Abstract
Objective:
This review aims to systematically summarize the available data on efficacy and safety of therapeutic enoxaparin in obese patients and to identify gaps to guide future research.
Data Sources:
Medline and Embase were systematically searched for eligible studies (last searched December 20, 2023). Studies were included if they reported on therapeutic dosing regimens, adverse bleeding, thrombotic outcomes, or antifactor Xa (AFXa) monitoring in obese adult patients.
Study Selection and Data Extraction:
The systematic review management tool Covidence was used to manage the study selection and data extraction process. The reference list from eligible studies was screened to determine any additional eligible studies.
Data Synthesis:
Sixteen studies were included in the analysis. Studies used a variety of doses, indications, and study designs making comparison difficult. Twelve studies reported the incidence of thrombotic events (median = 1.3% [interquartile range [IQR] = 0.3%-2.3%]) and all studies reported the incidence of bleeding events (median = 5.7% [IQR = 2.4%-14.5%]). Two of the 8 studies analyzing the influence of weight/body mass index (BMI) or dose per kg on AFXa levels reported statistically significant results. One study concluded that BMI did not affect achievement of target AFXa levels. However, the second study found that dosing using actual body weight was an independent predictor of supratherapeutic AFXa levels in the obese population.
Relevance to Patient Care and Clinical Practice:
This is the first comprehensive review with a focus on therapeutic dosing of enoxaparin in obesity and has been conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) 2020 statement. Seven of the included studies were published since 2018 indicating that new evidence on this topic is emerging.
Conclusion:
There was inadequate evidence to support an optimal dosing strategy in obese patients due to the heterogeneity of the studies. The AFXa monitoring may be appropriate to guide dosing in this population. Further research is required to determine a suitable dosing regimen.
Introduction
Thromboembolic conditions are a group of diseases that affect the vascular system and are among the leading causes of death globally. 1 They affect the arterial vasculature in the form of ischemic stroke and ischemic heart disease and manifest in the venous vasculature as deep vein thrombosis (DVT) and pulmonary embolism (PE).2,3 The most recent Lancet Global Burden of Disease data found that ischemic heart disease was the leading cause of mortality worldwide and the second-leading cause of lost disability-adjusted life-years (DALY). 2 Although less extensively evaluated, there is substantial evidence indicating that Venous Thromboembolism (VTE) is also a major contributor to the global disease burden.3,4 In low- and middle-income countries, VTE associated with hospitalization was the leading cause of DALYs lost, and in countries with high-incomes, it was listed as the second-leading cause. 3 There are many factors which can contribute to and increase risk of venous and arterial thrombosis including hospitalization, COVID-19 infection, smoking, immobility, acquired and inherited thrombophilia, and obesity.4-7
Obesity, defined as a body mass index (BMI) of greater than or equal to 30 kg/m2, is a condition that has now reached epidemic proportions in the global population. In 2022, the World Health Organization (WHO) 8 estimated that the worldwide prevalence of obesity has more than doubled since 1990 with more than 2.5 billion adults classified as obese or overweight. Obese patients pose a clinical challenge in many ways, including in the dosing of medications. These patients have a different body composition compared to their nonobese counterparts: they have higher proportions of adipose tissue and larger absolute lean body masses.9,10 Obesity is associated with physiological changes that may alter drug pharmacokinetics such as increased cardiac output, increased hepatic and splanchnic blood flow, and glomerular hyperfiltration caused by dysregulation of hormone and cytokine secretion in adipose tissue.10-12 Medications, such as low molecular weight heparins (LMWHs), which are usually dosed based on total actual body weight therefore may not have the same effect in this population using weight-based dosing convention. In addition, obesity is a strong independent risk factor for thrombotic disorders as it causes chronic inflammation which activates prothrombotic signaling pathways in vascular cells and impairs fibrinolysis. 13
The LMWH are used as first-line parenteral anticoagulants in the treatment of venous and arterial thromboembolisms. 14 They rapidly and completely absorbed after subcutaneous administration and at therapeutic doses have a half-life of approximately 3 to 5 hours.14,15 They are hydrophilic in nature and are therefore retained in extracellular fluid and distributed according to lean body mass. 14 The LMWH binds to the naturally circulating anticoagulant antithrombin, potentiating its inhibition of thrombin and thereby inhibiting thrombin and antifactor Xa (AFXa), thus inhibiting coagulation. 15 Side effects include bleeding, bruising, thrombocytopenia, and hyperkalaemia. 14 They include enoxaparin, dalteparin, and nadroparin and are dosed according to actual body weight. 14 For treatment, dalteparin is dosed at 100 IU/kg twice daily (BID) up to a maximum of 18 000 IU, enoxaparin is dosed at 1 mg/kg BID or 1.5 mg/kg once daily (OD) with no maximum dose listed in product information and nadroparin is dosed at 86 IU/kg BID or 171 anti-Xa IU/kg OD up to a maximum daily dose of 17 100 IU. 14 The Thrombosis and Haemostasis Society of Australia and New Zealand, the American Society of Hematology (ASH), and the European Society for Vascular Surgery guidelines recommend weight-based dosing using total body weight but do not provide guidance on dose modification in obese populations.16-18 Owing to the altered body composition of obese patients and the pharmacokinetics of LMWH, dosing based on total body weight may lead to supratherapeutic levels of LMWH in the obese population.14,15
There appears to be limited research examining dosing of LMWH in obese patients, with the majority of published data pertaining to enoxaparin. Furthermore, the definition of obesity varies across these studies, often deviating from the definition used by WHO. In addition, while dalteparin and nadroparin product information specify a maximum therapeutic dose, the enoxaparin product information provides no guidance on an upper limit. Therefore, this review aimed to systematically summarize the available data on efficacy and safety of therapeutic enoxaparin in obese patients and to identify gaps to guide future research.
Methods
Search Strategy
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) Statement 2020 was used to conduct this systematic review. 19 The clinical question was formulated using the “Population, Intervention, Comparator and Outcome” (PICO) method. For this question, “P” represented obese adults with an indication for therapeutic anticoagulation; “I” represented treatment with therapeutic enoxaparin; “C” (the comparator) was not applicable; and “O” represented adverse bleeding, thrombotic outcomes during course of treatment, and implications of AFXa biochemical monitoring regarding dose adjustments.
A systematic electronic search of the Ovid Medline database and Embase was conducted on March 20, 2023 and was last updated December 20, 2023 to identify any additional relevant articles, reporting on original studies evaluating outcomes and/or monitoring of therapeutic enoxaparin in patients with obesity without language or publication date restrictions. The references of eligible studies were screened to determine any additional eligible studies. The search terms used were: (Enoxaparin [MeSH] OR enoxaparin.mp) AND (Obesity [MeSH] OR Overweight [MeSH] OR obese.mp)
Owing to the large number of results, the additional search term “treatment.mp” was included in only the Embase search to further refine results for title and abstract review.
Eligibility Criteria
Studies were included in this review if they met the following criteria: (1) original data, (2) inpatients and/or outpatients with a BMI ≥30 kg/m2 or body weight of ≥100 kg, (3) adults >18 years of age, (4) therapeutic dosing regimen of enoxaparin, and (5) evaluation of adverse bleeding and/or thrombotic outcomes, and/or (6) assessment of AFXa levels. The following exclusions were applied: (1) case reports with less than 5 cases, (2) studies on pregnant/postpartum patients, (3) guidelines, conference abstracts, or expert commentaries, (4) studies on pediatric populations, (5) nonhuman studies, and (6) studies evaluating surgical dose adjustments.
Data Extraction
The systematic review management tool Covidence was used to manage the review process. All results from Embase and Ovid Medline were loaded into Covidence, and all duplicates were removed. The title and abstract of each study were independently screened by 2 reviewers (SK and JSR), where disagreements over study inclusion or exclusion occurred, and this article was independently reviewed by a third reviewer (SN). The full-text screening was performed independently by 4 reviewers (MA, SK, JSR and SN) and studies meeting all inclusion and none of the exclusion criteria were included in this review.
Three researchers (SK, JSR, and SN) then performed data extraction in Excel and extracted the following data: first author, research design, country, sample size, methods of patient recruitment, clinical baseline characteristics, dosing strategy, indication, follow-up period, safety, and outcomes data. Data extraction was independently checked (MA). The dose of enoxaparin was expressed as “mg” in all included studies. Although some jurisdictions use “IU” or “IU of anti-Xa activity” as dosing units, for consistency this review will use milligrams; 1 mg is equivalent to 100 IU of anti-Xa activity. 18
Data Analysis
Efficacy
Progression of the thrombus, formation of a new thrombus, ischemic stroke, and AFXa levels are often used to measure efficacy of enoxaparin.18,20,21 The AFXa is a pharmacodynamic marker showing a linear correlation with enoxaparin exposure in vivo.18,20 The target AFXa range for therapeutic enoxaparin in management of VTE has been defined by consensus of experts rather than clinical validation as the published data for the ranges originates from studies which only examined arterial disease.22,23 The suggested peak range for treatment doses is 0.6 to 1.0 IU/mL or 0.5 to 1.2 IU/mL for BID dosing and 1.0 to 2.0 IU/mL for OD dosing.23-25 In addition, although the recommendations from national guidelines to monitor AFXa levels for treatment of VTE in subpopulations such as those with renal impairment is clear, in obese patients it continues to evolve. The European Society for Vascular Surgery 2021 guidelines on the management of venous thrombosis recommend monitoring to these ranges in “special situations” but does not specifically state whether obesity would be considered a special situation. 17 The 2019 Thrombosis and Haemostasis Society of Australia and New Zealand guidelines do not provide any statement regarding AFXa monitoring in obesity. 16 Finally, the ASH guideline for the management of VTE: optimal management of anticoagulation therapy, published in 2018, does provided the following recommendation with regard to obesity and AFXa levels “the ASH guideline panel suggests against using AFXa concentration monitoring to guide LMWH dose adjustment (conditional recommendation based on very low certainty in the evidence about effects).” 26
Safety
Bleeding is frequently used as an indicator of excess anticoagulation during enoxaparin therapy.20,21 Various anticoagulation bleeding scoring systems are often used to assess the degree of bleeding. 27 These include the Thrombolysis in Myocardial Infarction (TIMI), Global Use of Strategies to Open Occluded Arteries (GUSTO), International Society on Thrombosis and Haemostasis (ISTH), and the Bleeding Academic Research Consortium (BARC) scales.27-29 Although these scales have commonalities, they were designed for use in different settings. For example, the ISTH scale was designed for VTE patients on long-term anticoagulation, whereas TIMI and GUSTO were designed for use in patients with ST elevated myocardial infarction (STEMI) post fibrinolytic therapy.28,29 Information on bleeding including decrease in hemoglobin, bruising, hematoma, and hemorrhage were therefore extracted from the included studies. In addition, peak AFXa levels are also used to assess safety of enoxaparin with the TIMI 11A trial demonstrating that peak anti-Xa concentrations greater than 1.0 IU/mL increased the incidence of bleeding. 22
Risk Assessment (Bias)
To determine the reliability and overall risk of bias of the included studies the Risk Of Bias In Non-randomized Studies–of Interventions (RoBINS-I) tool and the Cochrane risk-of-bias tool for randomized trials (RoB 2) were used.30,31 Coadministration of medications that affect bleeding risk (antiplatelets, aspirin, direct oral anticoagulants, glycoprotein IIb/IIIa inhibitors, non–steroidal anti-inflammatories, tissue plasminogen activator, and warfarin) was considered an important cointervention. Baseline serum creatinine levels and comorbidities that increase risk of thrombosis and bleeding (thrombophilic conditions, cancer, cirrhosis, history of gastrointestinal bleeding, and history of hemorrhagic stroke) were considered confounders. Risk of bias in individual studies was assessed by MA.
Results
The search in Ovid Medline and Embase resulted in 644 studies after removal of duplicates. After title and abstract screening and full-text review, 14 studies met the eligibility criteria (see Figure 1). Two additional studies were retrieved through screening the references of the included studies resulting in a total of 16 studies included in the analysis. The final search was performed on December 20, 2023 and while several new papers were identified, all were subsequently excluded. The PRISMA Chart (Figure 1) shows the selection process. The eligible studies included 21 431 participants with a median number of 118 participants (range = 26-19 061).32-47 There were 12 retrospective cohort studies,32-42 3 prospective cohort studies,43-45,47 and 1 randomized controlled trial. 46 The individual study characteristics are presented in Table 1, and the risk of bias is presented in Tables 2 and 3.
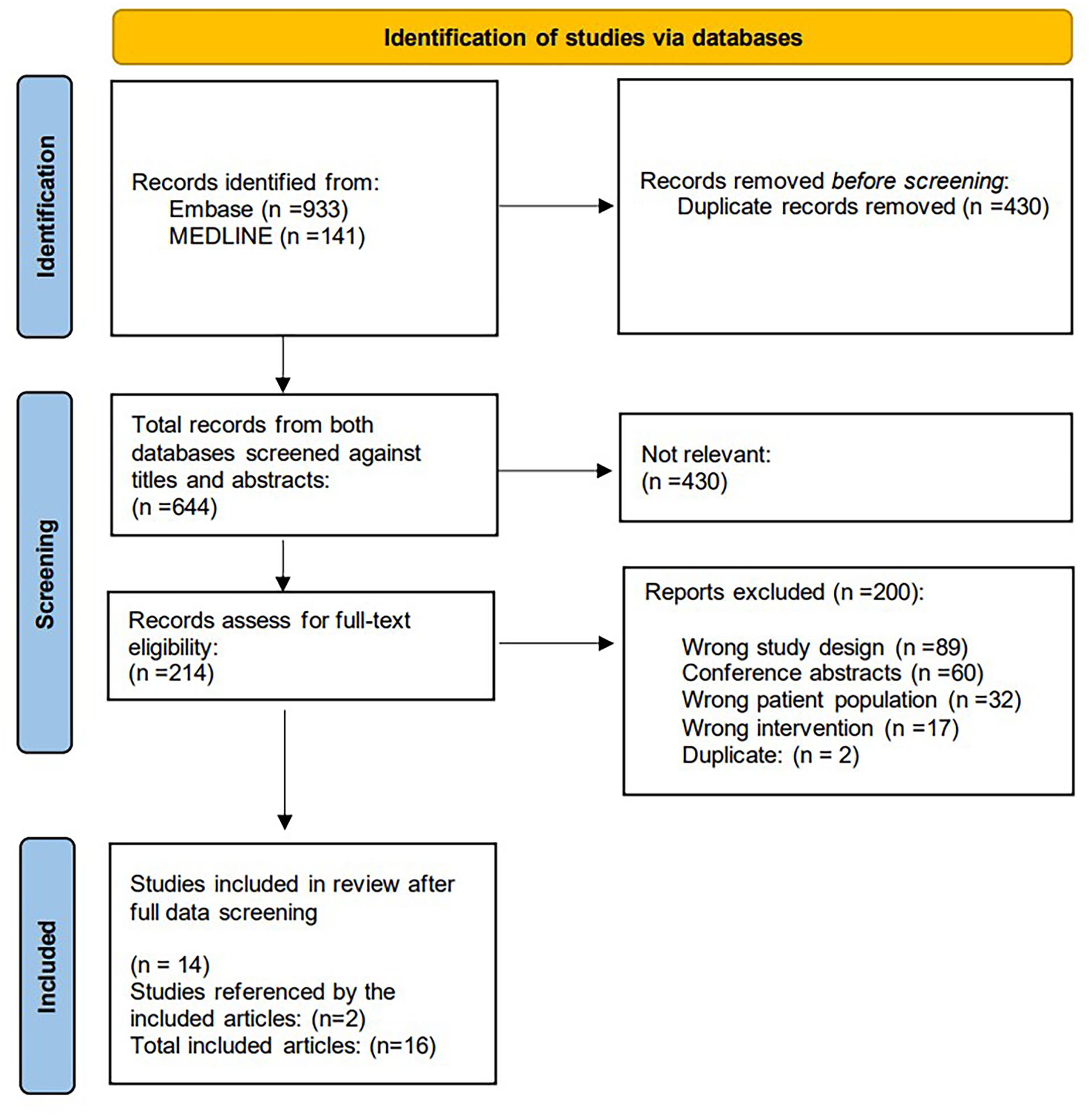
Flowchart of the Inclusion Criteria.

BMI = body mass index, BW = body weight, N.S. = not specified, N.R = not reported, AF = atrial fibrillation, MI = myocardial infarction, DVT = deep vein thrombosis, PE = pulmonary embolism, NSTEMI = non–ST elevated myocardial infarction, CABG = coronary artery bypass graft, BID = twice a day, OD = Daily, AFXa = antifactor Xa, SD = standard deviation, IQR = interquartile range, RCT = randomized control trial, Hb = hemoglobin, AOR = adjusted odds ratio.
Mean ± standard deviation or mean (range), unless otherwise reported.

L = low risk; ? = some concerns.
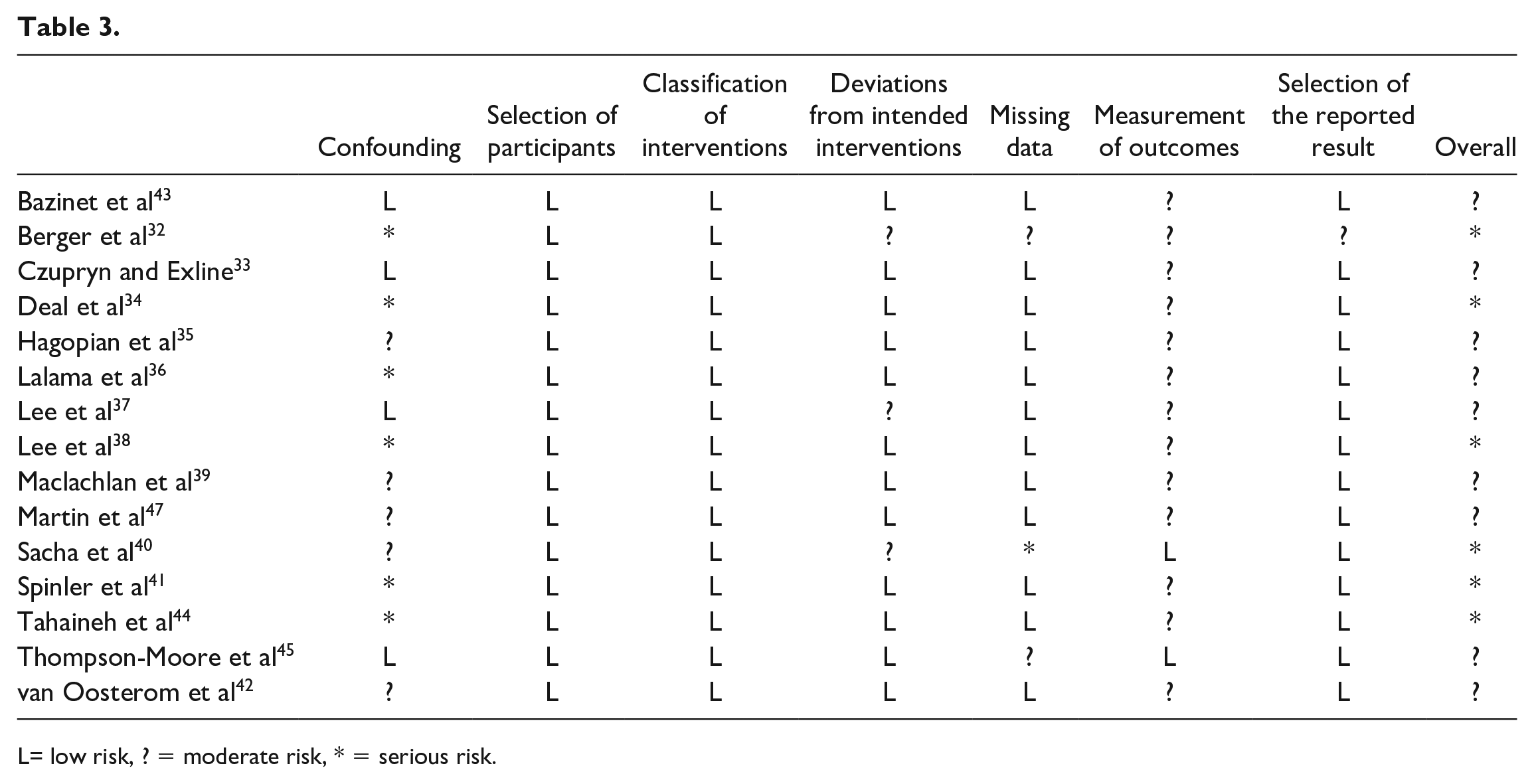
L= low risk, ? = moderate risk, * = serious risk.
The definition of obesity varied among the included studies. Four studies defined obesity as total body weight greater than a specific threshold,33,39,40,42 6 studies defined obesity as a BMI over a specific threshold32,34,35,43,44,46, whereas the remaining 6 studies used a combination of both parameters.36-38,4145,47 Although all included studies were conducted on inpatients, study design varied with 7 studies comparing therapeutic dosing in obese patients to nonobese patients35,37,39-41,47 and 9 studies examining dosing only in obese participants.32-34,36,38,42,44-46 Three of these studies compared obese participants dosed using conventional weight–based dosing to modified or reduced dosing strategies,33,43,46 whereas the remaining 6 studies were single arm with no intervention.32,34,36,38,42,44 Studies also differed in their inclusion criteria regarding the indication of therapeutic enoxaparin and the inclusion of renally impaired patients. Two studies44,45 did not provide information on indication and 10 studies (62.5%) listed “Atrial Fibrillation (AF)” as an indication, which can be considered a prophylactic indication.32-38,40,43,46 Out of the 5 studies34-36,38,47 that included renally impaired patients, only 1 study did not adjust the dose according to renal function. 34 Length of follow-up also varied greatly between studies. Four studies did not specify a follow-up period37,38,40,46 and the remaining studies measured the duration of follow-up using a variety of different definitions including number of days after cessation of therapy, days after admission to hospital, or days after discharge from hospital.32-36,39,41-45,47 These follow-up period were therefore not considered comparable.
Efficacy—Thrombotic Events
No specific information regarding formation of a new thrombus, progression of the existing thrombus or ischemic stroke was reported by 25% (n = 4) of the included studies.38,40,41,43 Thrombotic events were reported in 9 studies,32,33,35-37,39,42,44,47 whereas 3 studies reported that no thrombotic events occurred during the study or follow-up period.34,45,46 Across these studies, enoxaparin was prescribed for both arterial and venous indications. Of the 9 studies that reported these events,32-37,39,42,44-47 the incidence of thrombotic events was low (~1.8%) with the highest incidence of 3.8% in the small prospective cohort study by Tahaineh et al. 44 A total of 5 patients across 4 studies reported thrombotic events and had a corresponding AFXa level.36,37,42,44 These events all occurred in obese patients with 1 AFXa level reported as “nontherapeutic,” 3 levels reported as within therapeutic range, and 1 level reported as supratherapeutic (level = 1.13 IU/mL).36,37,42,44
Four of the studies33,35,37,39 that reported thrombotic events conducted statistical analysis with only 1 study, by Maclachlan et al, 39 reporting a statistically significant result. This study examined the difference between obese and nonobese groups who were dosed using 1 mg/kg BID based on actual body weight for treatment of VTE. 39 The 3 thrombotic events that occurred in this study occurred in the group which had a body weight of less than 100 kg which was a statistically significant difference when compared to the zero incidents in the obese participants group (weight >100 kg) (P = 0.049). 39 It should be noted that there were statistically significant differences in the baseline characteristics between the 2 groups with the healthy weight group being of older age, having a higher incidence of malignancy, renal impairment, and a higher incidence of provoked VTE as the indication for treatment. 39 In addition, one of the patients who experienced a thrombotic event had their dose reduced to prophylactic levels due to a bleed when on a therapeutic dose. 39 The 3 other studies that conducted statistical analysis examined enoxaparin dosing for both arterial and venous indications.33,35,37 Czupryn and Exline, 33 who examined dosing in patients weighing more than 120 kg, reported 5 thrombotic events, 2 were ischemic strokes, and 3 were VTE events. It reported that there was no statistically significant difference between the incidence of ischemic stroke or VTE in obese patients dosed with less than 90% of the Food and Drug Administration (FDA) approved dose when compared to those dosed with greater than 90% of the FDA approved dose (P = 0.52 and P = 0.60, respectively). Hagopian et al 35 compared patients with BMIs greater than 40 kg/m2 to patients with BMIs of less than 40 kg/m2 and reported 2 events in the obese group and 7 events in the nonobese group which was not statistically significant (P = 0.72). The final study to conduct statistical analysis, Lee et al, 37 determined that there was no statistically significant difference between the 1 thrombotic event that occurred in the BMI 40 to 50 kg/m2 group and the other BMI groups included in the study. The authors attributed this event to heparin-induced thrombocytopenia. 37
Safety—Bleeding Events
All 16 studies reported the incidence of bleeding events during the study and follow-up period.32-47 Studies included a variety of definitions and scales to measure bleeding. Six studies created their own unique definitions to measure and report the severity of bleeding events.34,35,38,41,45,47 Of the remaining studies, 2 used TIMI,33,40 3 used ISTH,32,37,39 1 used GUSTO, 36 1 used a modified version of the Warfarin-Optimized Outpatient Follow-up Study Classification 43 and 3 studies did not report how bleeding was defined.42,4446 Three of the included studies reported that no bleeding events occurred.38,44,46
Of those studies that did report bleeding events, 7 studies conducted statistical analysis to determine whether these events were associated with either weight/BMI35,37,39,47 or dose per kg.33,35,41,45,47 Three studies reported statistically significant results; however, it should be noted that 2 of these studies used unique definitions to measure and categorize bleeding events.35,41 Spinler et al, 41 who examined dosing of enoxaparin in obese patients with non–ST-segment elevation acute coronary syndromes (NSTEACS), determined that patients who weighed more than 150 kg who received a total daily dose of enoxaparin dosed at 1 mg/kg based on actual body weight had a greater than 2 times increased risk of bleeding when compared those who had their dose reduced (adjusted odds ratio [AOR] = 2.42; 95% confidence interval [CI] = 0.70-8.37). This study included the following adjusted variables in the model: age, sex, systolic blood pressure, history of renal insufficiency, and signs of heart failure. 41 Although this retrospective cohort study had a very large number of enrolled participants (n = 19 061), only 175 participants weighed more than 150 kg and 20% of these received the 1 mg/kg weight-based dose. 41 Maclachlan et al 39 only examined VTE indications and had 13 bleeding events and 6 of these events were classified as major bleeds and all events occurred in the healthy weight group. The authors concluded that there was a statistically significant difference in major bleeding events between obese and nonobese groups who received the 1 mg/kg dose (P = 0.003). 39 Hagopian et al 35 which compared obese and nonobese groups prescribed enoxaparin for both arterial and venous indications conducted a multivariate analysis to determine which factors were associated with an increase in bleeding events. This analysis concluded that morbid obesity was not an associated factor for bleeding but interestingly did find a statistically significant association between final enoxaparin doses of less than 0.9 mg/kg per dose and bleeding events (AOR = 2.35; 95% CI = 1.01 to 5.47, P = 0.04). 35 The authors proposed that this may be due to doses being reduced by prescribers due to perceived risk of bleeding in these patients. 35 In addition, durations of therapy beyond 48 hours (AOR = 2.42; 95% CI = 1.35 to 4.33; P < 0.01) and female gender (AOR = 2.05; 95% CI = 1.12 to 3.73; P = 0.02) also increased risk of bleeding. 35 This study concluded that the proportion of patients who experienced a bleeding event was not different between patients whose BMI was greater than or less than 40 kg/m2 (P = 0.30). 35
Monitoring: Antifactor Xa Levels
Fourteen studies recorded AFXa levels and specified a target range.32,34-40,42-47 Eleven studies defined separate target ranges for BID and OD dosing,32,34,35,37,39,40,42-44,46,47 whereas 3 studies used the same target regardless of dosing frequency.36,38,45 The most common target range was 0.5 to 1.0 IU/mL for BID enoxaparin dosing, this was used by 8 studies.32,34,35,37,39,40,42 Eight studies conducted statistical analysis to determine the influence of weight/BMI34,37-39,43,45 or dose per kg34,44,45,46 on AFXa levels with 2 studies reporting statistically significant results.43,45 Both studies examined enoxaparin treatment for both arterial and venous indications.43,45 Bazinet et al, 43 who included patients with BMIs greater than 18 kg/m2, found that obese patients (BMI >30 kg/m2) dosed with 1 mg/kg BID or 1.5 mg/kg OD had comparable AFXa levels when compared to their healthy weight counterparts (see Table 1). This study conducted multivariate logistic regression which determined that BMI was not a variable that affected achievement of target therapeutic AFXa activity for OD or BID dosing (odds ratio = 0.95; 95% CI = 0.521 to 1.73). 43 This study also determined that AFXa increases by 0.01 IU/mL for every increase in 1 kg/m2 (95% CI = 0.002 to 0.017); however, this increase was insufficient to result in supratherapeutic AFXa levels. 43 Thompson-Moore et al 45 conducted a prospective cohort study which examined patients with a BMI >40 kg/m2 or an actual body weight >140 kg to determine what dose of enoxaparin was required to achieve target AFXa levels in this population. This study used univariate logistic regression to determine that enoxaparin dosed using actual body weight was an independent predictor of supratherapeutic AFXa levels, odds ratio = 0.21 for enoxaparin dose at <0.95 mg/kg BID compared to ≥0.95 mg/kg BID (95% CI = 0.05 to 0.84, P = 0.02). 45 The median dose of enoxaparin that resulted in AFXa levels within the target range or above the target range at steady state was 0.83 and 0.98 mg/kg BID (−0.11; 95% CI = −0.20 to −0.01, P = 0.02), respectively.
Three studies conducted statistical analysis on the association between AFXa levels and bleeding risk with only Deal et al reporting a statistically significant result.34,45,47 Deal et al 34 examined 26 obese patients with BMI >40 kg/m2 who received enoxaparin doses of 1 mg/kg BID with doses capped at 150 mg for the following indications: VTE, AF, mechanical heart valve, or cerebral vascular accident (CVA). Six bleeding events occurred with 4 of these patients having AFXa levels that were supratherapeutic (>1.0 IU/mL) and 2 having subtherapeutic levels (<0.5 IU/mL). 34 This study reported that there was a statistically significant difference between the occurrence of bleeding events in patients who had supratherapeutic levels and those who had levels within the target range of 0.5-1.0 IU/mL (P = 0.033). 34 The enoxaparin doses of these patients who experienced a bleeding event ranged from 0.67 to 0.9 mg/kg based on actual body weight. 34
Reporting bias
Risk of bias for each study was assessed using the Cochrane ROBINS-I assessment tool and the revised Cochrane RoB 2 (see Tables 2 and 3). We concluded that the single randomized controlled trial 46 had some concerns regarding risk of bias. The observations studies were assessed with the ROBINS-I tool, and 9 studies were classified as moderate risk of bias and 5 studies as serious risk of bias.
Discussion
This systematic review summarized the available data on efficacy and safety of therapeutic enoxaparin in obese patients. Sixteen studies were included and there was a high degree of heterogenicity between the studies which made it difficult to aggregate the results. Although bleeding events were considered an endpoint by all studies, only 9 studies reported thrombotic events and 14 studies assessed monitoring of AFXa levels. There was a high degree of variability regarding the duration of follow-up periods between studies which may have resulted in the low number of thrombotic and bleeding events reported. In addition, many studies included a mix of arterial and venous indications further complicating data interpretation as the duration of treatment for each indication is significantly different. 18
This review found that the incidence of thrombotic events reported in the included studies was low. None of the studies which measured this endpoint performed power calculations to determine whether an association exists between subtherapeutic AFXa levels or dose and efficacy of enoxaparin treatment measured as formation of a new thrombus, progression of the existing thrombus, or ischemic stroke. The study by Maclachlan et al, 39 who only examined VTE indications, was the only study that demonstrated a statistically significant difference in thrombotic events between obese and nonobese patients who received 1 mg/kg BID dosing based on actual body weight. These events only occurred in the nonobese participants within the 30-day follow-up period, and by this time, only 67% of nonobese patients were still prescribed enoxaparin. 39 In addition, this study did not specify if the patients who experienced thrombus extension or recurrence were still prescribed enoxaparin at the time of the event, and this therefore limits interpretation of the results.
Furthermore, none of the included studies were able to determine a statistically significant difference between efficacy with standard dosing and reduced dosing, including dose capping, in the obese population. This could be attributed to the small number of participants in each study as well as the variety of indications and durations of treatment. These findings are consistent with studies which examine prophylactic enoxaparin dosing in the obese populations which also report low incidence of VTE and thus were unable to demonstrate an association between dosing strategies and efficacy.48-55 Historical data suggest that the incidence of VTE is 2.8% in the obese hospitalized population. 56 In the included studies in this literature review, treatment failure in obese patients on therapeutic enoxaparin was 1.7%. Therefore, to detect a 40% difference in efficacy with 80% power, a prospective study would require a sample size estimated to be 2853 participants per arm. Obtaining a sample size this large may be difficult or time-consuming as recent guidance from ISTH has indicated that the use of rivaroxaban or apixaban, direct-acting oral anticoagulants (DOACs), at standard doses is appropriate anticoagulant options regardless of high BMI and weight. 57 This is therefore likely to change prescription patterns and reduce the number of patients prescribed treatment enoxaparin. Alternatively, to overcome the issues of varying dosing strategies, various indications, and small sample size used between studies, it would be beneficial to create a larger data set using the individual deidentified data from each study. This would enable flexible and more detailed analysis of results which could provide greater insight into the effective dose of therapeutic enoxaparin in this population than could be obtained through reviewing aggregated data. 58 The individual participant data collected should include key information such as dose received, indication, patient outcomes, AFXa levels, and baseline characteristics such as gender, age, serum creatinine, and weight.
Bleeding events were considered a clinical endpoint by all included studies; however, a variety of tools were used to define these events. While 7 studies used validated tools,32,33,36,37,39,40,43 9 studies either created their own definition or did not report how bleeding was defined.34,35,38,41,42,44-47 This made comparison of this outcome between studies difficult. Only 3 studies had statistically significant results for bleeding; however, the results of each study were conflicting and the included indications for enoxaparin treatment varied for each study.35,39,41 Spinler et al 41 only examined arterial indications and demonstrated that obese patients were at an increased risk of bleeding when dosed with a total dose of 1 mg/kg based on actual body weight. Conversely, Hagopian et al 35 examined both arterial and venous indications and concluded that morbid obesity was not an associated factor that increased risk of bleeding. The final study, Maclachlan et al, 39 only included VTE indications, and concluded that nonobese patients were at an increased risk of bleeding compared to obese patients when dosed with 1 mg/kg BID on actual body weight. However, Maclachlan et al 39 acknowledged that there were significant clinical differences between the obese and nonobese patients included in their study which resulted in the nonobese group having a higher risk of bleeding. Furthermore, all 4 studies which conducted statistical analysis on this outcome but did not find statistically significant results concluded that their studies were too small or inadequately powered to detect a difference in clinical events.33,37,45,47 These conclusions are similar to those found in studies examining prophylactic enoxaparin doses in obesity which also found that the small number of participants in each study limited interpretation of outcome results.48-53,55,58 This further supports the suggestion that an individual participant data review would be beneficial as this type of review would not only enable pooling of patient data but could also enable standardization of how bleeding events were defined and categorized. 56
The majority of studies which assessed AFXa levels focused on assessing the probability of target attainment as a percentage of patients who achieved a level within the specified therapeutic range. Only 3 studies performed power calculations to determine a difference between dose and AFXa levels and all were underpowered.44,45,46 Although the target range for AFXa levels did vary between studies, 11 studies specified separate target ranges for BID and OD dosing which only varied by 0.1 IU/mL and were therefore considered comparable.32,34,35,37,39,40,42-44,46,47 The 2 studies which examined the influence of weight/BMI on AFXa levels and had statistically significant results included a mix of venous and arterial indications and came to different conclusions.43,45 Bazinet et al 43 concluded that obesity was not a variable that affected achievement of target therapeutic AFXa activity for enoxaparin dosed on actual body weight, whereas Thompson-Moore et al 45 concluded that dosing using actual body weight was an independent predictor of supratherapeutic AFXa levels in morbidly obese patients. The difference in these results may be due to the different minimum BMIs included in the studies. Bazinet et al 43 defined obesity as a BMI of >30 kg/m2, whereas Thompson-Moore et al 45 only included patients with a BMI of >40 kg/m2 or a weight >140 kg. This could potentially indicate that weight-based dosing is appropriate in patients with BMIs <40 kg/m2; however, further studies would be required to confirm this result.
Only 1 study, Deal et al 34 was able to demonstrate a statistically significant association between supratherapeutic AFXa levels and harm of intervention, measured as bleeding events, in the obese population (BMI >40 kg/m2). However, it should be noted that this was a small study with only 6 participants experiencing bleeds, and only 4 of these patients having a supratherapeutic level. Furthermore, the doses of enoxaparin these patients received varied greatly and all patients who experienced a bleed were dosed at less than 1 mg/kg BID based on actual body weight. 34
The aim of this systematic review was to examine therapeutic enoxaparin dosing in the obese population. Having a focused aim could be considered a limitation of this study as including thromboprophylaxis studies may have provided additional information regarding safety outcomes. However, while expanding the search to include other LMWH was considered to increase the scope of this systematic review, previous reviews examining this topic have found less than 5 studies examining the other agents.59,60 It was therefore decided that a focus on enoxaparin would provide more homogenous data and allow for more stringent analysis and robust results. Another potential limitation of this study was that the included studies were limited to adult populations. However, expanding the search to include pediatric populations would have further increased the heterogeneity of studies.
Relevance to Patient Care and Clinical Practice
The previous reviews on this topic have a broad focus with all examining both treatment and prophylaxis of low molecular weight heparins in obesity.59-62 This is the first comprehensive review with a focus on therapeutic dosing of enoxaparin in obesity and as such this focus has allowed for greater in-depth analysis including review of AFXa monitoring with clinical outcomes. It includes more studies than any previous review with 7 additional studies which were published since 2018. This indicates that new evidence on this topic is emerging.
While the results of this review may be used as a tool by clinicians, this study ultimately emphasizes the lack of consistent data available and has highlighted that several opportunities for future research into this topic exist. Only 6 of the included studies had statistically significant results with some reaching contradictory conclusions. While the studies were unable to determine an ideal dosing regimen, they do provide weak evidence that reduced dosing strategies may not increase the incidence of treatment failure in the obese population. Regarding safety outcomes, the studies were only able to provide limited evidence that conventional weight–based dosing increases the risk of bleeding in obesity. In addition, they were unable to clearly demonstrate that obesity was an independent variable that affected the achievement of target therapeutic AFXa levels; however, there was some evidence that morbid obesity (BMI >40 kg/m2) did increase the risk of supratherapeutic levels. Therefore, there is some evidence to support dose reduction in the obese population to reduce the risk of bleeding especially in those with a BMI >40 kg/m2. The key piece linking an ideal dosing regimen to efficacy, safety outcomes, and target AFXa levels remains unanswered. Future research should be conducted with a large enough sample size to answer this question.
Summary
This review provides a picture of heterogeneous therapeutic enoxaparin dosing strategies and attainment of AFXa target ranges. As a result, it is unable to provide specific recommendations regarding dosing, efficacy or safety in the obese population, and can only provide cautious guidance. Owing to the weak level of evidence, it may be appropriate to reduce the therapeutic dosage of enoxaparin in morbidly obese patients (BMI >40 kg/m2), and it may be appropriate to monitor AFXa levels to guide dose adjustment in obese and morbidly obese patients (BMI >30 kg/m2). An individual participant data review or a large prospective study could provide additional information to define an appropriate enoxaparin dosing regimen in this population.
Footnotes
Correction (July 2024):
Flow-Chart in the Figure 1 is updated.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by an Australian Government Research Training Program (RTP).
