Abstract
Objective:
There is limited knowledge about severe urinary tract infections associated with SGLT2i, despite this being the basis for the Food and Drug Administration (FDA) warning. We aim to provide real-world evidence to clarify this relationship further.
Data source:
A literature review was performed in PubMed and Embase for cohort studies published up to August 2024 using PICO-consistent terms.
Study selection and data extraction:
Cohort studies in English involving new users of SGLT2i that compare SGLT2i with glucagon-like receptor agonists (GLP-1RA), DPP4i, and other glucose-lowering medications and report severe urinary tract infection (UTI).
Data synthesis:
The random-effect model determined the odds ratio (OR) and 95% confidence interval (CI) for severe UTI. Subgroup analysis and meta-regression were used to identify sources of heterogeneity. In 11 cohort studies involving 679 617 individuals with type 2 diabetes mellitus and a median age of 64 (interquartile range [IQR] = 56-72) and 42% (IQR = 39%-51%) females, it was found that the use of SGLT2i was associated with a reduced risk of severe UTI compared with both composite glucose-lowering medications (OR = 0.73, 95% CI = 0.60-0.88) and DPP4i (OR = 0.48, 95% CI = 0.43-0.54). There was no significant difference in the risk compared with GLP-1RA (OR = 0.94, 95% CI = 0.78-1.14).
Relevance to patient care and clinical practice:
The lack of increased risk for severe UTI reassures physicians when assessing benefit-risk to continue SGLT2i after a severe UTI. This may enhance patient adherence and improve diabetes management. Furthermore, our findings show no significant risk increase in chronic kidney disease (CKD) patients who would benefit significantly from SGLT2i.
Conclusion:
SGLT2i does not appear to pose a greater risk of severe UTI than other oral glucose-lowering medications. This contributes to the existing literature on UTI, accounting for the event’s severity. However, more data are needed to assess the potential association between SGLT2i and life-threatening UTI events.
Keywords
Introduction
In 2015, the Food and Drug Administration (FDA) issued a drug safety communication regarding the increased risk of serious urinary tract infection (UTI) associated with sodium-glucose co-transporter 2 inhibitors (SGLT2is). 1
The impact of the glucosuric effect on UTI risk is debated. Previous meta-analyses show an insignificant association between SGLT2i use and UTI but are not entirely ruled out. 2
In a meta-analysis of 86 Randomized Controlled Trials (RCTs) enrolling 50 880 patients, the risk of urosepsis was insignificant with SGLT2i vs placebo. Still, a wide 95% confidence interval (CI) interpreted the results as inconclusive (Relative Risk = 1.41, 95% CI = 0.57-3.48, I2 = 0%). 3
Evidence concerning hospitalization due to severe UTI is limited. This definition aligns with the FDA’s characterization of serious UTI. The findings from pharmacovigilance analysis formed the basis for the FDA warning issued in 2015. 1 Therefore, our objective is to investigate the relationship between SGLT2i and severe UTI.
The extensive observational studies collected over the years have helped clarify the association, particularly for patient groups often excluded from RCTs, older populations, women, and high comorbidities. 2 Therefore, our meta-analysis includes observational studies over the years, explicitly analyzing severe UTI events.
According to studies, significant improvements in cardiovascular outcomes associated with SGLT2i could significantly reduce overall cardiovascular mortality and hospitalizations due to heart failure, particularly in patients with cardiovascular comorbidities.4 -6 A review published by the European Society of Cardiology addressed the controversial issue of cardiovascular safety concerning older glucose-lowering medications, such as dipeptidyl-peptidase-4 inhibitors (DPP4i), which are not recommended as a first-line treatment option for diabetic patients with heart failure. 7 In addition, sulfonylureas may increase the risk of heart failure, potentially leading to hospitalizations due to hypoglycemia. 8 It is well known that the early stages of sepsis can present as hypoglycemia and may be linked to the severity of the disease, resulting in poorer hospitalization outcomes.
This study is important for 2 main reasons. First, patients with type 2 diabetes mellitus (T2DM) who are hospitalized due to pyelonephritis or urosepsis face a higher risk of morbidity and mortality. 9 Second, studies on general urinary tract infections may not have a universally agreed-upon standard for diagnosis and sometimes need to differentiate between UTI with unspecific symptoms and asymptomatic bacteriuria based on subjective clinical judgment. 10 Hospitalization outcomes may be less susceptible to reporting or detection bias, for instance, severe UTI diagnosed by blood and urine bacterial cultures associated with sepsis criteria. 11
Methods
Meta-Analysis
This meta-analysis followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 12 The planned analysis was registered at the PROSPERO International Prospective Register of Systematic Reviews (CRD42024579358).
Data Source
Cohort studies investigating the safety or adverse events of SGLT2i in new users with T2DM were identified by searching the PubMed and Embase databases. All eligible studies in English published up to August 2024 were considered. Literature searches used specific keywords related to SGLT2i. The complete search strategies incorporated index terms, Medical Subject Headings (MeSH), and text words for the search in question are presented in Supplementary Table 2.
Inclusion Criteria
This meta-analysis included studies that met the following criteria. Patients with T2DM without any restrictions on age, sex, country, sample size, or ethnicity included in prospective or retrospective cohort studies that compare SGLT2i with other glucose-lowering medications and report severe UTI adverse events, urosepsis, pyelonephritis, and hospitalization due to UTI as a primary or secondary outcome.
Baseline information from the active and control groups had to be comparable, preferably by propensity score matching. Comorbidities or diabetes complications should be reported.
The observation group received new treatment with SGLT2i as a class or as individual agents compared with new users of active comparators, DPP4i, glucagon-like receptor agonists (GLP-1RA), or other guideline-directed glucose-lowering therapy.
Exclusion Criteria
The following criteria were established for the exclusion of studies in this meta-analysis: RCTs were excluded to ensure a focus on real-world data, as participants in RCTs are generally selected from a highly specific population. The analysis did not include systematic reviews, meta-analyses, pharmacovigilance disproportionality analyses, case reports, case studies, case series, letters, opinions, audits, protocols, and methodological papers. We excluded studies that lacked a control group, studies that did not report hospitalizations due to UTI, studies that demonstrated notable differences in follow-up duration between groups, and studies that did not employ propensity score matching or exhibited significant demographic disparities were also excluded. Furthermore, studies with potential immortal or lead-time bias were not considered.
These exclusion criteria were implemented to enhance the robustness and relevance of the analysis.
Data Extraction and Quality Assessment of the Studies
The studies underwent a rigorous screening process. First, duplicates were removed based on titles and abstracts. We then applied inclusion and exclusion criteria by assessing the methodology and demographic characteristics of the studies. This included considering new users of SGLT2i and a comparator, as well as considering comorbidities, follow-up time, long-term diabetes with previous medication use, and a glycosylated hemoglobin (HbA1c) greater than 7%. We also collected age, sex, study year, and studies on chronic kidney disease (CKD) and previous UTI. The outcomes collected were the events of severe urinary tract infections defined as hospitalization due to UTI, pyelonephritis, and urosepsis.
One reviewer (MA) identified the studies. Two reviewers (MA and YM) independently examined the articles for eligibility. We resolved disagreements by involving the principal investigator (EO).
Two authors independently assessed the studies’ quality using the Newcastle-Ottawa Scale. 13 This scale evaluates studies based on the following domains: selection, comparability, and exposure. The total score ranges from 0 to 9, with higher scores indicating higher study quality.
Studies were classified into poor quality (scores 0-3), moderate quality (scores 4-6), and good quality (scores 7-9).
Risk of Bias
We evaluated the risk of bias using 3 methods: first, considering the reported limitations mentioned in the studies. Second, the Joanna Briggs Institute (JBI) critical appraisal checklist for cohort studies was assessed using a score from 1 to 11. Supplementary Figure 4 shows the detailed bias assessment. 14 Finally, the reviewers found additional biases, such as lead-time bias, immortal time bias, confounding by indication, and unequal follow-up time, that were candidates for exclusion.
Outcomes
The primary outcome was the occurrences of severe urinary tract infections, defined as hospitalization due to UTI, urosepsis, or pyelonephritis, compared with GLP-1RA, DPP4i, and other glucose-lowering medications.
In the subgroup analysis, we evaluated how age impacted severe UTI. In addition, we compared other secondary outcomes, such as the influence of previous UTI, the presence of diabetes complications, the proportion of females in the studies, and the inclusion of CKD.
Statistical Analysis
Meta-analysis was performed using STATA version 18 and Review Manager 5.3 (RevMan 5.3). We conducted a random-effects meta-analysis to estimate the pooled treatment effect using odds ratio (OR) by the Hedges g’ model, considering sample size differences among the studies. For all dichotomous variables, ORs with 95% CIs were presented. Means and medians were calculated for all continuous variables. The P-value < 0.05 was deemed significant for all analyses.
The Cochran chi-square test, complemented by the I2 statistic, assessed statistical heterogeneity between studies. 15 I2 values of 25%, 50%, and 75% indicate low, moderate, and high heterogeneity, respectively.
We conducted a subgroup analysis based on the median age across the studies. This involved individuals older than the median age and used the following independent variables: prior UTI (defined in studies as occurring up to 1 year before the index date), studies reporting complicated diabetes, studies including CKD, and studies with over 50% female participation.
To identify the sources of heterogeneity, a meta-regression analysis was conducted to examine the impact of the contrast arm intervention, the proportion of females across studies, previous UTI, year, age, follow-up time, proportion of CKD, and sample size.
A sensitivity analysis was systematically performed to assess result stability, excluding 1 study at a time to examine its impact.
Ethics
Ethical approvals were not required, as the meta-analysis data was from published studies.
Results
A total of 2328 studies were retrieved, and 11 (n =679 617) were included in the analysis.16-26 Figure 1 shows the literature screening process.
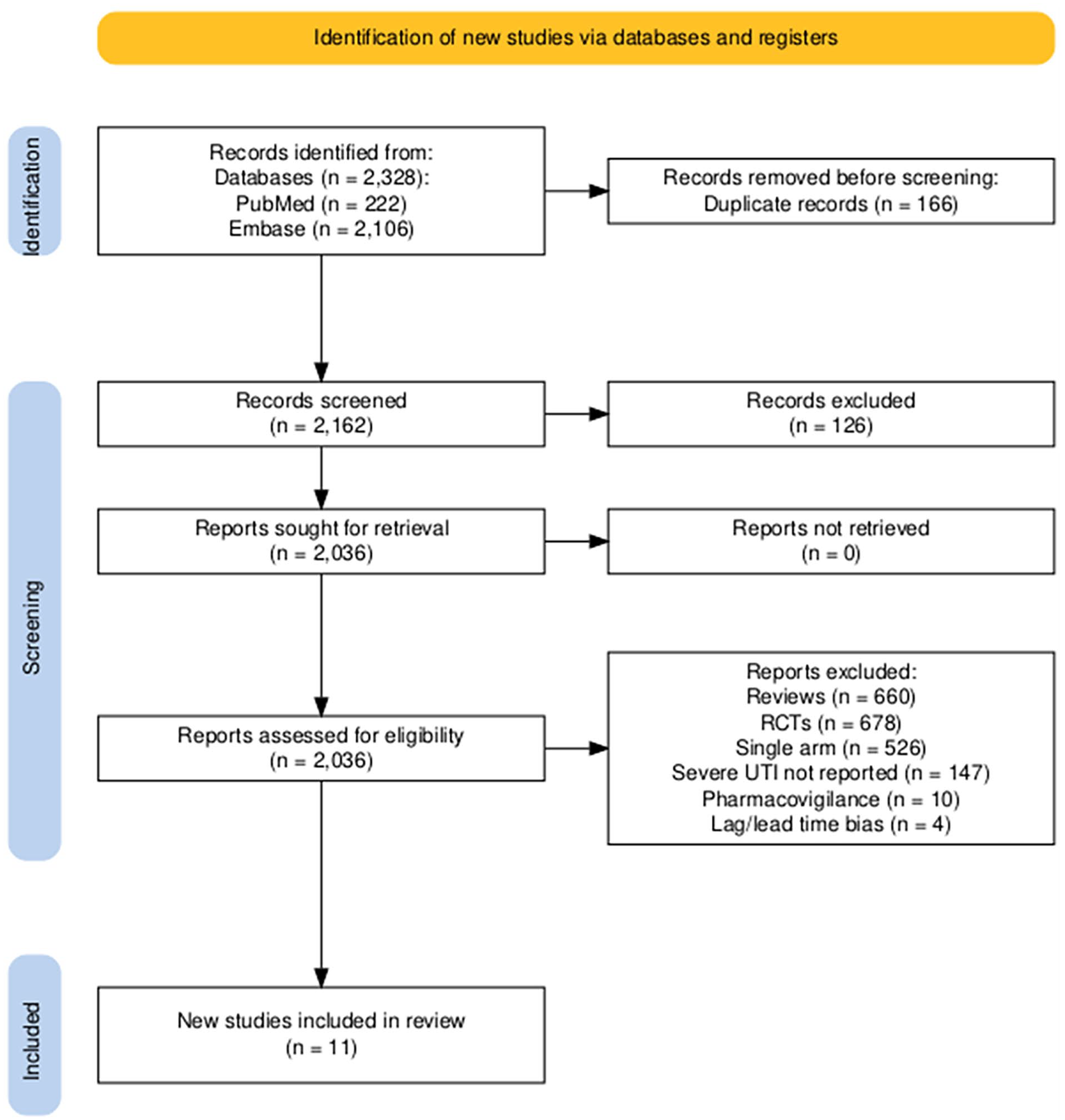
PRISMA flow chart.
The included studies were published from 2018 to 2024, with 9 published from 2020 onwards. Four were conducted in the United States, 2 in Europe, 1 in Canada, and 3 in Asia. One study included both US and European patients. All studies included patients with T2DM. Five included severe UTI as the primary outcome. The median age was 64 (interquartile range [IQR] = 56-72). The proportion of females in the studies was 42% (IQR = 39%-51%).
The median duration of follow-up was 10.5 months (range = 1-36 months). Baseline HbA1c was greater than 7% in all reported studies, and diabetes complications were reported in 5 studies. Table 1 summarizes the characteristics and limitations of the included studies.
Summary of Study Characteristics.
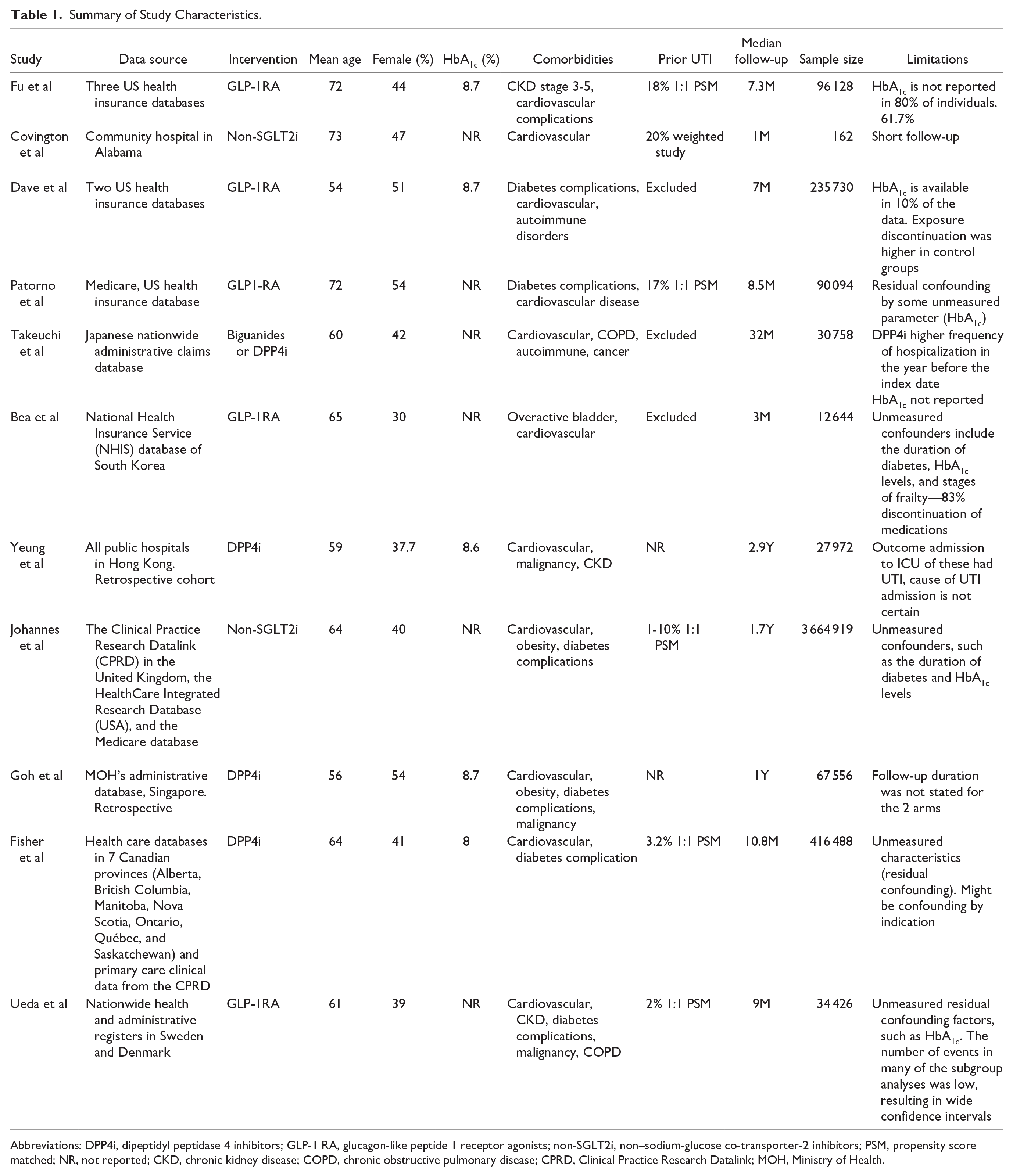
Abbreviations: DPP4i, dipeptidyl peptidase 4 inhibitors; GLP-1 RA, glucagon-like peptide 1 receptor agonists; non-SGLT2i, non–sodium-glucose co-transporter-2 inhibitors; PSM, propensity score matched; NR, not reported; CKD, chronic kidney disease; COPD, chronic obstructive pulmonary disease; CPRD, Clinical Practice Research Datalink; MOH, Ministry of Health.
In 11 studies, 45% compared SGLT2i with GLP-1RA and 55% compared SGLT2i with other glucose-lowering medications. The number of cases was 1636 among 177 732 patients treated with SGLT2i. In patients treated with GLP-1RA, 714 severe UTI cases were reported among 148 090 patients, and 3221 severe UTI cases were reported among 353 795 patients in the comparator group of various glucose-lowering medications. The SGLT2i did not show an increased risk of severe UTI than GLP-1RA (OR = 0.94, 95% CI = 0.72-1.21, P = 0.60, I2 = 81%). The SGLT2i class was associated with a reduced risk of severe UTI compared with DPP4i (OR = 0.48, 95% CI = 0.43-0.54, P < 0.001, I2 = 20%) and compared with other glucose-lowering medications (OR = 0.76, 95% CI = 0.66-0.89, P < 0.001, I2 = 0%). The overall effect size with all comparators favored the SGLT2i (OR = 0.73, 95% CI = 0.60-0.88, P < 0.001, I2 = 87%) (Figure 2).
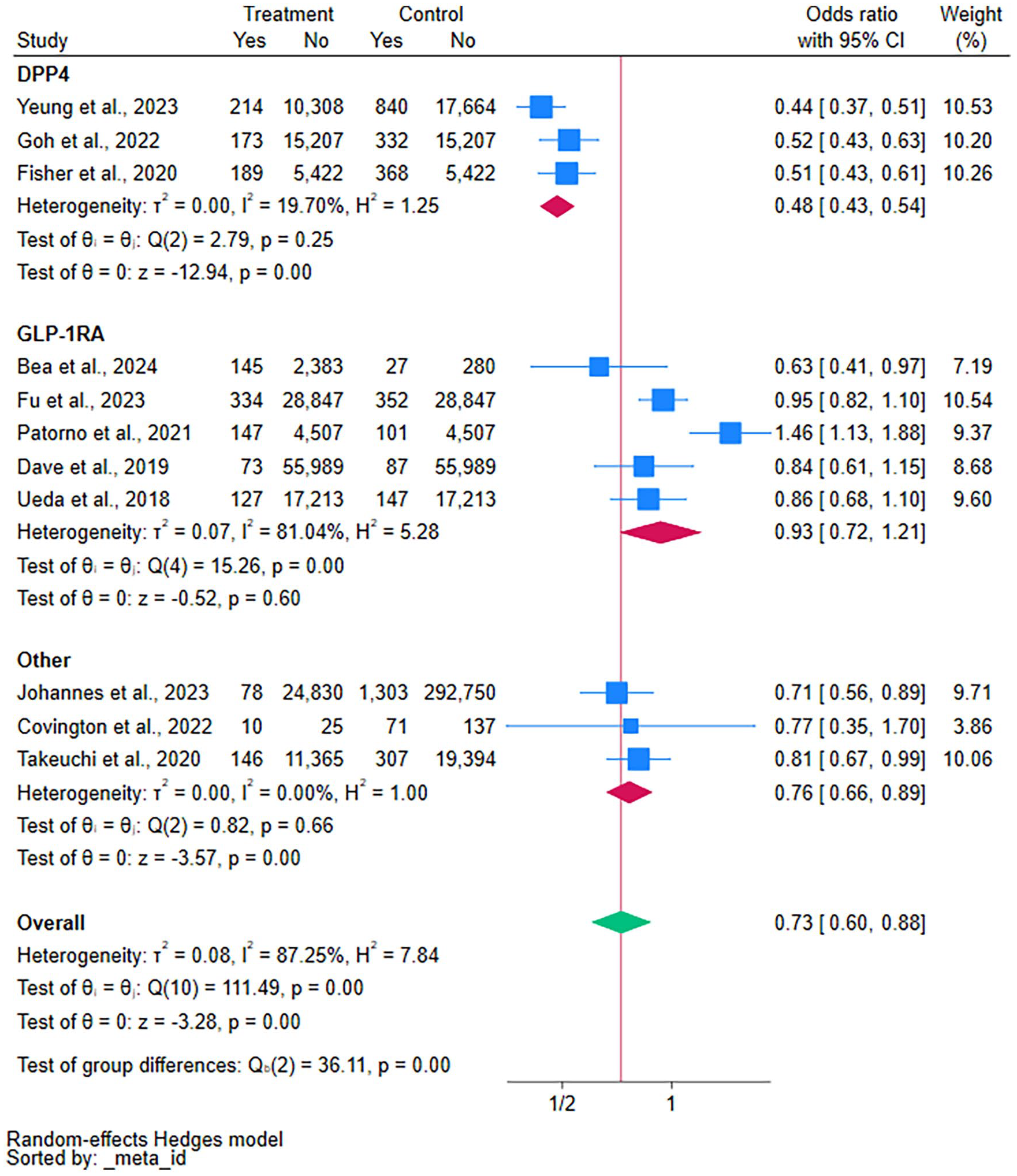
Severe UTI risk in SGLT2 inhibitors compared with GLP1-RAs, non-GLP-1RAs, and DPP4 inhibitors.
Secondary Outcome
Studies involving individuals under 64 years old showed a decreased risk of severe UTI (OR = 0.66, 95% CI = 0.50-0.87, P < 0.001, I2 = 89%). However, severe UTI in older age did not differ from the active comparator (Figure 3).
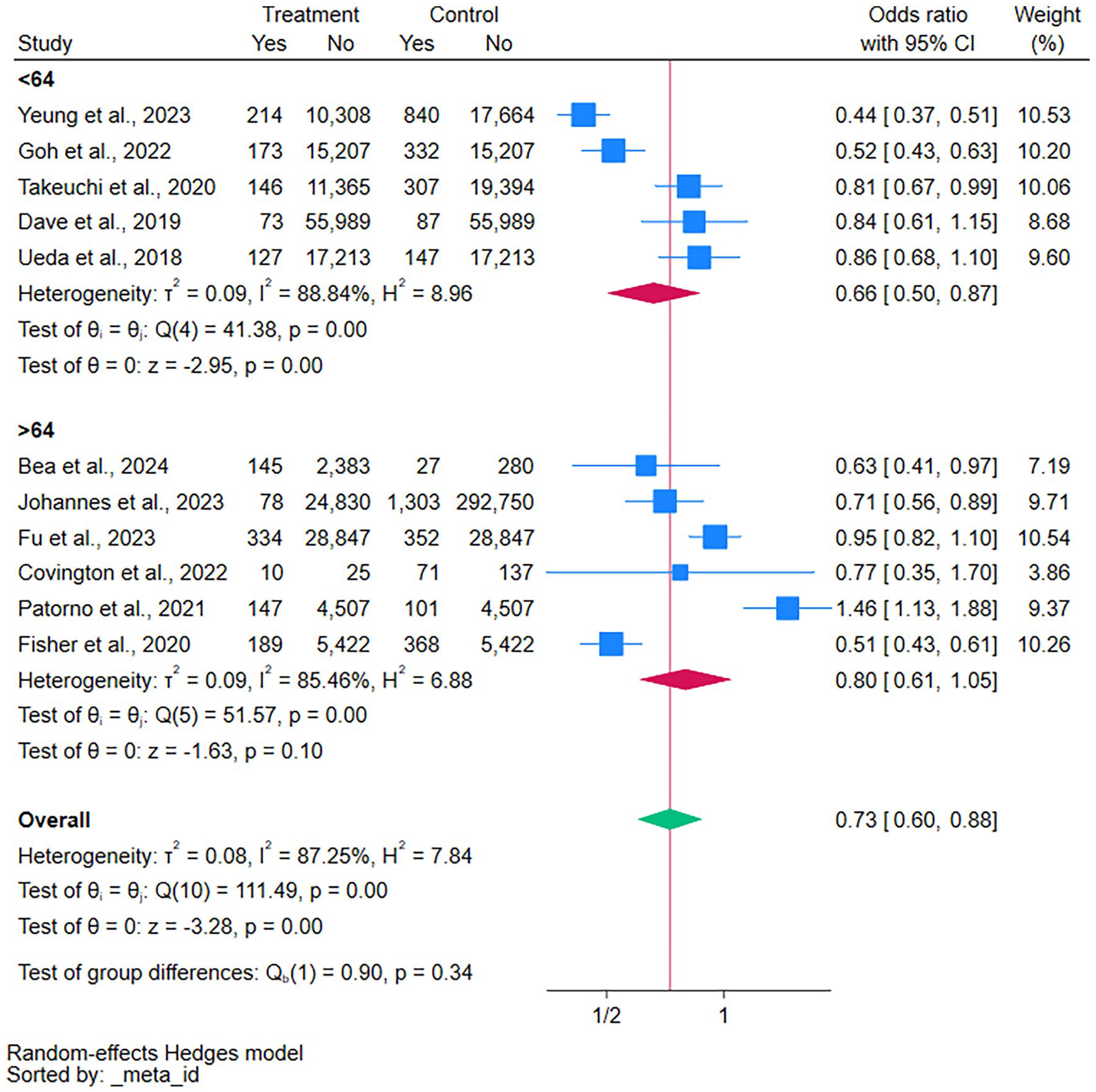
Age subgroup analysis of severe UTI in SGLT2 inhibitors.
Including more than 50% of females did not affect the results; both female groups (>50% or <50%) showed a lower association for severe UTI with SGLT2i compared with the active comparators (Figure 4).
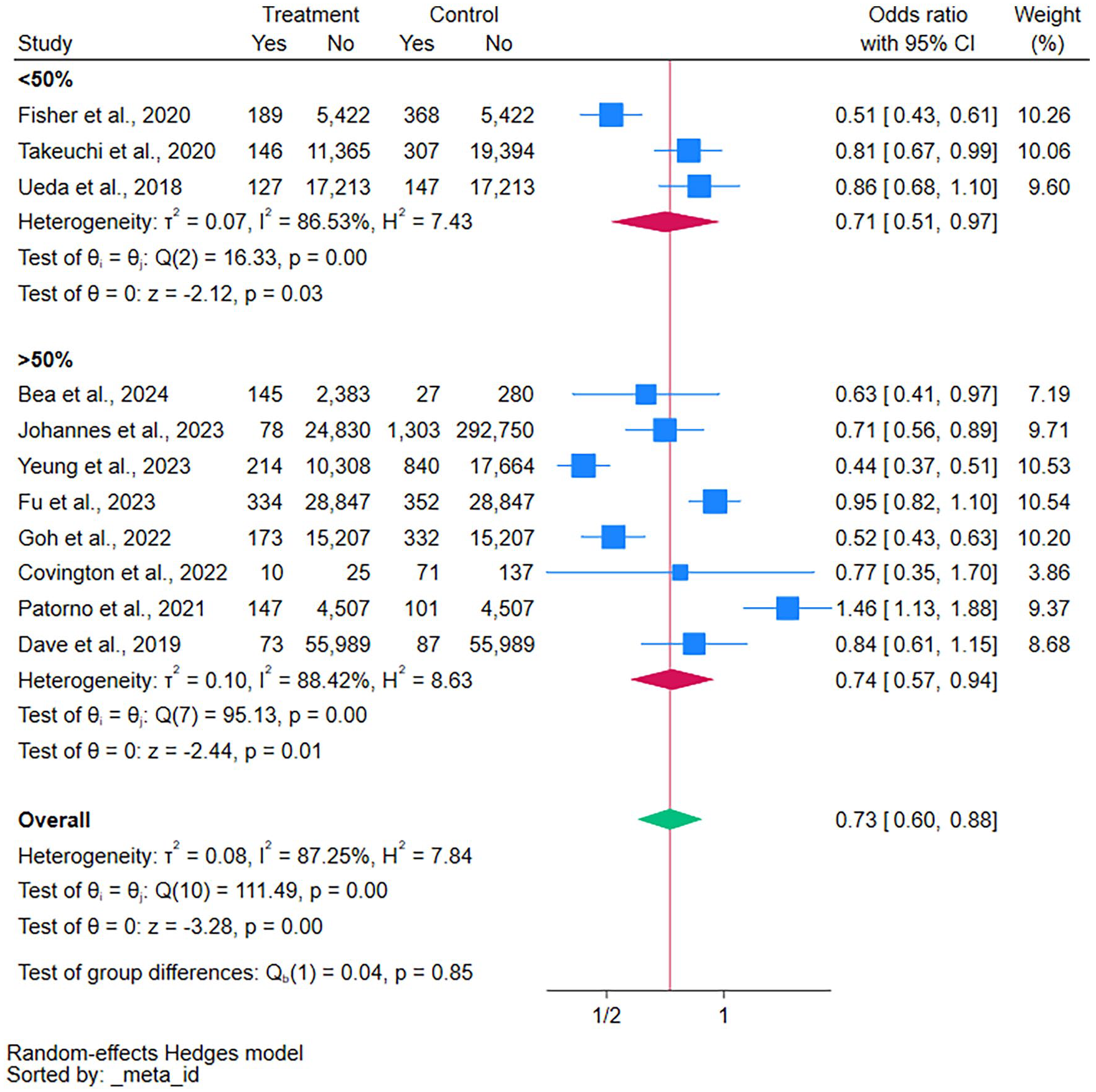
Female proportion subgroup analysis of severe UTI in SGLT2 inhibitors.
Subgroup analysis for studies that did not include patients with prior UTI demonstrated a lower risk (OR = 0.62, 95% CI = 0.48-0.81, P < 0.001, I2 = 85%) (Supplementary Figure 6). Studies excluding complicated diabetes demonstrated a lower risk (OR = 0.69, 95% CI = 0.55-0.86, P < 0.001, I2 = 77%) (Supplementary Figure 7). The CKD patients may also have a lower risk of severe UTI compared with the composite glucose-lowering medication (OR = 0.76, 95% CI = 0.60-0.95, P < 0.001, I2 = 86%) (Supplementary Figure 8). However, the groups did not differ from GLP-1RA.
Meta-Regression
Meta-regression analysis of the overall studies showed that the comparator intervention is the primary source of heterogeneity, explaining 65% of the heterogeneity between studies (R2 = 65%, Q = 8, df = 1, P = 0.005).
The independent variables, female proportion, diabetes complications, sample size, prior UTI, follow-up time, the percentage of patients with CKD, and the year of the study were not identified as the source of heterogeneity (Supplementary Table 9).
Sensitivity Analysis
Sensitivity analysis demonstrated that the combined effect values remained consistent before and after excluding any study for the above outcomes. This consistency suggests that the study results were stable.
Quality Assessment and Risk of Bias
The Newcastle-Ottawa Scale included moderate to good-quality studies (Supplementary Table 3). The median JBI score is 8 (IQR = 7-9), indicating a low risk of bias (Supplementary Figure 4).
Publication Bias Analysis
The funnel plot for severe UTI shows that the studies are distributed symmetrically about the combined effect size. Missing studies were in areas of statistical significance. In Egger’s test, the P-value for the primary outcome was P = 0.41, suggesting no significant publication bias (Supplementary Figure 10).
Discussion
This study is a meta-analysis of cohort studies that included severe UTIs as the primary outcome concerning SGLT2i treatment in patients with T2DM compared with other medications.
The results of this meta-analysis show that SGLT2is are not associated with severe UTI despite their glucosuric effect. The overall risk was lower than the composite comparators and did not differ when compared with GLP-1RA. The lower risk of severe UTI was also observed in subgroup analysis of patients without prior UTI, less than 64 years old, and with no diabetes complications. Notably, the sex composition of the studies did not influence the findings.
Compared with other medications, patients with CKD experienced a modest but significantly lower risk of severe urinary tract infections when treated with SGLT2i.
This is a noteworthy observation, given the historical concerns about increased severe UTI risk with SGLT2i by the FDA. 1 The debate about the risk of UTIs with SGLT2i is ongoing. However, studies consistently show no association.2,3 This study contributed evidence showing that meta-analyses of observational studies can reveal differences in severe and rare adverse events, such as severe UTI, while accurately reflecting real-world conditions of SGLT2i use. It is important to note that urinary tract infections are still common in clinical settings; however, our results focus explicitly on hospitalizations and the severity of UTI.
Several meta-analyses have examined the general association of urinary tract infections. However, regardless of severity, they typically focused on general UTI as their outcome.1 -3 Based on serious UTI signals, the FDA communication presents a contrasting perspective to most studies and meta-analyses. Similar to observational studies, signals by the FDA are also analyzed using real-world data but with reduced bias risk. Therefore, to examine the signals by the FDA, our study specifically delved into the association of SGLT2i with severe UTI reported by high-quality observational studies.
One of the significant challenges in diagnosing UTI in clinical studies or clinical settings is the need for a standard reference. The UTI mainly stems from subjective symptoms occurring in line with objective laboratory results, and this standard reference is crucial, especially when the patient presents with unspecific symptoms at an older age. 10
Our research findings demonstrate that individuals over the age of 64 using SGLT2i do not experience a heightened risk of severe UTI, thus reinforcing the validity of the results.
In 2019, Dave et al published the first large observational study of 235 273 1:1 propensity score-matched individuals, including severe UTI as the primary outcome in SGLT2i. In this study, the authors did not find an increase in risk for severe UTI events among new users of SGLT2i. Still, some evidence suggested a lower risk for UTIs with these agents vs GLP-1RA. 25 Fisher et al 24 found a lower rate of urosepsis with SGLT2i than DPP4i (adjusted hazard ratio = 0.58; 95% CI = 0.42-0.80). In 2022, Covington et al 23 published a smaller study that strengthened the results by Dave et al; SGLT2is were not associated with an increased risk of bacteremia.
Although these studies demonstrate a typical pattern of decreased incidence of severe UTI in SGLT2i, most did not reach statistical significance. Thus, meta-analysis is a robust tool for investigating the estimated effect size.
The benefits of SGLT2i on cardiac function may explain the reduced association in hospitalized UTI compared with DPP4i and other glucose-lowering medications. The SGLT2i users may be less prone to cardiac decompensation, hypoglycemia, and heart failure during complicated UTI.
The American Diabetes Association (ADA) views SGLT2i and GLP-1 RA as preferred treatments for managing T2DM. 27 In a meta-analysis of RCTs, GLP-1RA reduced the risk of all-cause mortality, hospital admission for heart failure, and worsening kidney function in patients with T2DM. 28 Although the mechanisms for improving glycemic control differ significantly, both preferred treatments may offer protection against acute events, such as severe UTI, compared with other glucose medications.
Additional hypotheses may include the biological plausibility of the protective effects of SGLT2i. Currently, some evidence is accumulating regarding the immunomodulatory properties of SGLT2i. An in-vitro study suggests that SGLT2i have direct anti-inflammatory effects by reducing the release of cytokines (TNF-α, IL-1β, IL-6, IL-8). Three human studies have shown lower IL-6 levels in SGLT2i users. Other studies suggest that SGLT2i may help reduce excessive nitric oxide release during sepsis.29 -31
These effects are significant in slowing the progression of renal disease and have some renal protective value, which could explain our results for the subgroup analysis in CKD patients.32,33 In this context, GLP-1RA may also have anti-inflammatory properties, which could account for the lack of difference between SGLT2i and GLP-1 RA. 34
The clinical translation of the SGLT2i immunomodulatory properties is still unclear, but they were demonstrated in other sources of infection. A nationwide registry study from Hong Kong revealed a lower risk of pneumonia-related death, sepsis-related death, and infection-related death compared with DPP4i. 35
In contrast, the DEFENDER trial, a recent open-label RCT that assessed the addition of dapagliflozin to standard care for critically ill patients, did not show improvement in mortality, hospital stay, or the need for renal replacement therapy. 36 However, 60% of the DEFENDER trial participants had acute organ dysfunction unrelated to infection and without reports of severe UTI.
This could also mean that the immunomodulatory properties are inadequate in the acute setting but do not rule out their long-term effect in prevention. However, more studies are required to prove this hypothesis.
Strengths and Limitations
Our investigation has a large cohort of 679 617 patients with T2DM and included data from studies conducted in various countries. Despite scarce evidence on severe UTI, rigorous inclusion and exclusion criteria ensured the study’s robustness. Studies were propensity score matched for demographic characteristics and comorbidities.
To address the study’s heterogeneity, we used meta-regression and subgroup analysis. The insignificant impact of the input variables in the meta-regression is in line with stringent inclusion criteria. As expected, most of the heterogeneity is explained by the variety of interventions in the contrast arm. Studies with poor quality on the Newcastle-Ottawa Scale were excluded. The JBI score indicated a low risk of bias. Using Egger’s test, we found no evidence of publication bias.
The comparison to GLP-1RA provides several advantages. First, the ADA considers SGLT2i and GLP-1RA as preferred therapies in T2DM. Hence, diabetes complications tend to be similar in GLP-1RA and SGLT2i. Second, this comparison potentially accounts for the socioeconomic differences in the 2 groups; both drugs are considered new and more expensive than older glucose-lowering medications, helping mitigate these differences.
Last, the pharmaceutical industry is more likely to promote studies in both medicines as they are considered new developments. This helps mitigate publication bias.
Our study has several limitations. These include unmeasured factors that may have impacted the heterogeneity of our results, potentially explaining the differences between studies. Using various large-cohort databases and including prospective and retrospective studies could also have affected the heterogeneity. There are also limited data for analyzing severe urinary tract infections with individual SGLT2i and reports of life-threatening UTI; further research is needed to confirm the safety of reinitiating SGLT2i treatment after a life-threatening UTI episode, particularly in cases involving septic shock due to a UTI.
Despite the high heterogeneity in our study, we opted to analyze real-world data to closely align with the disproportionality analysis of adverse events in pharmacovigilance. Our goal was to reduce bias and confounding factors that often complicate pharmacovigilance analysis, especially considering the FDA warning regarding serious urinary tract infections associated with SGLT2i compared with other medications.
Relevance to Patient Care and Clinical Practice
This real-world study provides insights into the FDA’s 2015 communication regarding SGLT2i and severe urinary tract infections, indicating that SGLT2i does not appear to increase the risk of severe UTIs compared with other glucose-lowering therapies. This is important as SGLT2i is considered by the ADA as the preferred treatment for T2DM, providing significant improvements in cardiovascular health compared with older-generation glucose-lowering medications.
The findings have important clinical implications, reassuring physicians and reducing treatment discontinuation after a severe UTI event, ultimately enhancing adherence, particularly for patients on the low-risk end. Also, it facilitates the assessment of benefits and risks for prescribing SGLT2i to CKD patients in greater need of its protective effects and enhances guideline implementation. 33
Unlike older-generation glucose-lowering therapies, SGLT2i effectively and preferably treats T2DM while providing cardioprotective benefits that may prevent decompensation during acute events. They may also have immunomodulatory effects, potentially explaining the reduced association in CKD.
As adverse events are a common reason for non-adherence, improving this aspect could help reduce the need for exogenous insulin, lower hospitalization rates, and decrease overall mortality. 37
Conclusion
The SGLT2i does not appear to present a greater risk of severe UTI when compared with other oral glucose-lowering medications. In addition, a decreased association was observed among specific patient subgroups, including individuals with CKD, women under the age of 65, and those without a history of prior UTI. Nonetheless, the study does not eliminate the possibility of UTI or severe UTI as complications in diabetic patients who are administered oral glucose-lowering medications. Instead, it indicates that SGLT2i may not exacerbate this risk.
Supplemental Material
sj-docx-1-aop-10.1177_10600280241312432 – Supplemental material for Sodium-Glucose Co-Transporter 2 Inhibitors and Severe Urinary Tract Infections: Real-World Meta-Analysis of Cohort Studies
Supplemental material, sj-docx-1-aop-10.1177_10600280241312432 for Sodium-Glucose Co-Transporter 2 Inhibitors and Severe Urinary Tract Infections: Real-World Meta-Analysis of Cohort Studies by Mohammed Aboukaoud, Yocheved Morhi and Ester Osher in Annals of Pharmacotherapy
Footnotes
Data Availability Statement
The main article provides all relevant material, as well as supplementary material from public databases and published research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval
This analysis did not require ethics approval and patient informed consent because it included only previously published research or data open to the public.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
