Abstract
Background:
Uncontrollable bleeding is a major cause of mortality and morbidity worldwide. Effective hemostatic agents are urgently needed. Red cell microparticles (RMPs) are a highly promising hemostatic agent. This study evaluated the safety profile of RMPs preliminary to clinical trial.
Methods and Results:
RMPs were prepared from type O+ human red blood cell by high-pressure extrusion. Male rats were treated with RMPs either a 1 × bolus, or 4 × or 20 × administered over 60 minutes. The vehicle-treated group was used as a control. Effects on physiological parameters were evaluated; namely, blood pressure, body and head temperature, hematocrit, and blood gases. We did not observe any adverse effects of RMPs on these physiological parameters. In addition, brain, heart, and lungs of rats treated with 4 × dose (bolus followed by infusion over 60 minutes) or vehicle were examined histologically for signs of thrombosis or other indications of toxicity. No thrombosis or indications of toxicity in brain, heart, or lungs were observed. Studies revealed that RMPs were distributed mainly in liver, spleen, and lymph nodes, and were potentially excreted through the kidneys.
Conclusions:
Our study indicates that RMP administration appears not to have any negative impact on the parameters studied and did not produce thrombosis in heart, brain, and lungs. However, more detailed long-term studies confirming the safety of RMP as a hemostatic agent are warranted.
Introduction
Uncontrolled bleeding from brain hemorrhage, trauma, or other causes have high mortality and morbidity. Brain hemorrhage and hemorrhagic shock account for up to 40% of trauma deaths worldwide. 1 Associated morbidity in survivors has lasting effects on quality of life owing to functional impairments. 2 Those with platelet disorders are at higher risk of excessive bleeding during surgical procedures. 3 Hemophilia A models have high rates of mortality from lethal bleeding in multiple tissues. 4 Death rates from intracranial hemorrhage in hemophilia A and B are as high as 20% and are especially problematic in neonates. 5 Many patients taking anticoagulant drugs for stroke or cardiovascular disease are at risk of excessive bleeding, and for some of the newer ones, there are no antidotes to control hemorrhagic events. 6,7
Bleeding in traumatic injury can usually be managed with proper therapeutic measures. 1,8 However, studies on existing hemostatic therapies for intracranial hemorrhage such as tranexamic acid, aminocaproic acid, and recombinant activated factor VII (rFVIIa) have shown negative effects that exceed their hemostatic benefits. Tranexamic acid and aminocaproic acid produce complications such as hydrocephalus and thrombosis, and rFVIIa may be associated with arterial and venous thromboembolisms at higher dosages. 9 -11 Because of such limitations, there is an urgent need for more effective hemostatic agents.
The proposed new hemostatic agent evaluated in this report is based on present knowledge of the procoagulant and hemostatic properties of circulating cell-derived microparticles. 12 -17 Cell-derived microparticles are small membrane vesicles released from a cell during activation or apoptosis. 12 Cell-derived microparticles from platelets, erythrocytes, granulocytes, and endothelial cells in circulation possess mild thrombotic properties that work by displaying procoagulant, negatively charged phospholipids in blood vessels upon cell activation. 13 These newly exposed phospholipids, such as phosphatidyl serine (PS), provide a substrate for clotting factors to bind, therefore accelerating hemostatic processes. 12 -15 More specifically, red cell microparticles (RMP), which contain the procoagulant phospholipid PS and act via the contact pathway independent of tissue factor (TF), were prepared ex vivo for use as a hemostatic agent. 12 -15,18 Infusion of RMP in models of bleeding in rats and rabbits substantially reduced blood loss. 18 In addition, blood samples from patients with a variety of bleeding disorders were largely corrected by addition of RMP, as evaluated by thromboelastography. 18 Unexpectedly, RMP also augmented platelet function (primary hemostasis). 18 We have developed a method to prepare high-pressure, extrusion-derived RMPs, so that it can be used as a therapy for bleeding disorders.
However, before the efficacy of RMP can be tested clinically, more rigorous studies are required. Preclinical studies of drugs require toxicological screening to determine the “No Observed Adverse Effect Level.” 19,20 Determination of the biodistribution of RMP can serve as an additional measure of safety. A recent international meeting discussing guidelines for gene therapy products emphasized the need for biodistribution studies to determine whether a drug is truly ready for clinical use. 21 These guidelines emphasize the need to determine organ distribution of RMPs postinjection. These assessments help regulatory authorities to determine the safety of a novel therapeutic agent. In view of these considerations, the goal of this study was to determine the safety and biodistribution profiles of RMP. We were interested in determining the safety of our RMPs when given at a pharmacological dose. The dose of RMPs was decided based on our recently published study, 22 where we reported the dose of RMP required to achieve its pharmacological concentration in the blood of rats. To confirm RMPs do not exhibit any negative impact on studied parameters at significantly higher dose, we also included a 20 × dose-treated group. This study is limited to the acute effects of RMP on various physiological parameters.
Materials and Methods
Preparation of RMPs
RMPs were produced using red blood cells (RBCs) obtained from blood (approved as per Food and Drug Administration -required tests for transfusion) drawn <10 days before (Rh+, type O, leukoreduced packed RBCs from OneBlood Center, Fort Lauderdale, FL). The RBC were washed twice with isotonic saline by centrifuging at 200g for 10 minutes and resuspending pellets in saline to hematocrit of 25%. The RBCs were converted to RMP by passage through a high-pressure extrusion device (Constant System Cell Disruptor, Northants, UK) twice at 35 000 psi. The RMP output was washed twice with saline by centrifuging at 25 000g for 30 minutes. The resultant RMP pellets were resuspended in saline to a total volume one-fourth of the original packed RBC used. For use, the lyophilized RMP were reconstituted with equal volume of distilled water, vortexed, then filtered through a 1.2-µm syringe filter to remove any microaggregates.
Particle Counting
A sample of 20 µL of RMP diluted 1:1000 in saline was mixed with 4-µL monoclonal anti-CD235a-PE, incubated for 20 minutes (60 rpm), then further diluted with a 976-µL volume of phosphate-buffered saline and run in flow cytometer (Beckman Coulter Model FC-500, Beckman Coulter Life Sciences, Indianapolis, IN). The flow cytometer was calibrated with Megamix (Stago, Parsippany, NJ ) and CountBright (Thermo Scientific, Waltham, MA) beads. Megamix beads were used to adjust the voltages of photomultiplier tube for forward scatter, side scatter, fluorescent parameter 1 (FL1), FL2, and boundaries of RMP distribution, as per the manufacturer’s instructions. CountBright was used for calibration of flow rate. The trigger (discriminator) was by fluorescent signal on FL2. Run time was 1 minute. Counts were enumerated by the following relation, where “flow rate” is mL/min determined by beads:
RMP/mL = (observed count) × (1 mL/flow rate) × dilution factor.
Animal Procedures
Experiments were conducted using male Sprague-Dawley rats (Charles River Laboratories International, Inc, Wilmington, MA) as per the Guide for the Care and Use of Laboratory Animals published by the National Institutes of Health. The protocol was duly approved by the University of Miami’s Animal Care and Use Committee. Induction of anesthesia was by 5% isoflurane in a mixture of 33% oxygen and 67% nitrous oxide and was maintained by 1% to 2% isoflurane in the same gas mixture. Rocuronium (10 mg/kg, intravenous [IV] every 20 minutes)-paralyzed rats were artificially ventilated while anesthesia was maintained as above. An incision was made in the flank region and a femoral artery and vein were cannulated. Physiological parameters were monitored at intervals; namely, blood pH, partial pressure of oxygen ( pO2), and partial pressure of carbon dioxide (pCO2) were measured in microsamples using an ABL80 FLEX blood gas analyzer (Radiometer America Inc, Brea, CA). Body temperature was monitored by rectal probe, inputting to a Yellow Springs Instruments model 73A 73ATA temperature controller (Houston, TX). Head temperature was monitored by a hypodermic needle probe (Omega, Stamford, CT) inserted into the temporalis muscle. Blood pressure was recorded throughout. Animals showing variations in these parameters outside the normal range during the baseline measurements were excluded.
Injection of RMP
RMPs were injected using an infusion pump (Stoelting Scientific Syringe Pump, Arlington, TX) into the cannulated femoral vein as per the protocol described in Figures 1, 2, and supplementary Figure 1. All bolus injections were given over 20 seconds. Groups were designated as follows:

Experimental design: Schematic representation of experimental protocol with dosing and sampling time points (A) paradigms used in safety study, and (B) paradigm used for thromboembolism study.

Experimental design biodistribution study: (A) Schematic representation of experimental protocol with dosing and sampling time points paradigm used for biodistribution study. (B) Fluorescence intensities of the overall dose of labelled RMPs injected per kg body weight to evaluate biodistribution at 15 minutes and 24 hours post-RMP injection.
A) Paradigms used for evaluating the effect of RMP injection on physiological parameters (Figure 1A):
B) Paradigms used for evaluating the effect of RMPs on thromboembolism (Figure 1B):
C) Paradigms used for the biodistribution study (Figure 2A):
To compensate for the loss of blood due to sampling, an IV infusion of normal saline was given between obtaining the first and last blood samples. The volume of saline injected via RMP injection and supplementary vehicle/saline injected to compensate for bloodletting was 3 mL/h.
Tissue Collection and Processing for Thromboembolism and Toxicity Studies
The rats were perfused transcardially at 4 hours post-RMP using saline and subsequently with formaldehyde-acetic acid-methanol (FAM) mixture (1:1:8) at constant pressure of 120 mm Hg. Brain, heart, and lungs were harvested and stored in FAM. These organs were processed, embedded in paraffin, sectioned, and stained with hematoxylin and eosin. Tissues were examined by a board-certified veterinary pathologist to determine thromboembolism and histological toxicity. Naive rats belonging to groups 3 and 4 were perfused at 4 hours postinjection. Tissues obtained from RMP-treated animals (n = 6, group 4) and saline-treated animals (n = 3, group 3) were evaluated in a blinded manner.
Assessment of Biodistribution
RMP were labeled with Alexa-488 dye as described earlier. 23 Unconjugated dye was removed using a microdialysis membrane (Spectra/Por Dialysis Membrane, Spectrum Laboratories, Inc, Rancho Dominguez, CA) and dialyzed against saline overnight with frequent saline changes. Dialyzed labeled RMPs were collected and purified using a 1.2-µm pore size filter before use. Labeled RMPs were injected by single bolus injection of the total 1× dosage at time 0 delivered over 20 seconds (groups 5 and 6). To confirm the systemic availability of RMPs, blood samples were obtained 0, 1, 3, 5, 10 and 15 minutes after treatment, and RMP levels were quantified in 15-minute group. Similarly, blood samples were obtained 0, 15 minutes, and 24 hours after treatment, and RMP levels were quantified in 24 hour group. Rats were then perfused with saline for 2 minutes to remove contamination of circulating labeled RMPs, at the end of either 15 minutes or 24 hours post-RMP injection. Blood was collected before perfusing rats. Brain, lung, heart, liver, spleen, kidney, bone marrow, lymph nodes, and muscles were harvested and frozen at −80°C until further analysis. Frozen tissues were thawed on ice and 8% to 10% homogenates were made for brain, liver, heart, kidney, lung, spleen, and muscle using glass Teflon homogenizer in radioimmunoprecipitation assay (RIPA) buffer (20 mM Tris-HCl pH 7.5, 150 mM NaCl, 1 mM EDTA, 1% NP-40, 1% sodium deoxycholate, 2.5 mM sodium pyrophosphate, and protease inhibitor cocktail); 5% homogenates were made for lymph node and bone marrow in RIPA buffer. Tissue homogenates were subjected to three 10-second cycles of sonication at 45% strength (1 minute interval between each cycle). Fluorescence of the Alexa-488 label (Excitation: 490 nm/Emission: 520 nm) was measured in resultant tissue homogenates as an indicator of the presence of RMPs in tissue. The results are expressed as mean fluorescence injected/kg (Figure 2B), mean fluorescence/g tissue for brain, liver, heart, kidney, lung, spleen, muscle, and lymph node (Figure 3A), mean fluorescence/bone marrow harvested from femur bone obtained from both the hind limbs (Figure 3A), and mean fluorescence/mL of blood (Figure 3B).

RMP biodistribution: Fluorescence intensities in (A) tissues (brain [n = 6], liver [n = 6], heart [n = 5], kidney [n = 6], lung [n = 6], spleen [n = 6], muscle [n = 6], lymph node [n = 5], and bone marrow [n = 5]), and (B) blood at 15 minutes and 24 hours post-RMP injection using 1 × bolus treatment paradigm. Fluorescence in all organs, except bone marrow, was normalized to g tissue weight. For bone marrow, fluorescence is normalized to total bone marrow harvested from femur bone obtained from both the hind limbs. *P < .05, **P < .01, and ***P < .001 compared with respective 24 hours values.
Statistics
All data points displayed in figures are mean ± standard error of mean (SEM), n = 10. Grubb test was used to identify and exclude significant outliers. Analysis of variance followed by Tukey post hoc test for multiple comparisons was used. A P value < 0.05 was considered statistically significant.
Results
Dosage Regimens
The total number of RMPs administered to groups 1, 2, 4, 5, and 6 were 6.07 × 1010, 1.21 × 1012, 2.43 × 1011, 6.07 × 1010, and 6.07 × 1010 particles/kg body weight (BW), respectively. Group 3 received 4 mL of saline/kg BW.
Effects on Physiological Parameters
A previous study has shown that blood levels of RMPs correlate with the risk of cardiovascular complications. 24 Therefore, to determine potential side-effects/toxicity of RMP injection, we monitored physiological parameters such as blood pressure, body and head temperature, hematocrit, and blood gases (pH, pCO2, and pO2) before and after RMP injection for all experiments (Figures 4 and 5). We did not observe any adverse effect of RMP injection on the abovementioned physiological parameters. However, we did observe a small but significant increase in blood pO2 (155 ± 12 mm Hg at 75 minutes post-RMP injection vs 125 ± 11 mm Hg at baseline) in 20 × RMP dose-treated animals (Figure 5).
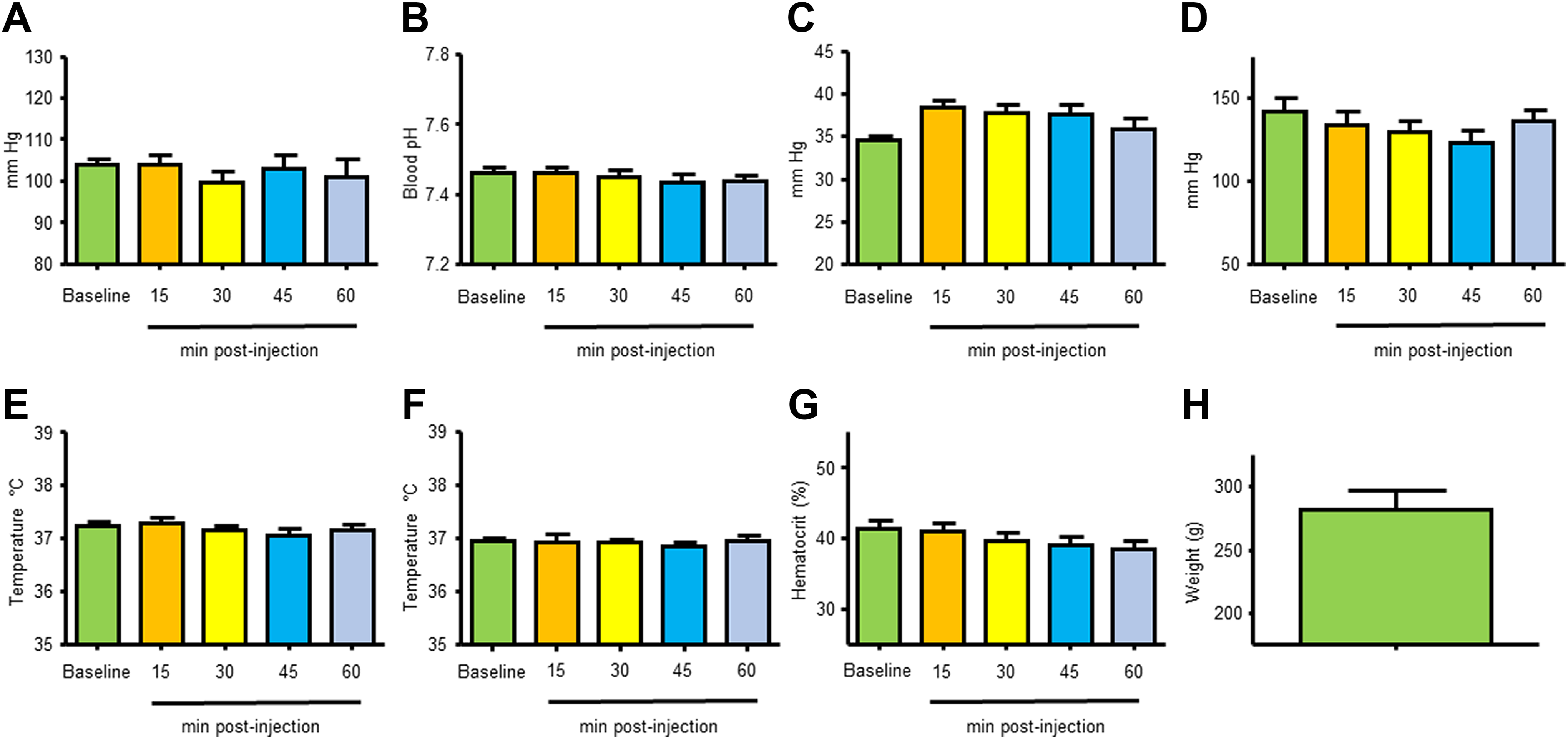
Physiological parameters: (A) mean arterial blood pressure, (B) blood pH, (C) blood pCO2, (D) blood pO2, (E) body temperature, (F) head temperature, (G) hematocrit, and (H) body weight for animals used in study using a single bolus regimen (group 1, n = 10).

Physiological parameters: (A) mean arterial blood pressure, (B) blood pH, (C) blood pCO2, (D) blood pO2, (E) body temperature, (F) head temperature, (G) hematocrit, and (H) body weight for animals used in study using 20 times the single dose infused over 60-minute regimen (group 2, n = 10). * P < .05 compared to baseline.
Evaluation of Potential Thromboembolism in Brain Following RMP Injection in Naive Rats
The goal of this experiment was to evaluate the potential side effect of RMP injection in naive rats, specifically, thromboembolic complications. We examined brains of 3 saline-treated and 6 RMP-treated rats for potential thromboembolism. Considering the balance of thrombosis and thrombolysis/fibrinolysis, we chose to evaluate brains at 4 hours post-RMP injection. RMP-treated rat tissues (n = 6) and saline-treated (n = 3) rat tissues were evaluated in blinded manner by a board-certified veterinary pathologist. No thrombosed blood vessels or indications of toxicity in any of the 9 brains, hearts, or lungs were observed. These results rule out the possibility of thrombosis as a potential side effect of RMP therapy to naive rats.
Blood Levels of Injected RMPs for Biodistribution Study
To determine the availability of RMPs in the body circulation following RMP administration, we monitored blood levels before and at various time points after RMP injection for biodistribution study (Figure 6). In 15-minute group, RMPs levels in blood were highest at 1-minute time point and progressively declined thereafter at 3, 5, 10, and 15-minute time points (Figure 6A). In 24-hour group, RMPs levels in blood, at 15-minute time point, were similar to what was observed for 15-minute group. These values declined at the 24-hour time point (Figure 6B). Blood RMPs level results for 15-minute group are also presented in terms of area under the curve (Figure 6C). These results confirm the systemic availability of RMPs in naive rats after IV administration of RMPs.

Time course of changes in blood levels of RMPs following a bolus dose for the biodistribution study at (A) 15 minutes and (B) 24 hours post-RMP injection. (C) Area under the curve of blood RMP levels for 15 minutes biodistribution group. Blood levels quantified using flow cytometry are represented in terms of number of RMPs per mL blood. Area under the curve was computed using the RMP concentration time curve.
Evaluation of Biodistribution Following RMP Injection in Naive Rats
Mean fluorescence for RMPs injected was 926 432 ± 72 643 arbitrary fluorescence units (AFU)/kg BW and 800 310 ± 25 554 AFU/kg BW for 15-minute and 24-hour groups, respectively. Major fluorescence was detected in liver (n = 6), and spleen (n = 6), while moderate fluorescence was detected in heart (n = 6), kidney (n = 5), lung (n = 6), and lymph node (n = 5) when measured at 15 minutes post-RMP injection (Figure 3A). Minor fluorescence was detected in brain (n = 6), muscle (n = 6), and bone marrow (n = 6) at the 15 minute postinjection time point (Figure 3A). A similar trend was observed when evaluated at 24 hours post-RMP injection (Figure 3A). Fluorescence in the 24 hours post-RMP injection group was significantly lower for liver (n = 6, P < .001), spleen (n = 6, P < .05), and bone marrow (n = 5, P< .01); while no significant difference was observed for kidney (n = 6), muscle (n = 6), and lymph node (n = 5) when compared to the 15 minutes/24 hours time point (Figure 3A). We observed mean fluorescence of 37 ± 23 and 287 ± 105 AFU/mL of blood when measured at 15 minute and 24 hours post-RMP injection, respectively (Figure 3B). Our results indicate that a very small fraction of injected RMPs is retained in the body at both 15 minutes and 24 hours post-RMP injection.
Discussion
Given the high morbidity and mortality of bleeding disorders and the current lack of efficacious treatment options, there is a need for new hemostatic agent to address shortcoming of present therapy. To meet this end, RMP have been evaluated in recent studies to assess their benefit as potential therapeutic hemostatic agents. RMPs are ideal candidates for therapeutic treatment as they have universal hemostatic activity since RMPs are able to correct hemostatic defect in both primary and secondary hemostasis. As reviewed previously, 12 they decrease lag time to initial fibrin formation and decrease bleeding time in rodent models. 18 RMP delivery is expected to have a safety profile like RBC transfusion. 18 While these studies demonstrate the hemostatic properties of RMPs, thorough preclinical studies assessing the toxicity, safety, and biodistribution of the particles are essential before they can be tested clinically. To evaluate the toxicity and side effects of this therapy, possible complications following RMP administration were gauged by assessing multiple physiological parameters, potential thrombosis, biodistribution, and mortality in a rodent model.
There are conflicting results on the potential effect of RMPs on hemostatic parameters. While some studies reported that RMPs possess antithrombin and fibrinolytic activity 25,26 as also reviewed previously, 27 others using a different methodology of functional assay demonstrated that RMPs inhibit fibrinolysis. 28 In addition, preliminary data from our laboratory shows that RMPs derived from high pressure extrusion inhibit fibrinolysis. 29,30 RMPs made using different methods may be structurally different, as our earlier study observed that high-pressure, extrusion-derived RMPs have lower levels of lipid rafts. 29 It is plausible that the method used to produce RMPs may dictate its anti-/prothrombotic properties. Some earlier studies indicated that transfusion of long-term stored packed RBC or blood may cause thromboembolic complications due to increased levels of microparticles or due to the release of other harmful mediators released in stored blood. 31,32 It should be noted that as mentioned in the methods section, we use < 10 days old blood to prepare our RMPs. We do not expect high number of microparticles in such short-term stored blood. Besides, we washed RBCs with saline repeatedly thrice prior to the generation of RMPs. We expect this washing to remove any potential contamination of microparticles or other harmful mediators.
Physiological parameters including blood gases, body temperature, hematocrit, and mean arterial blood pressure were chosen to evaluate toxicity and side effects of RMPs, as they are good measures of key organ function. Arterial blood gases (pO2 and pCO2) and pH were measured to assess adequacy of gas exchange occurring in the lungs. 33 Cardiovascular function was evaluated by measuring mean arterial blood pressure, and thus, the ability of the heart to perfuse organs, which is one of many important components of hemodynamic monitoring. 34,35 Given that RMPs elicit hemostasis, one of the concerning potential side effects would be thrombosis. Therefore, it was also important to evaluate the consequences that possible thrombosis would have on these physiological parameters. For example, a blood clot in the lungs would cause a mismatch between ventilation and perfusion, decreasing effective gas exchange and thus decreasing arterial pO2. 36 Our results did not indicate any adverse or toxic effects of RMP administration on arterial blood gases (pO2 and pCO2) and pH. We observed increased pO2 levels at 75 minutes postinitiation of the infusion in rats treated with 20 × RMP dose. Although RMPs are washed during preparation, it has a small amount of trapped hemoglobin. It is plausible that this increase in pO2 levels may be due to increased blood hemoglobin levels in rats treated with 20 × RMP dose. However, this hypothesis remains to be tested in future studies.
In addition to physiological parameters, the heart, lungs, and brain were examined histologically. While the role of RMPs in the coagulation cascade, production of thrombin, and increased rate of fibrin formation have been documented, 18,37,38 these particles elicit their hemostatic effect without possessing TF. 18 This property allows RMPs to stimulate clot formation to stop bleeding exclusively at the site of injury without causing thrombosis elsewhere in the body. To confirm this, animals were sacrificed 4 hours after injection of RMPs and tissues examined for thrombosed blood vessels by a blinded board-certified veterinary pathologist. The sacrificing time point of 4 hours was chosen to avoid potential natural thrombolysis/fibrinolysis. 39 -41 No evidence of thromboembolism or toxicity in either the brain, the heart, or the lungs was observed, suggesting that as predicted, RMPs did not cause hemostasis in the absence of vessel injury. A future avenue of investigation would be to evaluate the effects of RMPs on brain function and behavior. These outcomes would necessitate long-term survival of the animals, which was not explored in the present study. We do not expect RMPs to have any impact on brain function similar to heart and lung function. We did not observe any mortality when assessed up to 24 hours postinjection with RMPs. We evaluated the safety profile of human RMPs in rats. It is possible that this may lead to some interspecies compatibility issues. Not evaluating such compatibility issues is a limitation of our study. However, in an unpublished study, we evaluated similar paradigms of RMP injections in rats and found that all animals survived, at least, up to 1-month postinjection without any apparent impact on animals’ health. Besides, animals received RMPs over 20 seconds to 75 minutes and were not exposed to RMP injections over days. These results suggest the absence of any potential compatibility issue. However, more detailed long-term confirmatory studies are warranted. Earlier reports demonstrated that RMPs produced from long-term stored blood increases levels of inflammatory markers. 42 -46 The impact of high-pressure, extrusion-derived RMPs on inflammatory markers is not known. Not studying the impact of high-pressure, extrusion-derived RMPs on inflammatory pathways is another limitation of our study.
Biodistribution studies define the spreading of therapeutic agents in animals or human participants. To ensure safe and successful application of RMP-based therapies for blood hemostasis, a better understanding of the biodistribution of administered RMPs is required. Techniques with high signal-to-noise ratio and no impact on half-life of the therapeutic agent being tested are preferred for biodistribution studies. Some of the high resolution and sensitivity techniques used in such studies on exosomes employed the use of superparamagnetic iron oxide nanoparticles and radioisotopes. 47 -49 Alternatively, the advantage of using fluorescence labeling with Alexa-488 tags, as used in the current study, is that it forms a covalent bond with proteins. Such binding is extremely stable resulting in brighter and highly photostable conjugates. 50 Previous biodistribution studies have successfully employed Alexa-488 to tag polymer hydrogel capsules. 51 The biodistribution study for RMPs was performed by quantifying levels of fluorescently labeled RMPs in organs analyzed at 2 time points (15 minutes and 24 hours) after IV administration, similar to an earlier study on a biomaterial like exosomes. 52 Although the technique used for RMP tagging with a fluorophore produces stable conjugates, not confirming the presence of fluorophore on RMPs at the time of analysis is a limitation of the study. Future studies using a different method of measuring RMP biodistribution may help confirm our findings.
Our group has previously observed that RMPs possess a very short half-life in the systemic circulation (unpublished observation). 53 Therefore, the minor differences observed in the blood levels of RMPs at 15 minutes and 24 hours of a bolus dose administration indicate potential redistribution of RMPs among the different body compartments in an unknown manner. Given the short half-life of RMPs, they are apparently not retained in the blood for long. The absence of sustained accumulation of RMPs in key organs such as heart, brain, and lungs is indicative of their reduced chances of causing side effects in respective tissues. The high levels of RMPs seen in the liver, kidneys, and spleen show that these organs are potentially responsible for the clearance of RMPs from the systemic circulation. Our studies did not evaluate the mechanism or identify cellular/subcellular sites of their metabolism. An earlier study observed that lactadherin mediates clearance of platelet-derived microvesicles. 54 Kupffer cells in the liver are proposed to mediate the clearance of RBC-derived vesicles from the circulation via scavenger receptors. 55 We surmise that similar mechanisms may be involved in the metabolism/clearance of RMPs.
Overall, these results are encouraging for the safety of RMPs. Product safety is of paramount importance and must be established before clinical trials. Animal studies revealed no obvious adverse effects of RMP infusion in short-term observations, even at high doses. No accumulation of RMPs was observed in heart, lung, and brain as well as no evidence of thromboembolism was found in histology sections of various tissues (heart, lungs, brain). These promising results warrant further studies in an independent laboratory, as required by regulatory agencies such as the US Federal Drug Administration. 56 Following these guidelines, extensive future studies are merited given the supportive results found in this initial safety and toxicity screening study.
Supplemental Material
Supplemental Material, Supplementary_section - Preclinical Evaluation of Safety and Biodistribution of Red Cell Microparticles: A Novel Hemostatic Agent
Supplemental Material, Supplementary_section for Preclinical Evaluation of Safety and Biodistribution of Red Cell Microparticles: A Novel Hemostatic Agent by Ashish K. Rehni, Vibha Shukla, Hever Navarro Quero, Carlos Bidot, Conner R. Haase, Ensign Anise A. Crane, Shivam G. Patel, Sebastian Koch, Yeon S. Ahn, Wenche Jy and Kunjan R. Dave in Journal of Cardiovascular Pharmacology and Therapeutics
Footnotes
Acknowledgment
The authors would like to thank Dr Brant Watson for critical reading of this manuscript.
Author Contributions
A. K. Rehni contributed to acquisition, analysis, and interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy. V. Shukla, H. Navarro-Quero, and C. Bidot contributed to acquisition and analysis; critically revised the manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy. C. R. Haase and E.A.A. Crane drafted the manuscript; critically revised the manuscript; and gave final approval. Patel, S contributed to analysis; critically revised the manuscript; and gave final approval. S. Koch contributed to interpretation; critically revised the manuscript; and gave final approval. Y. S. Ahn contributed to conception and design; contributed to interpretation; critically revised the manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy. W. Jy and K. R. Dave contributed to conception, design, analysis, and interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring integrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RxMP Therapeutics provided the testing material for the study. Dr Ahn, Dr Jy, and the University of Miami have partial ownership in RxMP Therapeutics. Drs. Ahn and Jy are the inventors of 2 US patents related to red cell microparticles.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Health [grant number NS094896]. The funding agency was not involved in study design; in the collection, analysis, and interpretation of data; in the writing of the report; and in the decision to submit the article for publication. Drs Ahn and Jy also received a grant support from RxMP Therapeutics.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
