Abstract
Histoplasmosis is commonly a self-limited fungal disease that primarily affects the lung and reticuloendothelial system. Cardiac involvement by histoplasmosis is uncommon. In this report, we provide a detailed description of severe pulmonary histoplasmosis complicated by the disease involvement of the free wall of the right ventricle. A 55-year-old female presented with cough, fevers, dyspnea, and 30-pound unintentional weight loss in 6 months. Her past medical history was significant for supraventricular tachycardia with permanent pacemaker implantation. Imaging studies revealed an intracardiac mass accompanied by mediastinal lymphadenopathy and bilateral lung nodules. Endobronchial ultrasound-guided transbronchial needle aspiration of station 4R lymph nodes revealed numerous yeast forms, morphologically consistent with Histoplasma capsulatum. The diagnosis was further corroborated by the elevated titers of serum antibodies against Histoplasma capsulatum. The right ventricular mass debulking with biopsy showed necrotizing granulomatous inflammation involving nonvalvular endocardium and myocardium of the free wall of the right ventricle. The report documents an unusual presentation of pulmonary histoplasmosis accompanied by nonvalvular endocarditis and suggests a possible association between the site of the cardiac infection and the presence of a permanent intravascular pacer device.
Keywords
Background
Histoplasma capsulatum is a dimorphic fungus that propagates as mold in soil contaminated with bird or bat excrement. In the United States, Histoplasma capsulatum is endemic in the Ohio and Mississippi river valleys.1,2 Sporadic inhalational exposure to the disturbed soil in endemic areas can cause a self-limited histoplasmosis that is confined to the lung and reticuloendothelial system; however, extensive exposure and/or immunodeficiency can lead to disseminated infection.3–7 Acute granulomatous histoplasmosis involving the endocardium and/or myocardium is rare.8–10 We provide a detailed description of a severe form of pulmonary histoplasmosis complicated by granulomatous endocarditis involving the free wall of the right ventricle.
Case Presentation
A 55-year-old female former smoker sought medical attention for progressive cough, fevers, dyspnea, and 30-pound unintentional weight loss in 6 months. Her past medical history was significant for permanent pacemaker implantation for sinoatrial node dysfunction with ablation 8 years prior. A chest computed tomography at presentation showed mediastinal lymphadenopathy and variable in size upper lobe predominant 0.2 to 1 cm nodules, some with cavitation (Figure 1A and B). Cardiac images revealed a right ventricular mass (Figure 1C) associated with a pacemaker lead. Transesophageal echocardiogram documented a 3.1 × 4.1 cm heterogenous mass along the mid-apical region (Figure 2A and B). Notably, the right ventricular mass had been present for several years on prior transthoracic echocardiograms, however, it had enlarged over time. The right ventricle was moderately dilated producing mild systolic dysfunction. The left ventricular size and systolic function were normal with an estimated left ventricular ejection fraction of 55–60%. There was no hemodynamically significant valvular disease. There was no pericardial effusion. Endobronchial ultrasound-guided transbronchial needle aspiration of station 4R lymph nodes revealed necrotic lymph node tissue (Figure 3A) associated with numerous yeast forms, morphologically consistent with Histoplasma capsulatum (Figure 3B). The diagnosis was further corroborated by elevated serum antibodies against Histoplasma capsulatum by complement fixation (1:64, with normal reference range <1:8). Itraconazole was initiated however due to poor tolerance (nausea and vomiting) it was switched to Isavuconazole. On antifungal treatment, symptomatic improvement was noted at 10 weeks post presentation and at 14 weeks the patient underwent pacer electrode removal and right ventricular mass debulking with a new epicardial lead placement via sternotomy. At surgery, it became apparent that the mass was incorporated into the free wall of the right ventricle and into the tricuspid valve annulus, as well as completely encasing the preexisting right ventricular lead. Only partial debulking was completed due to mass adherence to nearly half of the right ventricular free wall. The pathological evaluation of the resected mass revealed florid necrotizing granulomatous inflammation involving the endocardium (Figure 3C and D) and the luminal aspect of the myocardium (Figure 3E and F). The patient showed symptomatic improvement however she experienced poor tolerance of oral antifungal therapy and at 6 months received induction dosing of amphotericin B. Since the patient had intolerance to antifungals and requested cessation of therapy she was discharged to hospice.
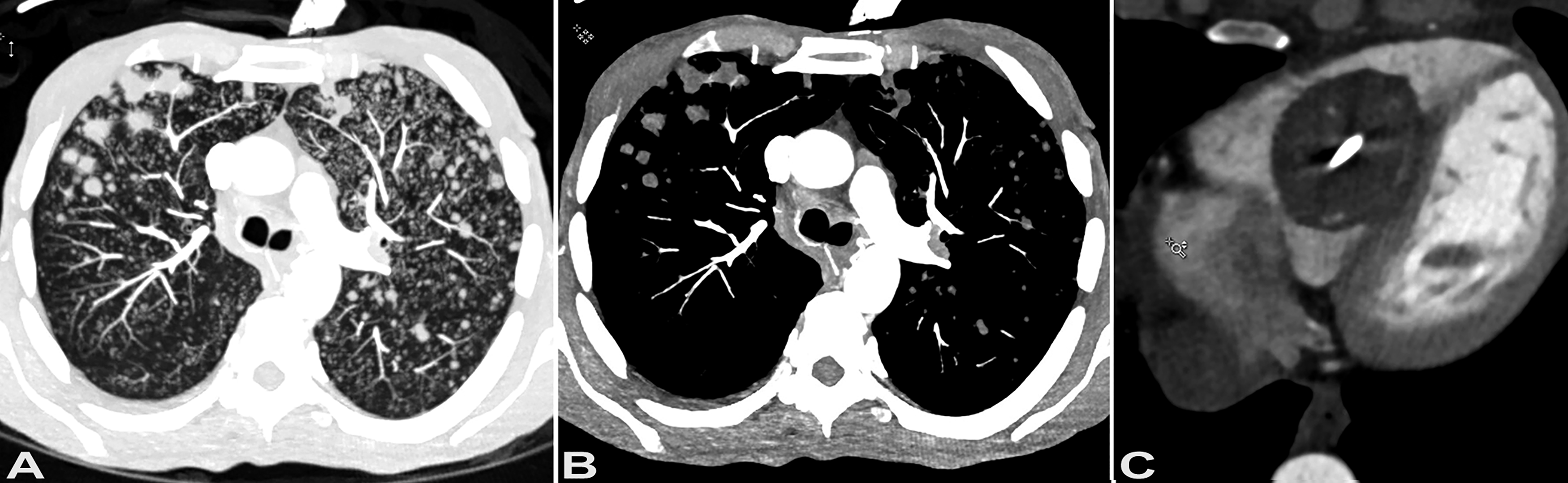
Computed tomography. Contrast-enhanced computed tomography images at the level of carina show multiple upper lobe predominant nodules and micronodules, measuring up to 1 cm in size (A, lung window; B, soft tissue window). Contrast-enhanced computed tomography images show large filling defects within the right ventricle (C), along the course of the pacemaker lead, with the right ventricular component measuring approximately 4.2 × 3.6 cm.
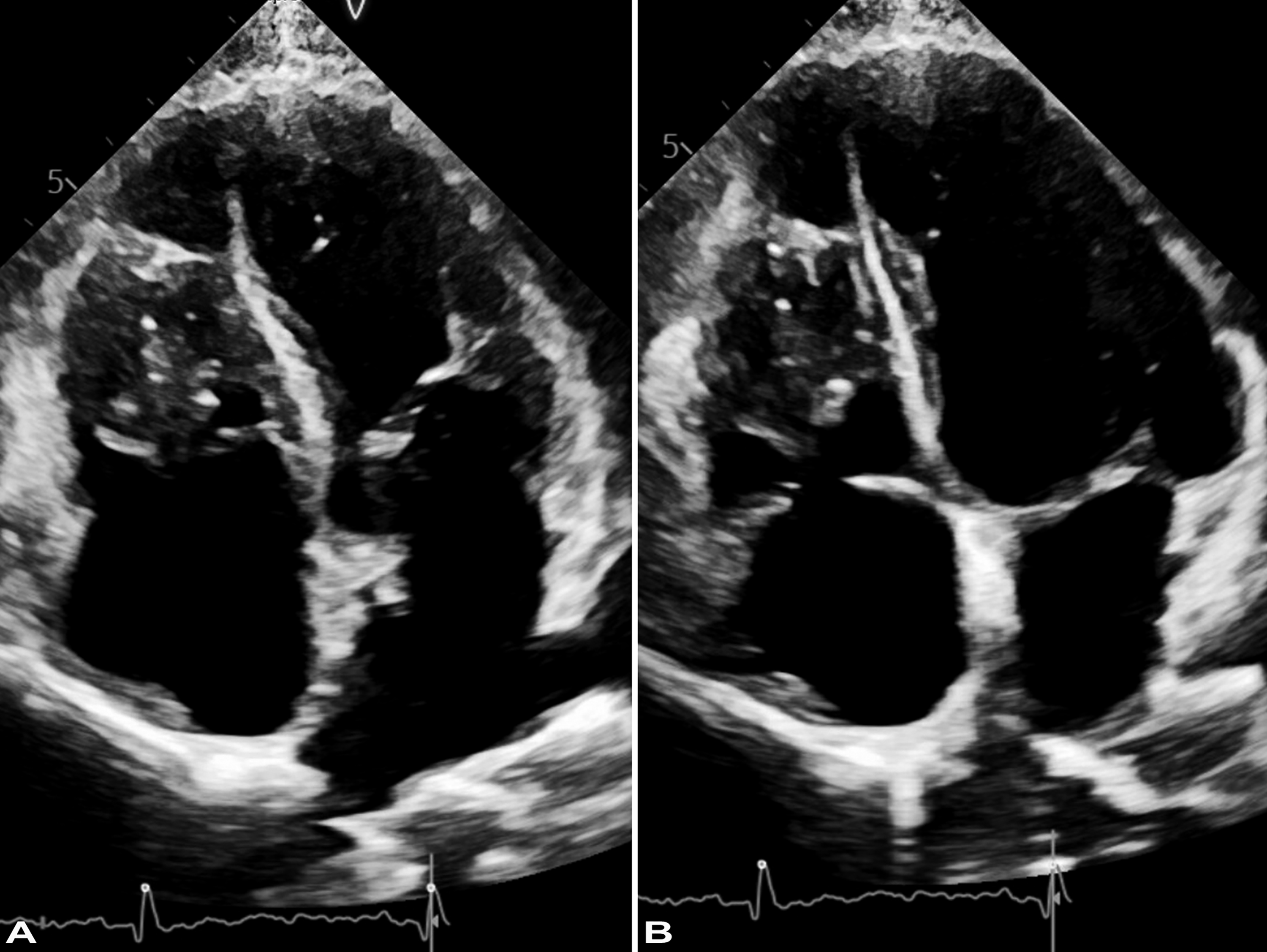
Transesophageal echocardiogram. Apical four-chamber view shows a heterogenous mass within the mid-section of the right ventricle, adherent to the interventricular septum (A), but separate from the tricuspid valve (B).
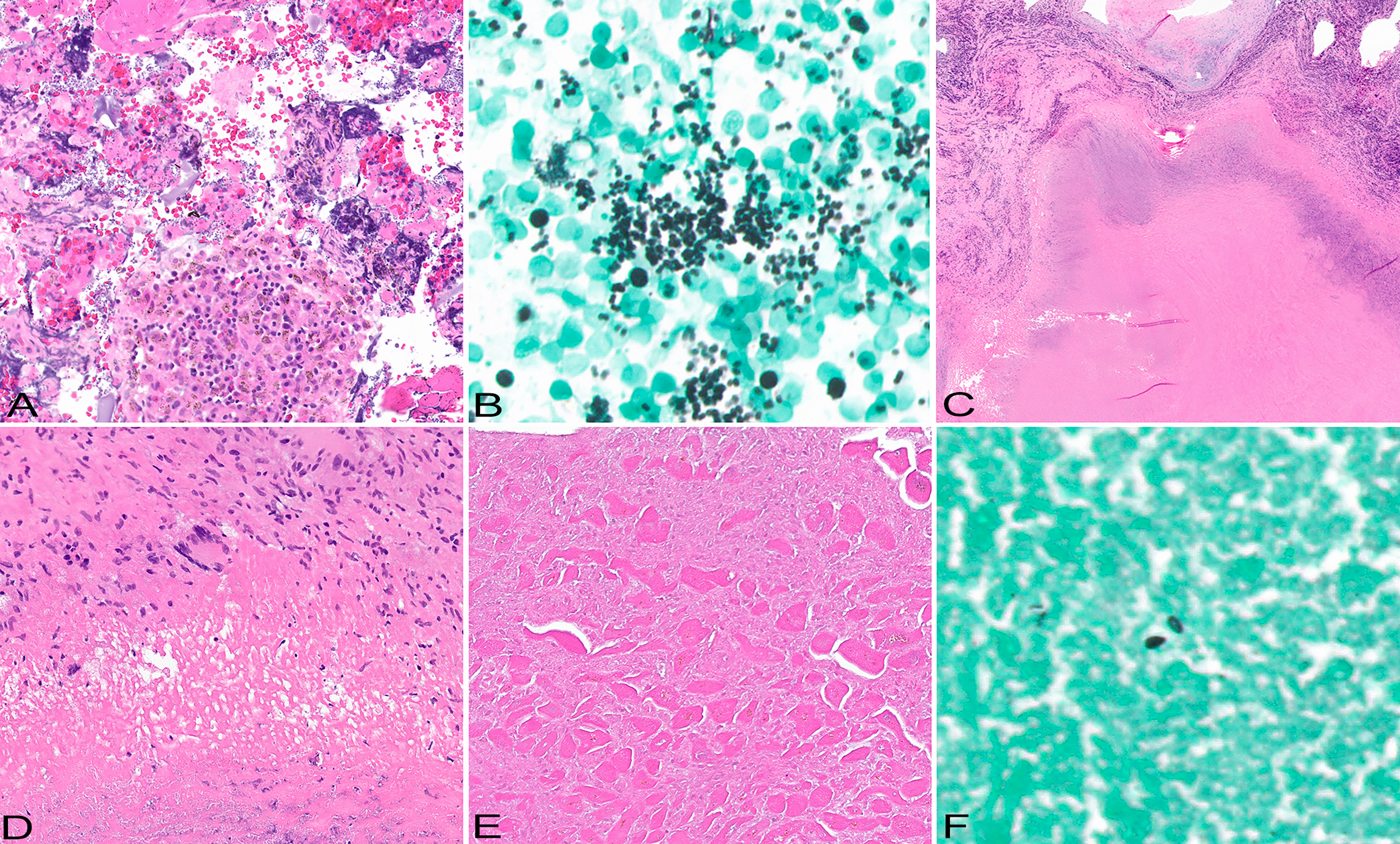
Pathological findings. Cell-block section of the endobronchial ultrasound-guided transbronchial needle aspiration of station 4R lymph node shows degenerated acellular debris and lymph node tissue (A), HE original magnification x20. Further evaluation of the material with Grocott's methenamine silver stain highlights numerous yeasts, morphologically consistent with Histoplasma capsulatum (B), HE original magnification x60. Right ventricular mass biopsy with debulking shows subendocardial necrotizing granulomas (C), represented by central necrotic core and peripheral rim of palisading histiocytes with occasional multinucleated giant cells (D), HE original magnification x2 and x20, respectively. The process involves right ventricular myocardium (E) showing myocyte necrosis; occasional yeast forms are apparent on Gomori methenamine silver stain (F), original magnification x20 and x100, respectively.
Discussion
Cardiac histoplasmosis is an uncommon complication of Histoplasma capsulatum infection that can occur via extrinsic or intrinsic route. In extrinsic cardiac histoplasmosis, there is direct involvement of the pericardium and myocardium by advancing granulomatous inflammation from the lung and mediastinal lymph nodes, in intrinsic, there is seeding of the cardiac lining from hematogenous spread that manifests as valvular or nonvalvular endocarditis.6,7,11,12 While extrinsic cardiac histoplasmosis (including secondary to fibrosing mediastinitis) and histoplasma endocarditis are well-documented, granulomatous histoplasmosis involving nonvalvular cardiac tissue is rare. The term “nonvalvular endocarditis” have been applied to cases where cardiac lining other than heart valves are affected by infection.11–13 Such cases have been reported in association with intravascular devices and intracardiac thrombi.11,14 Nonvalvular endocarditis is thought to be related to the potential damage to the endocardium produced by intravascular devices such as graft material or pacemakers.11,15 Most cases of endocarditis both valvular and nonvalvular are caused by bacteria and less commonly by fungus.10,13,16 The current case represents a unique manifestation of histoplasmosis as to the best of our knowledge no cases of nonvalvular granulomatous Histoplasma capsulatum endocarditis involving the free ventricular wall have been documented in the literature.
Pacemaker systems, battery and electrode, can serve as a conduit for the skin flora to enter the circulation and lead to infectious complications including sepsis and localized pockets of infection. 17 Recognized complications of infected endovascular leads include valvular and nonvalvular endocarditis, infected mural thrombi, localized abscesses, and late electrode perforation.17–20 The clinical manifestations of nonvalvular endocarditis somewhat overlap with that described as pacemaker endocarditis where both can be associated with the presence of a pacemaker device. However, in cases reported as pacemaker endocarditis, the device serves as a source or conduit of infection that may involve the adjacent vascular lining but invariably manifests by valvular endocarditis. 16 In this patient, the intravascular pacemaker system was likely responsible for the endothelial lining damage promoting infection at an unusual for histoplasmosis site rather than a source of infection. The initial clinical impression of a pacemaker associated thrombus on imaging did not readily correlate with the findings either at surgery or on pathological examination. A dense mass well incorporated into the right ventricular free wall was identified at surgery, while histologically it represented necrotizing granulomatous inflammation involving the endocardium and the adjacent myocardium. No histological evidence of organizing thrombus was apparent in the examined surgical material; nevertheless, it is difficult to completely exclude possibility of a histoplasma-infected thrombus akin to that described in rare instances of atrial myxoma superinfected with histoplasmosis. 21
Disease manifestations after inhalational exposure to Histoplasma capsulatum depend on the intensity of exposure and the immunity of the host. Acute illness ranges from asymptomatic infection to severe pneumonia; however, in most cases, it resolves without treatment. 22 Relatively common complications of pulmonary histoplasmosis include mediastinal granulomatous lymphadenitis, pulmonary cavitary disease, fibrosing mediastinitis, and broncholithiasis. 5 Hematogenous dissemination occurs in the acute phase of the disease, but it is rarely progressive, and in most cases the development of cell-mediated immunity to Histoplasma capsulatum leads to complete resolution of the disease. Occasionally, disseminated disease can lead to histoplasma endocarditis involving both native and prosthetic valves; reviewed in Bhatti et al 10 Similar to bacterial endocarditis, the left-sided valves are more commonly affected. Because bacterial cultures are negative, the diagnosis of histoplasma endocarditis tends to be delayed and if left untreated the disease is invariably fatal. Surgical intervention provides a better sample for diagnosis and decrease in the disease burden, and, in combination with Amphotericin B therapy, improves survival.10,15 Disseminated histoplasmosis occurs in immunocompromised individuals unable to produce a cell-mediated immune response to the organism and is fatal if untreated.3,22 There was no evidence of decreased cell-mediated immunity in this patient. An active lifestyle and certain occupations with outdoor exposures to disturbed soil in endemic areas are known risk factors for histoplasmosis and therefore an accidental inhalational exposure to Histoplasma capsulatum in the endemic region of the Ohio river valley where the patient resides is suspected. That is further supported by the fact that the patient's family member had recently been diagnosed and recovered from a limited form of histoplasmosis.
In conclusion, we report an unusual presentation of pulmonary histoplasmosis. As with other infections, in histoplasmosis, the damage of endothelial lining of heart chambers associated with a preexisting pacemaker can promote an infection at unusual sites manifesting as nonvalvular endocarditis.
Footnotes
Acknowledgments
The authors are thankful to Shawn Scally for excellent assistance in figures preparation.
Author Contributions
AMG and CCA participated in data collection; KS and NS wrote the manuscript text; AMG, SS, and KS edited the manuscript. The authors read and approved the final manuscript.
Consent for Publication
This study is exempt from the regulatory requirements of the National Institutes of Health (NIH) regarding studies on human subjects, based on the criteria described in section 4 of 46.101(b) of 45 CFR 46 “as research involving the collection or study of existing data, documents, records, if the information is recorded by the investigator in such a manner that subjects cannot be identified, directly or through identifiers linked to the subjects.” 23 According to the institutional guidelines, the patient's consent is not necessary for a case report with de-identified patient specific information.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
