Abstract
Villous adenoma (VA) of the renal pelvis is a rare intestinal epithelial neoplasm. It generally arises in elderly men and is frequently associated with chronic irritation, infection, or long-standing obstruction, most often due to nephrolithiasis or hydronephrosis. We report four previously unpublished patients diagnosed with VA of the renal pelvis. Each patient was evaluated clinicopathologically, radiologically, immunohistochemically to clarify morphologic diversity and biological behavior. We also review previously reported patients in the literature. All four patients were men, aged 45–70 years, each with common history of chronic infection or calculi. Radiological findings revealed advanced hydronephrosis and mucin-filled collecting systems (muconephrosis). Grossly, the lesions were mucinous and cystically dilated. Microscopically, the lesions from Patient 1 and 2 showed classic villiform mucinous epithelium without invasion; the lesion from Patient 3 exhibited serrated adenoma-like architecture with dysplastic nuclei; the lesion from Patient 4 demonstrated low-grade dysplasia with preserved crypt morphology. Immunohistochemically, all lesions expressed intestinal markers including KRT20, CDX2, SATB2, with variable KRT7 expression, wild-type p53 (TP53) expression. None of the lesions showed invasive growth or recurrence during follow-up. VA of the renal pelvis represents an intestinal-type lesion of upper urinary tract that develops in setting of chronic irritation and mucus accumulation and may be associated with intestinal metaplasia. Because its clinical and radiologic features often mimic inflammatory or cystic disease, histopathologic evaluation is essential for accurate diagnosis. Recognition of this rare entity is crucial to differentiate it from mucinous adenocarcinoma, as complete surgical excision is curative and long-term prognosis is excellent.
Keywords
Introduction
Villous adenoma of the renal pelvis is a rare epithelial neoplasm of the urinary tract, and is most often encountered in older men, typically presenting with nonspecific findings such as hematuria, flank pain, or mucusuria, frequently in association with hydronephrosis and nephrolithiasis.1,2 On gross examination, these lesions are often mucin-producing and may lead to marked cystic dilatation of the collecting system, resulting in so-called mucus-filled kidney or muconephrosis (ie, mucus-filled dilatation of the collecting system). 3 Since their first recognition, only a small number of patients have been reported in the literature, and most were incidentally discovered during evaluation or surgery for presumed inflammatory or obstructive renal disease. 4
The development of villous adenoma in the renal pelvis is believed to represent a reactive process that progresses through a metaplasia–dysplasia sequence. Chronic irritation, infection, and long-standing obstruction can induce intestinal-type metaplasia of the urothelium, which may subsequently undergo dysplastic change.1,5 Microscopically, these tumors exhibit villiform or papillary projections lined by columnar mucinous epithelium with goblet cells and most often low-grade dysplasia. Immunohistochemically, they commonly express intestinal markers such as KRT20, CDX2, SATB2, and CEA, with variable KRT7 expression. 6 Although the majority behave in a benign fashion, occasional patients with high-grade dysplastic or even infiltrative transformation or coexistence with mucinous adenocarcinoma have been documented. 5
Given the rarity of this entity, information regarding its clinical spectrum, histopathologic diversity, and biological behavior is scarce. We performed an extensive literature review and present four additional patients of villous adenoma arising in the renal pelvis, each with distinctive morphological and immunophenotypic characteristics. This series aims to enhance current understanding of the clinical presentation, histopathological features, and diagnostic considerations of this uncommon neoplasm.
Materials and Methods
This retrospective study included four patients diagnosed with villous adenoma of the renal pelvis, identified from institutional pathology archives. Inclusion was based on the availability of complete clinical, radiological, histopathological, and follow-up data. Clinical information and imaging studies, including ultrasonography, computed tomography (CT), and dimercaptosuccinic acid (DMSA) renal scintigraphy, were reviewed to assess renal pathology and to exclude evidence of a concurrent gastrointestinal primary tumor or contiguous spread to the urinary tract. All patients underwent nephrectomy with or without ureterectomy according to clinical indications, and written informed consent was obtained.
Resected specimens were fixed in 10% neutral-buffered formalin and examined grossly, with documentation of tumor size, macroscopic features, mucin accumulation, and associated calculi. Representative sections were processed for hematoxylin and eosin (H&E) staining, and all patients were extensively sampled to exclude invasive growth. Immunohistochemical studies were performed on selected sections using an automated platform, applying markers of intestinal differentiation and urothelial lineage; detailed antibody information is provided in Supplementary Table 1. p53 (TP53) immunohistochemistry was interpreted using pattern-based criteria, with a wild-type (non-aberrant) expression pattern defined by heterogeneous nuclear staining without diffuse overexpression or complete loss.
Results
Patient 1
A 55-year-old man presented with unexplained weight loss. During evaluation, right renal calculi were incidentally detected. The patient did not report flank pain or hematuria. Ultrasonography revealed a poorly defined right renal contour measuring approximately 76
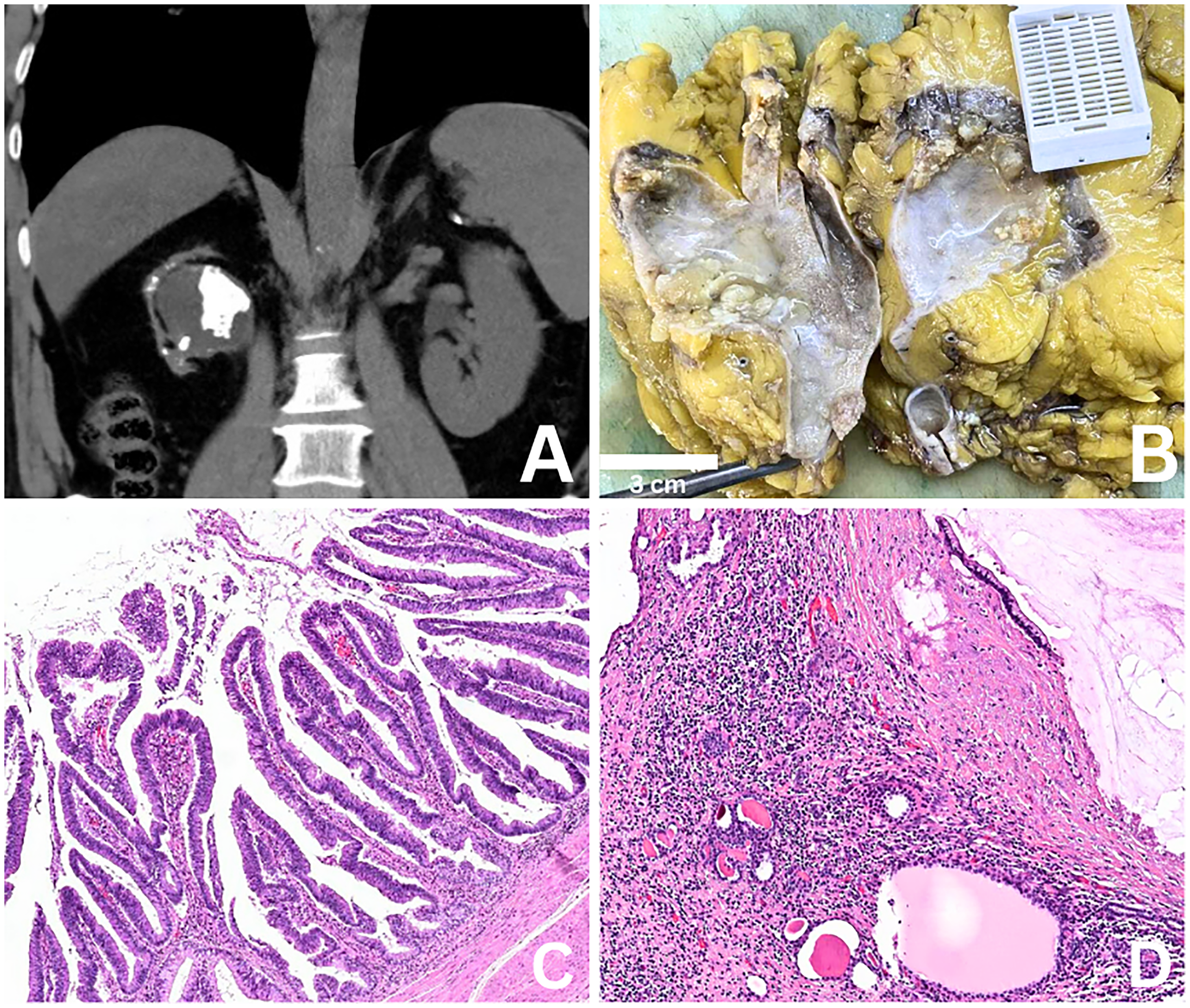
(A) Contrast-enhanced CT showing a severely hydronephrotic kidney with intrapelvic hyperdense material consistent with mucin accumulation and associated calculi. (B) Gross nephrectomy specimen demonstrating a markedly dilated pelvicalyceal system filled with abundant mucinous material and irregular villous mucosal projections. (C) Villous architecture composed of elongated papillary projections lined by intestinal-type columnar epithelium. (D) Extravasated mucin pools within the adjacent renal parenchyma accompanied by a fibroinflammatory reaction.
Patient 2
A 63-year-old man underwent right simple nephrectomy for a non-functioning kidney. Radiological evaluation demonstrated marked dilatation of the collecting system with a smooth, markedly expanded configuration, consistent with advanced hydronephrosis and mucin accumulation (Figure 2A). No radiological findings suggestive of a gastrointestinal primary tumor or secondary involvement of the renal pelvis were identified. Gross examination revealed a 25 × 15 × 12 cm cystic specimen with gray-white, mucinous, and papilliform areas on sectioning (Figure 2B). Microscopically, the pelvicalyceal system was markedly dilated and filled with abundant mucinous material. The lining epithelium displayed intestinal-type villous architecture composed of columnar cells with apical mucin and basally located nuclei (Figure 2C, D). There was no significant nuclear atypia, mitotic activity, or stromal invasion. The surrounding renal parenchyma was atrophic with interstitial fibrosis, consistent with chronic obstructive changes. Immunohistochemistry (IHC) showed nuclear positivity for CDX2, cytoplasmic positivity for KRT20, and PAX8, and a wild-type (non-aberrant) p53 (TP53) expression pattern. Alcian blue (pH 7.17) staining confirmed abundant acidic mucin production. The findings supported a diagnosis of villous adenoma of the renal pelvis without dysplasia or invasion.
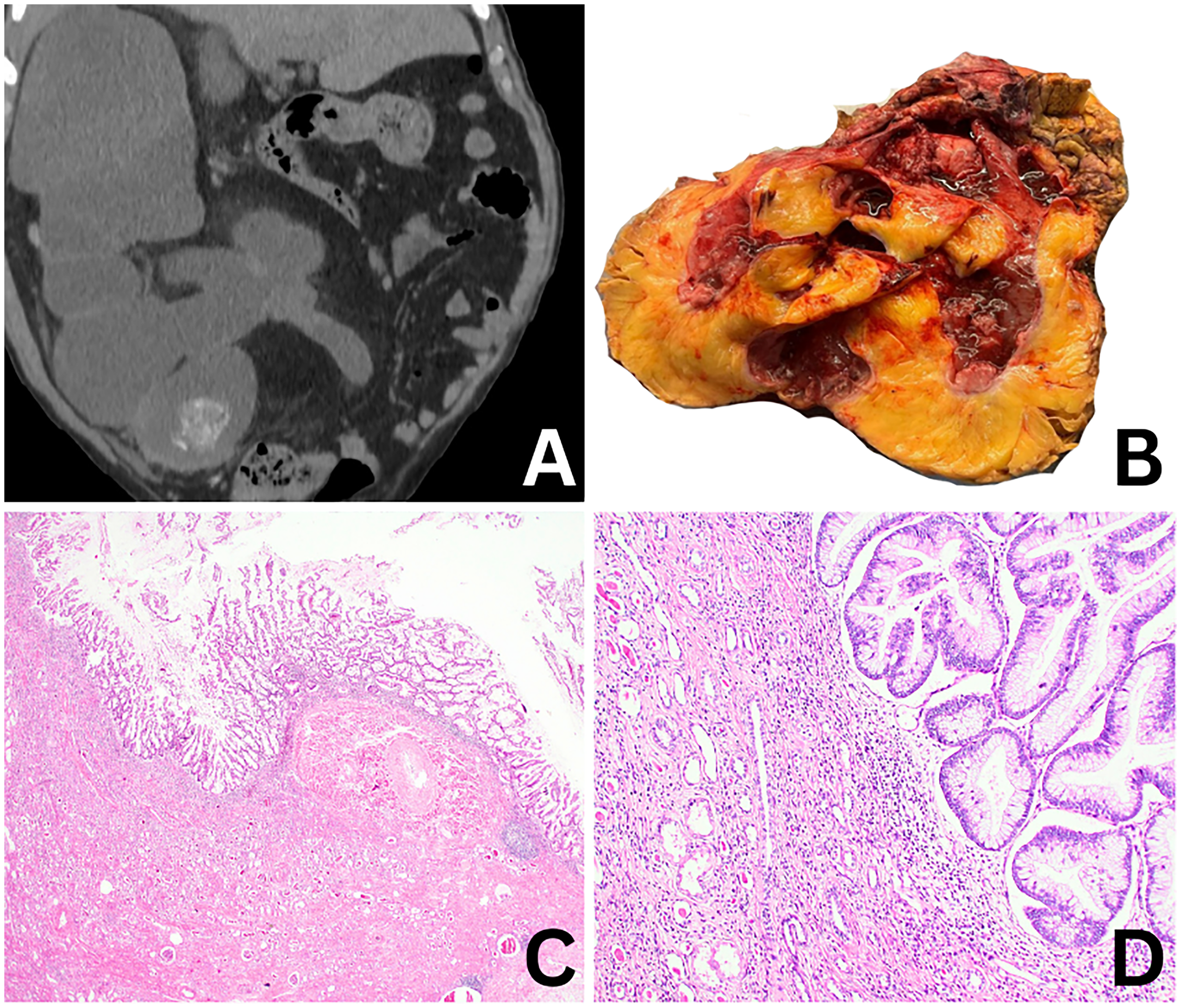
(A) Radiological imaging revealing a non-functioning kidney with advanced hydronephrosis and cystic dilatation of the collecting system. (B) Gross appearance of a cystically dilated kidney with gray-white mucinous and papilliform areas on cut section. (C) Villous adenoma characterized by prominent papillary projections lining the pelvicalyceal system. (D) Tall columnar epithelial cells with apical mucin and basally located nuclei without significant cytologic atypia or stromal invasion.
Patient 3
A 45-year-old man presented with right flank pain. CT demonstrated marked ureteral wall thickening, grade IV hydronephrosis, cortical thinning, and multiple cortical cysts, the largest measuring 13 cm (Figure 3A). The nephrectomy specimen measured 17 × 14 × 8 cm and included an 11.5 cm ureter. The pelvicalyceal system was markedly dilated and cystic. Within the renal pelvis, a 9 cm beige, papillary, exophytic mass extending into the ureter was identified. Microscopically, the tumor exhibited protuberant villiform growth with slit-like serrations, lined by pseudostratified columnar cells showing eosinophilic cytoplasm and dark, pencillate nuclei. Foci resembling serrated adenoma were present within the lesion (Figure 3B, C, D). The entire tumor was sampled, and no invasive component was identified. Immunohistochemically, the tumor showed diffuse nuclear positivity for CDX2, SATB2, and β-catenin (CTNNB1), cytoplasmic positivity for KRT20, and focal villin expression. KRT7, GATA3, estrogen receptor (ER) and vimentin were negative. p53 (TP53) immunohistochemistry demonstrated a wild-type (non-aberrant) expression pattern. The Ki-67 (MKI67) proliferation index was elevated within dysplastic epithelium. Despite the intestinal-type morphology, comprehensive clinical and radiological assessment did not demonstrate evidence of a primary gastrointestinal neoplasm or direct extension to the urinary tract. The overall findings supported the diagnosis of villous adenoma of the renal pelvis with serrated adenoma-like areas, extending into the ureter and showing intestinal differentiation without invasion. Detailed information on the immunohistochemical antibodies, including clones, dilutions, and manufacturers, was summarized in Supplementary Table 1.
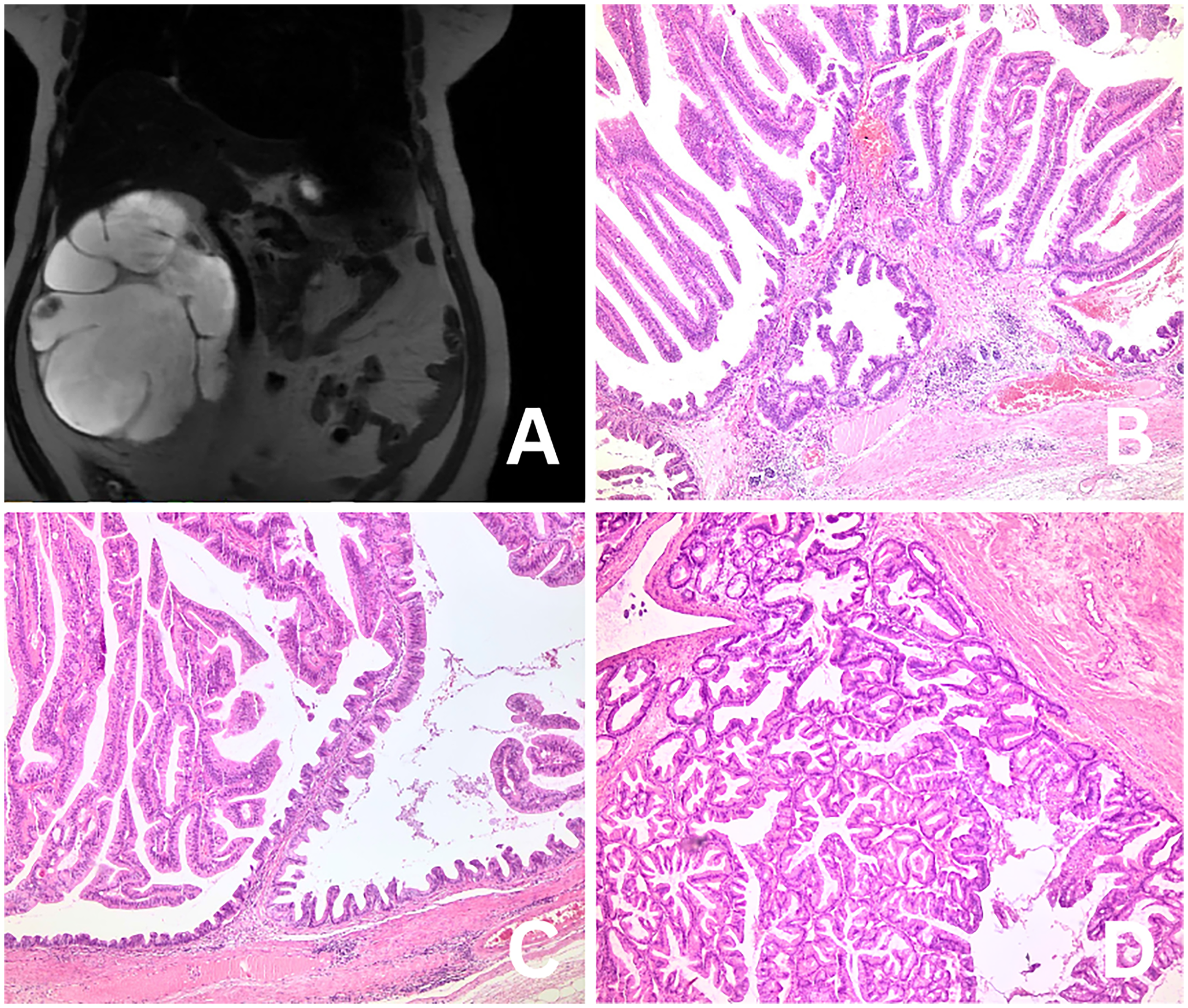
(A) CT image demonstrating marked hydronephrosis with cortical thinning and a large intrapelvic mass extending toward the ureter. (B) Prominent villous growth with slit-like serrations consistent with serrated adenoma-like architecture. (C) Pseudostratified columnar epithelium composed of cells with eosinophilic cytoplasm and elongated, pencillate nuclei. (D) Complex villous and serrated glandular architecture without evidence of invasive growth.
Patient 4
A 70-year-old man (ex-smoker) presented with a non-functioning left kidney. He reported hematuria and intermittent purulent urine for several weeks prior to surgery. Imaging revealed a completely cystic, mucus-filled kidney measuring 15 × 8 × 5 cm with multiple calculi (Figure 4A). DMSA scintigraphy demonstrated absence of cortical activity in the left kidney. The patient underwent simple nephrectomy, followed by ureterectomy and bladder biopsy. The latter revealed congested urothelial mucosa without dysplasia. Urine cytology was negative, and systemic imaging excluded additional primary or metastatic lesions. There was no clinical or radiological evidence of a gastrointestinal primary tumor or adjacent gastrointestinal involvement. The patient remained disease-free on follow-up. Grossly, the nephrectomy specimen was entirely cystic, filled with mucinous material, consistent with mucus-filled kidney (muconephrosis). Microscopically, the lesion exhibited epithelial finger-like projections composed of fibrovascular cores lined by dysplastic columnar epithelium showing villous architecture. The epithelium displayed crowded pseudostratification with elongated nuclei occupying the basal half of the cytoplasm. Low-grade dysplasia was the defining feature, characterized by minimal atypia, absence of pleomorphism, and rare mitotic figures (Figure 4B, C, D). Mild polarity loss and minimal stratification were present, whereas cribriform or complex glandular formations were absent. The crypts closely resembled normal colonic mucosa. No evidence of high-grade dysplasia or invasion was found. These findings were diagnostic of villous adenoma of the renal pelvis with low-grade dysplasia, most likely developing in the setting of chronic irritation and long-standing nephrolithiasis. The surrounding urothelial mucosa showed chronic inflammatory/obstructive changes, consistent with a long-standing irritation background.
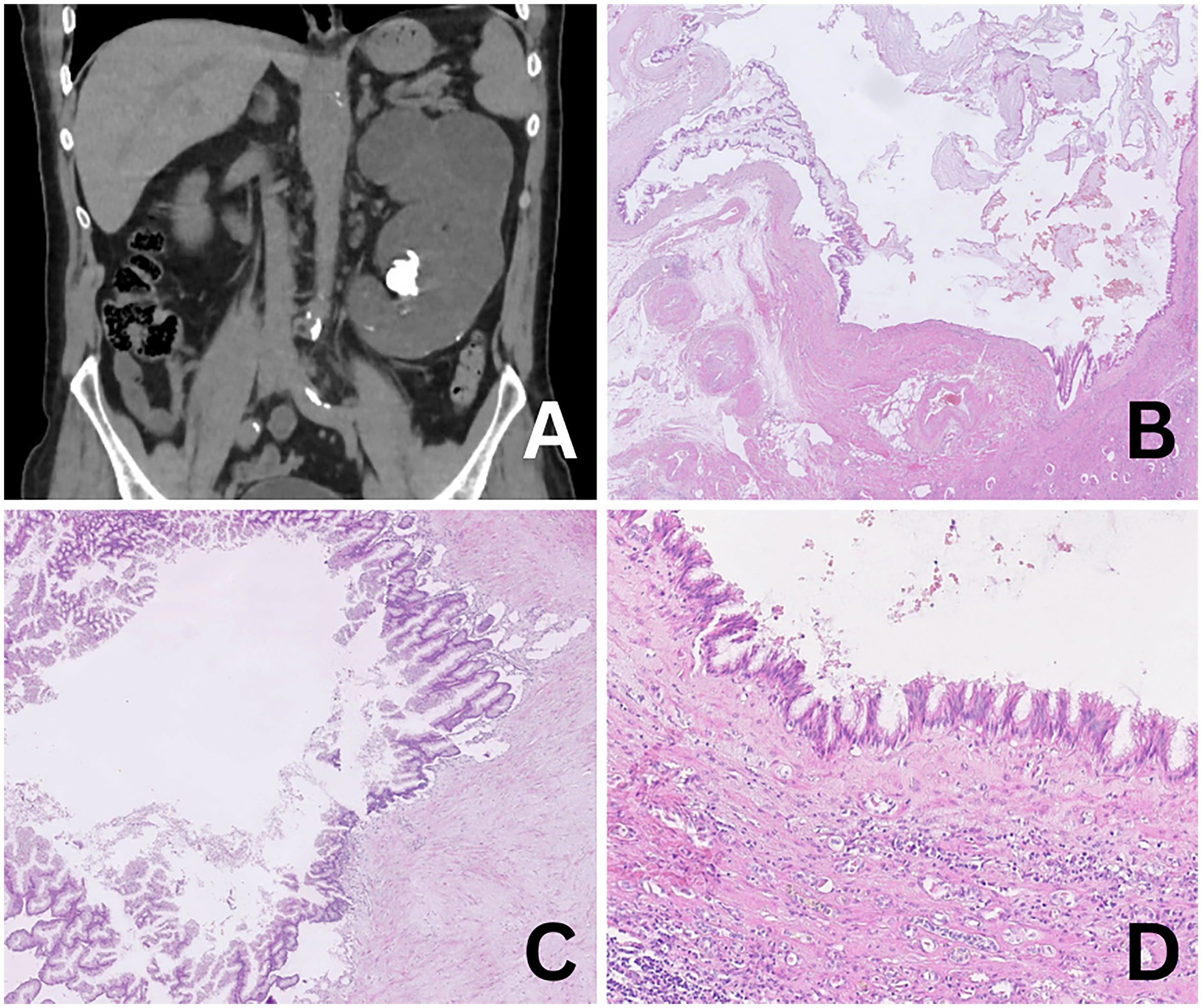
(A) Radiological imaging showing a completely cystic, mucus-filled non-functioning kidney associated with nephrolithiasis. (B) Villous projections lined by dysplastic intestinal-type epithelium within a markedly dilated pelvicalyceal system. (C) Preserved crypt architecture with orderly villous configuration. (D) Low-grade dysplasia characterized by mild nuclear stratification, preserved polarity, and absence of high-grade features or invasion.
Discussion
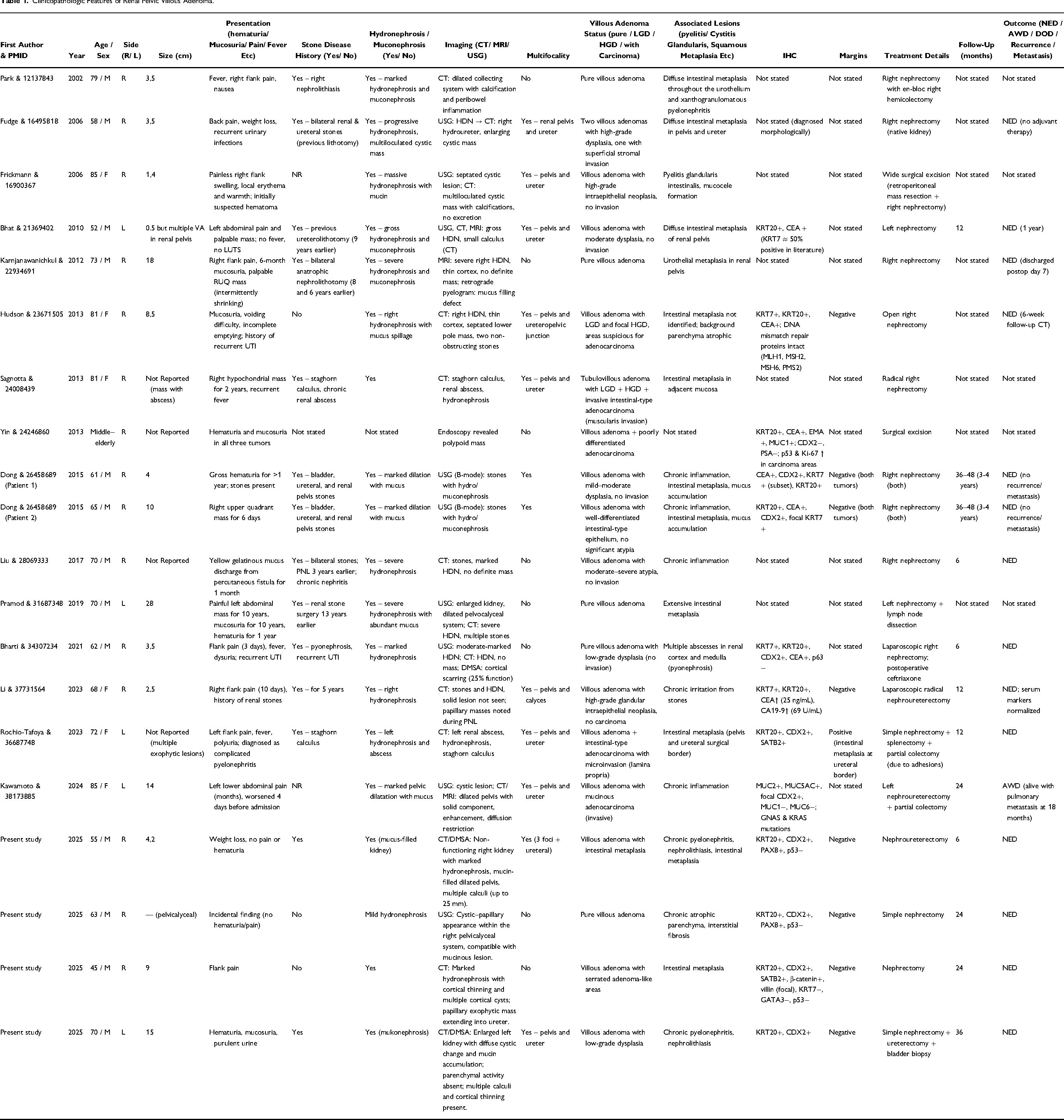
Villous adenoma of the renal pelvis is an exceedingly uncommon epithelial neoplasm of the urinary tract, with fewer than fifty patients described to date. It predominantly affects older men and is usually discovered incidentally or in association with hydronephrosis, nephrolithiasis, or chronic infection.7–9 Similar to previous reports, all four patients in our series were male and had a background of chronic inflammatory or obstructive pathology. Radiologically, severe hydronephrosis and mucus-filled cavities—a condition referred to as muconephrosis—were evident in three patients, mirroring the imaging findings emphasized by Park et al, Karnjanawanichkul et al, and Bharti et al 2,7,10 The clinicopathologic features of previously published patients are summarized in Table 1.
Clinicopathologic Features of Renal Pelvic Villous Adenoma.
Histologically, our patients encompassed the recognized morphological spectrum of villous adenoma of the renal pelvis. In two patients (Patients 1 and 2), classic villiform mucinous epithelium without invasion was present, similar to the lesions described by Hudson et al and Bhat and Chandran.8,11 The lesion from Patient 3 displayed serrated adenoma-like architecture with slit-like serrations and pencillate nuclei, resembling the dysplastic spectrum reported by Sagnotta et al, who proposed a stepwise evolution from intestinal metaplasia to dysplasia and, in rare instances, adenocarcinoma. 12 The lesion from Patient 4 exhibited low-grade dysplasia with preserved crypt architecture, closely paralleling the early-stage lesions documented by Liu et al 9 None of the four lesions revealed an invasive component, in keeping with the predominantly benign course noted in earlier series.5,13
Glandular lesions of the urinary tract, including villous adenoma and mucinous adenocarcinoma, have been proposed to arise through a metaplasia–dysplasia–carcinoma sequence in the setting of chronic irritation, infection, and long-standing obstruction. Prior reports have described intestinal metaplasia of the urothelium as a putative precursor, with progressive architectural and cytologic atypia and, in rare patients, coexistence with or evolution to adenocarcinoma.1–4,6–15 The presence of extensive intestinal metaplasia involving the renal pelvis and ureter (notably in Patient 1), together with low-grade dysplasia within the villous adenoma in Patient 4, is consistent with a proposed metaplasia–dysplasia continuum in glandular lesions of the upper urinary tract. Importantly, all patients were entirely submitted for histologic evaluation, and no invasive adenocarcinoma was identified.
Immunohistochemically, the tumors in our cohort consistently expressed markers of intestinal differentiation such as KRT20, CDX2, and SATB2, with variable KRT7 expression and a wild-type (non-aberrant) p53 (TP53) expression pattern—findings concordant with prior studies.1,6 The presence of diffuse intestinal metaplasia, chronic pyelonephritis, and mucin extravasation in two of our patients supports the concept that villous adenoma develops in a chronically inflamed milieu.14,15 Importantly, no recurrence or malignant transformation was identified during follow-up, consistent with the favorable outcomes previously reported.2,10 Altogether, our findings reinforce that villous adenoma of the renal pelvis represents a metaplasia-driven, indolent neoplasm that should be distinguished from mucinous adenocarcinoma through careful histopathologic and immunophenotypic assessment.
Although villous adenoma of the renal pelvis is often discussed through a metaplasia–dysplasia framework, molecular data for upper urinary tract patients remain very limited. In the published renal pelvis literature, only rare reports have included molecular testing; notably, a patient of villous adenoma with associated mucinous adenocarcinoma arising in a muconephrotic kidney demonstrated GNAS and KRAS mutations, suggesting that at least a subset may share oncogenic events seen in intestinal-type mucinous pathways. 5 In our series, we did not perform molecular profiling; however, the consistent background of chronic obstruction/inflammation and the spectrum from intestinal metaplasia to dysplasia support an irritation-driven process in which sustained injury, mucin stasis, and inflammatory signaling may create a permissive environment for clonal expansion and, in rare instances, acquisition of driver alterations. We have clarified this limitation in the manuscript and highlighted molecular characterization as an important next step for future multi-institutional cohorts.
Conclusion
Villous adenoma of the renal pelvis is a rare, benign epithelial tumor that typically develops on a background of chronic irritation, infection, or obstruction. Its nonspecific clinical and radiologic findings often mimic inflammatory conditions, underscoring the importance of histopathologic evaluation for accurate diagnosis. Recognition of this entity is essential to distinguish it from mucinous adenocarcinoma, as complete surgical excision is curative and the prognosis is excellent. Future studies incorporating targeted sequencing of larger patient series are warranted to better define the molecular landscape of renal pelvic villous adenoma and its rare progression to adenocarcinoma.
Supplemental Material
sj-docx-1-ijs-10.1177_10668969261422253 - Supplemental material for Villous Adenoma of the Renal Pelvis: Clinicopathologic Spectrum and Diagnostic Considerations
Supplemental material, sj-docx-1-ijs-10.1177_10668969261422253 for Villous Adenoma of the Renal Pelvis: Clinicopathologic Spectrum and Diagnostic Considerations by Busra Yaprak Bayrak, Meltem Oznur, Suheda Zeynep Seday, Meryem Aydın, Merve Meryem Kiran, Suleyman Altıntas, Nil Akcalı and Mahmut Akgul in International Journal of Surgical Pathology
Footnotes
Ethics Approval
Ethical approval for this study was obtained from the local ethics committee for non-interventional clinical research (GOKAEK-2026/86). Written informed consent was obtained from all patients included in this study.
Author Contributions
All authors collected the data. BYB, MA drafted and edited the manuscript, participated in the study design and coordination. All authors read and approved the final manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
All data generated or analyzed during this study are included in this published article. Data available on request due to privacy/ethical restrictions.
Supplemental Material
Supplemental material for this article is available online.
