Abstract
Introduction. Primary mucinous adenocarcinoma of the kidney is an exceedingly uncommon malignancy. It comprises a minority of renal malignancies and presents notable diagnostic complexities owing to its atypical manifestation and resemblance in clinical features to common urinary tract conditions. Surgical intervention is the preferred treatment modality, although the prognosis remains bleak, with high rates of recurrence. A definitive diagnosis is established through postoperative histopathological examination. Patient Presentation. Here, we present a 75-year-old man who reported complaints of a mass in the left flank with clinical findings suggestive of a nonfunctioning left kidney. Initial diagnostic imaging suggested a pelvi-uretric junction calculus-associated gross hydronephrosis. However, a subsequent nephrectomy was performed, and the specimen received was subjected to histopathological analysis, which revealed the diagnosis to be a primary mucinous adenocarcinoma involving the kidney. Conclusion. This clinical observation highlights the diagnostic challenge and importance of considering rare entities in patients with vague clinical presentations. Histopathological evaluation is crucial in accurately diagnosing and appropriately managing such rare malignancies. This report contributes to the limited literature on this entity, emphasising the need for awareness among clinicians.
Introduction
Adenocarcinomas originating in the renal pelvis constitute a minority, comprising <1% of total malignancies in this region. These tumors can be subdivided into tubulovillous, mucinous, and papillary nonintestinal types. 1 Ackerman LV was the first to document the occurrence of primary mucinous adenocarcinoma in the kidney in 1946. 2 This particular entity continues to be labeled as exceedingly uncommon, with under 100 patient reports described to date. The challenge lies in the preoperative identification of this condition, as it lacks distinctive symptoms and radiological signs. Furthermore, due to its rarity, there is no universally accepted standard treatment protocol. 3 Moreover, understanding of primary mucinous adenocarcinoma in the kidney is significantly lacking, and the World Health Organisation has not acknowledged these tumors in the 2022 classification of renal tumors. 4
Herein, we discuss comprehensively regarding an elderly man who came with complaints of a mass in the left flank and a nonfunctional left kidney who underwent radical left nephrectomy and, on subsequent histomorphological evaluation, was found to have primary mucinous adenocarcinoma in the kidney. The diagnostic approach and therapeutic interventions adopted are meticulously detailed, and an extensive review of pertinent literature is included.
Patient Presentation
A 75-year-old man who had a chronic history of pelvi-uretric junction (PUJ) obstructive symptoms persisting for 2 years presented in the outpatient section of the department of urology with complaints of progressive left-sided flank pain for 6 months. He had no history suggestive of any known comorbidities.
A general and systemic examination revealed a mass in the left lumbar quadrant measuring 20 cm, which was soft in consistency, nonballotable, and not associated with tenderness. Routine hematological and biochemical parameters were within normal limits. A computed tomography of the urinary tract revealed a calculus measuring 3.6 × 3.1 × 2.7 cm obstructing the PUJ and two calculi, each measuring 2.3 × 1.1 × 1.1 cm in the lower pole of the left kidney. The kidney measured 24.8 × 12.4 cm and exhibited signs of gross hydronephrosis, initially attributed to the calculus in the PUJ. There was evidence of a saccular aneurysm of the abdominal aorta and a well-defined collection along the left iliopsoas muscle measuring 17 × 9 × 6.7 cm, for which a radiological opinion of left-sided chronic psoas abscess was given. A diethylenetriamine pentaacetate scan performed subsequently identified the kidney as nonfunctioning due to obstructive nephropathy.
To remove the calculi, a percutaneous nephrostomy was performed under general anesthesia. However, this procedure drained a considerable amount of gelatinous material, raising the suspicion of an underlying neoplasm. Following this, a radical left nephrectomy was performed, and the resected kidney was submitted for histopathological examination.
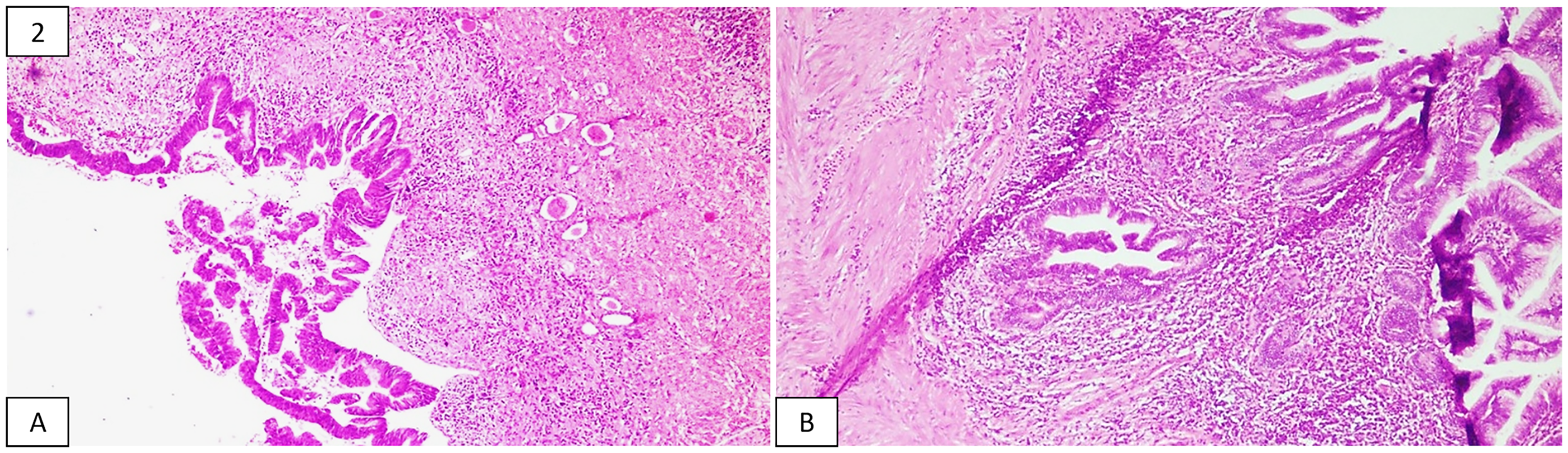
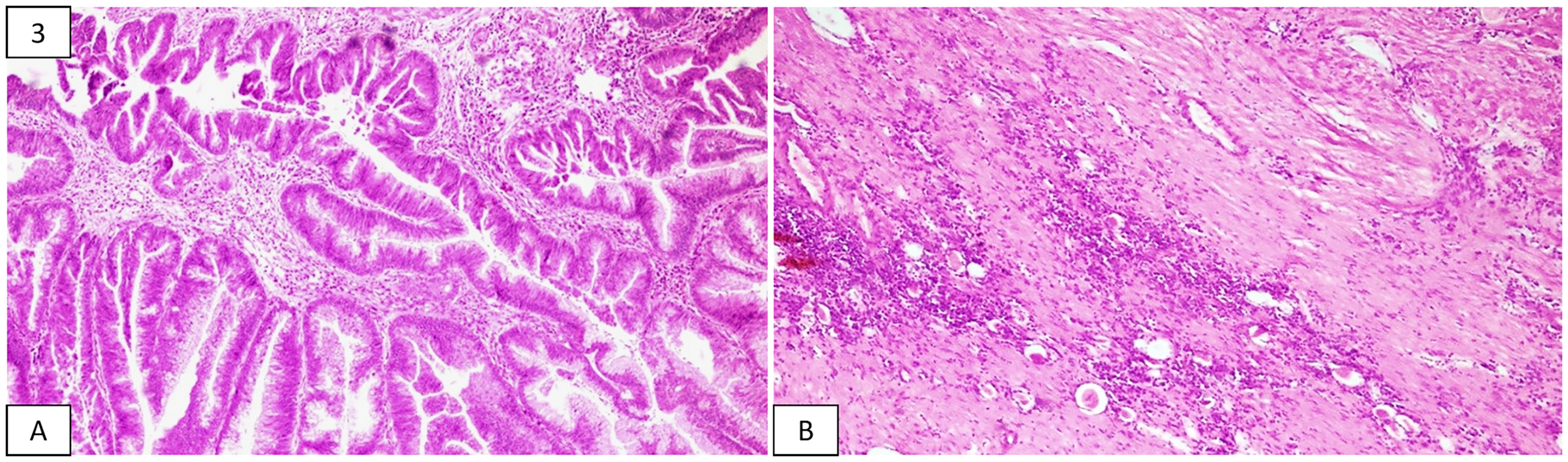
Grossly, the specimen of left radical nephrectomy, comprising an enormously cystic kidney and an adrenal gland, measured 17.5 × 11 × 5 cm and 2 × 1 × 1 cm, respectively. The outer surface of the kidney was bosselated and focally congested with multiple exudative areas. The cut surface showed cystic dilatation of the pelvicalyceal system with marked distortion of parenchymal architecture, with many cysts containing papillary excrescences (as shown in Figure 1). These cysts predominantly contained mucinous fluid. Neither areas of normal renal parenchyma nor visible infiltration of the ureter and renal vessels were appreciated macroscopically. The adrenal gland appeared unremarkable on examination. Representative sections, including solid and cystic areas, revealed an invasive tumor composed of pleomorphic columnar tumor cells in the form of complex glandular and papillary architecture bearing multiple villi (as shown in Figure 2). These cells were columnar and had cytoplasm containing abundant mucin, basally located hyperchromatic to vesicular nuclei, with a few showing prominent nucleoli (as shown in Figure 3). Large areas of mucin lakes containing floating tumor cells were appreciated. More than 60% of the tumor showed evidence of necrosis. The ureter, renal vessels, and adrenal gland were free of the tumor, while the adjacent renal parenchyma revealed features of chronic pyelonephritis. Based on these findings, diagnoses of mucinous adenocarcinoma in the kidney along with chronic pyelonephritis involving the left kidney were rendered and staged as pT1 according to the recent American Joint Committee on Cancer guidelines. An immunohistochemical analysis was done for further evaluation, wherein these tumor cells were found to express KRT7, KRT20, and CDX2, but were negative for PAX8. A positron-emission tomography and a bone scan were done for confirmation, which showed no evidence of metastasis and excluded the possibility of a primary malignancy elsewhere.
As per the opinion obtained from the medical oncologist, systemic chemotherapy comprising gemcitabine and cisplatin was initiated. The patient underwent 2 cycles of chemotherapy but discontinued the treatment, owing to its adverse effects.
Discussion
Although rare, most renal pelvic adenocarcinomas are documented in Asia. It is postulated that the etiology of renal adenocarcinomas can be attributed to ongoing inflammation resulting from chronic infection, obstruction, urinary calculi, or hydronephrosis. The prolonged injury caused by inflammation leads to glandular metaplasia of the urothelium, eventually resulting in dysplasia and the development of adenocarcinoma. Our patient presented with a prolonged clinical history indicative of PUJ obstruction, which may have played a significant role in the etiology of his condition. Alternative hypotheses regarding mucinous neoplasms associated with the kidney suggest a potential teratomatous or a coelomic epithelial origin, owing to their remarkable similarity to ovarian mucinous neoplasms. On the other hand, the tendency of this pathological lesion to appear in conjunction with renal anomalies has encouraged researchers to suggest that these neoplasms might stem from isolated renal pelvic epithelium found within the renal parenchyma, due to developmental abnormalities.5,6 However, the existing literature does not mention any characteristic clinical symptoms associated with these tumors.7,8
No distinctive radiological characteristics are identified for these tumors. 9 According to Li et al, 1 most of these patients presented with radiological features suggestive of hydronephrosis, while a few presented with findings suggestive of calculi, pyonephrosis, or PUJ obstruction. Hence, the challenge lies in establishing a diagnosis before surgery. 1
While tumor markers are not commonly employed for screening renal neoplasms, they can serve as a valuable adjunct for identifying these tumors. Elevated levels of carcinoembryonic antigen or CA19-9 have been documented in various instances; however, these markers are neither sensitive nor specific and are observed in only about 20% of patients.3,10
Histopathological analysis plays a pivotal role in recognizing these neoplasms, and the TNM staging at the time of diagnosis profoundly influences the overall prognosis. Grossly pronounced cystic dilatation of the renal pelvis and calyces is noted, typically containing a mucous-like material. Occasionally, the walls of these cysts may be thickened, while the renal parenchyma is often found to be thinned out and atrophied. Microscopically, the cysts contain mucin, which appears eosinophilic and are lined by mucinous epithelium. Typically, these tumor cells reveal a tall and columnar morphology, with nuclei found basally showing mild to moderate atypia and a mucinous cap on their apical surface. Additionally, abundant acellular mucin pools are frequently observed within the renal parenchyma and perinephric fat. 11
Metastasis of mucinous carcinomas from primary sites such as the urachus, prostate, colon, female genital tract, appendix, stomach, and breast must be diligently excluded before the diagnosis of this uncommon tumor can be established. A notable constraint in our case report was the absence of a thorough histopathological analysis of the appendix. Appendiceal mucinous neoplasms may sometimes manifest as occult neoplasms, which can pose challenges in gross identification, and frequently, a comprehensive histological assessment of the entire appendix is requisite to rule out a mucinous adenoma or a microscopic site of rupture. 12 Nevertheless, the lack of a clinically and radiologically identifiable primary lesion within the appendix while performing nephrectomy, coupled with the absence of related pathological entities such as pseudomyxoma peritonei in our patient, suggested the kidney as the plausible primary site of origin for the neoplasm. While KRT7 and KRT20 offer constrained discrimination, the utilization of CDX2 and β-catenin as biomarkers has been investigated. In the context of colorectal adenocarcinomas, immunohistochemical profiling typically reveals KRT20 positivity with concurrent loss of KRT7 expression; however, on further subtyping of the tumor, both KRT7 and KRT20 exhibited diffuse cytoplasmic positivity, a pattern that is not characteristic of metastatic colorectal adenocarcinoma and thus renders this diagnosis less likely on histopathological assessment. While suggestive, the positive expression of CDX2 is not conclusive, given its potential positivity appreciated in both primary and metastatic adenocarcinomas. The presence of nuclear expression of β-catenin, a feature seen in roughly 80% of adenocarcinomas derived from the gastrointestinal tract, confirms a metastatic origin of the tumor.13,14
Primary renal adenocarcinomas are usually highly aggressive and are classified as high-grade tumors. Prior research has shown that their survival outcomes are characteristically poorer when evaluated against those of nonmucinous adenocarcinomas, notably those of colorectal and ovarian origin.15,16 The absence of a standardized treatment protocol necessitates a tailored approach, with early radical surgery frequently being considered the optimal initial strategy, mirroring the therapeutic strategies applied to clear cell renal carcinomas. However, the prognosis is still bleak, with over half of the patients passing away within 2 years following the surgery. Within this specific context, the effectiveness of chemotherapy remains ambiguous; the methotrexate, vinblastine, adriamycin, and cisplatin treatment regimen, often utilized for papillary adenocarcinoma, has displayed restricted efficacy. Nevertheless, research has indicated that the prognosis for renal adenocarcinomas depends upon the histopathological subtypes. Tubulovillous adenocarcinomas are acknowledged as the most aggressive variant, associated with a 5-year survival rate of <30%. Mucinous neoplasms showed a survival rate of 67%, whereas the survival rate for papillary nonintestinal tumors reached close to 100%.17,18
Conclusion
Mucinous adenocarcinomas primarily arising in the renal pelvis are highly uncommon, distinguished by obscure clinical and radiological features, which results in frequent oversight of their preoperative recognition. The presence of gelatinous fluid during any minimally invasive urological procedure should raise the suspicion of a mucinous neoplasm. Nevertheless, the definitive diagnosis relies on histopathological examination. Our observation, along with the limited comparable instances documented in the established literature, suggests that these neoplasms represent a distinct clinicopathologic entity that merits inclusion in future classification frameworks for renal pelvic tumors. Nevertheless, nephrectomy remains the primary therapeutic approach. Although these tumors have been recognized for their relatively limited response to chemotherapy, additional research is imperative to elucidate the precise impact of chemotherapy in managing this rare tumor. Moreover, further research is warranted to identify this tumor's most suitable therapeutic approach.
Image of the cut surface of the kidney showing markedly dilated renal pelvis with foci of nodular excrescences (arrow) in the wall. Photomicrographs showing (A) tumor arranged in the form of glands and villiform architecture and (B) tumor glands invading the subepithelial connective tissue. Photomicrograph showing (A) columnar tumor cells containing mucin lining the glands and villi and (B) adjacent compressed renal parenchyma showing atrophic tubules.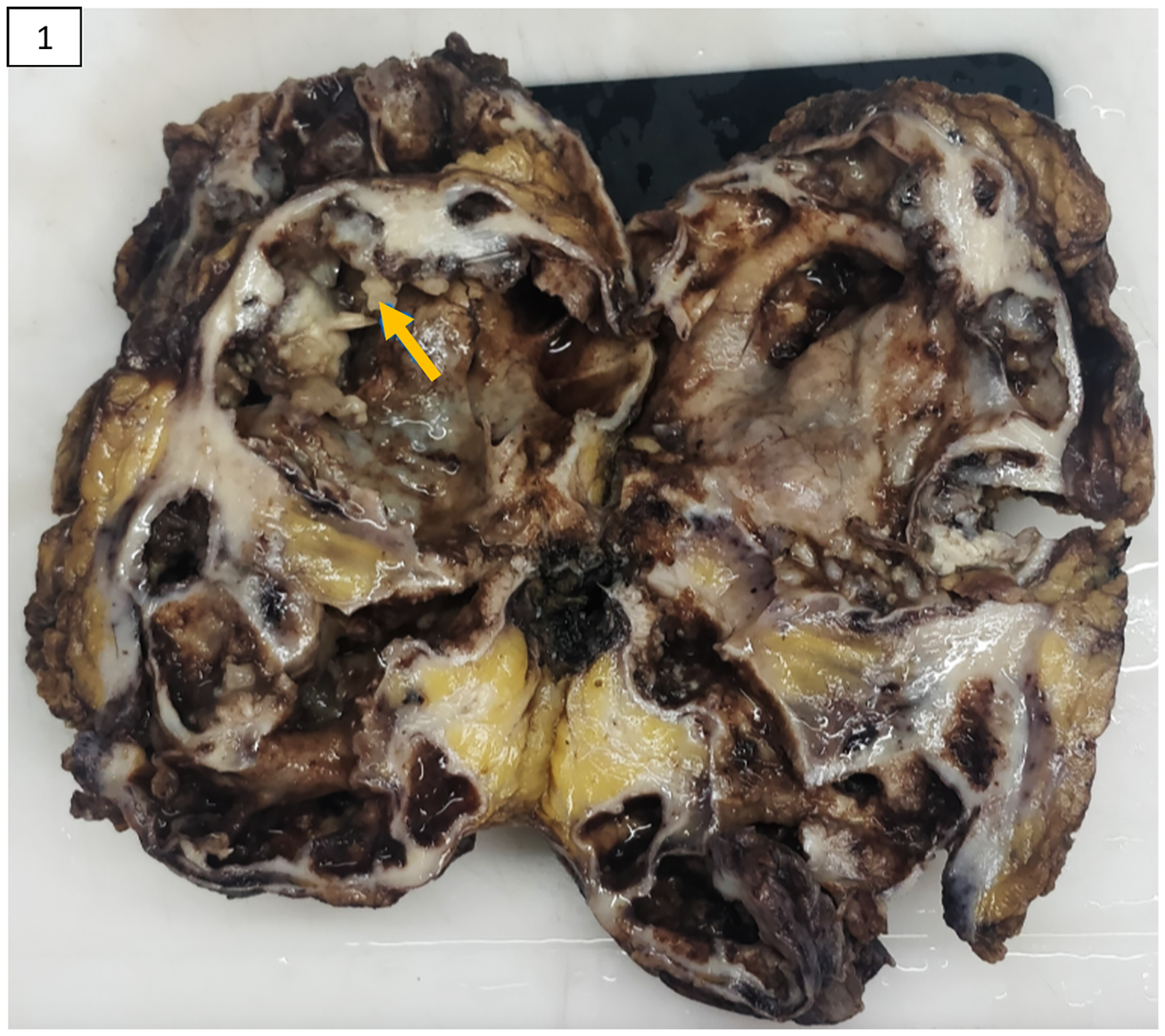
Footnotes
Author Contributions
The following authors were responsible for drafting the test, sourcing and editing clinical images, investigations, and critical revision for important intellectual content: SG, SR, and DASA. The following authors gave final approval: DASA and SP. SR was involved in drafting the manuscript and collecting data. DASA was involved in planning, coordinating, and reporting the patient. SP, TV, and CGV were involved in critically reviewing the article, collecting data, providing treatment, and caring for the patient.
Data Availability Statement
Data sharing does not apply to this article as no new datasets were generated or analyzed during the current study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Compliance
All procedures involving human participants in this study followed the institution's ethical standards, the 1964 Helsinki Declaration, and later amendments.
Trial Registration
Trial registration is waived off as this is a case report.
