Abstract

Dear Sir,
We have carefully read the interesting article “Mucinous Adenomyomatous Pulmonary Hamartoma: Clinicopathologic, Immunohistochemical, and Molecular Features of 6 Cases” 1 published in your journal. We recently observed a similar tumor involving a 76-year-old woman with no other remarkable history who was referred to us for an 8-year history of a solitary pulmonary nodule in the left lower lobe, which, in the last radiological check-up, had increased in size from 1.8 to 2.1 cm (Figure 1a). A positron emission tomography–computed tomography was performed following the assessment protocol for solitary pulmonary nodules. The lesion presented low radiotracer uptake, and malignancy could not be ruled out. Based on these results and not being able to perform a less invasive biopsy, we decided to address the lesion surgically. Due to the tumor's central location, we performed a left lower lobectomy.
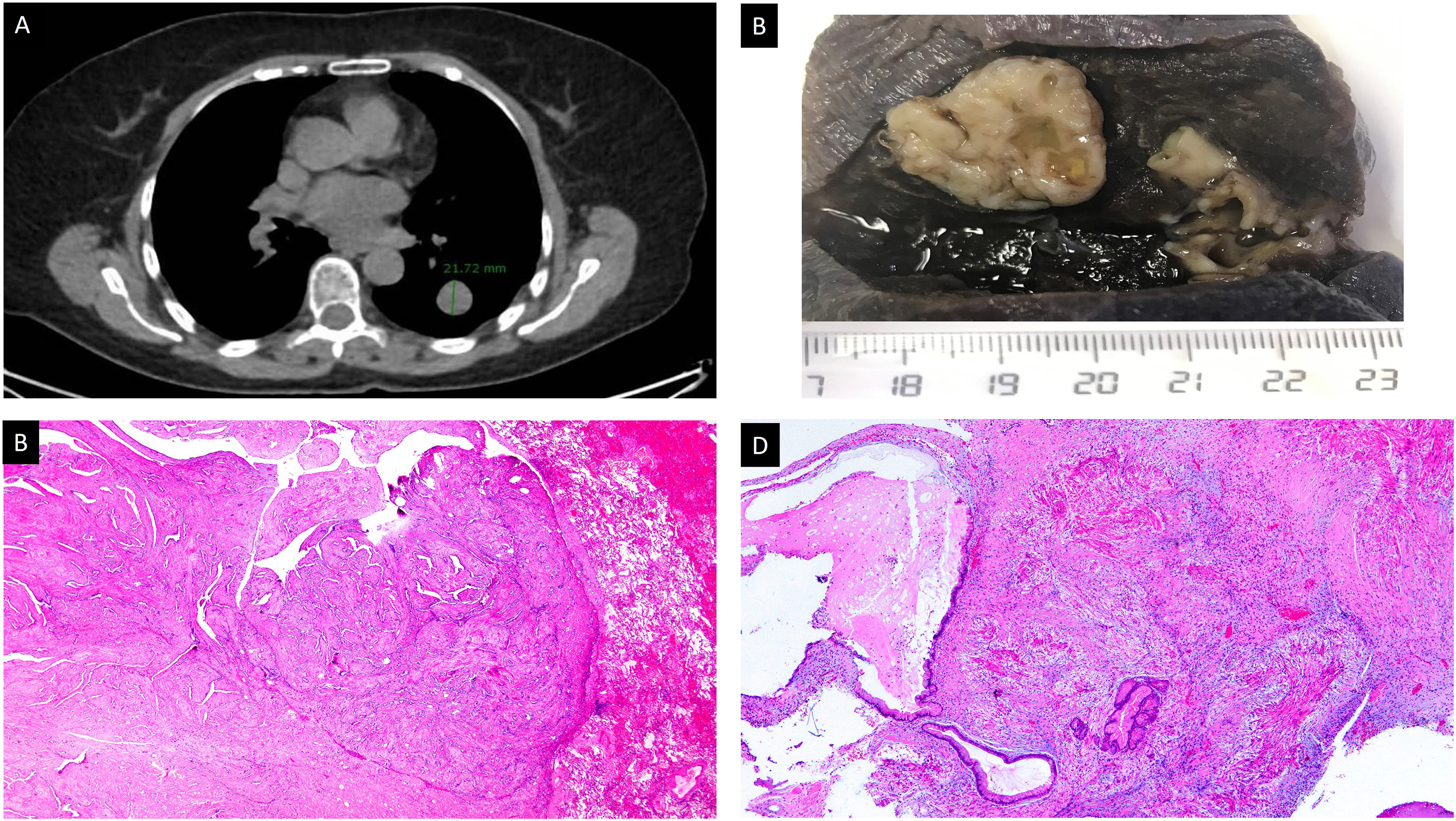
(A) Nodule measuring 21.72 mm in the lower lobe of the left lung (chest CT). (B) Macroscopic appearance of the specimen, showing a well-delimited, white lesion with a multicystic surface and mucinous areas measuring 2.3 cm. (C) Panoramic overview showing an expansive growth pattern and biphasic histology (HE; Panoramic). (D) Biphasic lesion with mucinous stromal and glandular proliferation (HE; 20×).
Macroscopically, the lesion was well-delimited and enucleated, measuring 2.1 cm, with a greyish surface and microcystic and mucoid areas (Figure 1b). Histologically, the lesion was biphasic with a stromal component consisting of smooth muscle tissue (desmin, calponin, and smooth muscle actin [SMA]-positive) and a glandular component (Figure 1c and d). We identified 3 types of glands: a number of small glands covered by cells with round nuclei and scarce cytoplasm; branching epithelial clefts; and a third type of gland covered by an epithelium with a broad mucinous cytoplasm, frequently dilated and with mucoid content. This last type of gland did not express transcription termination factor 1 (TTF1) nor did it present p63 expression because it lacked a row of basal cells, unlike the first 2 (Figure 2). The immunoreaction was positive in the epithelium of the 3 glandular types; we also observed immunostaining with KRT20 in the mucinous epithelium. CD34 and CDX2 were negative. With Ki67, we observed a low proliferation index in all components of the lesion.
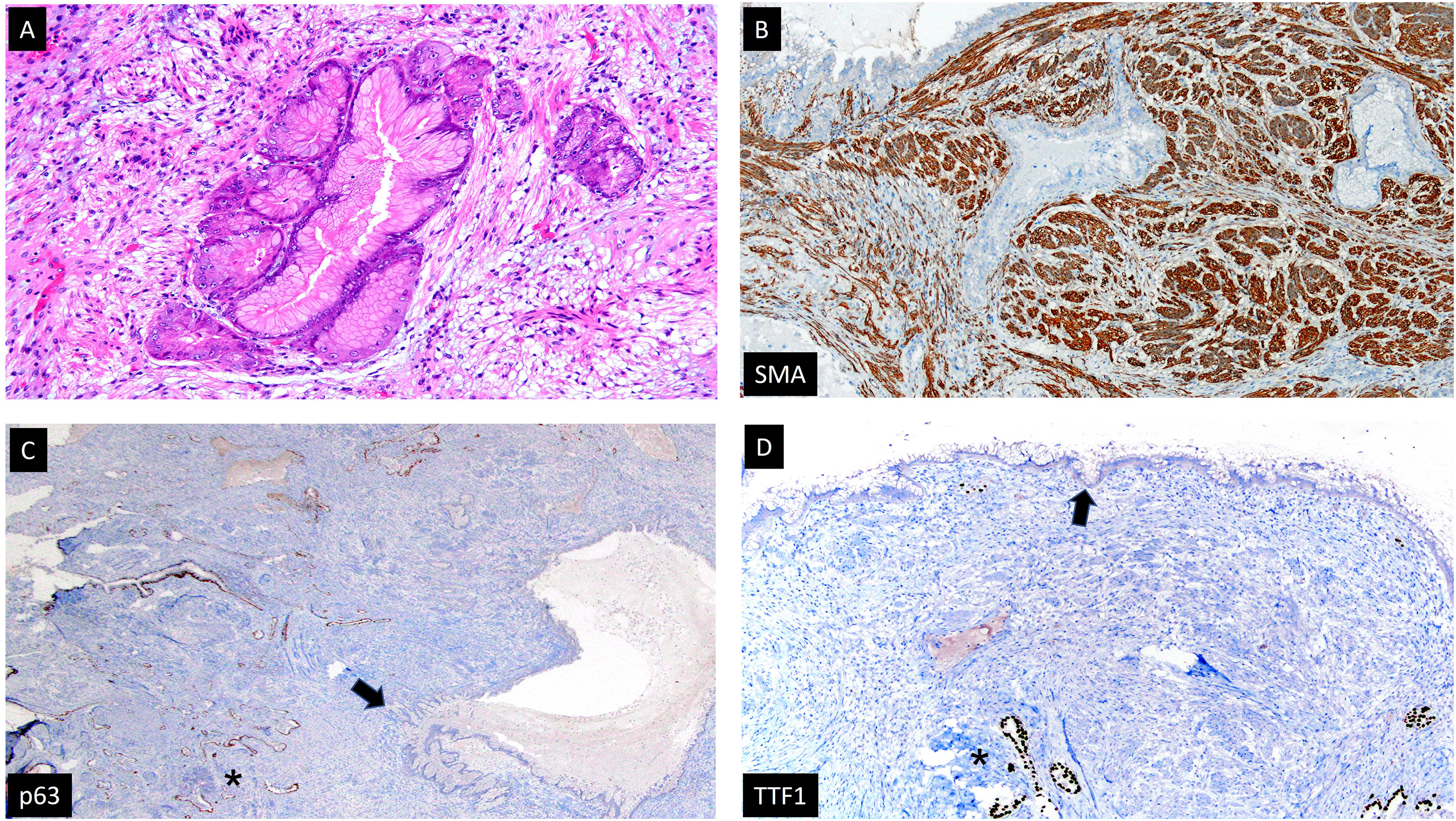
(A) Glandular proliferation of mucinous columnar epithelium without atypia or significant mitotic activity in the bed of a leiomyomatous stroma (HE; 100×). (B) The stroma presents diffuse nuclear immunostaining for SMA, confirming its muscle origin (SMA; 40×). (C) There is a row of p63 + basal cells in the epithelial cleft branches and small round glands (asterisk), which are not present in the mucinous glands (arrow) (p63; 20×). (D) There is no immunostaining against TTF1 in the mucinous glands (arrow), while there is an immunoreaction in the nonmucinous glandular epithelium (asterisk) (TTF1; 40×).
According to the literature, this lesion occurs almost exclusively in lower lobes, especially the left, as in our case. Unlike the one in our patient, these lesions usually appear in men in the sixth to seventh decade of life. 1
Unlike the 6 tumors published by Rossi et al 1 and others described in the literature,2,3 the glandular component in our patient was not exclusively mucinous, which, although uncommon, has also been reported in the literature, where branching epithelial clefts characteristic of adenofibroma were identified.4,5
Therefore, we believe that this type of lesion should be included in the same category. We propose a broader generic term for all of these lesions to avoid confusion in the terminology by overly extensive subclassifications. The term “adenomyomatous hamartoma” seems appropriate,2,3 specifying, when appropriate, mucinous differentiation.
Although the postoperative follow-up period is very short, the diagnosis should lean toward a benign or low-grade lesion if 8 years have passed with no substantial radiological changes or signs of dissemination, as has been reported in the literature.1–5
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
