Abstract
A 39-year-old woman originally from Northeast Africa sought medical attention for positional dyspnea. Computed tomography of the chest revealed an 8.5 cm hypodense anterior mediastinal mass with peripheral calcifications that raised a wide differential diagnosis including infectious and neoplastic lesions. Following surgical resection, a large cavitary necrotizing and calcified granuloma involving the thymus was identified on histopathological examination. The changes were associated with parasitic eggs that based on their morphology suggested infection due to trematode species. The diagnosis was further corroborated by identification of the increased IgG titers for Schistosoma species (ELISA Kit, NovaTec). The patient's symptoms improved following surgery and praziquantel therapy. This unique presentation emphasizes an unusual manifestation of schistosomiasis that can pose a diagnostic challenge, especially in non-endemic regions. It suggests that mediastinal involvement by schistosomiasis is likely due to an ectopic deposition of the parasitic eggs within a definitive host. Suspicion for schistosomiasis should be heightened based on patient demographics and travel to endemic areas.
Background
Schistosomiasis is a chronic parasitic infection that globally affects 250 million people, predominantly in distinct endemic areas (Sub-Saharan Africa, South America, Asia, and the Middle East). 1 Due to the absence of a suitable intermediate host, the disease is rarely seen in North America. 2 However, schistosomiasis can present in North American countries, particularly in patients that emigrate from or travel to endemic regions. The disease most commonly affects the bladder, intestine, hepatobiliary system, and lungs. 3 Despite the high prevalence of pulmonary involvement, mediastinal presentations of schistosomiasis are exceptionally rare. To the best of our knowledge, only 3 reports of mediastinal schistosomiasis have been documented in English language literature.4–6 Here we provide a detailed description of schistosomiasis presenting as a large mediastinal mass and contrast it with other types of mediastinal presentations in prior publications.
Case Presentation
A 39-year-old woman who recently immigrated from Northeast Africa to the United States sought medical attention for positional dyspnea. Computed tomography (CT) of the chest revealed an 8.5 cm hypodense anterior mediastinal mass with peripheral calcifications abutting the left atrial appendage and the pulmonary artery trunk without evidence of invasion (Figure 1). Clinical differential diagnosis at the time included infection (tuberculosis, histoplasmosis), cystic teratoma, infected foregut cyst, thymoma, and thymic carcinoma. Human immunodeficiency virus serology, QuantiFERON, sputum smears, and microbiological cultures for acid fast bacilli were negative. As initial percutaneous biopsy was inconclusive, a sternotomy for resection of the mass, which required an en bloc thymectomy and wedge resection of the left upper lobe of lung was performed. Gross examination showed a well-circumscribed mass-lesion measuring 7.6 × 4.8 × 4.1 cm with attached thymus and left upper lung lobe wedge. On cross-sectioning, the mass had mostly necrotic surfaces with a rim of preserved sub-capsular tissue (Figure 2A). Histopathologic examination revealed a large cavitary necrotizing granuloma abutting the left upper lobe of lung and the thymus (Figure 2B and C). There were occasional necrotizing granulomas within lung parenchyma (Figure 2D). Within necrotic debris there were degenerated parasitic eggs that lacked discernable morphological features (Figure 2E). Following serial sectioning in better preserved tissue there were parasitic eggs that based on their morphology suggested infection due to trematode species. At higher magnification, the eggs displayed a clearly visible slightly brown cell wall and partially preserved miracidium (Figure 2F). On examination of multiple histological step sections, no lateral or terminal spike or operculum was identified. The eggs showed no birefringence under polarized light. They measured up to 100 mkm in length and up to 40 mkm in width. However, these measurements likely underestimated the size as no complete longitudinal section of an egg was encountered on serial sections. Ziehl-Nielsen stain identified no acid-fast bacilli. Grocott's methenamine silver stain revealed no fungal organisms. The diagnosis was further corroborated by increased IgG titers for Schistosoma species (ELISA Kit, NovaTec Immunodiagnostica). Praziquantel treatment was initiated, and the patient's symptoms resolved within the following 4 weeks.
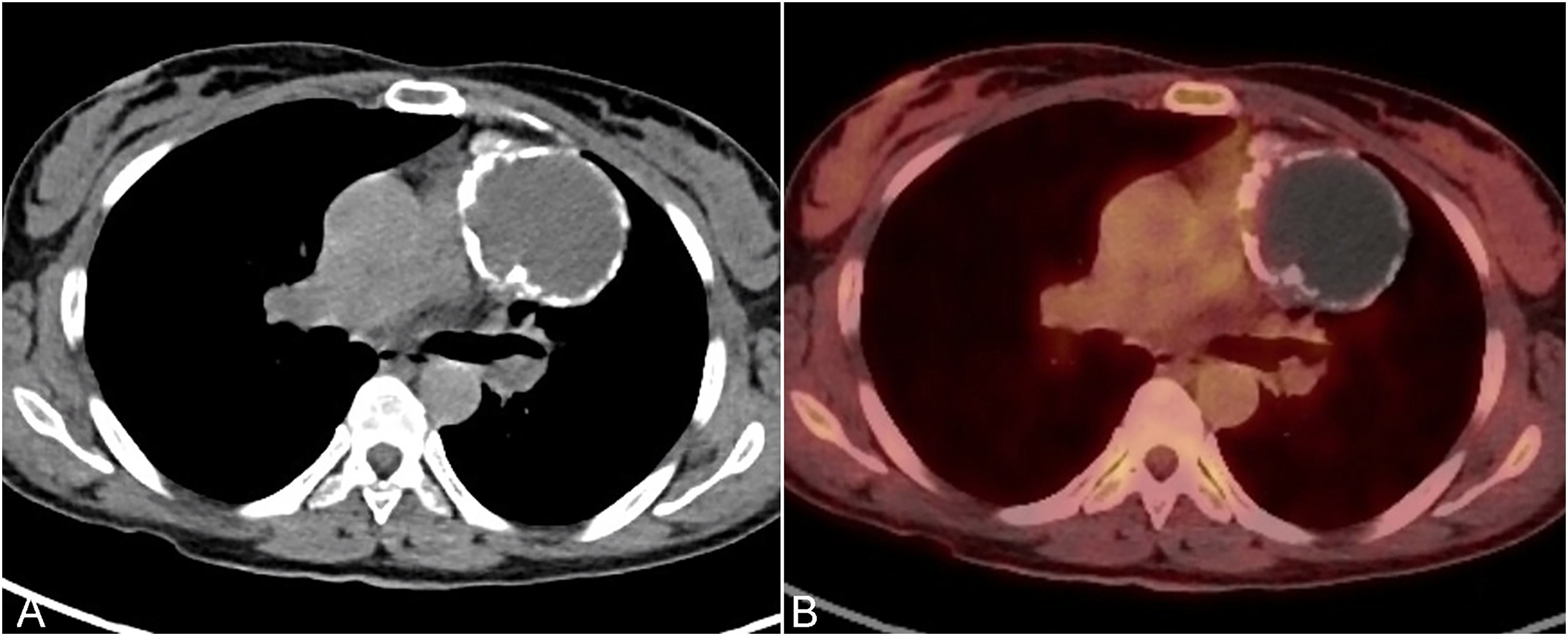
Imaging studies. (A) Contrast enhanced computed tomography imaging at the level of the carina shows an 8.5 × 6.8 cm peripherally calcified hypodense mediastinal mass that abuts the left atrial appendage and pulmonary trunk without evidence of invasion. (B) Corresponding fused positron emission tomography/computed tomography image shows a large partially calcified photopenic mass.
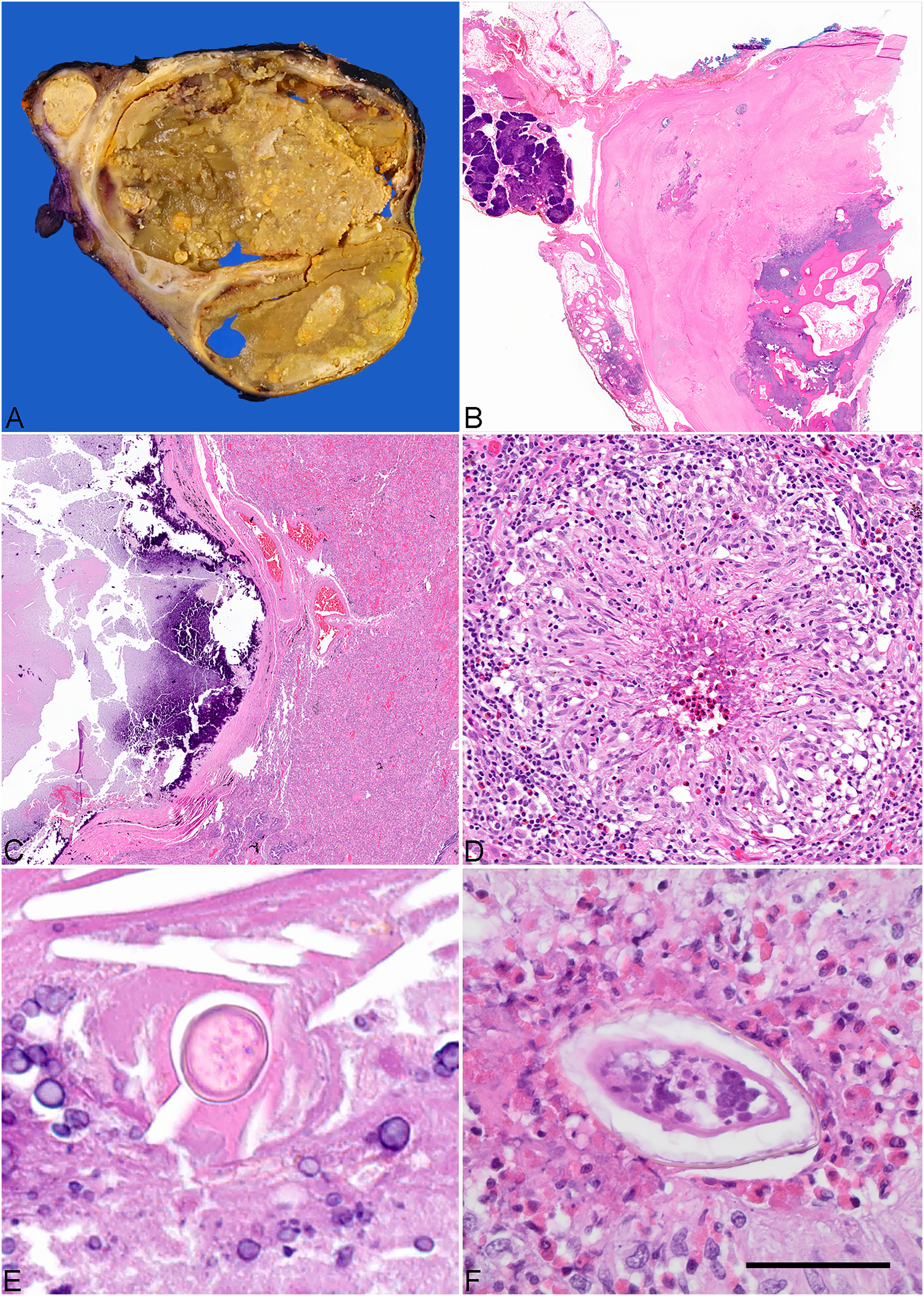
Pathological findings. (A) Cross-section of the resected 7.6 × 4.8 cm mediastinal mass shows central cavitation containing necrotic material, gross examination after overnight formalin fixation. (B) On light microscopic examination the mass is surrounded by thymic tissue and has thick fibrous wall with foci of metaplastic ossification, H&E original magnification ×2. (C) The mass abuts lung and contains acellular necrotic debris with calcifications, H&E original magnification ×2. (D) Within the mass and lung parenchyma there are necrotizing granulomas with eosinophils, H&E original magnification ×20. (E) Within necrotic debris there are degenerated parasitic eggs, H&E original magnification ×40. Occasional granulomas on serial sectioning reveal better preserved eggs that show slightly brown cell wall and miracidium with small nuclei, 40 mkm scale bar, H&E original magnification ×60.
Discussion
Schistosomiasis, or bilharziasis, is caused by species of trematodes in the Schistosoma genus. Human infection occurs after exposure to contaminated freshwater containing the parasite's intermediate host, infected snails.3,7 Excreted cercariae penetrate the human skin and then in a larval form enter the vascular system and migrate through the lungs. After maturation in the mesenteric or urogenital vasculature, the adult flukes release eggs that become trapped in various tissues. 8 The acute form of the disease is termed Katayama syndrome and is typically a flu-like syndrome with additional allergic symptoms including peripheral blood eosinophilia. 3 This acute phase is due to a hypersensitivity reaction to the worm migrating through the vasculature, approximately 2 to 8 weeks after infection. 3 Each major Schistosoma species has a prototypical organ it afflicts. Schistosoma mansoni and Schistosoma japonicum deposit eggs in the mesenteric venous system, while Schistosoma haematobium infects the vesical venous plexus. 9 The chronic forms of the disease are the result of the fibrosis and granulomatous inflammation triggered by the immune response to the parasitic eggs in the hepatobiliary or urogenital system, respectively. 10 However ectopic deposition of Schistosoma eggs can lead to the disease in unexpected sites, such as the mediastinum, gallbladder, ovary, testis, and cervix.4–6,11–13 Cerebral schistosomiasis is known to be more commonly associated with Schistosoma japonicum infections.7,14
Pulmonary Schistosomiasis
Clinical manifestations of pulmonary schistosomiasis are highly variable. Lung migration is estimated to last over multiple weeks after exposure. 8 The initial pulmonary manifestations occur when the metacercariae migrate through the lungs, producing mild and/or allergic symptoms. In cases of heavy infestation, granulomatous response to the parasite affects parenchymal or vascular compartments. The presence of a significant vascular component may ultimately lead to pulmonary hypertension. Pulmonary infarcts may result from adult worms occluding peripheral pulmonary artery branches.15,16 Chest CT during acute pulmonary schistosomiasis can include ground-glass opacities, small miliary nodules, reticular nodules, and pneumonic consolidations, while during the chronic phase, there are signs of pulmonary hypertension and fibrosis.15,17 The acute phase is commonly inconsequential and most of the host morbidity is due to granulomatous tissue reaction to the parasitic eggs in the chronic phase. 7 The species that most commonly affect the lung are Schistosoma mansoni and Schistosoma haematobium.8,18 Ova morphology can allow distinction of Schistosoma from other trematode infections and provide evidence to identify the species. 3 In tissue, if well preserved, Schistosoma eggs have a clearly visible cell wall that may contain slightly brown pigment, particularly in the area of spine. Serial sections maybe needed to identify the spine. The wall frequently collapses partially due to H&E processing, which creates spine-like artifacts. True spines are well defined and thickened, particularly the lateral spine of Schistosoma mansoni. In contrast to Paragonimus that can be seen in this location, Schistosoma eggs lack an operculum, their cell wall is not birefringent under polarized light and, if well preserved, the eggs display an internal nucleated miracidium.3,19,20 The Schistosoma eggs identified in urine or feces measure up to 170 to 175 mkm in length and 40–70 mkm in width. 3 However, formalin fixation can lead to tissue shrinkage, while tissue necrosis and eggs degeneration can make accurate size determination in tissue specimens impossible. Since Schistosoma mansoni and Schistosoma haematobium ova are relatively the same shape and size, the distinction between them in tissue and body fluids is made based on the position of the spine. The presence of a lateral spine is considered a diagnostic feature of Schistosoma mansoni, and the presence of a terminal spine is a feature of Schistosoma hematobium.3,21
Mediastinal Schistosomiasis
Although pulmonary involvement by Schistosoma species is common, mediastinal schistosomiasis is exceedingly rare. Our search of the English language literature using the key words “schistosomiasis,” “mediastinum,” “thymus,” “paraesophageal,” and “retrocardiac,” and dating back to 1960 revealed only 3 reports (Table 1). The most recent publication by Guermazi et al describes an unusual presentation of chronic hepatobiliary schistosomiasis in a 48-year-old Caribbean woman. 5 Following more than 15 years after the initial diagnosis, in the setting of liver cirrhosis and portal hypertension, she developed a lower mediastinal mass. Utilizing contrast enhanced CT and Doppler sonography technics, it was established that the mass was comprised of giant paraesophagial varices. While the report attributes her illness to hepatosplenic schistosomiasis due to Schistosoma mansoni, it lacks information on the diagnostic modalities including tissue diagnosis. Al-Fawaz et al reported mediastinal schistosomiasis in a 13-year-old girl from southwest Saudia Arabia. 4 Her symptoms included weight loss, fevers, hematuria, and abdominal pain. Her chest CT revealed mediastinal lymphadenopathy with pulmonary parenchymal opacities. A lung biopsy with mediastinal lymph node excision performed to exclude a neoplastic process revealed Schistosoma ova surrounded by granulomatous inflammation within the pulmonary parenchyma and reactive mediastinal lymphadenitis. Based on the ova morphology in the urine, the authors concluded the disease was due to Schistosoma haematobium. Nyman et al reported a retrocardiac mediastinal mass in a 60-year-old man from southwest Saudia Arabia. 6 With the help of different imaging modalities including ultrasonography, they defined the lesion as representing paraesophageal varices associated with periportal fibrosis. Due to positive serology and involvement of the hepatobiliary system, Schistosoma mansoni was presumed to be the most likely species. 22 Our patient was born in Eritrea and spent time throughout her life in different parts of Northeast Africa. Eritrea is an endemic country for both Schistosoma haematobium and Schistosoma mansoni. 23 The population-adjusted prevalence (in school-aged children) of both Schistosoma species in Eritrea was estimated to be 8.8% with Schistosoma mansoni having a slightly higher prevalence compared to Schistosoma haematobium (6.8% vs 2.2%, respectively). 23 The diagnosis of schistosomiasis was corroborated in our patient by identifying IgG-class antibodies to Schistosoma mansoni; however, due to known cross-reactivity to other Schistosoma species, the testing does not allow for a definitive speciation. The patient had no documented clinical signs of hepatobiliary or urinary disease to suggest either species as a cause of the disease. From an epidemiological perspective, the patient had a slightly higher risk to be infected with Schistosoma mansoni; however, the tissue assessment of the ova revealed a lack of the lateral or terminal spine. The lack of a visible spine does not rule out one species or another and the infecting species remains unknown. Direct involvement of the mediastinum by a large cavitary calcified granuloma containing Schistosoma eggs represents a unique aspect of our patient. The space-occupying lesions in the prior reports were reactive in nature comprising either mediastinal lymphadenitis or paraesophageal varices associated with portal fibrosis. Necrotizing granulomas were evident on histological examination of the lung in our patient; however, the presence of a large cavitary mass within the mediastinum is distinctly unusual. It likely represents an exuberant inflammatory reaction to the parasitic eggs that reached the mediastinum and thymus via systemic circulation. As no adult worms were identified, the possibility of an accidental entrapment of the parasite with direct release of the eggs in the mediastinum seems unlikely.
Examples of Mediastinal Schistosomiasis.
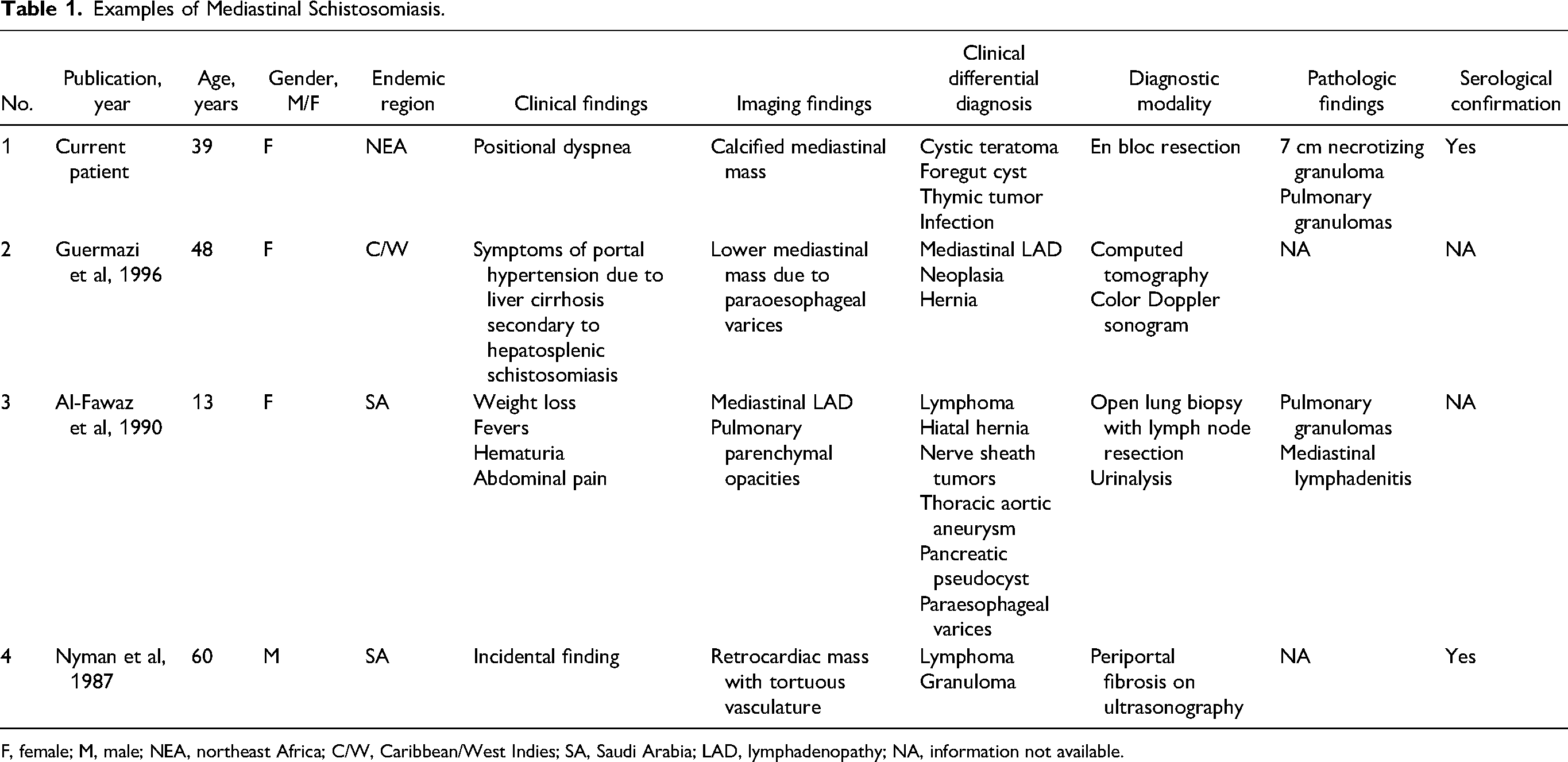
F, female; M, male; NEA, northeast Africa; C/W, Caribbean/West Indies; SA, Saudi Arabia; LAD, lymphadenopathy; NA, information not available.
In conclusion, this report emphasizes an unusual and rare manifestation of schistosomiasis that can pose a diagnostic challenge, especially in non-endemic regions. In rare occasions of ectopic deposition of Schistosoma eggs within a definitive host, schistosomiasis can present as a mass forming lesion within the mediastinum. Schistosomiasis should be entertained in the differential diagnosis for mass forming lesions in patients with a history of residence or travel to an endemic region.
Footnotes
Acknowledgments
The authors are thankful to Shawn Scally for excellent assistance in figures preparation and Shannon Yeh PA for excellent documentation of the gross findings.
Author Contributions
PK and WM participated in data collection; KS and LB wrote the manuscript text; PK, WM, and KS edited the manuscript. The authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent
This study is exempt from the regulatory requirements of the National Institutes of Health (NIH) regarding studies on human subjects, based on the criteria described in section 4 of 46.101(b) of 45 CFR 46 “as research involving the collection or study of existing data, documents, records, if the information is recorded by the investigator in such a manner that subjects cannot be identified, directly or through identifiers linked to the subjects.” 24 According to the institutional guidelines, the patient's consent is not necessary for a case report with de-identified patient specific information.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
