Abstract
Poorly differentiated colonic carcinoma with rhabdoid features is a rarely described entity. Our knowledge regarding the molecular phenotype of the tumor is evolving. We herein report a similar tumor with rhabdoid differentiation identified in the splenic flexure, which on histological examination showed a poorly differentiated phenotype with epithelioid to spindled morphology, tumor giant cells, and rhabdoid differentiation. The tumor was mismatch repair-proficient, deficient of INI1/SMARCB1, KRAS mutated (A146×), BRAFV600E mutated (c.1799T > A), and NRAS wild-type, indicating serrated differentiation in the tumor. The patient died after 3.5 months post-surgery. INI1-deficient poorly differentiated carcinoma of the colon is a rare, aggressive colonic malignancy showing a serrated phenotype. Routine identification and subtyping are important keeping in mind the distinct tumor phenotype, resistance to conventional chemotherapy, and dismal prognosis.
Introduction
Malignant rhabdoid tumor was originally described as renal tumors of infancy and childhood. 1 Subsequently, similar clinicopathological features were described in carcinomas arising from extrarenal sites, such as gastrointestinal tracts, central nervous system, and soft tissues, and were collectively named the malignant extrarenal rhabdoid tumors, with poor prognosis and widespread metastasis. 1 Due to rarity, our experience of this tumor and its phenotype are still evolving. INI1 (SMARCB1) is a tumor suppressor gene and is a member of the chromatin remodeling complex SWI/SNF (SWItch/Sucrose Non-Fermentable) complex located at chromosome 22q11.2,2–4 and ubiquitously expressed in all normal human tissue types and all neoplasms with intact SMARCB1 locus. 2 Mutations/deletions involving the SMARCB1 locus have been associated with poor tumor differentiation or rhabdoid differentiation. 2 Other members of the SWI/SNF complex can be mutated in several cancer types including SMARCA4 (BRG1), SMARCA2 (BRM), SMARCC1 (BAF155), and SMARCC2 (BAF170). 5 Currently, the main entities identified under the spectrum of “INI1-deficient neoplasms” are pediatric atypical teratoid/rhabdoid tumors, malignant rhabdoid tumors, of the kidney and extrarenal soft tissue sites, proximal and distal epithelioid sarcomas, renal medullary carcinoma, epithelioid malignant peripheral nerve sheath tumors, and subsets of extraskeletal myxoid chondrosarcoma/myoepithelial neoplasms of soft tissue. 2 However, the spectrum of SWI/SNF deficient tumors is expanding, especially among poorly differentiated gastrointestinal tract tumors. Such tumors can be treated with immune-checkpoint inhibitors, hence should be characterized fully. 6
In this article, we are reporting a poorly differentiated colonic carcinoma with rhabdoid features showing INI1 deficiency and its molecular phenotype to understand the background of its poor prognosis. To date, only 44 such tumors have been reported in the literature on extensive Google Scholar and PubMed database searches and analyses of published data by 2 individual authors.7,8 We have also summarized the salient findings of the full papers available in Table 1.
The Table Shows All Reported Cases of Colorectal Carcinoma With Rhabdoid Differentiation, Their Phenotype, and Clinical Outcomes.
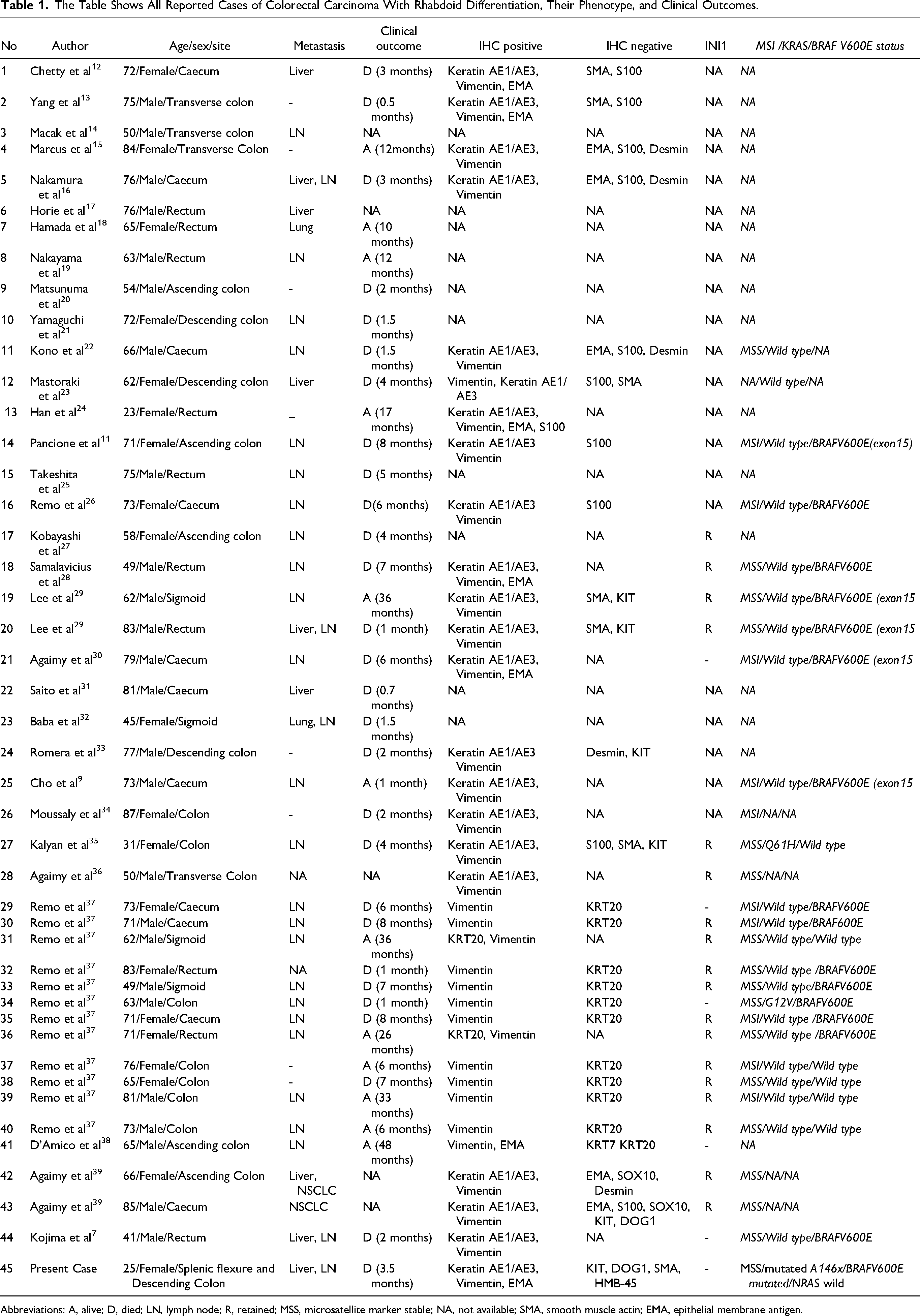
Abbreviations: A, alive; D, died; LN, lymph node; R, retained; MSS, microsatellite marker stable; NA, not available; SMA, smooth muscle actin; EMA, epithelial membrane antigen.
Case Presentation
A 25-year female patient presented with persistent weakness, pain in the left upper quadrant, and anemia for 5 months. On ultrasound and contrast-enhanced computer tomography, an ulceroproliferative growth was identified at the splenic flexure along with liver metastases and multiple small homogenously enhancing nodes, the largest measuring 7 mm in diameter. A short segment colo-colic intussusception was also identified (Figure 1A and B). Colonoscopic biopsy revealed features of a carcinosarcoma with pleomorphic spindled to epithelioid cells having positivity for keratin AE1/AE3 and vimentin. Stains for KIT, DOG1, KRT20, S100, ARG (Arginase1), smooth muscle actin (SMA), and HMB45 were negative. The tumor showed a mismatch repair (MMR) proficient phenotype. Soon after, she presented with large bowel obstruction and bleeding per rectum for 2 days and underwent a left hemicolectomy with a left-end colostomy. On gross examination, a large polypoidal growth, measuring 11 × 9 × 7 cm was identified at the splenic flexure obstructing the lumen, with transmural tumor invasion and serosal perforation (Figure 1C). The cut surface of the tumor showed hemorrhagic and necrotic areas. Microscopic examination revealed a spindled to epithelioid phenotype, with a prominent alveolar arrangement in the epithelioid tumor areas. Prominent rhabdoid differentiation and pleomorphic multinucleated tumor giant cells were identified (Figure 2A-C). Numerous mitotic figures, foci of necrosis, lymphovascular tumor emboli, and lymph node metastases (7/16) were identified. Tumor deposits were present in the submitted omentectomy specimen. Based on histomorphology, differential diagnoses of a gastrointestinal stromal tumor with rhabdoid differentiation, alveolar soft part sarcoma, clear cell-like soft tissue tumor of the gastrointestinal tract, PECOMA, poorly differentiated colonic carcinoma with rhabdoid differentiation and metastatic carcinomas with rhabdoid differentiation were considered. Detailed immunophenotyping showed positivity for KRT1B, vimentin, and epithelial membrane antigen, and the tumor cells were negative for KIT and DOG1. Focal positivity for SMA, desmin, and myogenin was noted. CD34, S100, synaptophysin, chromogranin, HMB45, and TFE3 stains were negative (Figure 2D-I). TFE3-break apart fluorescent in-situ hybridization (FISH) Probe analysis did not show TFE3 translocation in the tumor cells (Figure 2J). On further analyses, the tumor showed loss of SMARCB1/INI1, while SMARCA4/BRG1 stain was retained (Figure 2G and H). Hence, a final diagnosis of INI1-deficient poorly differentiated colonic carcinoma with rhabdoid differentiation was given. Like the preoperative biopsy, the resected tumor also showed MMR-proficient phenotype. On genetic analyses, the tumor showed a KRAS mutated (A146×), BRAFV600E mutated (c.1799T > A) and NRAS-wild phenotype, closely mimicking the serrated tumor phenotype of the colon.
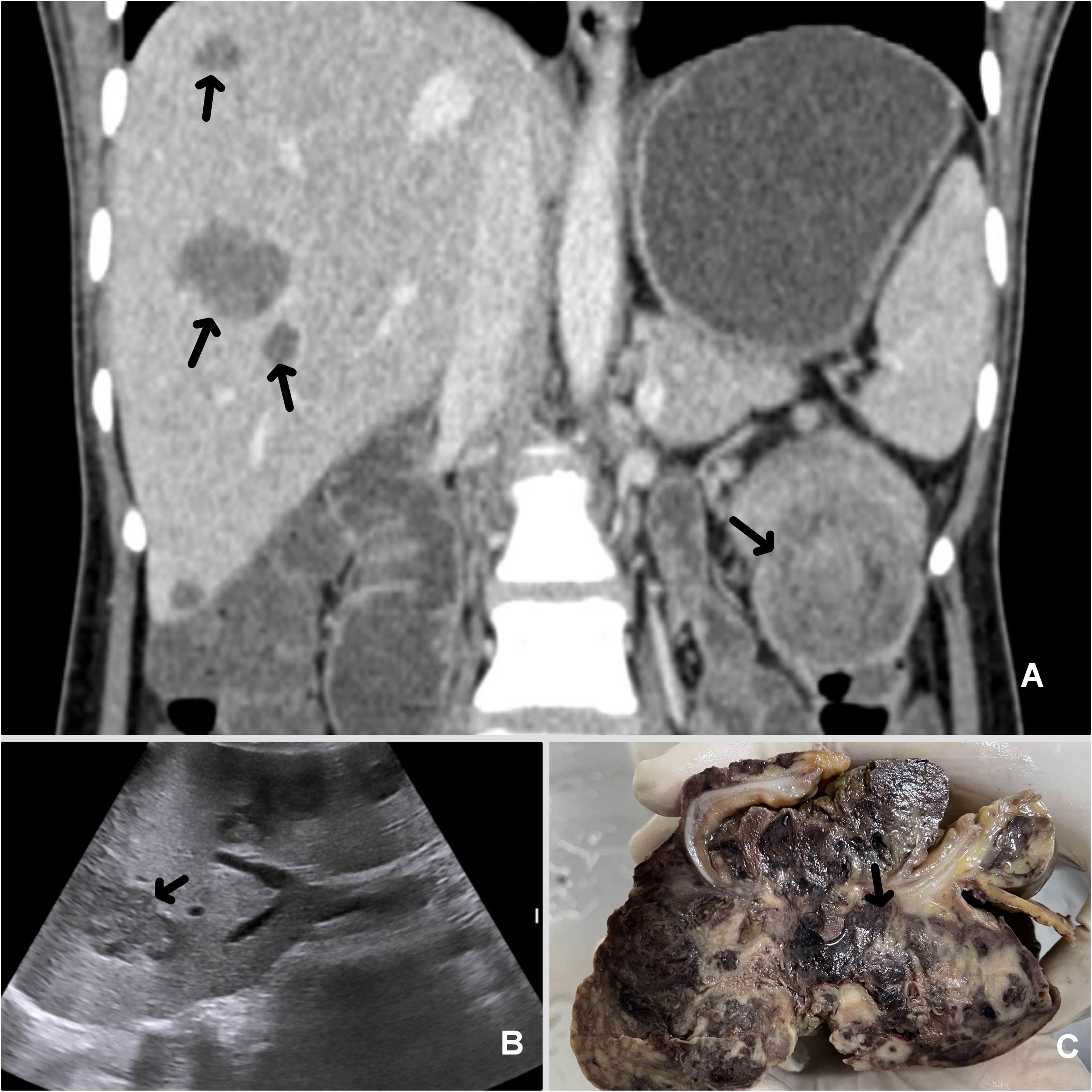
Coronal computed tomogram image shows a polypoidal growth at splenic flexure (A, arrow) of the colon with colo-colic intussusception and multiple foci of hepatic metastases (A, arrows). Ultrasonogram also showed hepatic metastases (B, arrow). The gross cut surface of the resected tumor showed a heterogeneous tumor mass with polypoidal projection into the lumen, transmural tumor infiltrates (arrow) with serosal perforation, and adjacent extension (C).
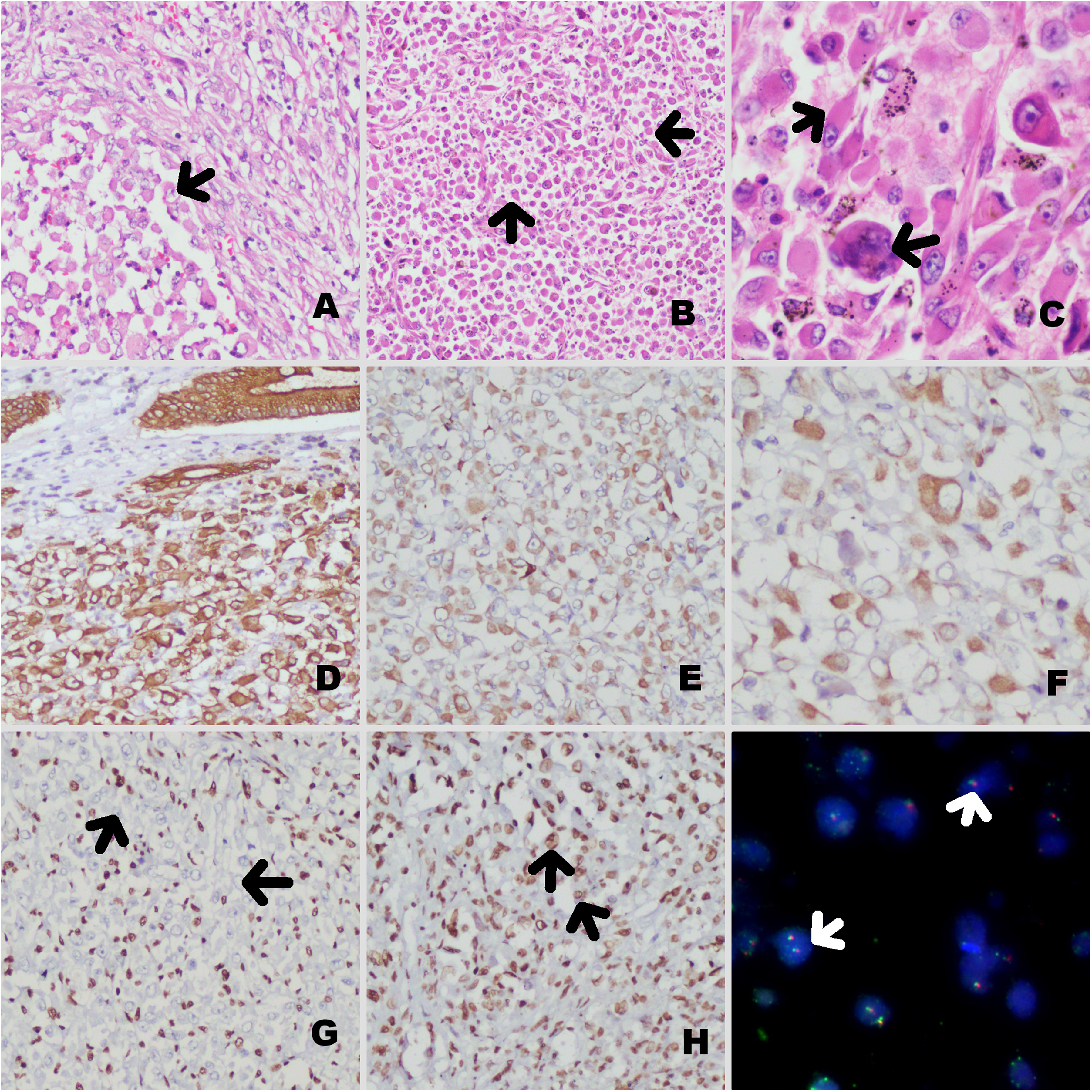
The tumor on histological examination showed spindled to epithelioid differentiation (arrow) [A×200]. In the epithelioid areas, tumor cells were arranged in a pseudo-alveolar pattern (arrows) [B×100]. The tumor cells were pleomorphic and showed multinucleated tumor giant cells and rhabdoid differentiation (arrows) [C×400]. The tumor cells were positive for KRT1B [D×200], vimentin [E×200], and desmin [F×400], with loss of INI1 stain in tumor cells (arrows) [G×200], retained BRG1 stain (arrow) [H×200], and no translocation on TFE3 FISH analysis (arrows) [I×400].
Three weeks post-surgery, the patient started having on-and-off episodes of bleeding per rectum, followed by bleeding from the midline wound site, and had a fall in hemoglobin to 7 g/dL and was readmitted after 2 months of surgery. CT angiography showed an 8.8 × 5.6 × 5 cm growth arising from the anterior abdominal wall with cystic changes but no point of active bleeding. The patient was planned for chemotherapy with capecitabine and oxaliplatin but succumbed to the disease 3.5 months after the operation.
Discussion
The present example emphasizes the need for detailed workup in colonic tumors showing poor differentiation, spindling, and rhabdoid differentiation, especially in terms of the status of SWI/SNF complex members, MMR markers, and molecular phenotype. It seems that this rarely reported tumor is another example of serrated colonic tumors, in addition to serrated colorectal adenocarcinomas. Not only the histomorphology and tumor phenotype are unique but also show a universally aggressive outcome and resistance to conventional chemotherapy regimens, unlike all other poorly differentiated adenocarcinomas of the colon.
Rhabdoid tumors are aggressive neoplasms, initially described by Beckwith and Palmer as a sarcomatoid rhabdoid variant of Wilms’ tumor.9,10 Tumors with similar clinicopathological characteristics were subsequently reported in several extrarenal sites with an unfavorable prognosis. 11 Of these, colorectal cancers with rhabdoid features are extremely rare, and to date, only 44 such tumors have been reported.11–39 Histologically, rhabdoid tumors are characterized by the unique morphological feature of proliferating rhabdoid cells, which have a polar large nucleus, prominent nucleoli, and typical eosinophilic cytoplasmic inclusion made up of aggregated intermediate filaments.1–3,7–9,11–39 All gastrointestinal tract tumors with similar histology have been reviewed and listed in Table 1.
The histology of the current tumor was elusive and appeared on pre-operative mucosal biopsies as a carcinosarcoma. The histology in the resected specimen was further striking, showing distinct pleomorphic spindled to epithelioid tumor cells, distinct alveolar arrangement, and associated prominent rhabdoid differentiation, giving rise to several differential diagnoses discussed earlier. This report shows that such tumors can be INI1 deficient, MMR-proficient, and have a distinct serrated molecular phenotype. Clinically and pathologically, the tumor had several poor prognostic factors including omental deposits, LVI, and multiple liver metastases. In most of the reported patients, mortality reported was around 61%, mostly within one year of diagnosis (mean 4 months), regardless of the surgical extension and radio/chemotherapy instituted. Metastasis in such tumors was correlated with the INI1 deficient phenotype, further stressing the need for a detailed workup. 40 The SWI/SNF complex regulates transcription, proliferation, and metastasis-related genes.5,30,41 Of all the 44 tumors reviewed, only in 23 tumors INI1 immunostaining was performed, out of which 5 (22%) showed INI1 loss. In INI1 deficient tumors, oncogenic transformation for several targets such as cyclin D1 and p16Ink4a has been identified. 41 However, it should be remembered that in a tumor with such histology, retained INI1 stain does not rule out an SWI/SNF tumor phenotype. Almost 29 genes have been identified in this complex, and these tumors can show mutation of other component genes such as BRG1 and BRM.6,42 The reason why such a workup is necessary is that of promising results with anti-PD-1 and anti-CTLA-4 antibodies in such tumors in clinical trials.
Regarding the molecular phenotype, out of the 44 reported tumors, in 23 tumors KRAS mutation was examined, of which 21 had wild-type genes. In the mutated tumors Q61H, G12 V mutations were identified. In our patient, a mutation in codon 146 was identified. NRAS gene mutation was, however, not identified. This information is important, as metastatic colonic tumors are eligible for anti-epidermal growth factor receptor therapy in combination with conventional chemotherapy if having the KRAS “wild type” gene. Out of the reported 44 tumors, in 21 cases BRAF-gene mutation was examined, of which, 15 had a BRAFV600E mutation. BRAF-mutated colonic carcinomas show aggressive tumor behavior. Also, out of only 26/44 tumors where microsatellite status was studied, 10 showed microsatellite instability. This indicates that a subset of poorly differentiated colonic carcinoma with rhabdoid differentiation shows characteristic serrated molecular phenotype (KRAS gene wild/mutated, BRAF-gene mutated, and MMR proficiency). Such tumors have been included in the WHO fifth edition as large cell carcinoma with rhabdoid features. 43 However, when such tumors are described as poorly differentiated carcinoma with rhabdoid differentiation with mention of INI1 status and serrated phenotype if any, reveals their true detailed phenotype.
Of the 44 reported patients, all received surgery as the initial treatment, and 20 of 29 of them also received neo/adjuvant radio/chemotherapy. The remainder had no detailed treatment data. Regional node metastasis was identified in 27 patients with detailed information (64%). Liver metastasis was diagnosed in 8 patients, including ours. Of the 44 patients, 27 (61%) died of disease within 1 to 12 months (mean 4 months). Our patient also died 3.5 months post-surgery. Only 11 patients with a follow-up of 6 to 36 months were alive and disease free at the last follow-up. In a study by Kojima et al, the authors identified an overall median survival of 3 months in patients having poorly differentiated colorectal carcinoma with rhabdoid differentiation out of 27 of their tumors, and 8 out of 9 of their patients with defined histology died within a year. 7
Conclusions
Poorly differentiated carcinomas of the colon with rhabdoid differentiation represent a rare group of SWI/SNF-mediated tumors, with an aggressive outcome. These tumors have a distinct MMR-stable, serrated phenotype. They are often resistant to conventional chemotargeted therapy and rarely do such patients survive over a year post-diagnosis; hence, deserve an early histological identification and characterization
Footnotes
Authors’ Contribution
Shiv, Suj, PD, and AB are responsible for the diagnosis and workup of the patient. SB, PD, AS, and AK are responsible for manuscript writing and reviews. JD, RS, AS, and AK are responsible for the clinical management of the patient. LM is responsible for performing wet lab investigations. PD is the guarantor of the article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
