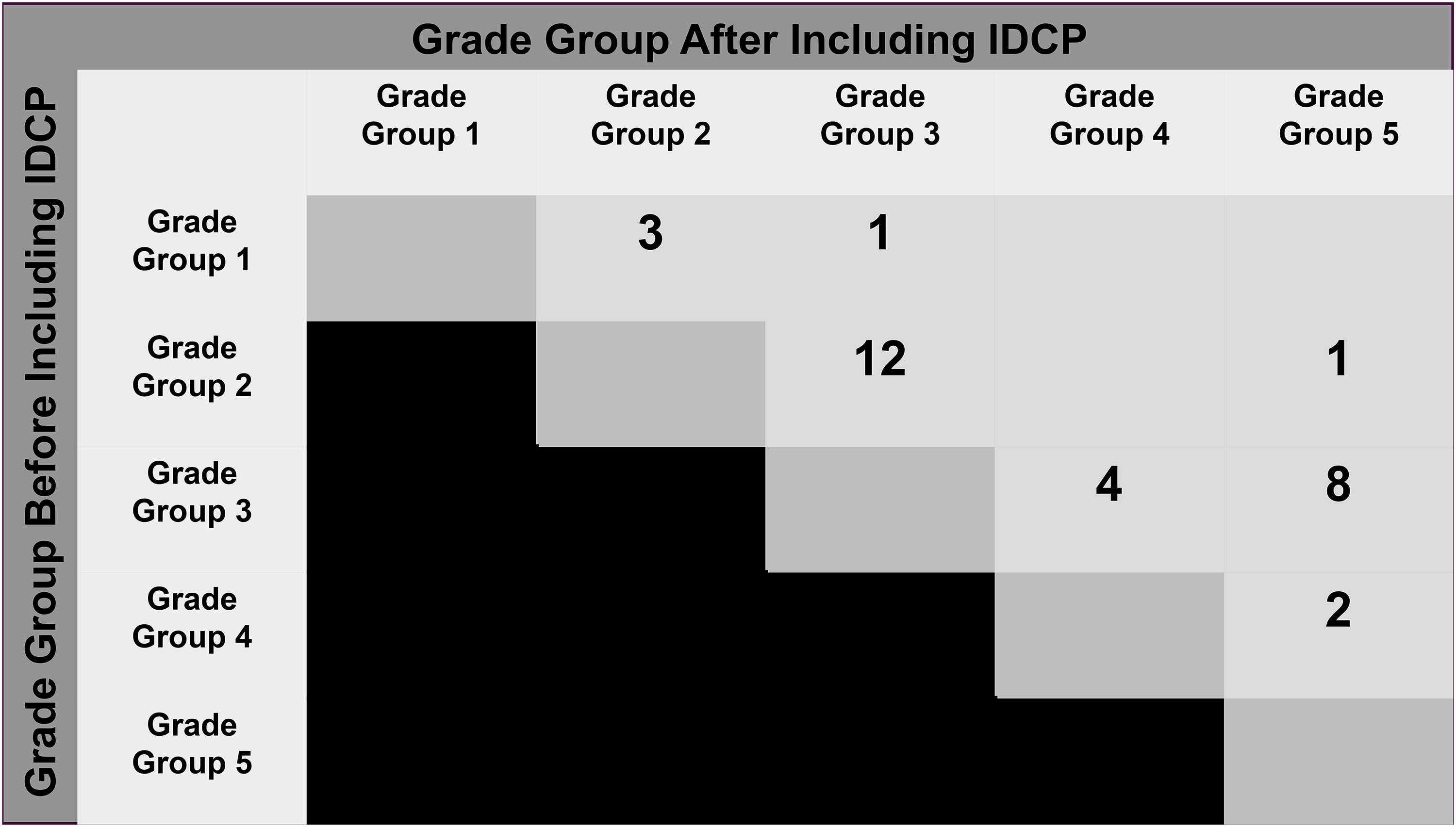
Changes in grade group when intraductal carcinoma of the prostate (IDCP) is included in grading. The grades of cases before and after inclusion of IDCP are listed on the left and top of the figure, with the number of cases with those grades shown in the row-column intersection.
Incorporating IDCP into grading impacted the NCCN risk group in 19 patients (19/31, 61.3% of the upgraded patients and 19/177, 10.7% of the total cohort). The original risk categories were changed in 3/3 low risk, 4/4 favourable intermediate risk, 12/19 unfavourable intermediate risk, 0/4 high risk and 0/1 very high risk groups. The NCCN changed by one group in 17/177 patients (9.6%), and by two groups in 2/177 patients (1.1%) of the cohort (Figure 2). Of the patients with change by 2 groups, 1 was due to comedonecrosis and 1 was GG1 with IDCP.
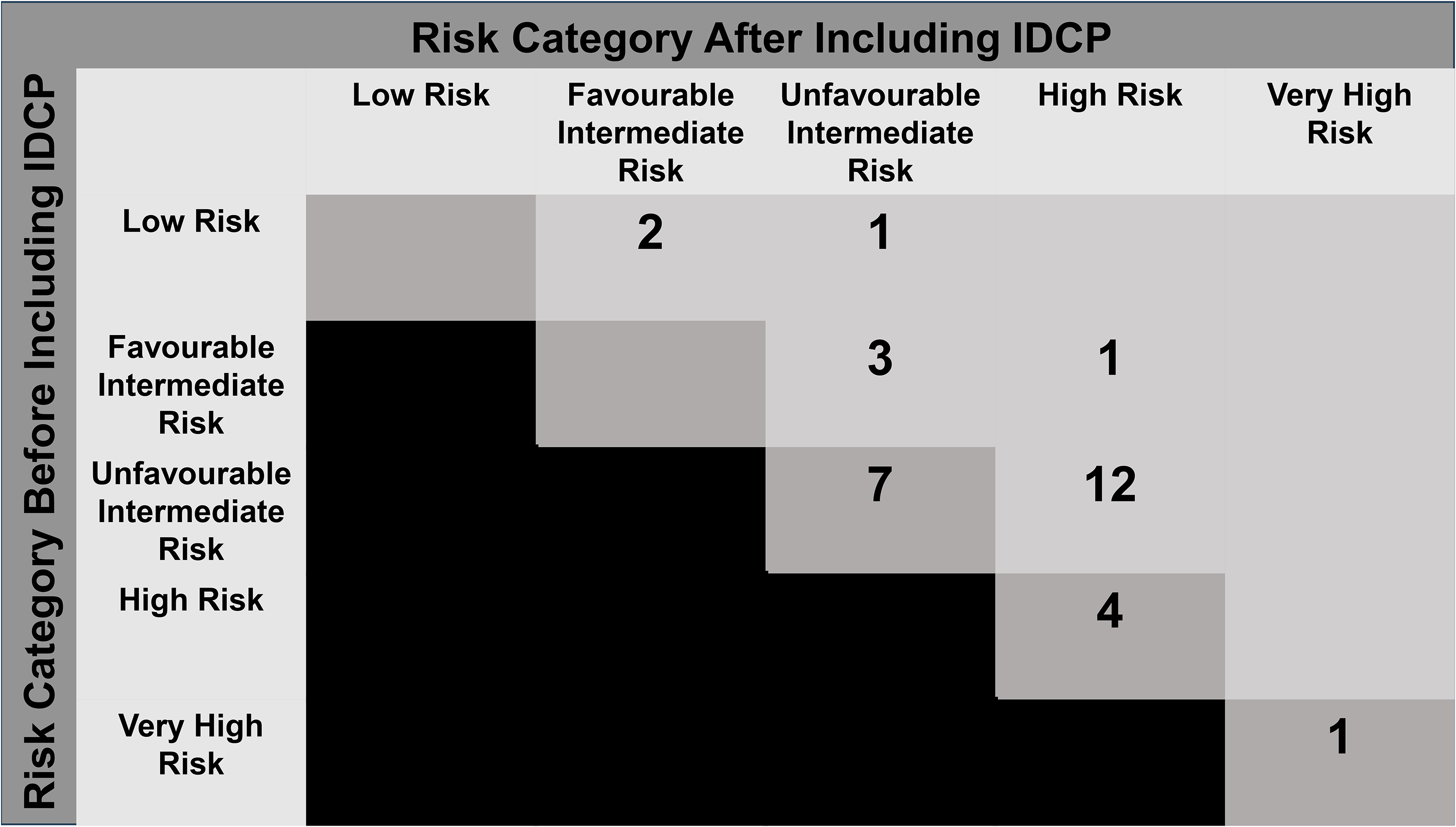
Changes in risk group when intraductal carcinoma of the prostate (IDCP) is included in grading. The risk groups of cases before and after inclusion of IDCP are listed on the left and top of the figure, with the number of cases with those risk groups shown in the row-column intersection.
Conflicting recommendations from genitourinary pathology associations have led to variability in how pathologists incorporate IDCP into the GS/GrG.3,4 Both organizations agree that isolated IDCP on biopsy should not be graded. However, when IDCP is present alongside invasive carcinoma, ISUP recommends including the IDCP component in grading, whereas GUPS advises excluding it. These differing guidelines have contributed to confusion among pathologists and treating clinicians.8,9
Previous studies have shown that alternative grading methods affect only 1-2% of all prostate biopsies with cancer, as most biopsies lack IDCP and those with Grade Group 5 disease remain unaffected.6,7 Among biopsies where grading could be impacted (ie, those with IDCP and invasive cancer ≤ Grade Group 4), variations in grading approaches alter the GrG in a subset of patients, with reported rates ranging from 12% to 27% in prior series.5–7 Consistent with these findings, the present study, which includes the largest biopsy cohort to date, found that the highest GrG changed in 17.5% of patients with IDCP. Importantly, clinical risk stratification incorporates multiple clinicopathologic parameters beyond GrG, which may further mitigate the impact of variations in grading practices. A previous study by Rizzo et al reported that grading IDCP altered the NCCN risk classification in only 4 of 48 patients with IDCP (8.3%). 5 The present study validated these findings in a larger cohort, observing a change in NCCN risk category in 10.7% (19/177) of patients with IDCP. While current ISUP and GUPS guidelines recommend that IDCP in the absence of invasive carcinoma should not be graded, it is not entirely clear if this applies to site-level or case-level reporting given that different biopsy sites may be sampling the same tumour. Future consensus recommendations may provide clarity on this point to further standardize reporting.
While different grading practices do not impact the vast majority of patients, for a small number the differences in GrG and/or clinical risk stratification may be significant. In the present study, the NCCN risk group changed by two categories in 2 patients, while Rizzo et al found that the NCCN group changed by 3 categories for 1 patient. 5 The greatest impact is in patients with low-grade (GrG1) invasive carcinoma with extensive IDCP, particularly with comedonecrosis. 10 These findings emphasize the need for clear communication with treating clinicians in these unusual scenarios pending large multi-institutional studies correlating grading methods with outcome in these patients. Although IDCP is not formally incorporated into clinical risk grouping, the most recent NCCN update recommends against active surveillance for patients with favourable intermediate risk prostate cancer. 1 Furthermore, both GUPS and ISUP concur that IDCP should be a contraindication to active surveillance.3,4 However, changes in risk categorization can also influence other management decisions such as the need for adjunct androgen deprivation therapy and additional staging or genetic investigations.
A limitation of the present study is that immunohistochemistry was not performed on all biopsy specimens and distinguishing IDCP from invasive cribriform carcinoma can be challenging. The identification of basal cells on H&E staining is not always reliable given that flattened tumour cells can mimic basal cells in invasive cribriform foci. 11 Nevertheless, the diagnosis of IDCP was based on careful morphologic assessment with IHC applied in the majority of patients. In real-world clinical practice, extensive immunohistochemical evaluation is generally not feasible.
In conclusion, our study found that variations in IDCP grading practices altered NCCN risk stratification in a minority of biopsies with IDCP. Although such circumstances are uncommon in clinical practice, clear communication with treating clinicians is essential for individual patient management. International consensus on grading of IDCP would allow for a consistent approach and facilitate future studies to determine how best to integrate IDCP into clinical risk models to optimize patient management.
Footnotes
Acknowledgements
We have no acknowledgements to declare.
Ethical Considerations
Institutional ethical approval was obtained (CREC reference ECM4(I)27/05/19).
All patient information was de-identified and patient consent was not required.
Consent to Participate
The requirement for informed consent was waived.
Author Contributions
SP designed the study. SP, SNM, and DM contributed to data acquisition and/or analysis. PM and SP conducted data visualization and prepared the manuscript.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
Data is available upon reasonable request.
