Abstract
Objective:
To evaluate the efficacy, safety, and clinical implications of insulin icodec, a novel once-weekly basal insulin for the treatment of type 1 diabetes (T1D) and type 2 diabetes (T2D), with an emphasis on its advantages and challenges in comparison with existing daily basal insulins.
Data Sources:
A literature search was performed using PubMed, Google Scholar, Embase, and ClinicalTrials.gov up to August 26, 2024, using the search terms icodec and ONWARDS trial. Studies involving patients living with T1D or T2D on once-weekly insulin icodec compared with once-daily insulins glargine U100, glargine U300, and degludec were considered for this review.
Study Selection and Data Extraction:
Relevant English-language studies and those conducted in humans were considered.
Data Synthesis:
Insulin icodec offers reduced dosing frequency and potentially superior glycemic management with a safety profile comparable to existing basal insulins.
Relevance to Patient Care and Clinical Practice:
Insulin icodec once-weekly dosing could significantly improve convenience and efficacy over daily basal insulins, representing a significant innovation in insulin therapy.
Conclusions:
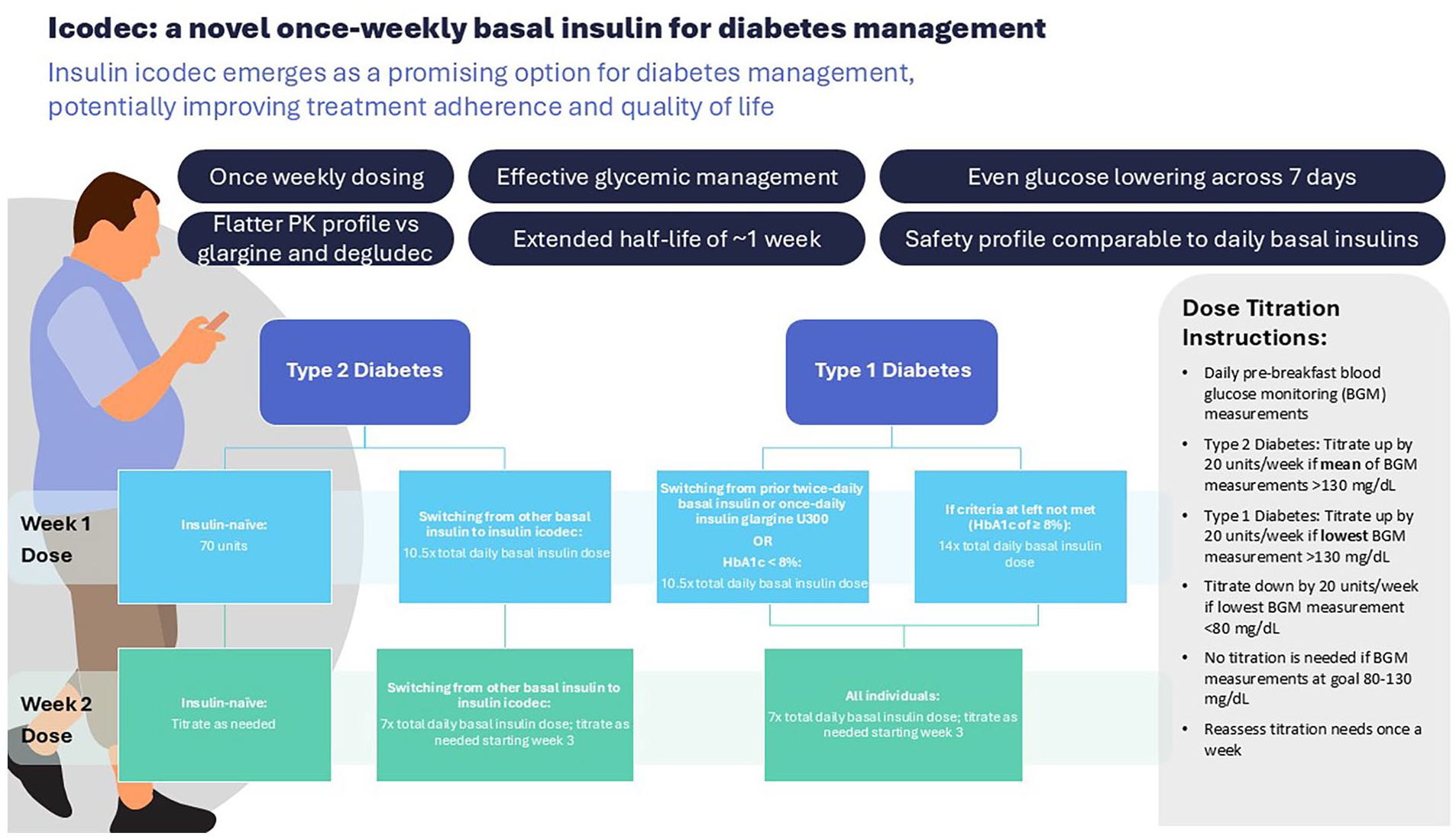
Insulin icodec emerges as a promising option for diabetes management, potentially improving treatment adherence and quality of life.
Graphical Abstract
Introduction
Diabetes has become a major worldwide health issue, affecting more than 537 million adults globally as of 2021. 1 Over the past 2 decades, there has been a significant increase in the prevalence of diabetes among adults in the United States. As of 2021, 38.1 million Americans aged 18 or older were living with diabetes, and an estimated 23% of type 2 diabetes (T2D) remain undiagnosed; approximately 1.7 million of those aged 20 or older have type 1 diabetes (T1D). 2 Diabetes leads to 6.7 million deaths per year globally and ranks as the eighth most common cause of death in the United States.1,2
Insulin is an essential treatment for diabetes, necessary for approximately 150 to 200 million individuals worldwide, and serves as the cornerstone in the management of T1D.3,4 Insulin plays a significant role in diabetes management in the United States, with 12.3% of adults with a diabetes diagnosis estimated to initiate insulin therapy within their first year of diagnosis. 2 Insulin glargine U100 was one of the first-generation long-acting basal insulin analogs approved for the treatment of T1D and T2D in 2000, 5 followed by second-generation, longer-acting insulin analogs, including insulin glargine U300 and insulin degludec, both US Food and Drug Administration (FDA)-approved as of 2015.6,7 For people with T2D, initiation of basal insulin is typically recommended when a combination of noninsulin therapies falls short of achieving glycemic goals or when severe hyperglycemia or catabolic symptoms are present.3,8 However, the initiation of any insulin therapy in patients with T2D is frequently delayed, often due to hesitancy on the part of both patients and healthcare providers to initiate insulin treatment. 9 This hesitation is rooted in concerns related to injection discomfort, potential adverse effects such as hypoglycemia and weight gain, diminished quality of life, and worries about treatment adherence when glycemic targets remain unmet. 9
Following initiation of insulin therapy, several obstacles associated with basal insulin treatment in T2D may hinder glycemic target achievement, with the most common challenges reported by physicians or patients being the frequency of injections, fear of hypoglycemia, adherence to prescribed schedules, and difficulties in adjusting insulin doses. 10 Early implementation of pharmacotherapy aimed at achieving glycemic goals has a demonstrable impact on mitigating microvascular complications in individuals with both T1D and T2D. 11 Evidence supports the early initiation of insulin treatment as a preventive measure against diabetic-related complications. 12 Furthermore, nonadherence to insulin therapy among patients with diabetes has been unequivocally correlated with increased rates of all-cause mortality and hospitalization. 13
Considering that one-third of individuals receiving insulin therapy exhibit nonadherence—typically defined as less than 80% of prescribed doses taken—the availability of once-weekly insulin formulations holds promise in addressing various challenges associated with adherence to insulin regimens.10,14 A once-weekly basal insulin could reduce the burden of daily insulin injections and potentially increase adherence with treatment, 15 similar to the advantages observed with once-weekly versus daily glucagon-like peptide-1 receptor agonists (GLP-1 RAs).16,17 Three phase II and 6 phase III trials have evaluated the safety and efficacy of insulin icodec, a human insulin analog formulation administered subcutaneously once weekly.18-26 Icodec has a prolonged half-life of 196 hours, approximately equivalent to 7 days, and reaches a steady state after 3 or 4 once-weekly injections.27,28
Data Sources
This review was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) checklist. 29 The search strategy aimed to locate published primary studies, meta-analyses, and systematic reviews. Using Population, Intervention, Comparison, Outcomes and Study (PICOS) criteria, 30 only studies involving people living with T1D or T2D on once-weekly insulin icodec compared with once-daily insulin glargine U100, glargine U300, and degludec were considered for this review. All publications related to insulin icodec that did not compare it with basal insulin analogs were excluded. Dissertations, conference abstracts, pharmacokinetic/pharmacodynamic studies, and publications pending publication were also excluded. The following databases were searched: PubMed, Google Scholar, Embase, and ClinicalTrials.gov (through August 26, 2024), and the following search terms were used: icodec and ONWARDS trial. The search was limited to the “English” language and “human trials,” exclusively focusing on trials assessing the efficacy or safety of icodec compared with basal insulins currently approved by the FDA and available for use. The bibliographies of all retrieved articles were reviewed for additional relevant citations.
Efficacy outcomes of interest included information related to continuous data at the baseline and at the endpoint of the study in relation to glycated hemoglobin (HbA1c, %) change from baseline, fasting plasma glucose (FPG, mg/dL) change from baseline, and time in range (TIR, %). Safety outcomes of interest included information related to body weight (kg) change from baseline, overall clinically significant hypoglycemia, and severe hypoglycemia reported in each trial.
Data Synthesis
Twenty-seven citations were found, and 9 unique studies, 3 meta-analyses, and 1 systematic review were reviewed and included. The flowchart for the selection of articles is presented in Figure 1.

Flowchart of selection of articles.
Pharmacokinetic Evolution of Basal Insulin
Basal insulin formulations have undergone significant transformations in the quest for optimal glycemic management and a reduction in hypoglycemic episodes. Initial insulin preparations required multiple daily injections due to their short duration of action, prompting the development of insulin formulations with prolonged biological availability and the potential for less frequent, once-daily administration. Earlier formulations involved the complexing of insulin with protamine or zinc to delay absorption. Still, these methods proved inadequate due to the occurrence of prominent insulin peaks following injection, variability in absorption rates, and an insufficient duration of action. 31
The inherent peaks observed with neutral protamine Hagedorn (NPH) or lente insulins increased the risk for hypoglycemia and did not possess the required duration of action suitable for once-a-day administration. 32 This led to the development of first-generation basal insulin analogs, including insulin glargine U100 and insulin detemir, aiming to eliminate insulin peaks and extend the duration of action up to 24 hours.33,34 Subsequently, second-generation basal insulin analogs, exemplified by insulin degludec U100 and U200 and insulin glargine U300, brought further improvements, minimizing peaks with flatter insulin profiles and sustained action exceeding 24 hours.35,36
Further evolution led to a once-weekly formulation where the glucose-lowering effect of insulin icodec exhibits a uniform distribution across the entire 1-week dosing period, suggesting that a peak following a dose is minimal because insulin icodec is strongly but reversibly bound to albumin, acting as a depot for the drug.15,37 These advancements represent milestones in the refinement of basal insulin therapy to enhance glycemic control and reduce the risk of hypoglycemia. Table 1 summarizes the key pharmacokinetic differences among basal insulin formulations.
Key Pharmacokinetic Features in the Evolution of Basal Insulin.

Abbreviations: FDA, US Food and Drug Administration; NPH, neutral protamine Hagedorn; U, units per milliliter.
Detemir is not included because all insulin detemir formulations will no longer be available for use in the United States after December 31, 2024. 44
Icodec Pharmacokinetic and Pharmacodynamic Considerations
Three amino acid substitutions (TyrA14Glu, TyrB16His, and PheB25His) in the structural engineering of insulin icodec result in improved molecular stability, reduced enzymatic degradation, and slower, insulin-receptor-mediated clearance. 15 The extended half-life of icodec was also achieved by incorporating a 20-carbon icosane fatty diacid (icosanedioic acid). These modifications facilitate strong but reversible binding to albumin, rendering the insulin inactive while bound.15,37 The formation of inactive albumin-bound icodec, coupled with its lower affinity for insulin receptors, leads to a gradual, continuous release that results in prolonged, consistent activation of insulin receptors and clearance of icodec from the bloodstream.15,37 This, in turn, results in a stable insulin action maintained throughout the 1-week dosing interval, with the mean glucose-lowering effect being close to evenly distributed over the entire week. 15 The serum concentration of insulin icodec at steady state is significantly lower than the serum albumin concentration, resulting in an abundance of albumin binding sites (>2000:1) compared with insulin icodec, making displacement of insulin icodec from albumin unlikely to have a clinically relevant effect on its activity, even in cases of higher icodec dosing or factors like hypoalbuminemia or competitive protein binding. 15 Furthermore, an analysis of 2 open-label, parallel-group trials found that neither renal nor hepatic impairment affected the shape of the pharmacokinetic profile of insulin icodec and that no specific icodec dose adjustment is required in people with renal or hepatic impairment. 45
Clinical Trials
The clinical efficacy and safety of treatment with icodec have been examined in one phase IIIa trial among people with T1D and several trials among individuals with T2D (3 phase II and 5 phase IIIa). Only the phase IIIa trials are included in the Once-Weekly Non-acute-Interventional Research in Diabetes (ONWARDS) program.18,20-27
Phase II Trials
The ONWARDS phase II trials are summarized in Table 2. Individuals with T2D treated with once-weekly icodec insulin have demonstrated favorable tolerance and glucose-reducing benefits. In individuals who were insulin-naïve, the administration of icodec, either alone or alongside other glucose-lowering agents, showed comparable effectiveness to once-daily insulin glargine U100, evidenced by reductions in baseline HbA1c levels (icodec, −1.33%; glargine U100, −1.15%; between-group difference: −0.18%; 95% confidence interval [CI]: –0.38 to 0.02, P = .08). 23 In addition, the incidence rate of combined clinically significant and severe hypoglycemia was similar in the groups treated with either insulin icodec or insulin glargine U100 (estimated rate ratio, 1.09; 95% CI: 0.45-2.65 [P value not reported]). 23
Insulin Icodec Phase II and III Trials Key Study Design Elements.
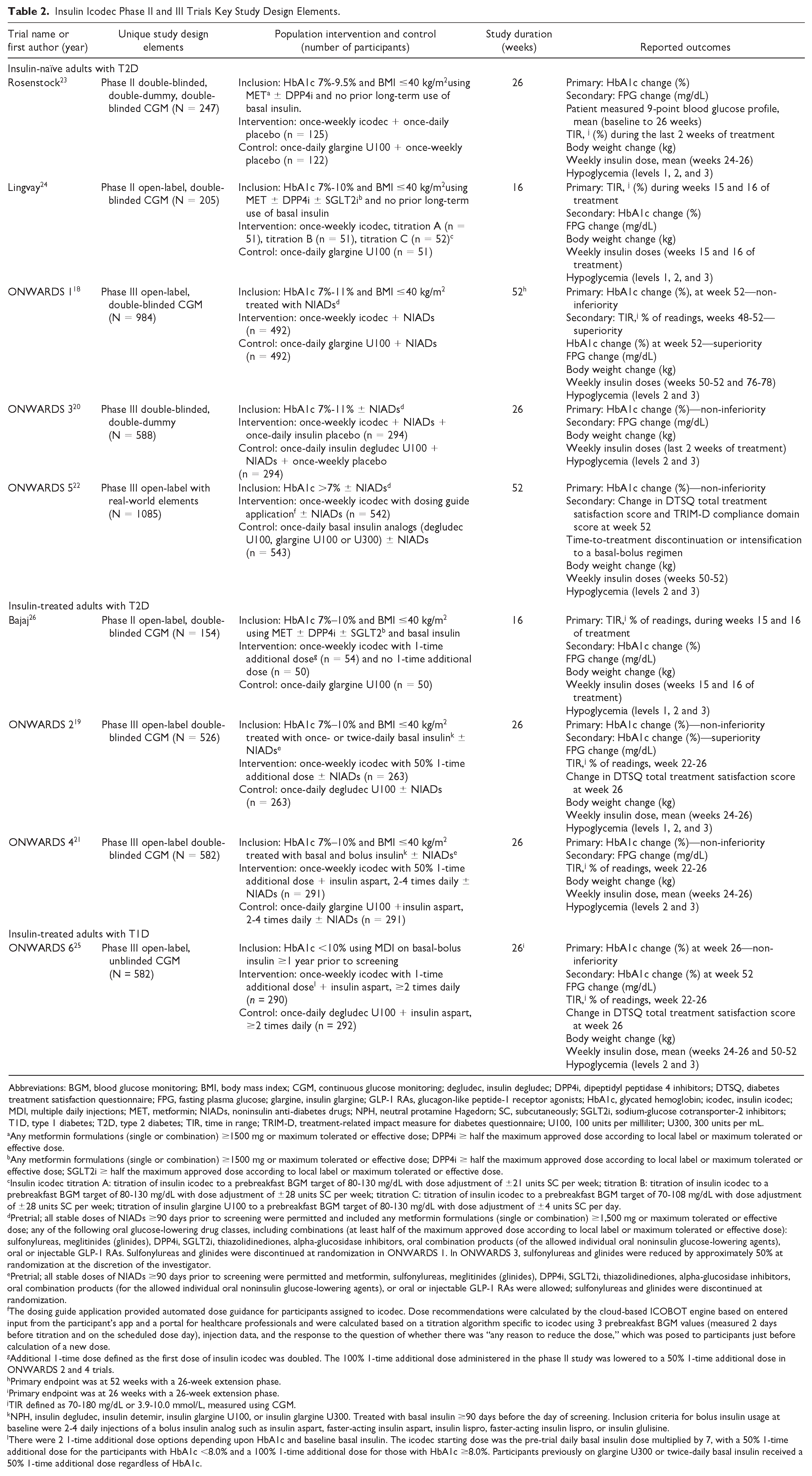
Abbreviations: BGM, blood glucose monitoring; BMI, body mass index; CGM, continuous glucose monitoring; degludec, insulin degludec; DPP4i, dipeptidyl peptidase 4 inhibitors; DTSQ, diabetes treatment satisfaction questionnaire; FPG, fasting plasma glucose; glargine, insulin glargine; GLP-1 RAs, glucagon-like peptide-1 receptor agonists; HbA1c, glycated hemoglobin; icodec, insulin icodec; MDI, multiple daily injections; MET, metformin; NIADs, noninsulin anti-diabetes drugs; NPH, neutral protamine Hagedorn; SC, subcutaneously; SGLT2i, sodium-glucose cotransporter-2 inhibitors; T1D, type 1 diabetes; T2D, type 2 diabetes; TIR, time in range; TRIM-D, treatment-related impact measure for diabetes questionnaire; U100, 100 units per milliliter; U300, 300 units per mL.
Any metformin formulations (single or combination) ≥1500 mg or maximum tolerated or effective dose; DPP4i ≥ half the maximum approved dose according to local label or maximum tolerated or effective dose.
Any metformin formulations (single or combination) ≥1500 mg or maximum tolerated or effective dose; DPP4i ≥ half the maximum approved dose according to local label or maximum tolerated or effective dose; SGLT2i ≥ half the maximum approved dose according to local label or maximum tolerated or effective dose.
Insulin icodec titration A: titration of insulin icodec to a prebreakfast BGM target of 80-130 mg/dL with dose adjustment of ±21 units SC per week; titration B: titration of insulin icodec to a prebreakfast BGM target of 80-130 mg/dL with dose adjustment of ±28 units SC per week; titration C: titration of insulin icodec to a prebreakfast BGM target of 70-108 mg/dL with dose adjustment of ±28 units SC per week; titration of insulin glargine U100 to a prebreakfast BGM target of 80-130 mg/dL with dose adjustment of ±4 units SC per day.
Pretrial; all stable doses of NIADs ≥90 days prior to screening were permitted and included any metformin formulations (single or combination) ≥1,500 mg or maximum tolerated or effective dose; any of the following oral glucose-lowering drug classes, including combinations (at least half of the maximum approved dose according to local label or maximum tolerated or effective dose): sulfonylureas, meglitinides (glinides), DPP4i, SGLT2i, thiazolidinediones, alpha-glucosidase inhibitors, oral combination products (of the allowed individual oral noninsulin glucose-lowering agents), oral or injectable GLP-1 RAs. Sulfonylureas and glinides were discontinued at randomization in ONWARDS 1. In ONWARDS 3, sulfonylureas and glinides were reduced by approximately 50% at randomization at the discretion of the investigator.
Pretrial; all stable doses of NIADs ≥90 days prior to screening were permitted and metformin, sulfonylureas, meglitinides (glinides), DPP4i, SGLT2i, thiazolidinediones, alpha-glucosidase inhibitors, oral combination products (for the allowed individual oral noninsulin glucose-lowering agents), or oral or injectable GLP-1 RAs were allowed; sulfonylureas and glinides were discontinued at randomization.
The dosing guide application provided automated dose guidance for participants assigned to icodec. Dose recommendations were calculated by the cloud-based ICOBOT engine based on entered input from the participant’s app and a portal for healthcare professionals and were calculated based on a titration algorithm specific to icodec using 3 prebreakfast BGM values (measured 2 days before titration and on the scheduled dose day), injection data, and the response to the question of whether there was “any reason to reduce the dose,” which was posed to participants just before calculation of a new dose.
Additional 1-time dose defined as the first dose of insulin icodec was doubled. The 100% 1-time additional dose administered in the phase II study was lowered to a 50% 1-time additional dose in ONWARDS 2 and 4 trials.
Primary endpoint was at 52 weeks with a 26-week extension phase.
Primary endpoint was at 26 weeks with a 26-week extension phase.
TIR defined as 70-180 mg/dL or 3.9-10.0 mmol/L, measured using CGM.
NPH, insulin degludec, insulin detemir, insulin glargine U100, or insulin glargine U300. Treated with basal insulin ≥90 days before the day of screening. Inclusion criteria for bolus insulin usage at baseline were 2-4 daily injections of a bolus insulin analog such as insulin aspart, faster-acting insulin aspart, insulin lispro, faster-acting insulin lispro, or insulin glulisine.
There were 2 1-time additional dose options depending upon HbA1c and baseline basal insulin. The icodec starting dose was the pre-trial daily basal insulin dose multiplied by 7, with a 50% 1-time additional dose for the participants with HbA1c <8.0% and a 100% 1-time additional dose for those with HbA1c ≥8.0%. Participants previously on glargine U300 or twice-daily basal insulin received a 50% 1-time additional dose regardless of HbA1c.
In another 16-week phase II trial, insulin icodec was tested in 205 insulin-naïve adults with T2D using 3 different titration algorithms alongside once-daily insulin glargine U100. The study found that once-weekly icodec was effective and well tolerated across all titration methods, with titration A (fasting blood glucose target 80-130 mg/dL; ±21 units/week) providing the optimal balance between glycemic control and hypoglycemia risk. 24 For individuals previously treated with insulin, introducing a 1-time additional dose of icodec when switching from daily basal insulins to once-weekly icodec maintained effective glycemic control. 26 This approach prevented an increase in fasting glucose levels during the transition and also did not heighten the risk of clinically significant hypoglycemia, compared with daily insulin glargine U100. 26 A recently published meta-analysis encompassing the 3 phase II insulin icodec trials reported no significant difference observed between icodec and insulin glargine U100 with regard to FPG, body weight change, hypoglycemia, or any adverse events evaluated. 28
Onwards, Phase III Trials
Drawing on the insights from the 3 phase II trials, the phase IIIa ONWARDS clinical trial program was designed to further explore the effectiveness and long-term safety of once-weekly icodec (700 units/mL; prefilled pen injector), extending the research initiated in the phase II trials. 27 All ONWARDS trials were phase IIIa, 2-arm, randomized, active-controlled, treat-to-target, multicenter, multinational trials with the primary objective of establishing noninferiority to the comparator through the primary outcome of percent change in HbA1c from baseline. 27 In treat-to-target trials, insulin doses are titrated to established fasting blood glucose targets for glycemic control.46,47 The unique study design elements for the ONWARDS phase IIIa trials are summarized in Table 2.
Efficacy
In insulin-naïve patients diagnosed with T2D participating in ONWARDS 1, 3, and 5, as well as in insulin-treated patients with T2D in ONWARDS 2, there was a greater estimated mean change in HbA1c from baseline with insulin icodec in comparison with insulin degludec, glargine U100, and glargine U300 (Figure 2).18-20,22 In addition, these analyses confirmed the attainment of both noninferiority and superiority with insulin icodec over the referenced comparators.18-20,22 Findings from the ONWARDS 4 trial demonstrated that insulin icodec exhibited noninferiority compared with insulin glargine U100 in individuals with T2D with prior basal-bolus insulin use. 21
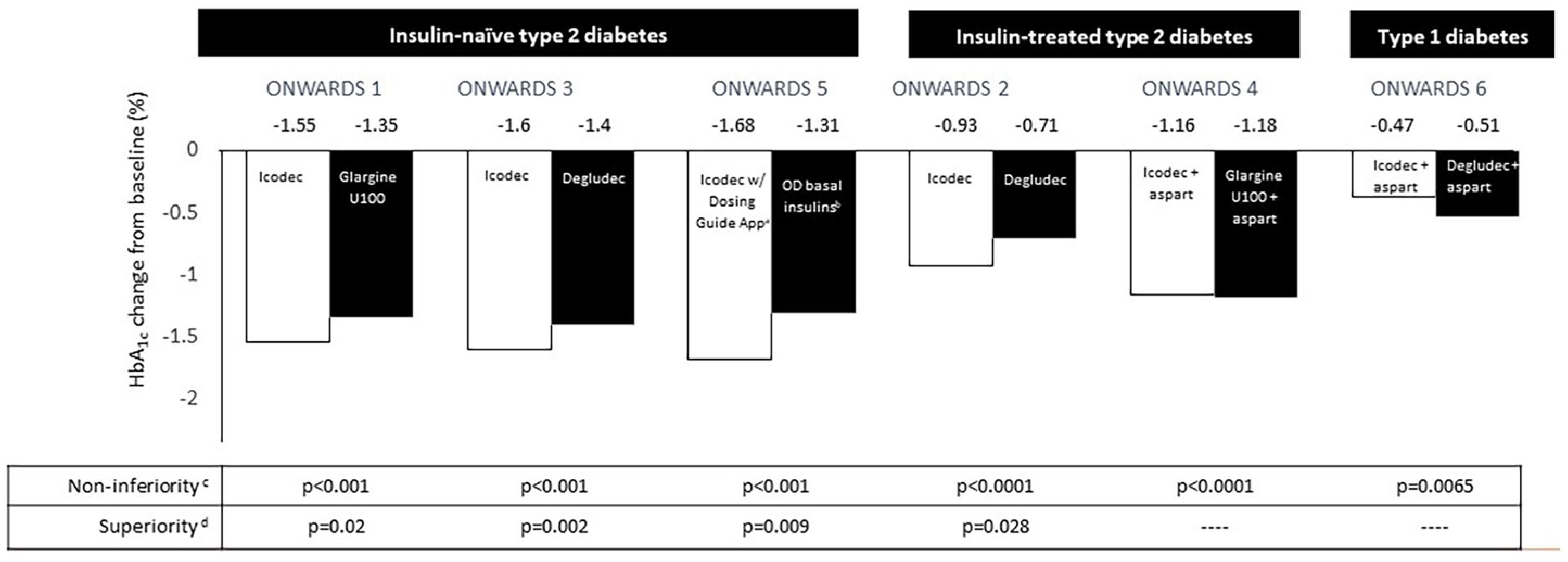
HbA1c percent change from baseline reported in ONWARDS trials.
Several recently published meta-analyses have reinforced the findings on glycemic management in T2D, indicating statistically significant improvements in reductions in HbA1c and TIR with insulin icodec compared with other basal insulins.28,48 A meta-analysis by Dutta et al 49 found that icodec significantly reduced HbA1c at 26 (4 studies, n = 2045) and 52 weeks (2 studies, n = 2069) compared with other basal insulins studied (26 weeks, –0.19%; 95% CI: –0.35 to −0.03; P = .02; 52 weeks, –0.28%; 95% CI, –0.45 to −0.12; P = .0008), while maintaining similar FPG at 26 weeks. Similar findings were reported in another meta-analysis by Shetty et al 50 of the ONWARDS 1 through 5 trials in those with T2D. In addition, higher proportions of patients achieved HbA1c levels below 7% (odds ratio [OR]: 1.51; 95% CI: 1.14-1.99; P = .004) and HbA1c achievement <7% without hypoglycemia (OR: 1.45; 95% CI: 1.26-1.67; P ≤ .00001) with insulin icodec. 50 In the ONWARDS 6 trial, which focused on individuals diagnosed with T1D, noninferiority of insulin icodec in comparison to insulin degludec was established with regard to reductions in HbA1c levels during the main phase of the study, baseline to week 26. 25 An estimated mean HbA1c reduction that was statistically significantly lower with icodec than degludec at the end of the extension phase (week 52) was observed (estimated treatment difference 0.17%; 95% CI: 0.02-0.31; P = .021). 25
Safety
In general, insulin therapy aimed at optimizing glycemic control may result in undesirable adverse events, including hypoglycemia and weight gain. Consequently, achieving a harmonious equilibrium between effective glycemic management and minimizing hypoglycemia and weight gain is essential. The manifestation of hypoglycemia within any diabetes therapeutic regimen is a major concern. Its ramifications extend from diminishing patient trust in their treatment regimen and adversely influencing their quality of life to contributing to heightened mortality rates and cognitive decline. 51
In the ONWARDS trials, hypoglycemic events were classified as alert level 1 (≥54 to <70 mg/dL, confirmed by blood glucose meter), clinically significant level 2 (<54 mg/dL, confirmed by blood glucose meter), or severe level 3 (associated with severe cognitive impairment requiring external assistance for recovery), with nocturnal hypoglycemic events accounting for those occurring between the hours of 00:01
Notably, similar rates of combined level 2 or level 3 hypoglycemia were observed in ONWARDS 1 through 5 in patients with T2D (Figure 3) for the entire study duration.18-22 A higher rate of combined level 2 or 3 hypoglycemia was not noted between insulin icodec and daily basal insulin comparators from baseline to week 26 in the ONWARDS 1, 2, 4, and 5 trials.18,19,21,22 In the ONWARDS 3 study, there was a statistically significant difference in hypoglycemia among those treated with icodec at week 26, but this was not evident at the conclusion of the study at week 31. 20 A recent participant-level post hoc meta-analysis of the ONWARDS 1 to 5 trials found a similar incidence of clinically significant hypoglycemia for the icodec and comparator groups and a low but significantly higher rate of events in the icodec group (estimated rate ratio 1.51 [95% CI 1.24, 1.85]); however, there were fewer severe hypoglycemic episodes among those treated with icodec. 52
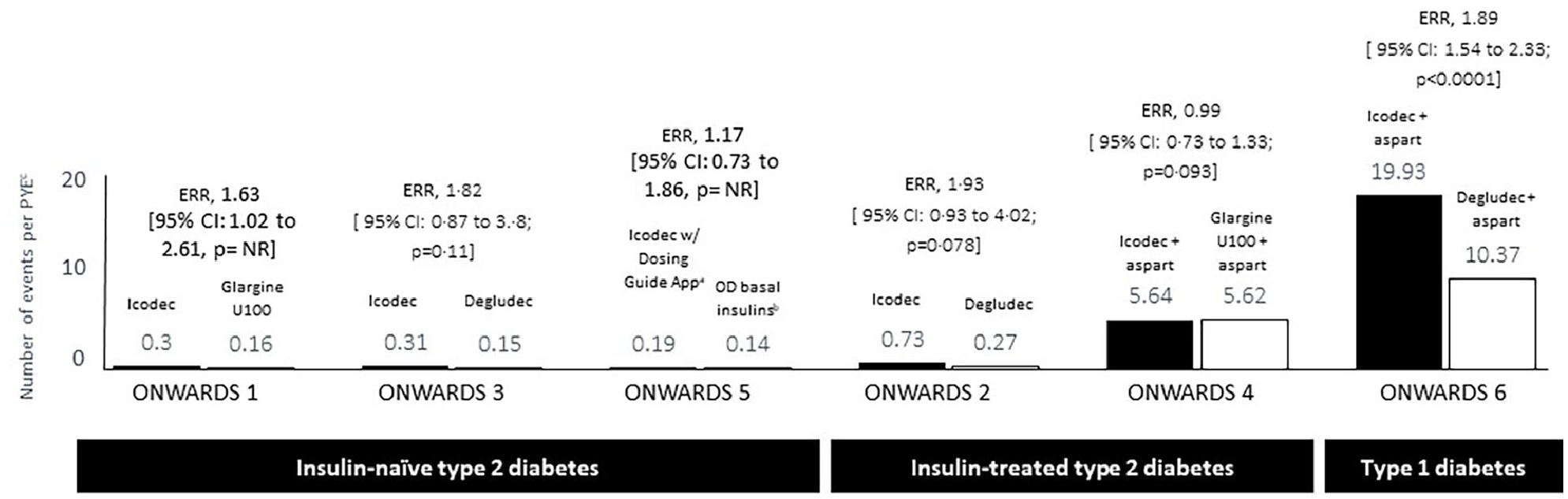
Estimated rate of level 2 or 3 hypoglycemia in ONWARDS trials.
Furthermore, meta-analyses present mixed findings regarding the hypoglycemia safety profile of insulin icodec in T2D.28,48 Although certain meta-analyses suggest that insulin icodec may have a comparable safety profile regarding hypoglycemia to other basal insulin analogs,28,49 one meta-analysis points to an increase of 24% (95% CI: 1.02, 1.50, P = .03) in the overall incidence of hypoglycemia associated with insulin icodec. 48 However, another recent meta-analysis by Shetty et al 50 reported that insulin icodec had a higher incidence of level 1 hypoglycemia compared with insulin degludec (OR: 1.56; 95% CI: 1.21-2.01, P = .0007) but not compared with insulin glargine U100 (OR: 1.12; 95% CI: 0.67-1.87; P = .67). The incidence of level 2 hypoglycemia was also higher with insulin icodec than with insulin degludec (OR: 2.03; 95% CI: 1.30-3.18; P = .002) and showed no significant difference when compared to insulin glargine U100 (OR: 0.89; 95% CI: 0.69-1.16; P = .39). The analysis found no significant difference in level 3 hypoglycemia incidence between insulin icodec and either insulin degludec (OR: 0.33; 95% CI: 0.01-8.22; P = .50) or glargine U100 (OR: 0.96; 95% CI: 0.17-5.48; P = .96). 50
The mixed findings in these meta-analyses can be attributed to differences in study design and overall quality. Mukhopadhyay et al 48 reported on level 1 and level 3 hypoglycemia events but did not report on clinically significant level 2 events. In addition, Mukhopadhyay et al 48 and Shetty et al 50 claimed to report the number of hypoglycemic events in the trials analyzed, but the meta-analyses figures represent the number of participants reporting events, instead. Event rates were reported at different trial time points across the meta-analyses, with some reporting the results at the end of the study duration and others at the end of the extension phase.50,48 Furthermore, each meta-analysis included different trials, contributing to the variability. Ribeiro et al 28 included only the 3 phase II trials, limiting its utility given the availability of phase IIIa trial data. Mukhopadhyay et al reported the results from the phase II trials and the ONWARDS 1 to 4 phase IIIa trials, excluding ONWARDS 5 and 6. In addition, Mukhopadhyay et al 48 included only one icodec arm (titration C) from the phase II study by Lingvay et al, which is the most aggressive dosing algorithm and was not used in the phase IIIa ONWARDS trials. Dutta et al 49 analyzed the phase II trials and data from the ONWARDS 1 to 5 trials, while Shetty et al 50 only included ONWARDS 1 to 5 phase IIIa trials. Therefore, the differences in design, hypoglycemia events reported, and trial time points considered may have contributed to the mixed findings regarding the hypoglycemia safety profile of insulin icodec.
Individuals using continuous glucose monitoring (CGM) as part of their diabetes management have well-defined therapeutic goals for glycemic metrics (Table 3). Time below range (TBR), a metric monitored to evaluate hypoglycemia, calculates the percentage of time when glycemic values fall within 2 specified intervals: level 1 hypoglycemia (glucose readings of 54-69 mg/dL or 3.0-3.8 mmol/L) and level 2 hypoglycemia (glucose readings below 54 mg/dL or <3.0 mmol/L).11,53 The target goals established to mitigate the risk of hypoglycemic events for combined TBR level 1 and level 2 is less than 4% of the time and for TBR level 2 under 1% of the time.11,53
Dosing, Titration Algorithm, and CGM Metrics Used in ONWARDS Trials for Patients With Type 1 and Type 2 Diabetes.

Abbreviations: BGM, blood glucose monitoring; CGM, continuous glucose monitoring; HbA1c, glycated hemoglobin; TAR, time above range; TBR, time below range; TIR, time in range.
For ONWARDS 1-5, weekly dose adjustment is based on 3 prebreakfast BGM values, measured 2 days prior to and on the day of titration. If any of the 3 prebreakfast BGM values were below the lower limit of the target range, titration was based on the lowest recorded value. If all 3 BGM values were above the lower limit of the target range, titration was based on the mean of the 3 measurements. For ONWARDS 6, weekly dose adjustments were based on 3 prebreakfast BGM values, measured 2 days prior to and on the day of titration, with titration based on the lowest recorded value.
Percentage of time was defined as 100 times the number of recorded measurements in a given range, divided by the total number of recorded measurements.
Level 1 hypoglycemia, 54-69 mg/dL (3.0-3.8 mmol/L); level 2 hypoglycemia, <54 mg/dL (<3.0 mmol/L); level 1 hyperglycemia, 181-250 mg/dL (10.1-13.9 mmol/L); level 2 hyperglycemia, >250 mg/dL (>13.9 mmol/L).
In the ONWARDS trials evaluating CGM, the goal of maintaining TBR level 1 below 4% was consistently achieved, aligning with the respective comparator daily basal insulin groups and exhibiting TBR level 1 of no more than 2.8% during weeks the 4 weeks of data analysis.18,19,21,25 Regarding TBR level 2, all ONWARDS trials evaluating CGM parameters achieved the target goal of below 1% except for ONWARDS 6, which reached the threshold of 1% at 26 weeks and was below 1% (0.8%) at 52 weeks.18,19,21,25 A recent meta-analysis reported that the percentages of time spent in TIR were similar between insulin icodec and the comparator groups in the 3 ONWARDS trials in participants with T2D that included CGM (mean difference 2.42%; 95% CI: 0.01-4.84; P = .05) but was significantly higher with insulin icodec than with insulin glargine U100 in the ONWARDS 1 trial (4.27% estimated between-group difference [95% CI, 1.92-6.62]; P < .001).18,50
In patients with T1D, the combined clinically significant or severe hypoglycemic incidence rate was higher for insulin icodec compared with daily insulin degludec (estimated rate ratio, 1.89; 95% CI: 1.54-2.33; P ≤ .0001) in the ONWARDS 6 trial, despite similar glycemic control. 25 Although the insulin icodec group experienced a higher frequency of severe hypoglycemic episodes, the incidence was low and the same in both the insulin icodec and insulin degludec cohorts (9 individuals [3%] in each arm) and could be related to the “treat-to-target” study design as the baseline mean HbA1c level was 7.6%. 25
Based on a published ONWARDS trial study protocol, mild hypoglycemia was managed by patients self-administering oral glucose or carbohydrate-containing products. Treatment for severe hypoglycemia required the administration of glucagon (0.5-1 mg) via intramuscular or subcutaneous injections by trained personnel or with intravenous glucose by medical professionals. If there was no improvement in severe hypoglycemia within 10 to 15 minutes following glucagon treatment, intravenous glucose was given, and patients who had lost consciousness were given oral carbohydrates upon regaining consciousness to mitigate the risk of relapse. 18 In the ONWARDS 6 trial, which included patients with insulin-treated T1D, severe hypoglycemic episodes were reported by 13 participants (at the end of the extension phase, week 57) in the insulin icodec arm (4.5% of 290) and 12 in the insulin degludec arm (4.1% of 292) with 8 individuals from each arm managing their symptoms with carbohydrate ingestion. Treatment with glucagon or intravenous glucose was necessary for 2.7% of those in the insulin icodec group (8 out of 290) and 1.4% in the insulin degludec group (4 out of 292). All patients who experienced a severe hypoglycemic episode reported feeling better after receiving treatment. 25
Weight gain frequently accompanies the use of basal insulin, attributed not just to the anabolic properties of insulin but also to behaviors like defensive eating that patients adopt to counteract hypoglycemia.51,54 In the ONWARDS trials, the data regarding weight changes when comparing insulin icodec with other basal insulins presented heterogeneous results.18-22,25 While all ONWARDS trials reported weight gain, the estimated mean change in body weight was not statistically significant except in ONWARDS 2. In the ONWARDS 2 trial, a statistically significant difference was observed, but this was primarily driven by an unexpected weight loss in the degludec arm (Table 4).18-22,25 A recent meta-analysis that included the ONWARDS 1 to 5 trials also found no significant difference in weight when comparing insulin icodec individually with glargine U100 (mean difference: 0.49 kg; 95% CI: –0.00 to 0.98; P = .05) and degludec (mean difference: 1.03 kg; 95% CI: –0.18 to 2.24; P = .10). 50 However, contrasting these findings, 2 recent meta-analyses found that insulin icodec led to marginal weight gain (0.61 kg [95 % CI: 0.38–0.84; P < .00001, at week 26]; to 0.64 kg [95% CI: 0.61-0.67]) compared with other once-daily basal insulins studied.49,48
Estimated Treatment Differences in Mean Body Weight in ONWARDS Trials.

Further long-term studies are needed to determine whether the use of insulin analogs like insulin icodec may be beneficial in managing insulin-related weight gain. Adding certain antihyperglycemic agents, such as GLP-1 RAs, to the treatment of patients with T2D can assist with weight control or even facilitate weight loss. The addition of a GLP-1 RA to a basal-bolus regimen has been associated with improvements in mean HbA1c, body weight, hypoglycemic events, and reductions in insulin requirements by decreasing the frequency of bolus injections needed.55,56 In the ONWARDS trials for T2D, baseline use of concomitant GLP-1 RAs varied: 16.9% and 18.7% in ONWARDS 1, 26% in both arms of ONWARDS 2, 21.8% and 16.3% in ONWARDS 3, 13% and 12% in ONWARDS 4, and 27.3% and 29.1% in ONWARDS 5, reflecting a consistent but varied use of these agents across the studies.18-22 Although the ONWARDS trials did not evaluate the effect of concomitant use of GLP-1 RA and insulin icodec, the 52-week COMBINE 3 phase IIIa trial demonstrated that a fixed-ratio combination of once-weekly basal insulin icodec and semaglutide was noninferior to daily insulin glargine U100 and insulin aspart in reducing HbA1c in those with T2D. 57 The icodec and semaglutide combination achieved a 1.47% reduction in HbA1c, comparable to −1.40% with the daily insulin regimen. It offered superior outcomes in weight management, with a −3.6 kg change versus a +3.2 kg gain and lower hypoglycemia rates, at 0.26 versus 2.18 events per patient-year of exposure. 57
Dosing and Administration
It is important to note that 1 unit of insulin icodec produces an equivalent glucose-lowering effect as 1 unit of the reference basal insulin. 27 When switching from once- or twice-daily basal insulin to once-weekly insulin icodec, the insulin icodec dose corresponds to 7 times the once-daily dose of the comparator basal insulin; the initial dose of insulin icodec needed can be calculated by multiplying the daily basal insulin dose by a factor of 7. 27
Across all ONWARDS trials for T2D, individuals who were insulin-naïve were started on a weekly subcutaneous dose of 70 units of insulin icodec. For individuals already being treated with basal insulin, the initial dose was 7 times their daily basal insulin dose pre-trial, with an additional 1-time 50% dose given (equivalent to 10.5 times the pretrial insulin dose; Table 3). 27
In the ONWARDS 6 trial for patients with T1D, the starting dose for insulin icodec was also 7-fold higher than the pretrial basal insulin dose. However, there was a special provision for the first dose, requiring a 1-time additional 50% increase, which equates to multiplying the daily basal insulin dose by a factor of 10.5 for the week 1 dose for individuals with T1D who had prior twice-daily basal insulin or once-daily insulin glargine U300 use or HbA1c of <8%. In those with HbA1c of ≥8%, the first week’s dose was 14 times the total basal insulin dose or 2 times the calculated weekly dose (100% 1-time increase). 25 Subsequently, the weekly dosage aligned with 7 times the previous daily basal insulin dose starting from week 2 onward (Table 3). 25
Since insulin icodec reaches a steady state around 3 to 4 weeks, discontinuation of a previous basal insulin could decompensate glycemic control. The higher initial dose is part of the dosing strategy to facilitate the transition to once-weekly dosing and ensure effective blood glucose control. 28 In a previous phase II study, transitioning from once-daily basal insulin to once-weekly insulin icodec therapy, participants with T2D who received an additional, 1-time dose of insulin icodec demonstrated effective glycemic control, no transient increase in FPG levels and no elevated risk of clinically significant hypoglycemia when compared with insulin glargine U100. 26 Insulin icodec injections may be administered at any time of the day on the same day each week, but administration day can be changed by up to 3 days (with a minimum of 4 days between injections) in patients with T2D. 27 If a dose of insulin icodec is missed within 3 days of the scheduled dosing date, patients should administer the missed dose as soon as possible. If the missed dose extends beyond 3 days, patients should wait to resume dosing on the next scheduled injection day. 20
Dose Titration Considerations
For diabetes management, achieving glycemic targets relies on glucose monitoring, which includes using capillary (finger stick) devices for blood glucose monitoring (BGM) or incorporating CGM alongside routine HbA1c testing. 11 Continuous glucose monitoring technology is recommended for those requiring insulin because it allows for continuous monitoring for insulin adjustments, reduces the need for frequent BGM, and has been shown to lower HbA1c and improve treatment satisfaction. 58 Clinical trials specifically highlight the benefits of CGM for those with T2D on basal insulin, showing increased TIR, improved HbA1c, and a reduction in hypoglycemia, including severe hypoglycemic events. 8 Individuals with T1D and T2D using insulin should self-monitor their blood glucose levels daily for dose adjustments and prevention of hypoglycemia, with prebreakfast levels primarily for basal insulin adjustments. 27 Additional BGM is recommended if hypoglycemia is suspected. 27 The prebreakfast BGM target values for dose adjustments in the ONWARDS trials were established in accordance with the American Diabetes Association guidelines, which recommend a preprandial plasma glucose target range of 80 to 130 mg/dL (4.4-7.2 mmol/L). 27
In the ONWARDS trials for patients with T2D, insulin icodec doses were adjusted weekly, with titration based on the analysis of 3 prebreakfast BGM values collected during the 2 days leading up to the titration day and on the titration day itself. 27 If any of the 3 prebreakfast BGM values fell below the lower limit of the established target range, the titration was determined based on the lowest recorded value. Conversely, if all 3 BGM values exceeded the lower limit of the target range, the titration was calculated based on the mean of these 3 measurements. Weekly insulin icodec dose adjustments were 20 units per week (Table 3). 27 For the ONWARDS 6 trial in patients with T1D, weekly dosage adjustments were based on 3 prebreakfast BGM values measured 2 days before titration and on the day of the titration. Titration was always based on the lowest BGM value. 25
Relevance to Patient Care and Clinical Practice
The introduction of insulin icodec represents a significant advancement in the treatment of T2D. It mirrors the impact of weekly GLP-1 RA therapy by reducing the need for daily insulin injections and impacting medication adherence, glycemic control, and overall quality of life for patients. In addition, weekly insulin administration is promising for those with T1D and T2D, offering a simplified approach to insulin management and greater dietary flexibility. However, further research is imperative to fully understand and optimize its use in T1D.
Insulin icodec has several advantages, as it may likely be the first once-weekly basal insulin available for the treatment of diabetes. Additional benefits include the infrequent dosing, which may be more advantageous when prescribed concomitantly with a once-weekly GLP-1 RA, allowing for ease of administration. Although adherence issues are always a concern for any therapy, in addition to being preferred by patients with T2D,59-61 once-weekly dosing of GLP-1 RAs has been associated with greater adherence than 1-daily treatments.16,17,62,63 Insulin icodec has shown to be an effective therapeutic agent in glycemic management, with demonstrated superiority to other basal insulins while maintaining comparable rates of severe hypoglycemia.
Disadvantages of insulin icodec include uncertainty in the safety profile regarding hypoglycemia, especially in T1D, as a slight increase in overall hypoglycemic events was observed in the ONWARDS 6 clinical trial. Additional disadvantages include the need for further studies to investigate areas with current knowledge gaps such as evaluating the long-term safety and effectiveness of insulin icodec in a real-world setting across a diverse patient population. Furthermore, understanding the change from daily to once-a-week dosing may pose a challenge to prescribers and increase the risk of a dosing error. It is critical for pharmacists in all areas of practice and all prescribers to understand how to dose icodec for safety and efficacy.
In addition, there is a need to investigate further the hypoglycemia risks associated with insulin icodec and potential underlying contributing factors. Future studies are warranted to explore the implications of insulin icodec in special patient populations such as older adults, those with comorbid conditions, specific ethnic groups, as well as patients with quickly changing insulin needs, such as those receiving glucocorticoids, and those experiencing hormonal shifts as seen in menstrual hormonal fluctuations, which often cause varying insulin requirements. This gap prevents a comparative analysis with the control regarding episode duration, treatment approaches, and symptom resolution. Additional research can help provide a comprehensive understanding of the advantages and limitations of insulin icodec in these distinct populations.
Conclusions
In conclusion, insulin icodec presents as a promising and safe option for managing diabetes, notably reducing the frequency of basal insulin injections, which is likely to be well-received by patients and healthcare professionals. Insulin icodec maintains consistent efficacy in glycemic management, with a possibility of superior efficacy compared to basal insulin analogs currently available for use. The occurrence of clinically significant hypoglycemia in patients with T2D using insulin icodec was comparable to that observed with daily basal insulin analogs, though more research is warranted in patients with T1D. Nonetheless, the development of a once-weekly insulin regimen that achieves effective glycemic control with comparable hypoglycemia rates represents a significant advancement in basal insulin therapy.
Footnotes
Acknowledgements
The authors thank Ladan Panahi, PharmD, BCPS, and Rebecca Hahn, MPH, of KJT Group, Inc., Rochester, NY, for providing medical writing support, which was funded by Novo Nordisk Inc., Plainsboro, NJ, in accordance with Good Publication Practice (GPP 2022) and International Committee of Medical Journal Editors (ICMJE 2023) guidelines.
Author Contributions
JG, DI, and CT were responsible for manuscript design, drafting/revising the manuscript, and reviewing/approving the final version for submission.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JG, DI, and CT have served as consultants for Novo Nordisk Inc. JG has served on the Speaker’s Bureau for Novo Nordisk, Eli Lilly, Sanofi, Xeris, CeQur, Abbott Diabetes, and Amgen. DI has served as a speaker for Eli Lilly, Sanofi, CeQur, Dexcom, Abbott Diabetes, Insulet, and Mannkind. CT has served on the Speaker’s Bureau for Novo Nordisk-Ozempic/Rybelsus.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Novo Nordisk Inc., Plainsboro, New Jersey.
