Abstract
Thymolipoma is a rare benign tumor of the anterior mediastinum. Only a few reports describing thymoma arising within a thymolipoma have been documented in the literature. We report herein a detailed description of thymolipoma giving rise to 2 thymomas of different histological subtypes. A 74-year-old male with history of metastatic papillary thyroid carcinoma gradually developed 2 soft tissue nodules within a large right hemithorax fatty mass that was present for the past 20 years. Computed tomography (CT)-guided needle biopsy revealed one of the soft tissue nodules to be a thymoma, and the entire mass was surgically resected. Final pathological examination demonstrated the mass to be a thymolipoma containing a micronodular thymoma with lymphoid stroma as well as a WHO type B1 thymoma. No evidence of disease recurrence was seen at the time of his 7-year follow-up. This case documents a rare presentation of thymolipoma harboring 2 thymomas of different histological subtypes and highlights the need for early surgical resection of thymolipomas, as they may harbor malignant nodules.
Introduction
Thymolipoma is a rare benign tumor of the anterior mediastinum composed of predominantly mature adipose tissue with interspersed nonneoplastic thymic tissue.1,2 Thymoma is an epithelial tumor with a variable immature T lymphocyte component. Since it shows potential for invasion and metastasis, all thymoma subtypes are considered potentially malignant. 2 Thymoma is most commonly treated by surgery if deemed surgically resectable at the time of diagnosis. 2 Only a few cases of thymoma arising in thymolipoma have been documented in the literature.3–7 We report herein a detailed description of thymolipoma giving rise to 2 thymomas of different histological subtypes.
Case Presentation
A 74-year-old male with a history of metastatic papillary thyroid carcinoma gradually developed 2 soft tissue nodules within a large right hemithorax fatty mass that was present for the past 20 years. His past clinical history was significant for BRAF V600E mutant papillary thyroid carcinoma with local recurrence followed by lung metastases. He was treated with thyroidectomy, neck dissection, radioactive iodine, and later metastasectomy. Additional comorbidities included congestive heart failure, deep vein thrombosis complicated by pulmonary emboli, and atrial fibrillation requiring pacemaker placement. The patient had no symptoms of myasthenia gravis (MG). Computed tomography (CT) of the chest confirmed a large fatty mass in the right lower hemithorax containing 2 soft tissue nodules (Figure 1). CT-guided needle biopsy of one of the 2 soft tissue nodules revealed the presence of a thymoma. It was evident at surgical resection the mass did not involve the lung but was widely adherent to the entirety of the right pericardium up to the junction of the innominate vein and superior vena cava. Despite the size and adherence to the pericardium, the mass was completely resected. Gross examination showed an encapsulated mass weighing 1060 g and measuring 23.2 × 19.2 × 6.7 cm (Figure 2A). The cut surfaces were lobulated and yellow, and they revealed 2 indurated nodules measuring 5.3 × 4.3 × 2.3 cm and 4.5 × 3.9 × 2.5 cm. The light microscopic examination of the resected mass revealed a predominance of mature adipose tissue (Figure 2B) associated with streaks of thymic tissue demonstrating features of involution (Figure 2C), the findings typical of thymolipoma. Pathological examination of the indurated nodules within the thymolipoma identified 2 thymomas of different histological subtypes. On light microscopic examination, the larger 5.3 cm nodule showed micronodular and trabecular arrangement of epithelioid and spindle cells within lymphoid stroma (Figure 3A to C). The epithelioid and spindle cells were immunoreactive with keratin AE1/AE3 (Figure 3D) and keratin 5/6, while lymphoid stroma was immunoreactive with CD20 (Figure 3E) and TDT (Figure 3F), supporting the diagnosis of micronodular thymoma with lymphoid stroma. On light microscopic examination, the smaller 4.5 cm nodule showed predominantly lymphoid proliferation (Figure 4A) separated by fibrous septae that resembled normal thymic cortex. There were subtle foci of medullary differentiation (Figure 4B to C). Scattered within lymphoid cells were occasional epithelioid cells (Figure 4D) forming a delicate cytoplasmic meshwork (highlighted with keratin AE1/AE3, Figure 4E). TDT stain labeled densely packed immature T cells (Figure 4F), supporting the diagnosis of WHO type B1 thymoma. The patient's postoperative course was unremarkable, and he was discharged 5 days after surgery. The patient has been followed with yearly surveillance chest CT with no evidence of thymoma recurrence at 7-year follow-up.
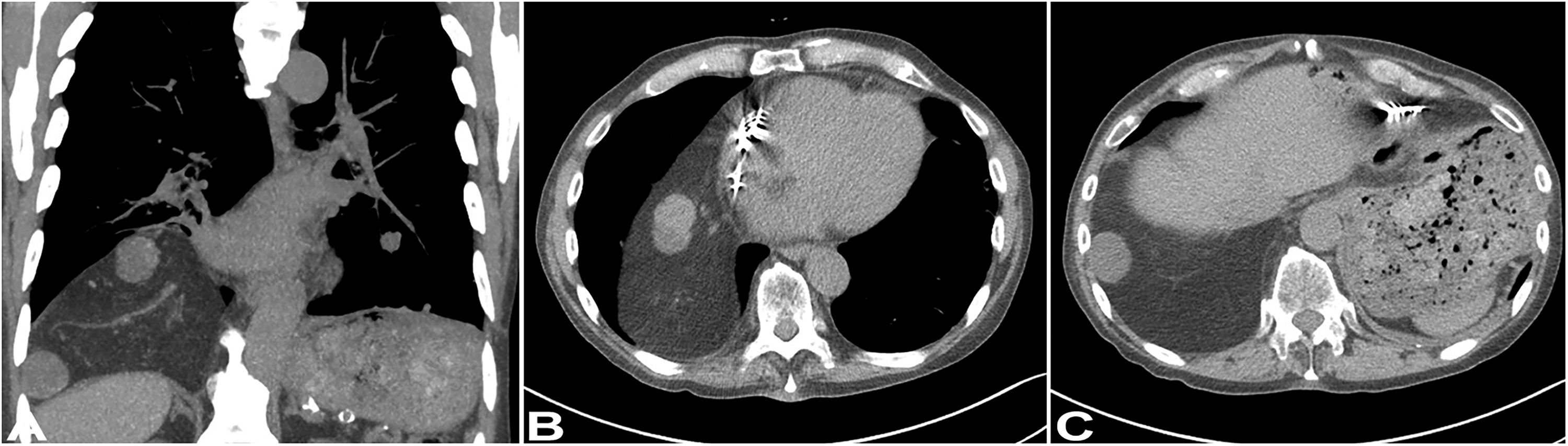
Computed tomography (CT). Chest images show a large up to 20 cm right lower hemithorax mass with a density similar to subcutaneous adipose tissue containing 2 soft tissue density nodules. (A) Coronal and (B-C) sagittal images.
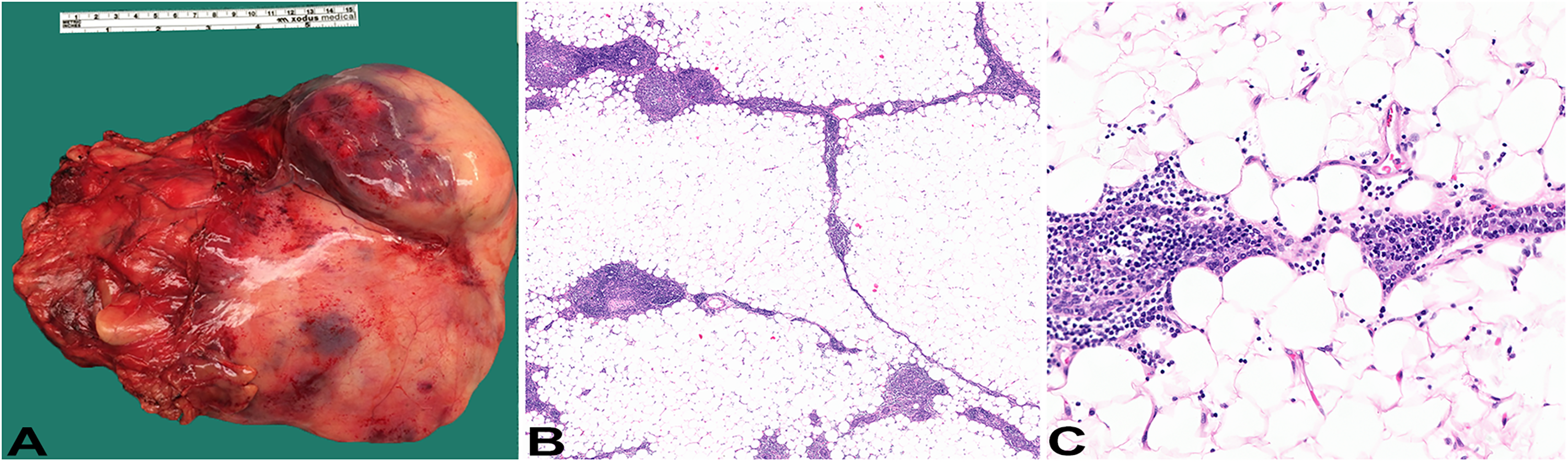
Pathologic features of thymolipoma. (A) Gross examination shows a large encapsulated 23 cm mass. (B) Light microscopic examination shows predominantly adipose tissue interspersed with streaks of involuted thymic tissue; H&E, original magnification 2x. (C) The adipose tissue lacks cytological atypia while thymic tissue has paucity of lymphocytes; H&E, original magnification: 20x.
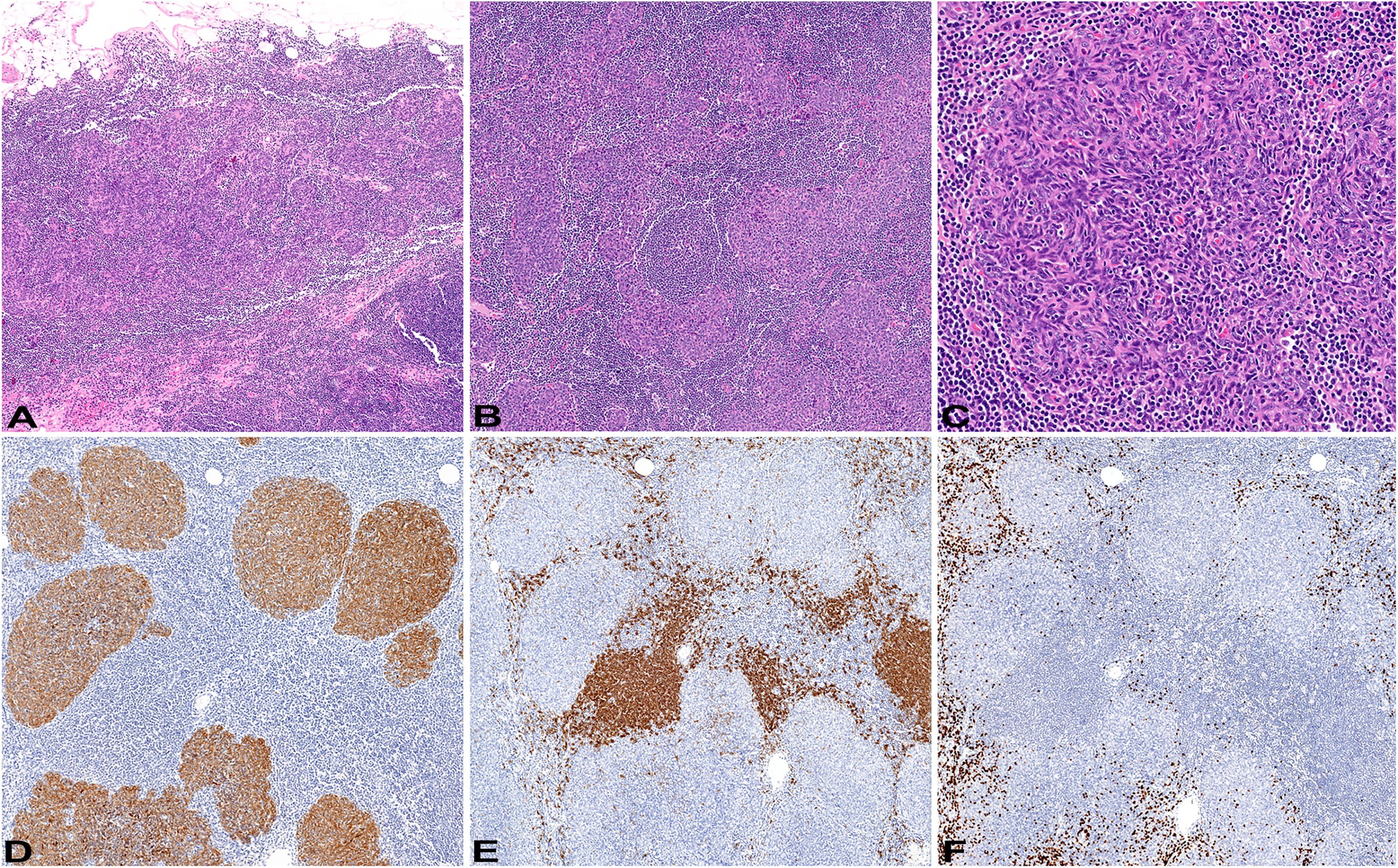
Pathological features of micronodular thymoma with lymphoid stroma. (A-B) Light microscopic examination shows varying in size epithelioid cell nodules within lymphoid stroma; H&E original magnification: 2x and 5x, respectively. (C) The epithelioid nodules are composed of spindled to epithelioid cells; H&E original magnificatio:n 20x. (D) The epithelioid nodules are immunoreactive with keratin AE1/AE3, while lymphoid stoma is immunoreactive with CD20 (E) and TDT (F); immunohistochemistry, original magnification: 10x.
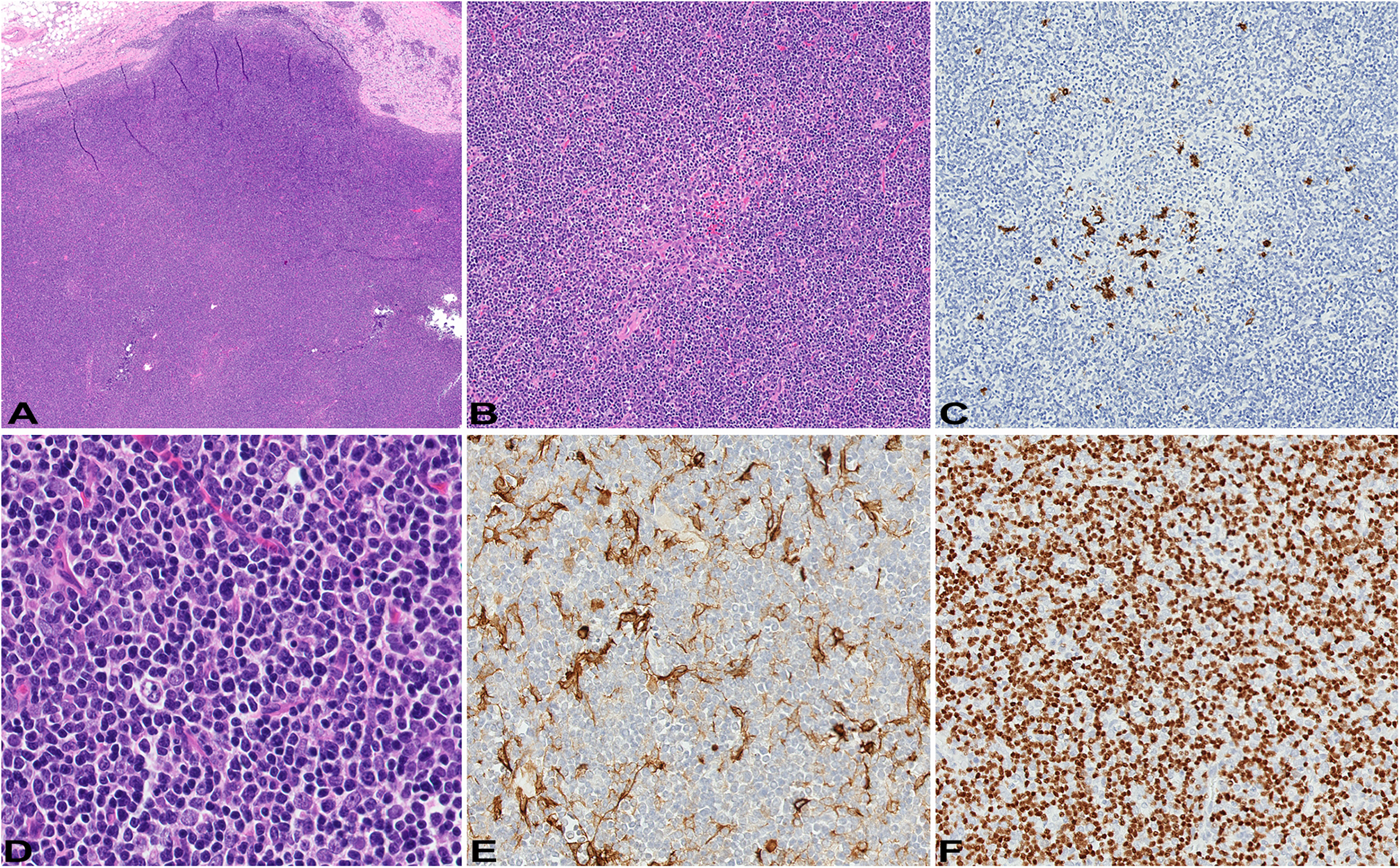
Pathological features of WHO type B1 thymoma. (A) Light microscopic examination shows predominantly lymphoid cells arranged in sheets that resemble the cortex of normal thymus; H&E original magnification: 2x. There are subtle areas of medullary differentiation (B) containing occasional clusters of B-lymphocytes (C); H&E and immunohistochemistry for CD20 original magnification: 10x. (D) The neoplastic epithelioid cells are associated with numerous lymphoid cells; H&E original magnification: 40x. The epithelioid cells are immunoreactive with keratin AE1/AE3 (E) while lymphoid cells are immunoreactive with TDT (F); immunohistochemistry, original magnification: 20x, respectively.
Discussion
Thymolipomas are rare benign neoplasms of uncertain histogenesis that occur in the anterior mediastinum. Likely due to their slow growth they may achieve significant size, conforming to the shape of adjacent structures without eliciting symptoms. 8 If symptoms are present, they are likely related to MG. The reported incidence of MG in thymolipoma patients varies with some series reporting as high as 80%. 9
Several hypotheses of thymolipoma histogenesis have been suggested. It has been viewed as a regression of thymic hyperplasia, involuted thymoma, hamartoma, and lipoma of thymic fat.10–12 More recently, a translocation involving the HMGA2 gene on chromosome 12q15 has been reported in a thymolipoma. 13 Since chromosomal aberrations of 12q13-15 occur in a significant percentage of soft tissue lipomas, 14 the observation of a chromosomal translocation adds further evidence to the notion that thymolipoma is a neoplasm of thymic fat, while the thymic epithelial component represents nonneoplastic tissue. 13 This cytogenetic profile is different from those reported in thymomas that show chromosomal imbalances. 15 The commonest recurrent genetic alteration in thymomas, a missense mutation in GTF2I, has been reported in as many as 38% of cases.16–18 The GTF2I mutation has been associated with WHO type A or AB thymomas, less advanced Masaoka stage, and the absence of MG. 16 GTF2I mutation appears specific to thymomas and thymic carcinomas and has not been reported in other tumors.16,17
There have been only rare reports of thymoma arising in thymolipoma documented in the literature3–7. Their clinicopathological features are summarized in Table 1. These patients were predominantly females (female-to-male ratio of 2:1) of a wide age range (range: 23-74 years; mean age of 51.3 years). Five out of 6 patients presented with a large intrathoracic mass (more than 20 cm and/or more than 1 kg) occupying nearly the entire hemithorax. All 6 thymomas were classified as WHO type B and 5 out of 6, including the tumor in the current report, were represented by a lymphocyte-rich subtype with the presence of either B1 or B2 component or combination of both. All patients were treated with surgery and showed no evidence of disease recurrence with long-term follow-up (mean: 50.1 months).
Clinicopathological Findings.

Pathological diagnosis according to the current WHO classification.
50% of thymolipoma.
A combined maximum dimension of B2 thymoma and thymic carcinoma was 20.0 cm.
Abbreviations: F, female; FUO, follow-up in months and outcome; M, male; MG, myasthenia gravis; MN, micronodular thymoma with lymphoid stroma; N, no; NA, not available; NED, no evidence of disease; TL, thymolipoma; Y, yes.
One of 6 patients (Table 1, patient 5), in addition to WHO B2 thymoma, also had a second malignancy represented by a high-grade thymic carcinoma. 4 In our patient, in addition to WHO B1 thymoma, the thymolipoma harbored a micronodular thymoma with lymphoid stroma, a rare thymoma subtype presumably originating from thymic medullary epithelial cells and with good prognosis. 19 While it is not possible to completely exclude local metastasis (a combined tumor with WHO B1 thymoma and micronodular thymoma components), based on the presence of 2 separate distinct masses, lack of histological overlap and relatively similar size, they were interpreted as 2 independent primaries.
Patients with WHO B1-B3 thymomas on average have a 2-fold higher incidence of MG compared to patients with WHO type A thymoma,20,21 however, none of the reported cases of thymoma in thymolipoma (Table 1) had association with MG, suggesting that autoimmunity has no contribution to their tumorigenesis.
Over the years, the patient had developed 3 types of neoplasia including papillary thyroid carcinoma, thymolipoma, and thymoma. Although rare, this association does raise the question of a possible link between the tumors. Indeed, occasional reports document multiple tumors in the setting of thymoma with colorectal and thyroid carcinomas being the most frequent.22,23 Such occasions allow to speculate that there is a syndromic association between a thymoma and secondary malignancies. 23 Nevertheless, since secondary malignancies are diagnosed both before and after the diagnosis of thymoma, it remains unknown if genetic predisposition or thymoma-associated immune disbalance plays a role in developing multiple neoplasia.22–24
The current report documents a unique occurrence of 2 thymomas of different histological subtypes arising within a large thymolipoma. To the best of our knowledge, the histology of one of the thymomas representing micronodular thymoma with lymphoid stroma has not been reported previously in this setting. The lack of association with MG in this and previous reports suggests that genetic predisposition not MG or autoimmunity may play a role in the development of secondary malignancy in patients with thymolipoma. This case reinforces the need for active clinical follow-up of patients with thymolipoma and early surgical intervention as they may harbor occult malignancy.
Footnotes
Acknowledgments
The authors are thankful to Shawn Scally for excellent assistance in figures preparation.
Author’s Contribution
VCD participated in data collection; KS wrote the manuscript text; VCD and KS edited the manuscript. The authors read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Consent for Publication
This study is exempt from the regulatory requirements of the National Institutes of Health (NIH) regarding studies on human subjects, based on the criteria described in section 4 of 46.101(b) of 45 CFR 46 “as research involving the collection or study of existing data, documents, records, if the information is recorded by the investigator in such a manner that subjects cannot be identified, directly or through identifiers linked to the subjects”. 25 According to the institutional guidelines, the patient's consent is not necessary for a case report with de-identified patient-specific information.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
