Abstract
Combined hepatocellular-cholangiocarcinoma with a ductal plate malformation pattern is an extremely rare entity with unelucidated pathogenesis. We present the case of a 60-year-old male patient who underwent a sectionectomy for pre-operative diagnosis of hepatocellular carcinoma based on clinical and image findings. Gross examination of the specimen revealed a well-defined tumor with cystic change measuring 6.7 × 6.2 cm. Microscopically, the lesion had classical features of hepatocellular carcinoma and intrahepatic cholangiocarcinoma exhibited neoplastic glands with irregular-sized dilated lumens, resembling a ductal plate malformation. Postoperative diagnosis was combined hepatocellular-cholangiocarcinoma with ductal plate malformation pattern. Next-generation sequencing revealed genomic alteration in 15 genes: CDKN2A, CHD4, CYP2D6, ERBB3, KIR3DL1, KRAS, MDM2, PIM1, STAT6, TPMT amplification, FANCD2, FAT1, FLT4, RASA1, and TP53 point mutation. This is the first case report of molecular alteration in combined hepatocellular-cholangiocarcinoma with ductal plate malformation pattern.
Introduction
Primary liver cancer is common and is the third leading cause of cancer-related mortality worldwide. 1 The incidence of combined hepatocellular-cholangiocarcinoma among primary liver cancers varies according to study (ranges from 0.4% to 14.2%). 2 Hepatocellular carcinoma (HCC) is a malignant tumor with hepatocellular differentiation, and intrahepatic cholangiocarcinoma is a malignant tumor with biliary epithelial differentiation. There are several subtypes of intrahepatic cholangiocarcinoma based on histopathological phenotype. Recently, a variant of intrahepatic cholangiocarcinoma with a ductal plate malformation pattern has been reported. 3 Combined hepatocellular-cholangiocarcinoma with ductal plate malformation pattern is an extremely rare entity. Since 2012, only 4 cases including the present case have been reported.3–5 The clinicopathological features of combined hepatocellular-cholangiocarcinoma with ductal plate malformation pattern have not been clarified. Analyzing the molecular features of combined hepatocellular-cholangiocarcinoma with ductal plate malformation pattern may be helpful to elucidate the pathogenesis of this entity. Here, we report a case of combined hepatocellular-cholangiocarcinoma with ductal plate malformation pattern and a result from next-generation sequencing (NGS) analysis. In addition, we briefly review the literature for clinical and histopathological features of combined hepatocellular-cholangiocarcinoma with ductal plate malformation pattern.
Case Report
A 60-year-old male patient presented to the surgery department for evaluation of an incidentally detected liver mass. He had a history of untreated hepatitis B and chronic alcohol intake. Laboratory examinations were as follows: serum tumor markers, alpha-fetoprotein (AFP): 2577.2 ng/mL [normal < 8.1 ng/mL] and protein induced by vitamin K antagonist-II (PIVKA-II): 94 mAU/mL [normal < 40 mAU/mL]. Liver function test showed elevated aspartate transaminase 37 IU/L [normal 12-33 IU/L], alanine transaminase 58 IU/L [normal 5-35 IU/L], and gammaglutamyl transferase 120 IU/L [normal 12-73 IU/L]. Alkaline phosphatase level was 99 IU/L [normal 45-129 IU/L]. Contrast-enhanced computed tomography (CT) demonstrated a 7.7 cm-sized mass in right lobe of the liver. The mass was composed of a solid portion and a nonsolid portion. The solid portion of the mass showed heterogeneous nonrim arterial hyperenhancement (Figure 1A) and delayed washout (Figure 1B), while the nonsolid portion (Figure 1A and B), which was regarded as fat, showed subtle progressive enhancement throughout the delayed phase. Based on CT reading, hypervascular HCC with a portion of fatty change was suspected. For further evaluation, the patient underwent gadoxetic acid-enhanced magnetic resonance imaging. The nonsolid portion of the mass demonstrated bright hyperintensity on T2-weighted imaging with a portion of multilocular cystic appearance and some fluid–fluid levels (Figure 1C and D), which was regarded as necrotic change with hemorrhage. After injection of gadoxetic acid, the solid portion showed strong nonrim hyperenhancement, followed by washout and capsule appearance. Based on these clinical findings, the final impression was hypervascular HCC with necrotic change. In addition, the diagnostic evaluation was based on clinical and radiological findings which revealed no evidence of fibropolycystic liver diseases. Hence, the patient underwent sectionectomy for diagnosis and treatment and experienced no recurrence or metastasis for over 6 months.
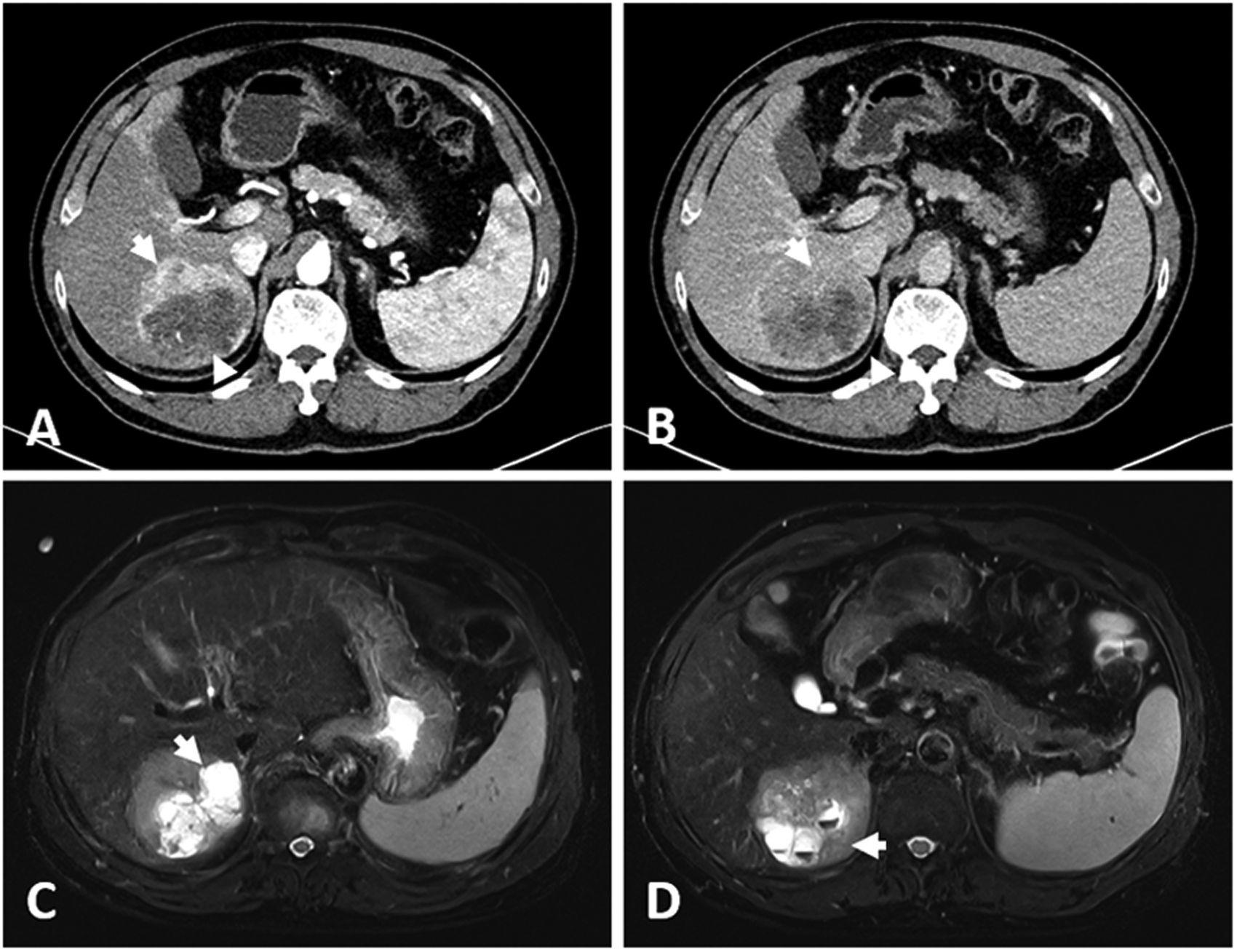
Radiological findings of combined hepatocellular-cholangiocarcinoma with ductal plate malformation pattern. Contrast-enhanced computed tomography demonstrated a 7.7 cm-sized mass in the right lobe of the liver. (A) The solid portion of the mass showed heterogeneous nonrim arterial hyperenhancement (arrow) and (B) delayed washout (arrow). The nonsolid portion (arrowheads), which was regarded as fat, showed subtle progressive enhancement throughout the delayed phase. (C) On gadoxetic acid-enhanced magnetic resonance imaging, the nonsolid portion of the mass demonstrated bright hyperintensity on T2-weighted image with a portion of multilocular cystic appearance (arrow). (D) The nonsolid portion of the mass showed some fluid–fluid levels (arrow).
Gross examination of the specimen revealed a well-defined tumor measuring 6.7 × 6.2 cm. The visible cystic area ranged from 0.2 to 1.1 cm in diameter (Figure 2A). Microscopic findings of a resected specimen revealed the following 2 components: HCC and intrahepatic cholangiocarcinoma with ductal plate malformation pattern. Histologically, the tumor gradually transitioned from the HCC component to intrahepatic cholangiocarcinoma with ductal plate malformation pattern. The HCC component showed a trabecular pattern and significant pleomorphism (Figure 2B). The histopathological finding confirmed moderately differentiated HCC. There was also a transition between HCC component and intrahepatic cholangiocarcinoma component. The intermediate cell component was identified (Figure 2C). The cholangiocarcinoma component revealed irregular haphazard gland differentiation. The ductal plate malformation pattern component was composed of an irregular haphazard glandular structure resembling ductal plate malformation. Tumor cells were cuboidal to low-columnar cells and demonstrated significant cytologic atypia and mitoses (Figure 2D). The pathological findings of background liver revealed hepatitis B virus-related chronic hepatitis. There was no ductal plate malformation lesion in the background liver.
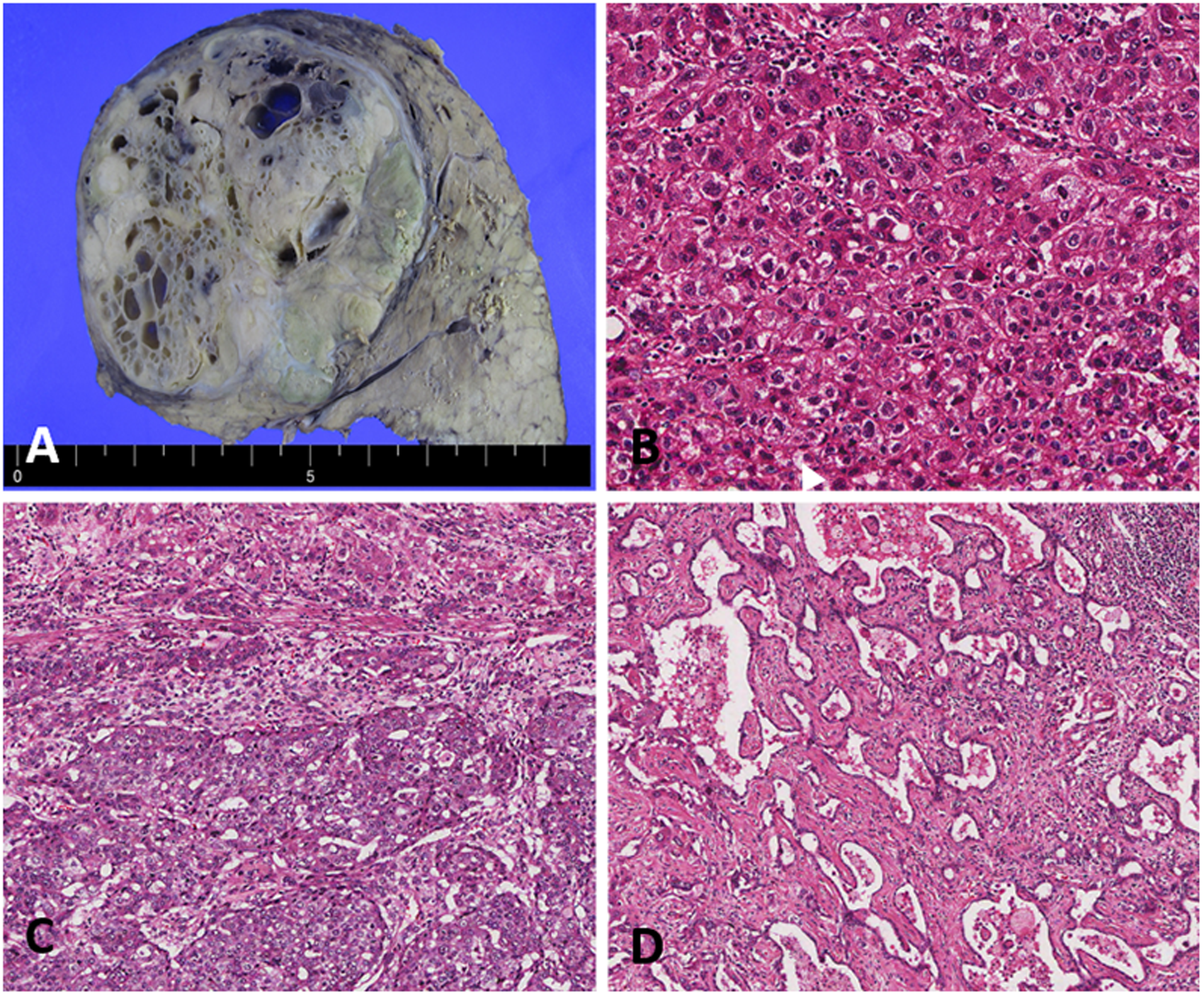
Histopathological findings of combined hepatocellular-cholangiocarcinoma with ductal plate malformation pattern. (A) The resected specimen revealed a well-defined tumor measuring 6.7 × 6.2 cm. (B) The HCC component demonstrated a trabecular pattern and significant pleomorphism (H&E ×100). (C) The intermediate cell component was identified between the HCC component and intrahepatic cholangiocarcinoma component (H&E ×100). (D) The ductal plate malformation pattern component. Cuboidal tumor cells are forming irregularly dilated and coalesced spaces, resembling developmental anomaly of ductal plate (H&E ×80). Abbreviations: HCC, hepatocellular carcinoma; H&E, hematoxylin and eosin.
HCC and intrahepatic cholangiocarcinoma with ductal plate malformation pattern showed opposing staining for hepatocyte antigen (HepAg) and keratin 19 (KRT19). Both HepAg and KRT19 were positive in the intermediate cell components (Figure 3A and B). Immunohistochemical staining showed strong positivity for KRT19, focal positivity for C-reactive protein, and negativity for S100 calcium-binding protein in the ductal plate malformation pattern component (Figure 3C-E). The tumor epithelial cell component was basolateral positive for neural cell adhesion molecule (Figure 3F). The pathological diagnosis confirmed the mass to be a combined hepatocellular-cholangiocarcinoma with ductal plate malformation pattern.
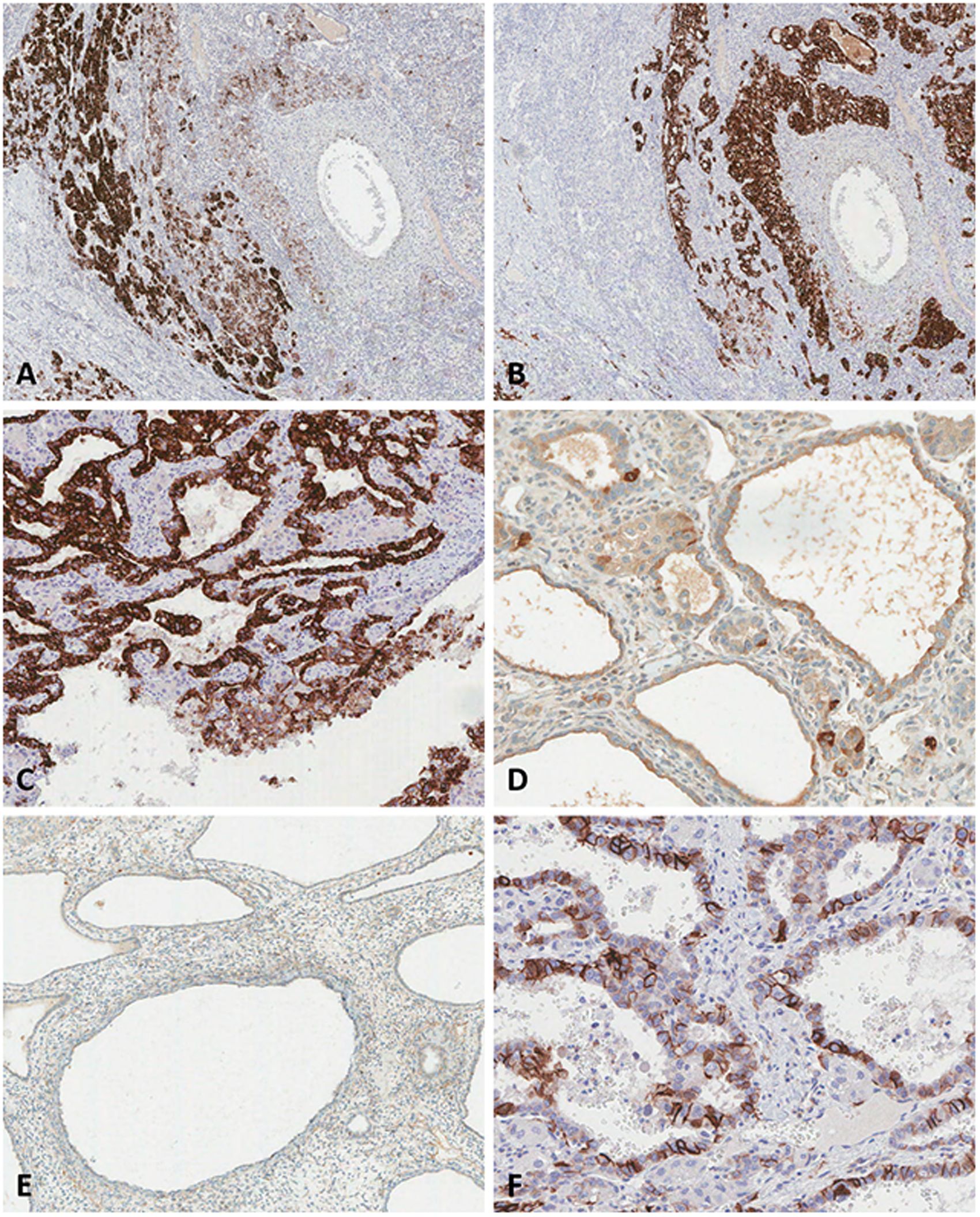
Immunohistochemical findings of combined hepatocellular-cholangiocarcinoma with ductal plate malformation pattern. (A) The HCC component and intermediate cell component were positive for HepAg (original magnification ×40). (B) Cholangiocarcinoma component and intermediate cell component were positive for KRT19 (original magnification ×40). (C) In the ductal plate malformation pattern component, KRT19 staining was strongly positivity (original magnification ×100), (D) showed focal positivity for CRP (original magnification ×200), and (E) was negative for S100 calcium-binding protein (original magnification ×80). (F) The tumor epithelial cell in the ductal plate malformation pattern component was basolateral positive for NCAM (original magnification ×200). Abbreviations: CRP, C-reactive protein; HCC, hepatocellular carcinoma; HepAg, hepatocyte antigen; KRT19, keratin 19; NCAM, neural cell adhesion molecule.
We performed targeted NGS-based genomic profiling of the resected specimen using the NGS gene panel, Oncomine® Comprehensive Assay Plus. This analysis identified genomic alterations in the following from over 500 tumor-related genes: CDKN2A amplification, CHD4 amplification, CYP2D6 amplification, ERBB3 amplification, KIR3DL1 amplification, KRAS amplification, MDM2 amplification, PIM1 amplification, STAT6 amplification, TPMT amplification, FAT1 (c.7130C > T, p.Thr2377Met) variant, FLT4 (c.2710G > A, p.Gly904Ser) variant, FANCD2 (c.2102C > T, p.Pro701Leu) variant, RASA1 (c.2032A > C, p.Ser678Arg) variant, and TP53 (c.746G > C, p.Arg249Thr) variant.
Discussion
Ductal plate malformation is the basic component of congenital disorders of the intrahepatic bile ducts, such as Caroli's disease, congenital biliary atresia, congenital hepatic fibrosis, and von Meyenburg complex. It is well-known that ductal plate malformation of the bile duct is a precursor to biliary intraepithelial neoplasia that can transform to an invasive carcinoma.5,6 Intrahepatic cholangiocarcinoma with ductal plate malformation pattern was first recognized as a distinctive variant by Nakanuma in 2012. 3 Intrahepatic cholangiocarcinoma with ductal plate malformation pattern is rare, with a low incidence in the literature. Thus, the present case of combined hepatocellular-cholangiocarcinoma with ductal plate malformation pattern is an extremely rare entity. Only 4 cases including the present case have been reported, and clinical data including serum tumor marker, tumor size, and tumor location of four cases are listed in Table 1. Three patients diagnosed with combined hepatocellular-cholangiocarcinoma with ductal plate malformation pattern were female, although the patient of the present case was male. The mean age of all patients was 61.0 years (range 50-70 years). Serum tumor markers were tested in the present case only. Serum AFP and PIVKA-II levels were elevated. Well-differentiated small duct-type cholangiocarcinoma can be challenging differential diagnosis of cholangiocarcinoma with ductal plate malformation pattern. Well-differentiated small duct-type cholangiocarcinoma is tubular, with papillary architecture and cuboidal cells. Well-differentiated small duct-type cholangiocarcinoma glands are dispersed randomly in a desmoplastic stroma. Intrahepatic cholangiocarcinoma with ductal plate malformation pattern is characterized by irregularly dilated lumens with intraluminal papillary projections and bridge formation. Genetic alterations of conventional small duct-type cholangiocarcinoma were detected in IDH1/2 mutation, FGFR2 fusion. In cholangiocarcinoma with ductal plate malformation pattern, genetic alteration was found in PTPRT, CDKN2A, KDM6A, and IRS1. 7
Clinicopathological Features of 4 Cases of Combined Hepatocellular-Cholangiocarcinoma With Ductal Plate Malformation Pattern.
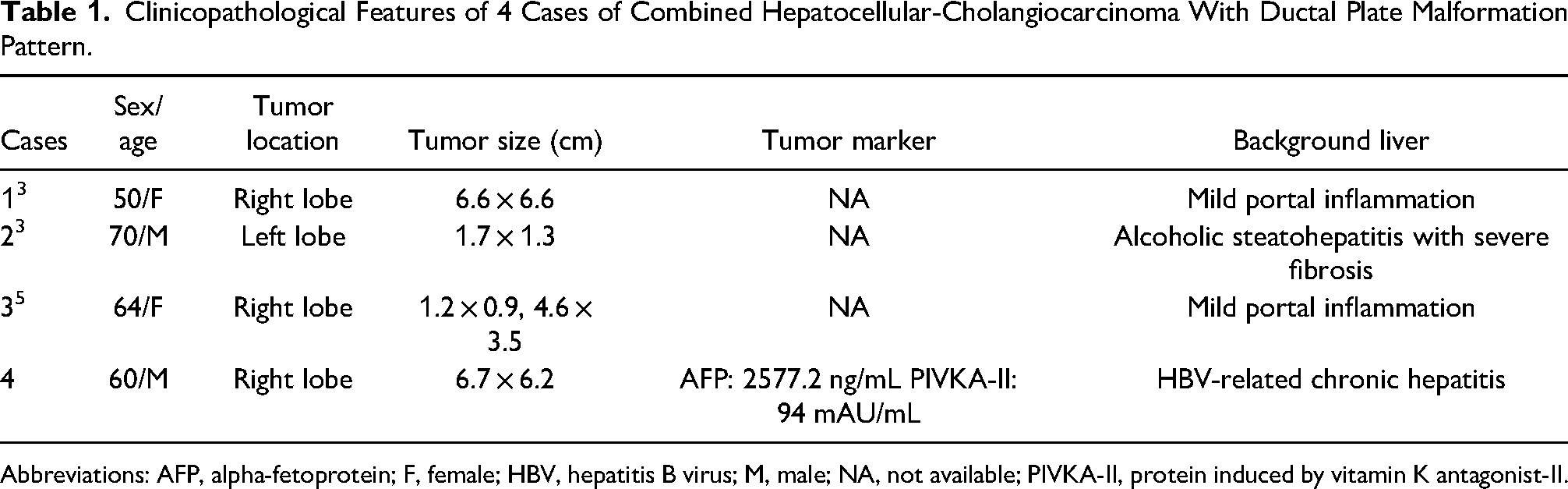
Abbreviations: AFP, alpha-fetoprotein; F, female; HBV, hepatitis B virus; M, male; NA, not available; PIVKA-II, protein induced by vitamin K antagonist-II.
In the present case, the patient showed single nucleotide variants (SNVs) in FAT1, FLT4, FANCD2, RASA1, and TP53. Genetic alteration was found in FAT1 (5.88%), FLT4 (3.05%), FANCD2 (4.16%), RASA1 (1.97%), and TP53 (32.56%) of all cancer. In addition, NGS analysis exhibited structural variants in CDKN2A, CHD4, CYP2D6, ERBB3, KIR3DL1, KRAS, MDM2, PIM1, STAT6, and TPMT amplification. Gene amplification was found in CDKN2A (0.11%), CHD4 (0.99%), ERBB3 (0.30%), MDM2 (2.85%), PIM1 (0.23%), and STAT6 (0.20%) of all cancers. 7 Of the 15 genetic mutations, several novel genetic alterations were detected, including TP53 SNV and MDM2 amplification.
TP53 is a well-known tumor suppressor gene and can control DNA repair, cell cycle, senescence, and apoptosis. TP53 mutation was present in 30% and 21% of HCC and cholangiocarcinoma samples of previous studies, respectively.8,9 A previous study on large-scale genetic alteration of combined hepatocellular-cholangiocarcinoma showed TP53 to be the most commonly mutated gene (49% of the entire cases) in combined hepatocellular-cholangiocarcinoma cases. 10 TP53 mutation of the present case is consistent with findings of previous studies. MDM2 is a proto-oncogene encoding E3 ubiquitin ligases and is a hallmark of tumorigenesis. 11 MDM2 amplification has been identified in various tumor types including soft tissue sarcoma, breast cancer, oral cancer, and glioblastoma.12,13 MDM2 amplification acts as an oncogenic role in several malignant tumors. 14 This is the first case report in which MDM2 amplification has been found in combined hepatocellular-cholangiocarcinoma. Thus, the pathogenic significance of MDM2 amplification in combined hepatocellular-cholangiocarcinoma has not been elucidated. Recent study indicates that tumors harboring MDM2 amplification could benefit from MDM2 inhibitors. MDM2 amplification also has been associated with hyperprogression (acceleration in tumor progression) of patients treated with immune checkpoint inhibitors. 12
It is widely accepted that the important regulator of TP53 is MDM2. 15 MDM2 overexpression yields p53-dependent growth arrest and apoptosis in a variety of cell lines.16,17 Dysfunction of the MDM2-p53 axis plays a critical role in tumorigenesis.13,15 Understanding of MDM2's interaction with p53 may be useful. Management of combined hepatocellular-cholangiocarcinoma based on genetic alteration has not been standardized. The molecular analysis of the present case can help to establish treatment guideline in patients exhibiting MDM2 amplification and TP53 mutation. In the present case, NGS analysis was not performed separately in each compartment; HCC and intrahepatic cholangiocarcinoma with ductal plate malformation pattern components. Despite this limitation, to our knowledge, this is the first case report of molecular alteration in combined hepatocellular-cholangiocarcinoma with ductal plate malformation pattern. Further molecular studies of this entity can help to develop optimization of targeted therapy.
Footnotes
Authors’ Contributions
Woo Sung Moon, Kyoung Min Kim and Ae Ri Ahn summarized the pathological and genomic data, Kyoung Min Kim and Ae Ri Ahn analyzed and interpreted the sequencing data, Ji Soo Song and Hee Chul Yu summarized the clinical data, Woo Sung Moon, Kyoung Min Kim and Ae Ri Ahn contributed as pathologists, Woo Sung Moon, Kyoung Min Kim and Ae Ri Ahn organized the study and wrote the manuscript. All authors read and approved the final manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This paper was supported by the Fund of Biomedical Research Institute, Jeonbuk National University Hospital.
Ethics
This case report was approved by Jeonbuk National University Hospital Institutional Review Board (approval no. IRB 2022-02-031). This case report was conducted in accordance with the Declaration of Helsinki of 1975.
Informed Consent
Patient consent was obtained for publication.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
