Abstract
Undifferentiated carcinomas are highly aggressive tumors with a dismal prognosis. A subset of these tumors has been associated with inactivation or mutations of the Switch/Sucrose Nonfermenting (SWI/SNF) remodeling complex. Our understanding of the relationship between the clinicopathological features and molecular profiling of SWI/SNF-deficient undifferentiated carcinoma is still evolving due to its rarity. We herein present a rare tumor of undifferentiated carcinoma with SMARCB1/INI1 deficiency arising from the colon. The histology revealed a tumor composed of sheets of discohesive, high-grade epithelioid cells with rhabdoid morphology along with anaplastic giant cells. Additionally, there was a significant infiltration of inflammatory cells in the background. Immunohistochemical (IHC) analysis supported the diagnosis of carcinoma with loss of INI1 expression, the tumor was mismatch repair protein proficient. Molecular analysis demonstrated an oncogenic KRAS mutation (p.G12D), whereas it was wild-type BRAF, and wild-type NRAS. The diagnosis of SWI/SNF-deficient undifferentiated carcinoma can be challenging. Correlation with clinical findings, in conjunction with IHC work-up and molecular analysis, is of utmost importance to arrive at the appropriate diagnosis and exclude potential mimics.
Introduction
Undifferentiated carcinomas, defined by a lack of definite histological features of squamous, glandular, or neuroendocrine differentiation, are rare and aggressive malignancies associated with poor prognosis. 1 Often displaying rhabdoid features, these tumors have been linked to the inactivation of the Switch/Sucrose Nonfermenting (SWI/SNF) chromatin-remodeling complex, composed of a number of core subunits including SMARCB1. SMARCB1, acting as a tumor suppressor and found on chromosome 22q11.2, 2 is normally expressed in all human tissues. However, its loss, leading to SMARCB1/INI1 deficiency, is frequently observed across a variety of human tumors, ranging from benign to highly aggressive forms. 3
SMARCB1/INI1-deficient undifferentiated/rhabdoid carcinomas affecting the gastrointestinal (GI) tract are extremely rare, only several dozen tumors have been documented thus far.4,5 In this report, we present an undifferentiated colonic carcinoma demonstrating INI1 deficiency and its molecular profiles to highlight the diagnostic challenges. Additionally, we conducted an extensive review of the literature to summarize clinicopathological and molecular characteristics observed in similar tumors.
Case Report
A 70-year-old man with no significant medical history presented with a sudden onset of generalized abdominal pain. Bowel obstruction was suspected, and subsequent abdominal computed tomography scan was performed. This revealed circumferential wall thickening in the distal right colon, raising suspicion of a neoplastic process. Consequently, the patient underwent a right hemicolectomy at an outside hospital. Upon gross examination of the resected specimen, a mass measuring 7.3 × 4.7 × 3.2 cm was identified within the ascending colon. The mass appeared near circumferential, extending through the muscular wall and potentially involving the serosa. Computed tomography chest revealed no evidence of metastatic disease within the chest and a dominant thyroid nodule in the left lobe.
Histologic sections of the tumor demonstrated sheet-like poorly cohesive cells with prominent tumoral inflammatory cell infiltration, consisting of neutrophils and lymphocytes (Figure 1A). The tumor lacked intervening stroma and neoplastic cells exhibited medium to large-sized rhabdoid morphology characterized by polygonal configuration and large vesicular eccentrically located nuclei with prominent nucleoli (Figure 1A). The cytoplasm contained large hyaline paranuclear inclusions that pushed the nucleus to the periphery of the cells. Binucleated cells, multinucleated giant cells, or anaplastic giant cells with similar nuclear and cytoplasmic features were also common, with some resembling hallmark cells, a characteristic feature of anaplastic large-cell lymphoma (Figure 1A). Notably, no definitive gland formation or precursor lesions were identified in the colonic mucosa (Figure 1A). The neoplasm extended from the mucosa through the muscularis propria into the pericolic fat. High mitotic activity, focal areas of necrosis, and evidence of lymphovascular invasion were noted. However, no lymph node metastases were detected among the 22 lymph nodes submitted for examination.
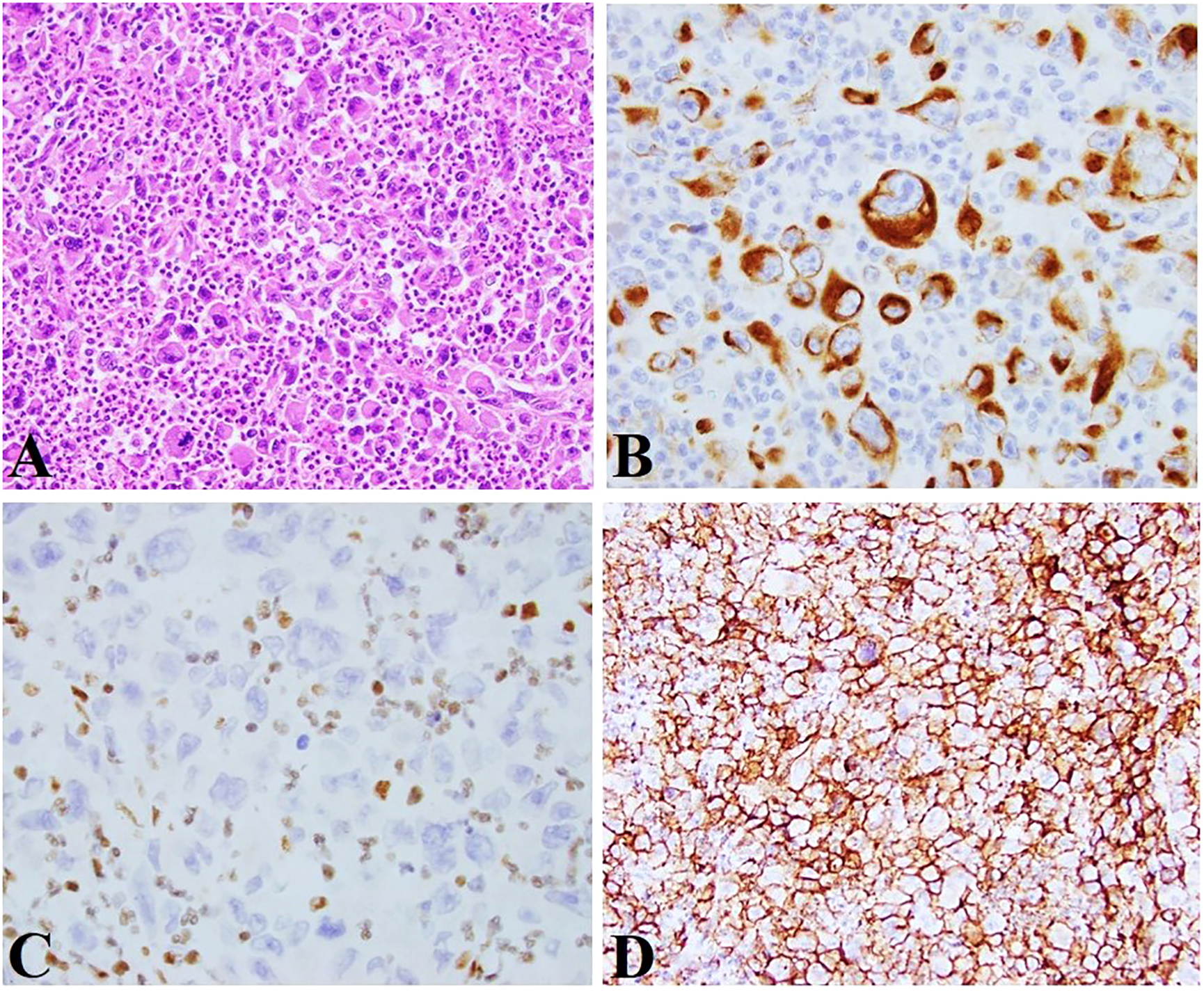
(A) Poorly cohesive growth pattern was seen with medium-large rhabdoid tumor cells and a prominent inflammatory cell infiltrate (×200); (B) The tumor cells were positive for keratin AE1/AE3 (×400); (C) The tumor cells demonstrated loss of INI1 expression (note the positive internal control within the surrounding inflammatory cells) (×400). (D) The tumor cells showed diffuse cell membrane staining with claudin 4 (×200).
Based on histomorphology, broad differential diagnoses were considered including gastrointestinal stromal tumor (GIST) with rhabdoid differentiation, melanoma, anaplastic large cell lymphoma, rhabdomyosarcoma, malignant extrarenal rhabdoid tumor, epithelioid sarcoma, mesothelioma, undifferentiated colonic carcinoma with rhabdoid features, and metastatic carcinoma with rhabdoid differentiation. Immunohistochemical (IHC) stains showed the tumor cells were positive for keratin AE1/AE3 (Figure 1B), with a weak, patchy expression of CD30 and PAX8, whereas they were negative for KIT, S100, SOX10, Melanin A, MITF, CD45, desmin, keratin 7, keratin 20, CDX2, GATA3, keratin 5/6, MOC31, calretinin, CD31, CD34, and OCT3/4. These combined findings argued against GIST, melanoma, metastatic urothelial carcinoma, and mesothelioma. The weak positivity of PAX8 and CD30 raised the possibility of renal origin and lymphoma. Therefore, CD10, CA9, PAX5, ALK1, CD4, and CD7 were tested at our institution which demonstrated weak, patchy nonspecific positivity for CD10 and CA9, with negative expression for ALK1, CD4, PAX5, and CD7, excluding these entities. Additional stains were performed at our institution and were all negative to exclude other rare possibilities including TTF1 and thyroglobulin to exclude thyroid origin as the patient reportedly had a left thyroid nodule, MYOD1, and myogenin to exclude rhabdomyosarcoma, ERG, CD31, and CD34 to exclude epithelioid angiosarcoma, and CD68 and CD163 to exclude histiocytic neoplasms. INI1 demonstrated a loss of expression within the neoplastic cells (Figure 1C) which express claudin 4 (Figure 1D). Hence, a final diagnosis of SMARCB1/INI1-deficient undifferentiated colonic carcinoma was rendered, staged as pT3N0 according to the American Joint Committee on Cancer Eighth Edition.
Analysis for the mismatch repair proteins MLH1, MSH2, MSH6, and PMS2 showed retained expression, indicating the tumor was proficient mismatch repair (pMMR). Molecular testing revealed an oncogenic KRAS mutation (p.G12D), whereas it was wild-type BRAF and wild-type NRAS. No additional mutations were detected in our 35-gene solid tumor next-generation sequencing panel (note, the panel at this time did not have any SWI/SNF genes included).
Detailed clinical management and follow-up information for this patient were unavailable as the patient was receiving care from an external facility.
Discussion
SMARCB1/INI1 loss was first discovered in pediatric malignant rhabdoid tumors. 6 Since then, this gene has been implicated in the pathogenesis of an increasingly diverse group of human tumors including undifferentiated carcinoma. 7 However, studies have shown that SMARCB1/INI1-deficient undifferentiated colon carcinoma is exceedingly rare, constituting only 0.4% to 0.7% of all colorectal carcinomas.5,8
A comprehensive search using PubMed and Google Scholar was conducted, utilizing the terms “SWI/SNF,” “colorectal,” and “carcinoma,” which yielded 40 additional tumors originating from the colon. Hence, including our presented tumor, a total of 41 detailed descriptions of undifferentiated colorectal carcinomas with SWI/SNF deficiency have been reported. The key clinicopathologic characteristics are summarized in Supplemental Material 1.4,9–21 Among the patients, there were 25 men and 16 women, with a mean age of 60 years (ranging from 25 to 85 years old). The majority of tumors (66% 25/38) were located in the right and transverse colon, whereas 13 (34%) were situated in the left colon. Tumor size ranged from 2 to 12 cm, with a mean size of 7 cm. Histologically, these tumors consistently exhibited a medullary-like growth pattern, tumor cell discohesion, and anaplastic and pleomorphic histology. Rhabdoid morphology was frequently observed in 26 of the reported tumors (87%, 26/30). Notably, definite glandular differentiation was mostly absent, with only minor areas suggestive of abortive gland formation detected in 6 tumors (20%, 6/30). This absence of definitive glandular features can pose a diagnostic challenge in identifying this as carcinoma. However, it is important to consider that this may be attributed to sampling issues, where a minor epithelial component can be easily overlooked. Therefore, thorough sampling is recommended in these challenging tumors. 14 Moreover, our tumor and similar tumors from the literature did not show remnants of any epithelial in situ components. 14 Differentiating between primary and metastatic undifferentiated carcinoma can be challenging, necessitating careful examination of clinical history and imaging data. Immunohistochemical analysis revealed that the tumor cells often expressed epithelial markers, with epithelial membrane antigen (EMA) being universally expressed in all tumors (100%, 5/5), and variable expression of keratin AE1/AE3 (91%, 20/22), CDX2 (25%, 4/16), keratin 20 (17%, 5/29), and keratin 7 (7%, 1/14). These findings indicate that EMA can be valuable in recognizing the epithelial origin, particularly in tumors with limited keratin expression. Furthermore, vimentin coexpression was observed in nearly all tumors (97%, 29/30).
Agaimy et al 13 were among the first to illustrate the close link between SWI/SNF deficiency and the aggressive rhabdoid phenotype. Subsequently, a series of studies have been published, revealing that loss of at least one subunit of the SWI/SNF complex, either in isolation or in various combinations, is commonly encountered in this highly aggressive tumor. Importantly, it has been noted that the inactivation of SMARCA4 and SMARCB1 is mutually exclusive.5,14,17,20 Our review of 41 tumors further supports these findings, identifying a complete loss of expression for SMARCB1/INI1 in 37% of tumors (15/41), SMARCA4/BRG1 in 25% (4/16), SMARCA2/BRM in 79% (11/14), and ARID1A in 33% (4/12). Notably, none of the 11 tumors with all 4 aforementioned SWI/SNF components tested demonstrated complete retained expression for all components. No SMARCB1-deficient tumors co-occurred with loss of SMARCA4, consistent with the previous findings that deficiency of SMARCB1 and SMARCA4 was mutually exclusive. Additionally, the loss of SMARCA2 was observed in combination with loss of SMARCA4 (n = 3), SMARCB1 (n = 1), or ARID1A (n = 2). It is worth noting that not all undifferentiated carcinomas with rhabdoid morphology show loss of SMARCB1/INI1, therefore, it is imperative to exclude the possibility of loss of an alternative SWI/SNF subunit in the work-up of these tumors.
Regarding the molecular profile, SWI/SNF-deficient undifferentiated carcinoma has been associated with various gene mutations commonly found in conventional colorectal carcinoma. These include KRAS mutations, BRAF mutations, DNA mismatch repair deficiency (dMMR), CpG island methylator phenotype, and TP53 mutations.14,16 In the reported tumors, 36 tumors underwent mismatch repair (MMR) testing, with 11 (31%) exhibiting MMR deficiency, all of which originated from the right colon and transverse colon. In line with previous observations, 16 where BRAF mutations emerged as the prominent molecular feature of rhabdoid undifferentiated carcinoma, this was detected in 15 tumors (15/25, 60%) and was frequently associated with pMMR (10/15, 67%). Moreover, TP53 alterations were observed in 8 tumors (8/12, 67%) with a high concurrence rate of BRAF mutations (3/6, 50%). Additionally, KRAS mutations were detected in 6 tumors (6/24, 25%), all of which exhibited proficient MMR status. However, the precise clinical significance of these molecular associations remains controversial.5,16,17 Therefore, further comprehensive molecular profiling is necessary to gain a deeper understanding of the interplay between the clinicopathological features of undifferentiated carcinoma and their molecular profile.
Rhabdoid undifferentiated carcinomas have shown resistance to conventional therapies commonly used for GI neoplasms, such as the FOLFOX (folinic acid, fluorouracil and oxaliplatin) regimen combined with monoclonal antibodies. Furthermore, anthracycline-based regimens typically employed in sarcoma treatment have demonstrated limited efficacy. 16 However, as our understanding of SWI/SNF biology continues to evolve, numerous potential therapeutic targets have been identified. Among these targets, inhibitors of PRC2 (Polycomb Repressive Complex 2), such as tazemetostat, have been developed to specifically inhibit EZH2 (enhancer of zeste homolog 2), the catalytic subunit of PRC2. Tazemetostat has gained FDA approval for the treatment of SMARCB1-deficient epithelioid sarcoma.2,22–24 Additionally, emerging evidence suggests that SWI/SNF components play crucial roles in the regulation of anti-tumor immunity, indicating that SWI/SNF deficiency could be targeted therapeutically by immune checkpoint inhibitors. 25 Clinical trials are currently underway to investigate the potential clinical benefits of immune checkpoint inhibition in SMARCB1-deficient cancers.22,26
SWI/SNF-deficient undifferentiated carcinomas are clinically characterized by their aggressive behavior and poor prognosis. Among the reviewed tumors, all but 2 patients presented with metastasis either synchronously or shortly after diagnosis. Thirty-four patients had follow-up data ranging from 1 month to 216 months (median: 7 months). Among these patients, 13 individuals (35%) were still alive at the time of follow-up, whereas 21 patients (65%) succumbed to disease progression (median survival: 6 months, range 1-27 months). Notably, the majority of deaths occurred within one year after diagnosis, highlighting the rapid disease progression and the challenging nature of managing these aggressive tumors. Kaplan-Meier analysis revealed that SMARCB1 deficiency (P < .01, HR 3.0 (1.3-7.2) and BRAF mutation (P < .05, HR 3.4 (1.1-10.7)) were 2 significant risk factors predictive of poor prognosis (Figure 2). Given the rarity of this tumor, further evaluation is needed to explore the underlying genetic risk factors.
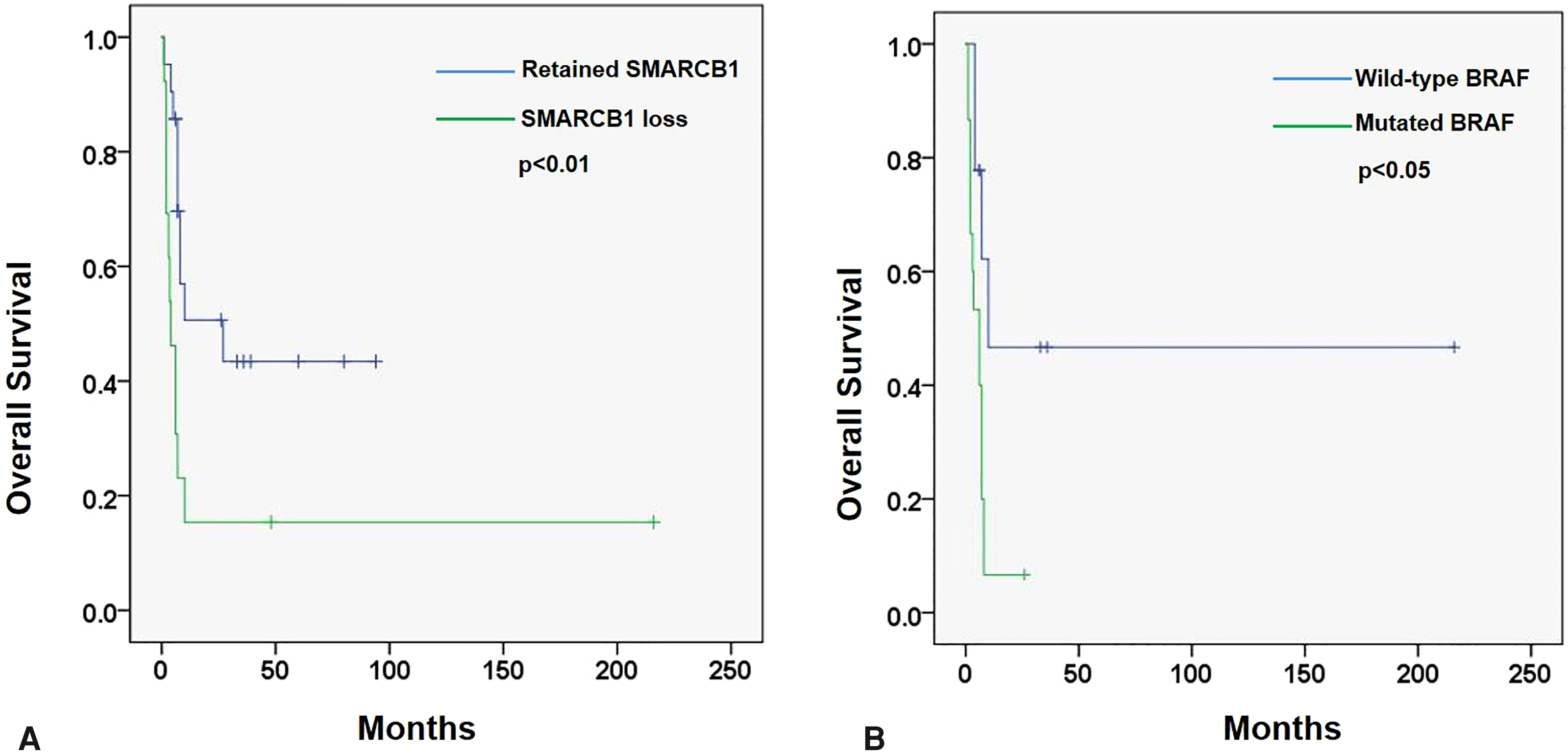
Kaplan-Meier analysis demonstrated that SMARCB1/INI1 loss (A) and BRAF mutation (B) were significantly associated with poor overall survival in patients with rhabdoid undifferentiated colonic carcinoma.
A timely and accurate diagnosis of SWI/SNF-deficient undifferentiated carcinoma is crucial due to its clinical and therapeutic significance. The undifferentiated nature of these tumors necessitates a comprehensive differential diagnosis. Histologically, metastatic melanoma with rhabdoid features can closely resemble undifferentiated carcinoma, making clinical history and IHC staining with melanoma markers essential for establishing the correct diagnosis. In rare tumors where melanoma is undifferentiated with loss of expression of commonly used melanoma markers, performing SWI/SNF immunohistochemistry stains or genotyping the tumor may be necessary to resolve diagnostic ambiguity.27,28 Additionally, aggressive hematological malignancies expressing EMA, such as anaplastic large cell lymphoma with rhabdoid morphology and plasmablastic malignancies, can exhibit similar histological features and should be argued against using hematological markers. 27 Besides undifferentiated carcinoma, rhabdoid features with loss of SMARCB1/INI1 are frequently associated with epithelioid sarcoma and malignant rhabdoid tumors. The distinction between those entities may be challenging as the histological, IHC, and molecular patterns of those sarcomas are nearly identical to undifferentiated carcinoma. However, tumor localization in the GI tract and their demographic features are similar to common GI cancer and clearly different from those of epithelioid sarcoma and malignant rhabdoid tumor. Additionally, the presence of a focal glandular component or abortive gland formation is useful, along with the presence of common colorectal genetic aberrations by molecular analysis. In a study assessing claudin 4 expression in SWI/SNF complex-deficient neoplasms, it was found that 80% of SWI/SNF complex-deficient undifferentiated carcinomas exhibited membranous expression of claudin 4. Conversely, claudin 4 expression was absent in other soft tissue tumors, suggesting that claudin 4 expression is strongly associated with true epithelial differentiation. 29 This finding suggests that claudin 4 could serve as a useful marker to distinguish SWI/SNF complex-deficient undifferentiated carcinomas from epithelioid sarcomas, as demonstrated in our tumor.
In summary, undifferentiated carcinoma with SMARCB1/INI1 deficiency in the colon is an exceptionally rare and aggressive malignancy associated with unfavorable outcomes. The presence of a discohesive growth pattern and rhabdoid morphology should raise suspicion for this specific entity, necessitating further investigation. The recognition of this rare variant holds important therapeutic implications and prognostic significance, emphasizing the need for accurate identification and appropriate management strategies.
Supplemental Material
sj-docx-1-ijs-10.1177_10668969241232699 - Supplemental material for Elusive and Aggressive: Unraveling SMARCB1/INI1-Deficient Undifferentiated Carcinoma With Rhabdoid Features Arising From the Colon: A Case Report and Comprehensive Literature Review
Supplemental material, sj-docx-1-ijs-10.1177_10668969241232699 for Elusive and Aggressive: Unraveling SMARCB1/INI1-Deficient Undifferentiated Carcinoma With Rhabdoid Features Arising From the Colon: A Case Report and Comprehensive Literature Review by Xiaoqin (Lucy) Liu and Diana Agostini-Vulaj in International Journal of Surgical Pathology
Supplemental Material
sj-xlsx-2-ijs-10.1177_10668969241232699 - Supplemental material for Elusive and Aggressive: Unraveling SMARCB1/INI1-Deficient Undifferentiated Carcinoma With Rhabdoid Features Arising From the Colon: A Case Report and Comprehensive Literature Review
Supplemental material, sj-xlsx-2-ijs-10.1177_10668969241232699 for Elusive and Aggressive: Unraveling SMARCB1/INI1-Deficient Undifferentiated Carcinoma With Rhabdoid Features Arising From the Colon: A Case Report and Comprehensive Literature Review by Xiaoqin (Lucy) Liu and Diana Agostini-Vulaj in International Journal of Surgical Pathology
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
