Abstract
Background:
Direct-acting oral anticoagulants (DOACs) are first-line therapy for atrial fibrillation and for venous thromboembolism prevention and treatment. In addition, proton pump inhibitors (PPIs) are recommended by the ACC Expert Consensus for patients receiving DOAC.
Objective:
To determine the risk of clinically important thromboembolic and bleeding events and death in patients concomitantly prescribed DOACs and PPIs compared with DOAC alone.
Methods:
We undertook a population-based cohort study of Ontario residents aged 66 years or older who were newly dispensed a DOAC from 2009 to 2020. The primary outcome was a composite of clinically relevant bleeding, thrombotic events, and all-cause death. We then used a time-dependent Cox regression model to estimate the adjusted hazard of outcomes during time periods where patients were exposed vs not exposed to PPI.
Results:
We included 283 771 new DOAC users (mean age 78.3 years, 49.4% female). The age/sex-standardized outcome event rate of the composite outcome was higher in the DOAC-PPI cohort [20.2 (95% CI 20.0-20.5) per 100 person-years] than in the DOAC-alone cohort [15.2 (95% CI 15.1-15.4) per 100 person-years]. In time-dependent Cox regression analyses examining time periods during DOAC-PPI co-therapy vs DOAC alone, risks were elevated for all outcomes during periods of PPI co-therapy, suggesting residual confounding. In a secondary analysis of patients receiving DOAC-PPI co-therapy (n = 115 493), periods of DOAC-PPI co-therapy were associated with a greater hazard for death (HR 2.24, 95% CI 2.14-2.35), but a lower risk for both thrombosis (HR 0.93, 95% CI 0.89-0.96) and clinically relevant bleeding (HR 0.79, 95% CI 0.76-0.81).
Conclusion and Relevance:
Our cohort study of older adults suggests that DOAC-PPI co-therapy is associated with decreased upper GI bleeding but increased mortality. Since the implications are major and may be due to residual confounding, we recommend that the findings be verified in randomized trials before clinical application.
Keywords
Introduction
Direct acting oral anticoagulants (DOACs) are first-line therapy for atrial fibrillation and for venous thromboembolism prevention and treatment.1 -4 One of the proposed advantages of DOACs compared with warfarin has been fewer clinically significant drug interactions based on limited evidence.2 -4 Proton pump inhibitors (PPIs) are one of the drug families commonly co-administered with anticoagulants, as is recommended by the ACC Expert Consensus for patients receiving DOAC co-prescribed with antiplatelets or agents with antiplatelet properties.5,6 PPIs are effective for managing acid-related gastrointestinal (GI) disorders including peptic ulcer disease and related GI bleeding, and gastroesophageal reflux disease, since they have a favorable safety profile, evidence has been extrapolated to support their off-label use for prevention of DOAC-related GI bleeding. 7 ,8 -12 It was reported that the utilization of DOACs in the United States has increased significantly for annual prescription numbers from 1079 in 2011 to 1.5 million in 2019, 13 and the PPI absolute number of units purchased from September 2018 to August 2019 reached 99 021 030. 14 Similarly, in 2018, the direct factor Xa inhibitors, and PPIs were each among the 10 drug classes topping public drug program spending on seniors in Canada: $316.2 million and $180.8 million, respectively. 15
According to a recent systematic review, PPIs have a protective effect against warfarin-related GI bleeding (OR = 0.69; 95% CI 0.64-0.73), with no significant effect on thromboembolic events or mortality. 16 Population-based studies by Ray et al11,17 have reported that PPI co-therapy was associated with reduced risk of OAC (including DOAC)-related upper GI bleeding. However, evidence for the co-therapy of PPIs and DOACs that includes the outcomes of thromboembolic events and mortality is lacking. Accordingly, controversy remains regarding the necessity of combining PPIs with DOACs, even if it is recommended by the ACC Expert Consensus for patients receiving ≥2 antithrombotic agents.5,6 For instance, 2 studies have reported no evidence of a protective effect of PPIs against dabigatran-related GI bleeding.18,19 The only relevant randomized trial showed that pantoprazole given concomitantly with low-dose rivaroxaban did not reduce upper GI bleeding. 20 Co-prescription of PPIs may reduce the efficacy of dabigatran due to an increase in gastric pH that may reduce drug absorption. 21 In the RE-LY trial, concomitant use of PPIs reduced dabigatran serum levels by 15%, but there was no significant impact on dabigatran efficacy in terms of plasma concentrations. 22 A pilot RCT reported that a 2-week period of PPI withdrawal led to a significant increase in dabigatran trough and peak plasma levels in patients with AF. 23
It is important for clinicians to know whether there are clinically relevant interactions between PPIs and DOACs, particularly beyond the data on protection from upper GI bleeding.11,24,25 For example, could the trade-off from upper GI bleeding rates be increased thromboembolic events, or deaths from other causes. The objective of this study was to determine the risk of clinically important thromboembolic and bleeding events and death in patients concomitantly prescribed DOACs and PPIs compared with those prescribed DOACs alone.
Methods
Study Design and Data Sources
Our study was a population-based cohort study of administrative healthcare data from Ontario, Canada’s most populous province. The detailed study protocol is published.
26
The databases we used are listed in Table A1. This report follows the REporting of studies Conducted using Observational Routinely collected health Data (RECORD-PE) guidelines (
The study was performed at ICES (www.ices.on.ca), where Ontario’s administrative healthcare databases are held securely and linked at the person level using a coded version of the provincial health insurance number. We used prescription drug claims in the Ontario Drug Benefit Database, which contains comprehensive records of prescriptions dispensed to all Ontarians aged 65 years or older; the Canadian Institute for Health Information (CIHI) Discharge Abstract Database, which captures diagnostic and procedural information about hospital admissions; the Ontario Health Insurance Plan (OHIP) Registered Persons Database, which contains demographic and vital status data; and the OHIP Claims History Database to identify physicians’ services. Researchers routinely use these databases to study the clinical consequences of drug-drug interactions.28,29 International Classification of Diseases, 9th and 10th Revision, codes were used to capture the clinical diagnoses associated with healthcare encounters (Table A1).
Study Population
We studied Ontario residents aged 66 years or older who were newly dispensed a DOAC (dabigatran, rivaroxaban, apixaban, or edoxaban) from 1 June 2009 to 31 March 2020. As prescription drug insurance is available for all adults from their 65th birthday, we included individuals aged 66 years or older to ensure complete medication records with a 1-year look back. We excluded patients with: (1) a missing or invalid Ontario Health Insurance Plan (OHIP) number; (2) missing age or sex; (3) age < 66 years at entry (the date of the index dispensation for DOAC); (4) a history of fewer than 365 days of OHIP coverage at entry; (5) prescription claims for multiple different DOACs at entry; (6) a prescription claim for a DOAC in the 365 d preceding the index date; (7) a prescription claim for warfarin or heparin in the 100 days preceding or including the index date; and (8) patients who received their initial DOAC prescription within 7 days of total joint arthroplasty, on the grounds that such courses was likely to be short perioperative therapy. A study flow diagram is provided in Figure 1.
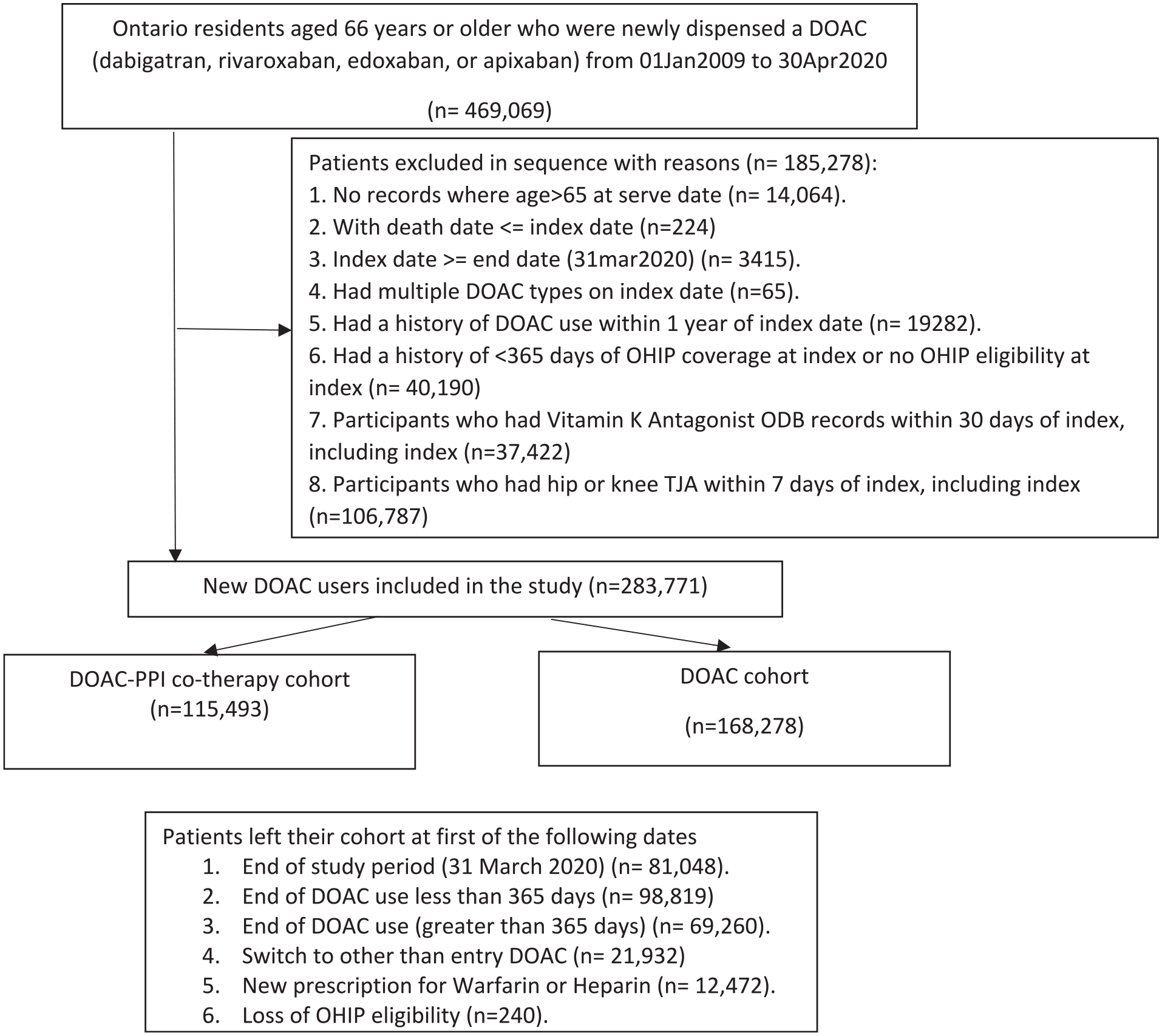
Study flow diagram.
Study Outcomes
The primary outcome was a composite of all clinically relevant bleeding (defined as bleeding bringing the patient to an Emergency Department [ED]), thrombotic events, and all-cause death. The diagnosis and procedure codes used to define the outcomes can be found in Table A1. Thrombotic events were defined as a hospitalization or ED visit for any thromboembolic event, including myocardial infarction (MI), TIA, systemic embolism, peripheral embolism, ischemic stroke, deep vein thrombosis (DVT), pulmonary embolism (PE), coronary artery bypass graft surgery (CABG), and percutaneous coronary interventions, as captured in the hospital discharge abstracts (CIHI-DAD) or ED records (NACRS). GI bleeding included either upper or lower GI sources. Secondary outcomes included all clinically relevant bleeding, including GI, intracranial, and other bleeding that required hospitalization or an emergency room visit.
Study Drug Exposures
Exposures to PPIs and DOACs were treated as time-varying variables, with each day assessed for exposure. The drug exposure period was defined according to the date the prescription was filled and the prescription duration (dispensed days of supply). We identified a period of continuous DOAC use for each patient, beginning with the first pharmacy claim for a DOAC following the patient’s 66th birthday (index date). Our definition of continuous use was a subsequent prescription within 1.5 times the days supplied of the previous DOAC prescription, using a minimum grace period of 30 days. The risk of DOAC-related bleeding, thromboembolic events, or death was captured only while patients were taking the index DOAC. Thus, all study analyses were restricted to periods of anticoagulant treatment during follow-up.
Co-Variables
The potential confounders included patient demographics, inferred indications for DOAC therapy, comorbidities, components of HAS-BLED
30
and CHA2DS2-VASc Scores,
31
and the Deyo-Charlson Comorbidity Index.
32
Other variables included previous use of warfarin, PPI use at baseline and potentially interacting drugs during the follow-up period. The potentially interacting drugs were treated as time-varying exposures, with continuous use defined as subsequent prescription within 1.5 times the days’ supply of the previous prescription, plus a grace period of 30 days. Potentially interacting drugs included amiodarone, antiplatelet agents, nonsteroidal anti-inflammatory drugs, statins, antimicrobials, and selective serotonin receptor inhibitors. Detailed information on covariates can be found in the protocol.
26
The details of the co-variables can be found in
Statistical Analysis
Patients were followed from DOAC initiation until: (1) discontinuation of the index DOAC, defined as 1.5 times the days’ supply plus a grace period of 30 days with no following prescription; (2) switching to another DOAC, warfarin or heparin; (3) loss of OHIP coverage; (4) the date of a study endpoint (bleeding, thrombosis or death); or (5) the last day of the study period (March 31, 2020), whichever occurred first.
The categorical variables are presented as counts and percentages and the continuous variables as means and standard deviations (SD). We compared baseline characteristics of the exposure groups—those exposed to DOAC-PPI co-therapy at any time during study follow-up versus those exposed to DOAC alone—using standardized differences, with values >0.1 considered important. We compared outcomes during time periods during which patients were exposed vs not exposed to PPI as a time-varying variable.
A time-dependent Cox regression model was used to estimate the adjusted hazard of outcomes expressed using hazard ratios (HRs) and 95% confidence intervals (CIs). A secondary analysis was performed among the subset of DOAC patients who were exposed to PPIs at some point during the study period, comparing risks during the days when patients were exposed versus unexposed to PPIs. All statistical analyses were performed at ICES McMaster using SAS version 9.4 (Statistics Analysis System, Inc., Cary, North Carolina, USA).
Use of the data in this project was authorized under section 45 of Ontario’s Personal Health Information Protection Act, which does not require review by a Research Ethics Board.
Results
Participants
We identified 469 049 patients with a DOAC prescription during the 11-year study period. After applying the exclusion criteria, 283 771 new DOAC users remained—details appear in the Study Flow Diagram (Figure 1). A total of 140 280 (49.4%) patients were female and the mean (SD) age of patients at entry was 78.3 (7.7) years. Most lived in urban centers (246 118/283 771, 86.7%). The numbers receiving Apixaban, Dabigatran, Edoxaban, and Rivaroxaban were 115 701 (40.8%), 46 387 (16.3%), 3030 (1.1%), and 118 653 (41.8%), respectively. More than half of the study patients (184 165/283 771, 64.9%) had AF. Patients were followed for an average (SD) 514.0 (609.4) days. Patients’ sociodemographic and clinical characteristic details are presented in Table 1 and Table A2.
Baseline Characteristics of Patients Taking DOAC-PPI Co-Therapy Versus DOAC Alone.
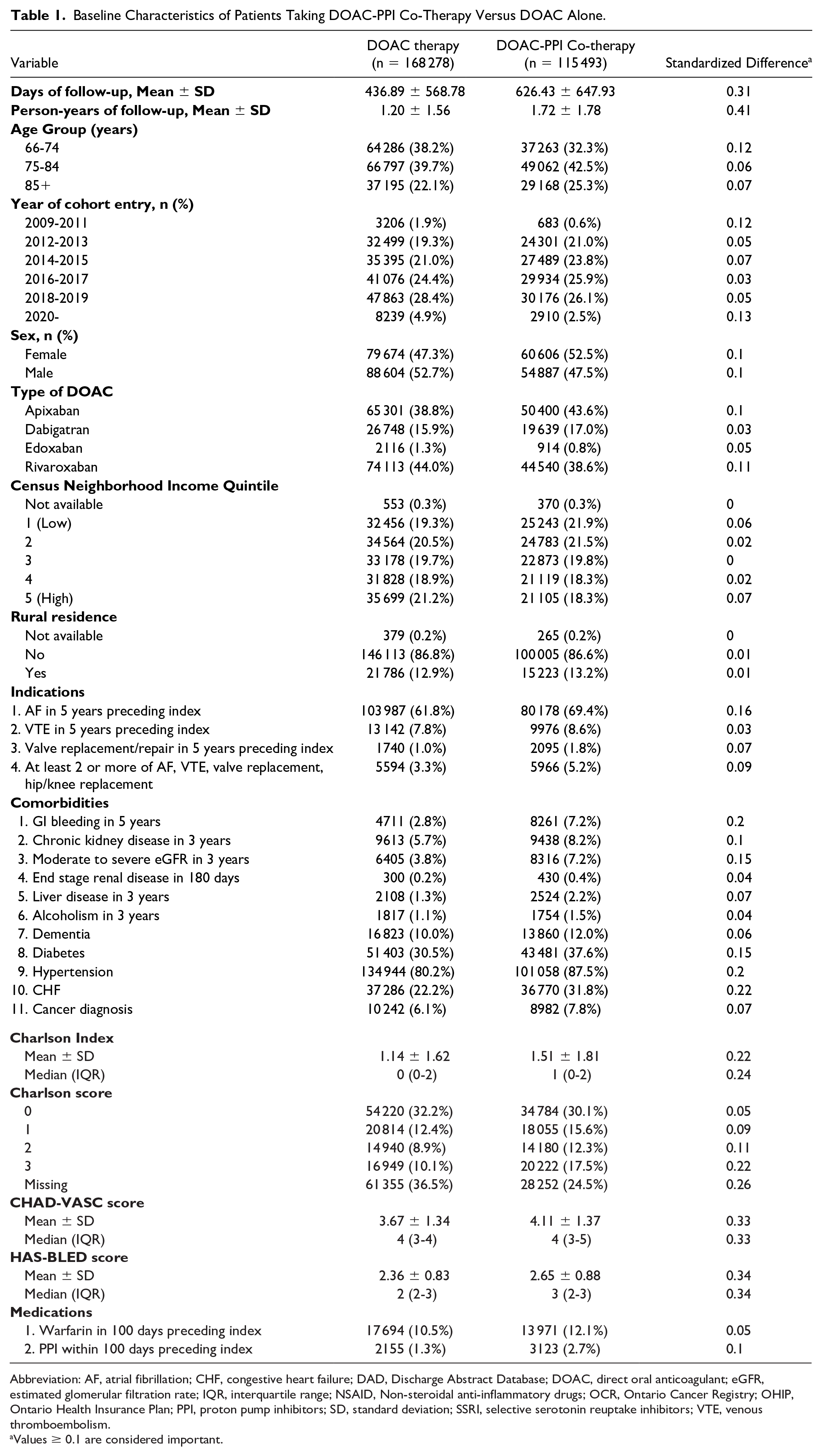
Abbreviation: AF, atrial fibrillation; CHF, congestive heart failure; DAD, Discharge Abstract Database; DOAC, direct oral anticoagulant; eGFR, estimated glomerular filtration rate; IQR, interquartile range; NSAID, Non-steroidal anti-inflammatory drugs; OCR, Ontario Cancer Registry; OHIP, Ontario Health Insurance Plan; PPI, proton pump inhibitors; SD, standard deviation; SSRI, selective serotonin reuptake inhibitors; VTE, venous thromboembolism.
Values ≥ 0.1 are considered important.
We identified 115 493 and 168 278 patients in the DOAC-PPI (defined as patients taking a PPI at any point during their available follow-up) and DOAC-only cohorts, respectively. The DOAC-PPI cohort of 115 493 patients contains 198 648 person-years of observation of patients taking DOAC and PPI together. The 279 670 person-years of observation for no PPI co-therapy included a total of 332 342 patients, 167 672 patients in the DOAC-only cohort, as well as 74 497 patients in the DOAC-PPI cohort who had periods where they had DOAC without PPI. A comparison of baseline characteristics between the DOAC-PPI treated and DOAC only treated patients in Table 1 shows expected differences for multiple potential confounders, including age, history of AF, history of bleeding, Charlson Comorbidity Index score, CHA2DS2-VASC score, and HAS-BLED score, with the PPI-treated patients being older, with greater comorbidity burden and having characteristics that put them at relatively greater risk for subsequent bleeding, thrombosis, and death.
Age/Sex-Standardized Outcome Event Rates
A total of 60 639 composite outcome events occurred during follow-up, with 23 903 and 36 736 events during the DOAC-PPI observation times and DOAC-only observation times, respectively. The age/sex-standardized composite outcome event rate of the composite outcome was higher during the DOAC-PPI observation times [20.2 (95% CI 20.0-20.5) per 100 person-years] than that during the DOAC-only observation times [15.2 (95% CI 15.1-15.4) per 100 person-years]. Results for the other outcomes are presented in Table 2. The age/sex-standardized outcome event rates were consistently greater during DOAC-PPI co-therapy.
Age/Sex-Standardized Outcome Event Rates During DOAC-PPI Co-Therapy Versus DOAC Alone.
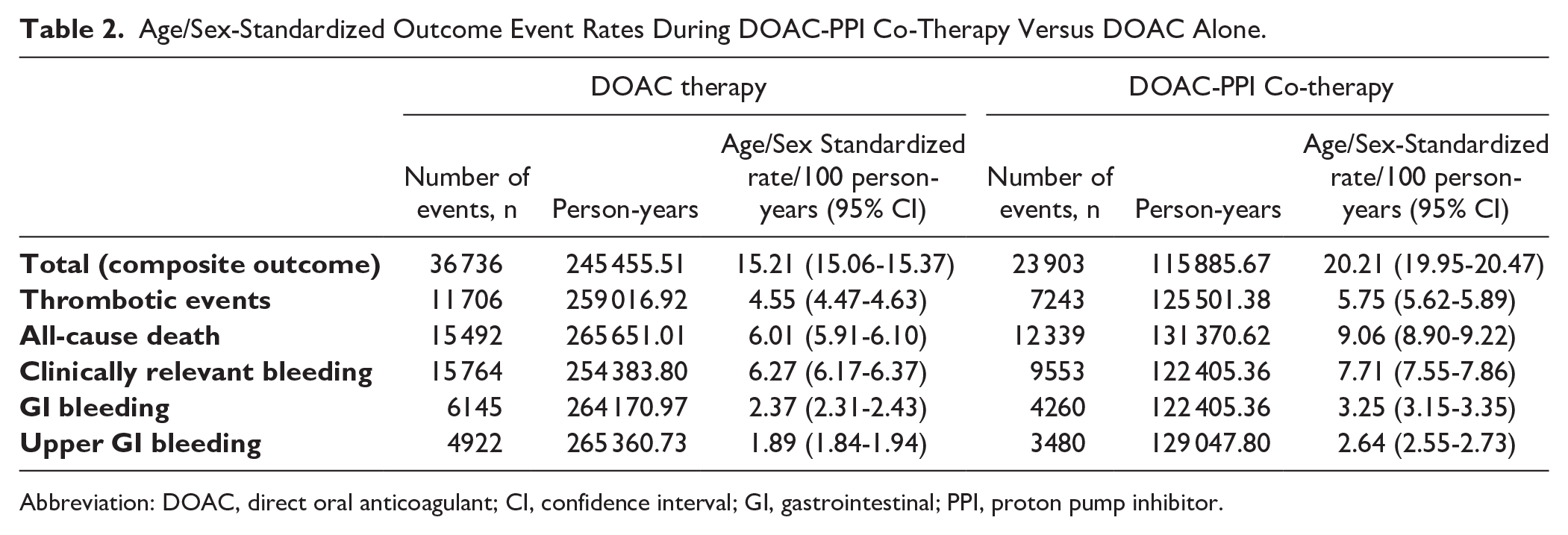
Abbreviation: DOAC, direct oral anticoagulant; CI, confidence interval; GI, gastrointestinal; PPI, proton pump inhibitor.
Time-Dependent Cox Regression Analyses
In the fully-adjusted time-dependent Cox regression analyses comparing patient time periods exposed to PPIs with periods exposed to DOAC alone, risks were elevated for all outcomes during periods of PPI co-therapy, with the adjusted hazard ratios (HR,95% CI) ranging from 1.04 (95% CI 1.02-1.07) for clinically relevant bleeding to 1.20 (95% CI 1.16-1.22) for all-cause death (Table 3).
Adjusted Hazard Ratios (95% CI) for Study Outcomes for DOAC-PPI Co-Therapy.
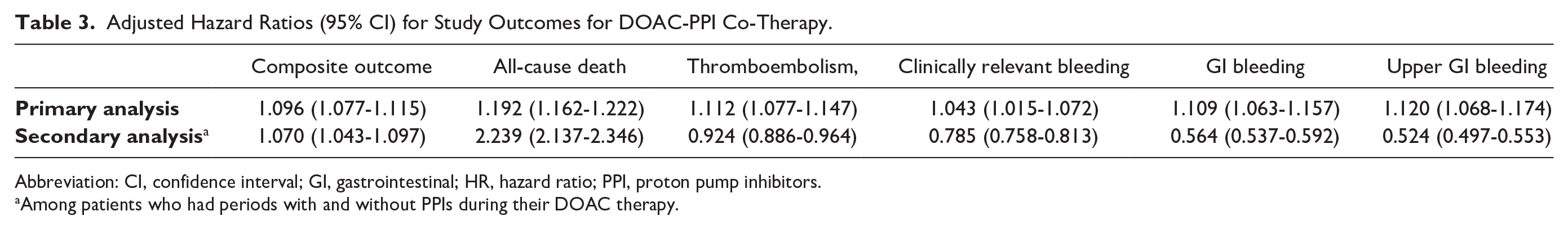
Abbreviation: CI, confidence interval; GI, gastrointestinal; HR, hazard ratio; PPI, proton pump inhibitors.
Among patients who had periods with and without PPIs during their DOAC therapy.
In the secondary analysis of the subset of 115 493 patients who received at least 1 day of DOAC-PPI co-therapy, compared with periods when patients were not exposed to PPI, periods of DOAC-PPI co-therapy were associated with a greater hazard for death (HR 2.24, 95% CI 2.14-2.35), but a significantly lower risk for both thrombosis (HR 0.93, 95% CI 0.89-0.96) and clinically relevant bleeding (HR 0.79, 95% CI 0.76-0.81), risk reductions of 7 and 21%, respectively (Table 3).
Discussion
In this population-based cohort study, DOAC-PPI co-therapy was associated with increased risk for all study outcomes, independently and collectively. This suggests important residual confounding. In our secondary analysis of patients who were exposed to DOAC-PPI co-therapy at some time during the study period, periods of PPI co-therapy were associated with fewer clinically relevant bleeds and thromboembolic events, but with double the risk of death. The relatively greater risk of death during periods of DOAC-PPI co-therapy suggests that PPI treatment was a surrogate for periods of higher risk likely due to worse illness beyond bleeding and thromboembolic events. More detailed data including laboratory results, markers of frailty, residence in long term care, use of chemotherapy, socioeconomic factors, and so on would help clarify prognosis but might require electronic medical record data.
The main purpose for using PPIs in patients with DOACs is to decrease the risk of potential OAC-related GI bleeding while having a neutral effect on thromboembolic events. There is no evidence or pharmacologic rationale including drug interaction with DOACs for PPIs causing an increase in thromboembolic events or death while reducing GI bleeding. The pharmacokinetic interactions proposed mainly affect dabigatran which is no longer commonly used, and the interaction is mainly theoretical and would decrease the DOAC effect. 33 Multiple guidelines based on trials and long term safety evidence, recommend PPIs to decrease GI bleeding and gastroesophageal reflux disease (GERD) events such that the drugs are amongst the most widely used families in the world.34,35
Other studies have addressed DOAC—PPI interactions and their impact on bleeding. However, none has targeted the comparison of all of the key clinical events related to oral anticoagulant use—thromboembolic, hemorrhagic, and death.9,11,20,22,24,25,33 A Korean population-based study published in 2022 found that PPI co-therapy with rivaroxaban for patients with AF who had previously had a GI bleed, was associated with a significantly lower risk of major GI bleeding, but in dabigatran, apixaban, and edoxaban users, PPI co-therapy did not show a significant reduction in the risk of major GI bleeding. 24 Ray et al 11 examined the incidence of upper GI bleeding hospitalizations for individual anticoagulants without and with PPI co-therapy and found the risk of upper GI tract bleeding hospitalizations to be lower with PPI co-therapy (IRR, 0.66 [95% CI 0.62-0.69]). Recently published systematic reviews which summarize the evidence on association of anti-secretory drugs with upper GI bleeding in patients using OACs suggest that patients taking OAC and PPI co-therapy achieved a lower total, major, or upper GI bleeding compared with OAC alone.36,37 These findings are similar to our secondary analysis of DOAC patients who served as their own controls by having periods with and without DOAC-PPI co-therapy. However, it is unclear what is contributing to the greater risk of death during periods of DOAC-PPI co-therapy. Death rates for patients with disorders requiring OAC therapy can be substantial—from 7% over 2 years in patients with AF to 8% to 10% in-hospital mortality in patients with cancer.38,39 A recent systematic review suggests that the most frequent causes of death in patients taking DOACs are cancer (42%), venous thromboembolism (20%), infections (13%), bleeding (6%), heart disease (4%), and stroke (2%). 40 These are common causes of death, given the diseases that patients taking DOACs tend to have. We are not aware of data that DOACs themselves increase mortality rates from cancer, infections or vascular events.
Strengths of our study include its large, ethnically diverse population-based cohort in Ontario (approximately 250 ethnic origins were reported by Ontarians in the 2016 Census) 41 and rigorous methodology. Our study also has important limitations. First, as a retrospective cohort study of administrative data, it suffers from the intrinsic problem of confounding, including unmeasured confounding. Second, our study concentrated on seniors since they are the most relevant population for this question and, until recently, the only segment of the Ontario population with universal drug coverage. Consequently, we are uncertain about the generalizability of the results to younger people. Third, some PPIs and other relevant drugs (eg, Aspirin and NSAIDs) are available over the counter, leading to possible information bias, although coverage of PPI by the provincial drug plan makes it less likely PPI would be purchased out of pocket. Finally, more sophisticated methodologies beyond the scope of this article (such as propensity score-matching) might have helped reduced confounding.
In terms of the clinical and policy implications of our findings, they suggest clinicians and policymakers need not make important changes in practice. However, future studies, preferably randomized trials, on this topic are a high priority given the huge population at risk of potentially severe clinical outcomes.
Conclusion and Relevance
In this population-based cohort study of older adults, analyses including self-controlled time dependent analyses showed that periods of DOAC-PPI co-therapy were associated with reduced GI bleeding and a small decrease in thromboembolic events, but increased mortality compared with periods of DOAC alone. Since the implications are major and may be due to residual confounding, we recommend that the findings be verified in randomized trials before clinical application.
Footnotes
Appendix A
Acknowledgements
This study was supported by ICES, which is funded by an annual grant from the Ontario Ministry of Health and the Ministry of Long-Term Care. This study used data adapted from the Statistics Canada Postal Code Conversion FileOM, which is based on data licensed from Canada Post Corporation, and/or data adapted from the Ontario Ministry of Health Postal Code Conversion File, which contains data copied under license from ©Canada Post Corporation and Statistics Canada. Parts of this material are based on data and/or information compiled and provided by the Ontario Ministry of Health and the Canadian Institute for Health Information. We thank IQVIA Solutions Canada Inc. for the use of their Drug Information File. The analyses, conclusions, opinions, and statements expressed herein are solely those of the authors and do not reflect those of the funding or data sources; no endorsement is intended or should be inferred.
Author Contributions
Conceptualization: MW, AH, and MP; Data curation: FN; Formal analysis: FN; Funding acquisition: MW and AH; Investigation: All authors; Methodology: MW, AH, FN, and MP; Project administration: AH and MP; Resources: MP and FN; Software: MP and FN; Supervision: AH and MP; Validation: DS, AH, and MP; Writing—original draft: MW; Writing—review and editing: All authors.
Data Availability
The data set from this study is held securely in coded form at ICES. While legal data-sharing agreements between ICES and data providers (eg, health care organizations and government) prohibit ICES from making the data set publicly available, access may be granted to those who meet pre-specified criteria for confidential access, available at ![]() (email:
(email:
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funded by the Canadian Institutes of Health Research (CIHR) Award Number 365834 to Dr Anne Holbrook, a studentship award to Mei Wang from the Research Institute of St. Joe’s Hamilton and a CanVECTOR Research Start-Up Award.
Ethical Considerations
Use of the data in this project was authorized under section 45 of Ontario’s Personal Health Information Protection Act, which does not require review by a Research Ethics Board.
