Abstract
Objective:
The objective of this review is to discuss acid-base physiology, describe the essential steps for interpreting an arterial blood gas and relevant laboratory tests, and review the 4 distinct types of acid-base disorders.
Data Sources:
A comprehensive literature search and resultant bibliography review of PubMed from inception through March 7, 2023.
Study Selection and Data Extraction:
Relevant English-language articles were extracted and evaluated.
Data Synthesis:
Critically ill patients are prone to significant acid-base disorders that can adversely affect clinical outcomes. Assessing these acid-base abnormalities can be complex because of dynamic aberrations in plasma proteins, electrolytes, extracellular volume, concomitant therapies, and use of mechanical ventilation. This article provides a systematic approach to acid-base abnormalities which is necessary to facilitate prompt identification of acid-base disturbances and prevent untoward morbidity and mortality.
Relevance to Patient Care and Clinical Practice:
Many acid-base disorders result from medication therapy or are treated with medications. Pharmacists are uniquely poised as the medication experts on the multidisciplinary team to assist with acid-base assessments in the context of pharmacotherapy.
Conclusion:
The use of a systematic approach to address acid-base disorders can be performed by all pharmacists to improve pharmacotherapy and optimize patient outcomes.
Introduction
Acid-base abnormalities are common in the intensive care unit (ICU) and can substantially impact patient outcomes. While most acid-base disorders are mild and self-limiting, some can be severe, leading to adverse consequences and even mortality. The etiology of acid-base disorders is multifactorial and can include complex disease processes like shock, invasive mechanical ventilation strategies, fluid resuscitation and electrolyte concentrations, organ failure, intoxications, and pharmacotherapy. Pharmacists play an essential role as the medication expert on the multidisciplinary team and can provide important contributions in the management of acid-base disorders (Table 1). For example, many commonly used medications in the ICU can lead to acid-base disorders, either directly via their mechanism of action or as an over-extension of their pharmacologic effect. Many acid-base disorders are iatrogenic which can be minimized or prevented through careful monitoring and evaluation of medical therapies. Finally, acid-base disorders are often treated with medications, thereby highlighting the importance of routine pharmacist assessment. This narrative review provides a practical foundation for new practitioners, residents, or students, who practice in the ICU regarding acid-base physiology, essential steps for interpreting an arterial blood gas and a review of the 4 distinct types of disorders. For a complete review on treatment of acid-base disorders, the reader is referred elsewhere. 1
Importance of Pharmacist’s Assessment of Acid-Base Status.
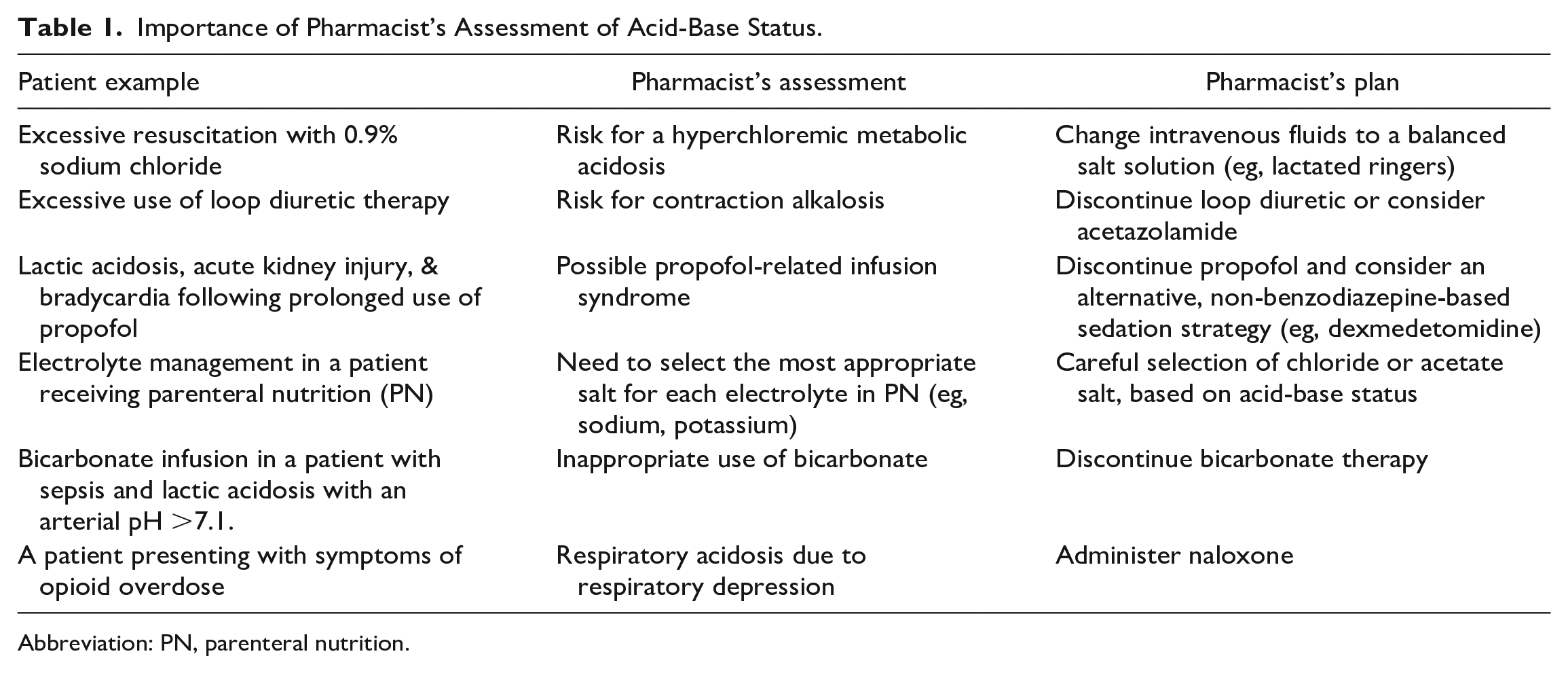
Abbreviation: PN, parenteral nutrition.
Data Sources
A comprehensive literature search was conducted using PubMed from inception through March 7, 2023. Articles were screened for content, and bibliographies were reviewed to identify any articles that may have been missed by the original literature search. Animal studies were excluded along with articles that were not available in English.
Acid-Base Physiology
Acid-base homeostasis is an essential requirement for normal cellular and metabolic function. An acid is a substance that can donate a proton; a base is a substance that can accept a proton. Every acid has a corresponding base, and every base has a corresponding acid. Common acid-base pairs are carbonic acid/bicarbonate, ammonium/ammonia, and lactic acid/lactate.
The acidity of a body fluid is determined by the concentration of hydrogen ion. Normal hydrogen ion concentration in plasma is approximately 40 nEq/L. 2 Because this is an extremely small unit of measurement, hydrogen ion concentration is expressed on a logarithmic scale using pH units. Normal blood pH ranges between 7.35 and 7.45, with 7.4 as the threshold for normal. Acid-base disorders considered incompatible with life are those where pH is <6.8 or >7.8 which corresponds to a hydrogen ion concentration of >160 nEq/L and <16 nEq/L, respectively. This is a very narrow range (16-160 nEq/L) given the large amounts of acid that are generated each day through normal metabolic processes.
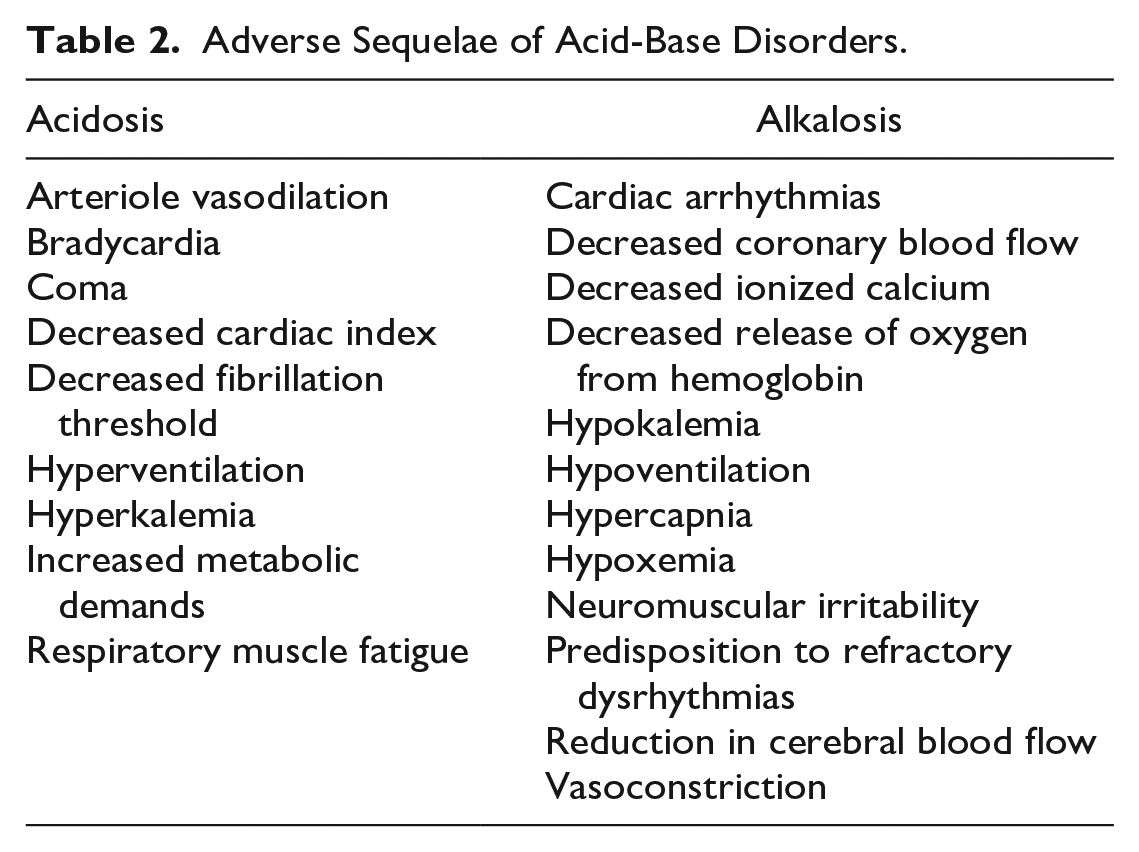
The terms acidemia or alkalemia are used to refer to an abnormal pH in the blood; acidemia when pH is < 7.35, alkalemia when pH is > 7.45. The terms acidosis or alkalosis describe the physiologic process that causes acid or base to accumulate. It is therefore possible to have, for example, an acidosis without an acidemia provided an alkalosis concurrently exists. In general, the body typically tolerates an acid load better than a base excess. 3 This is because changes in hydrogen ion concentration are not linearly related to changes in pH. Specifically, with alkalemia, small changes in hydrogen ion are associated with large changes in pH. With acidemia, on the other hand, larger changes in hydrogen ion are required to influence pH. In addition, with alkalemia, the oxy-hemoglobin dissociation curve will shift to the left, where the affinity of oxygen to hemoglobin is stronger resulting in inefficient release of oxygen to the tissues. Other adverse sequelae associated with acid-base disorders are displayed in Table 2.
Adverse Sequelae of Acid-Base Disorders.
Sources of acid production include end products of normal cellular metabolism (eg, carbon dioxide [CO2]), metabolism of dietary nutrients (eg, sulfuric acid, phosphoric acid), and incomplete metabolism of neutral substances (eg, glucose → lactic acid, fatty acids → acetoacetic acid and β-hydroxybutyric acid). Most acid production is in the form of CO2 whereby approximately 15 000 mmol are produced each day. 4 In critical illness, nonvolatile acids such as lactic acid or keto acids can be produced in pathologic amounts and are often the primary source for an acid-base disorder with some disease states (eg, shock, diabetic ketoacidosis).
For normal cellular and metabolic function to occur, acid-base balance must be maintained. There are 3 mechanisms the body uses to maintain the narrow therapeutic range for hydrogen ion: buffers, respiratory, and renal regulation. A buffer is a substance that can absorb or donate hydrogen ions to resist changes in pH in the presence of a strong acid or base. Buffers are the first line of defense when an acid-base imbalance is present. The dominant extracellular buffer system is the bicarbonate/carbonic acid system, defined using the following equation:
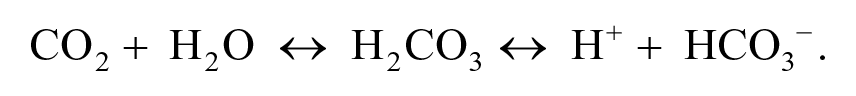
In this equation, reactions can flow in both directions depending on the concentration of each component and the imbalance that exists. Carbonic acid (H2CO3) is in equilibrium with bicarbonate (HCO3-) and hydrogen ions (H+). In the presence of carbonic anhydrase, carbonic acid is converted to CO2 and water (H2O). Because carbonic acid is directly proportional to the amount of CO2 in the blood, CO2 is considered an acid. Carbon dioxide is regulated through the lungs and can be controlled with either hyper- or hypoventilation. Bicarbonate concentrations are regulated through the kidney. Thus, disorders with HCO3- are usually thought of as metabolic while disorders of CO2 are respiratory. There are other important buffer systems that exist, such as the phosphate buffer system and intracellular and extracellular proteins, but these are more so reflective of intracellular systems.
The second mechanism used to maintain acid-base balance is respiratory regulation. Chemoreceptor cells located in the medulla oblongata are sensitive to pH and control alveolar ventilation. Hyperventilation will lead to increased elimination of CO2 (ie, respiratory acid) which will increase pH. Hypoventilation on the other hand will cause CO2 retention and decrease pH. In the presence of a metabolic disorder, the lungs will compensate by altering the respiratory rate to adjust CO2 levels to restore pH toward neutral (ie, 7.4). These changes in respiratory rate occur quickly where a new steady-state CO2 is reached within hours. 2
The third mechanism for maintaining acid-base homeostasis is renal regulation which involves regulating the concentration of bicarbonate in the blood (Figure 1). Approximately 4500 mEq of bicarbonate are filtered by the kidney each day. 4 Most of this is reabsorbed in the proximal tubule (approximately 80%) with the remaining occurring in more distal segments. 5 Filtered bicarbonate combines with hydrogen ions secreted by the renal tubule cell to form carbonic acid. In the presence of carbonic anhydrase, carbonic acid is converted to CO2 and H2O. Carbon dioxide readily crosses the cell membrane and enters the renal tubule cell where it is converted back to carbonic acid by intracellular carbonic anhydrase. Carbonic acid dissociates into hydrogen ion (which is later secreted into the tubular lumen) and bicarbonate which is reabsorbed into the bloodstream. In addition, the kidneys also excrete the daily load of nonvolatile acids through ammoniagenesis, regeneration of new bicarbonate, and distal tubular hydrogen ion secretion. Renal regulation of acid-base status begins within approximately 6 to 12 hours, but it can take 3 to 5 days for full compensation to occur.
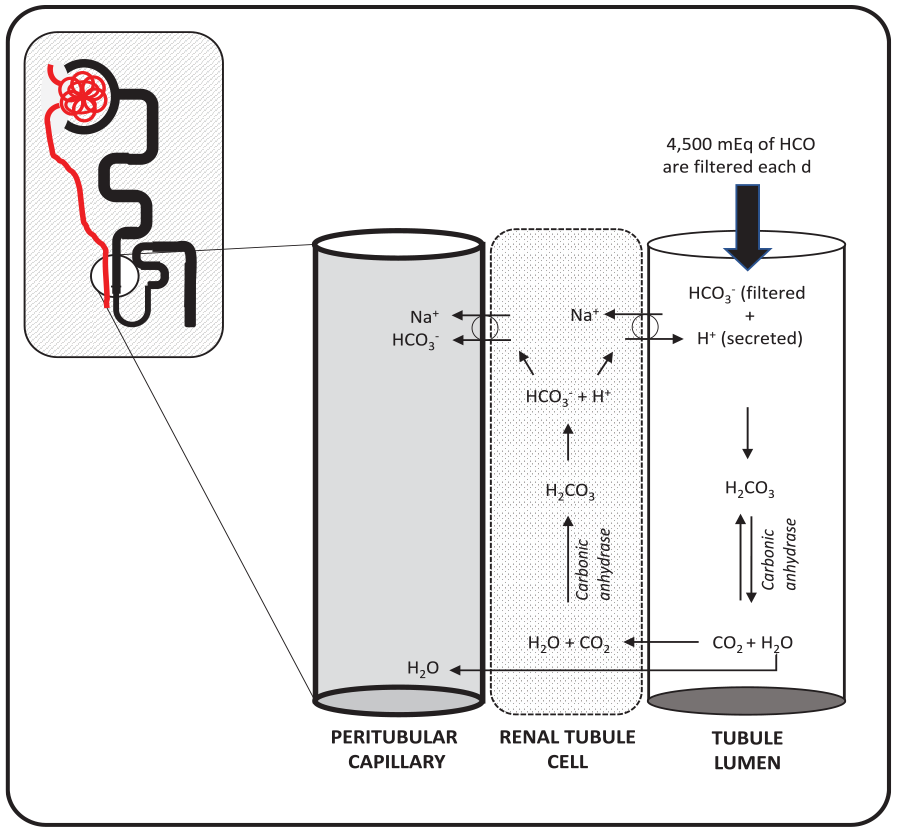
Renal regulation of acid-base homeostasis.
Arterial Blood Gas Assessment
Acid-base assessment is performed using an arterial blood gas (ABG). Arterial blood can be obtained through a percutaneous needle puncture or from an indwelling arterial catheter. When assessing a blood gas result, it is important to distinguish the type of sample collected (ie, arterial vs venous) because there are differences in the normal values between the two (Table 3). Venous blood gas sampling may be acceptable in some clinical scenarios (eg, a hemodynamically stable patient or emergency settings) given the correlation that exists between venous and arterial values, specifically pH and partial pressure of CO2. 7 In patients with shock or post-cardiac arrest, however, arterial blood samples are recommended.
Normal Values From Adult Blood Gas Analysis. 6

Abbreviations: HCO3-, bicarbonate; PCO2; partial pressure of carbon dioxide; PO2, partial pressure of oxygen.
Values obtained from an ABG include pH, partial pressure of arterial carbon dioxide (PaCO2), partial pressure of arterial oxygen (PaO2), bicarbonate (HCO3-), base excess, and oxygen saturation. The variables that directly influence acid-base status are pH, PaCO2, and HCO3-. Arterial pH will define the primary acid-base disorder and is the first variable to evaluate. PaCO2 represents the amount of respiratory acid present and provides information about how effectively the lungs are excreting CO2. Bicarbonate on an ABG reflects the amount of metabolic acid and is regulated by the kidneys. It is important to note, the concentration of HCO3- reported from an ABG is not a direct measurement but a calculated value using the Henderson-Hasselbalch equation. Under normal circumstances, this value will differ from the total CO2 content value obtained from a venous basic metabolic panel by about 1.5 to 3 mEq/L. This is because total CO2 content consists of all carbon dioxide present in the blood and includes bicarbonate, dissolved CO2, carbonic acid, and carbamino compounds. 3 Approximately 95% of total CO2 content is in the form of bicarbonate; thus, many clinicians refer to this value as “serum bicarbonate” in practice. This terminology is typically preferred to avoid confusion with PaCO2 on an ABG (Figure 2).

Pitfalls to avoid when evaluating acid-base homeostasis and an arterial blood gas analysis.
Approaches to Acid-Base Analysis and Interpretation
Management of acid-base disorders requires astute understanding of the physiologic variables that contribute to acid-base homeostasis. Described below are the 3 approaches to acid-base analysis and interpretation (Table 4).
Comparison of Models on Acid-Base Disorders.
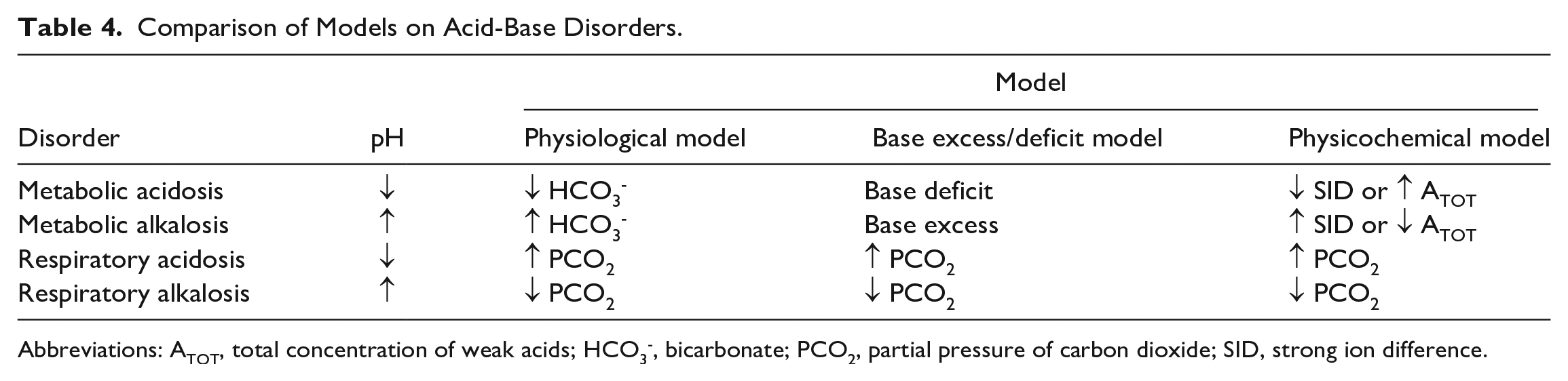
Abbreviations: ATOT, total concentration of weak acids; HCO3-, bicarbonate; PCO2, partial pressure of carbon dioxide; SID, strong ion difference.
The Physiologic Approach (Henderson-Hasselbalch)
The physiologic approach is based largely upon a single buffer system composed of 2 components: carbonic acid and HCO3-. This approach aims to describe the relationship between blood pH and the primary acid-base buffering properties of CO2 and HCO3- quantified by the Henderson and Hasselbalch formula 8 :
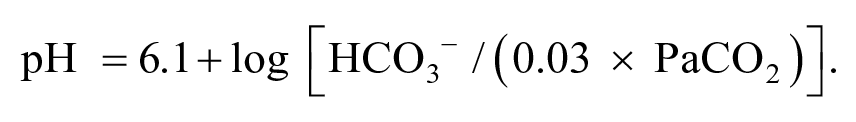
The Henderson-Hasselbalch equation assumes the carbonic acid-bicarbonate buffer system is the main determinant of acid-base status within the body.8,9 The blood pH calculated from the Henderson-Hasselbalch equation considers both respiratory (partial pressure of carbon dioxide [PCO2]) and metabolic (serum HCO3-) contributors, not by their individual absolute values, but by the ratio of the components. Therefore, a patient with multiple contrasting acid-base disturbances may possess a pH within normal limits. 10 The carbonic acid/bicarbonate pair is used as the major buffering system within the physiologic model because of their abundance and ability to be regulated via the respiratory and metabolic systems. 10 Each primary disorder is resultant of a change in serum HCO3- or PCO2 in conjunction with an appropriate secondary or compensatory response of the opposing variable (Table 5). The absence of a secondary response typically indicates the existence of multiple disorders or a mixed-acid-base disorder. 10 Supplemental variables can be incorporated for more accurate clinical diagnosis. Interpretation of a patient’s serum anion gap (AG), the difference between the measured cations and anions, is useful to determine whether additional unmeasured anions may be contributing to the primary acid-base disturbance. 11 Based on the principle of electroneutrality, AG assumes the concentrations of negatively charged particles (anions) must be equal to the concentration of positively charged particles (cations). Many substances in smaller quantities though are not routinely measured (or accounted for in standard acid-base analysis), and these unmeasured anions constitute the AG. 2 Laboratories may have different normal values for the AG, but a normal upper limit of 10 to 12 mEq/L is often reported. 12 Most clinicians would consider an AG in excess of 16 mEq/L clinically important and represent substantial anion accumulation. Occasionally, the concentration of potassium is included in the AG equation, which would make the normal AG approximately 4 mEq/L higher. Because potassium values do not generally deviate significantly from 4 mEq/L, it is usually simplest and most straightforward to eliminate it from the analysis. AG can be calculated using the following formula:
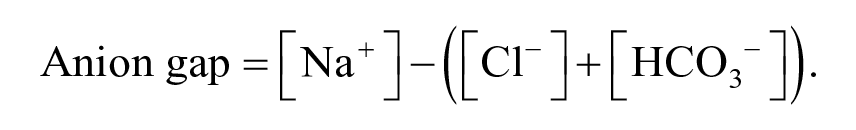
Expected Compensatory Responses to Acid-Base Disturbances.
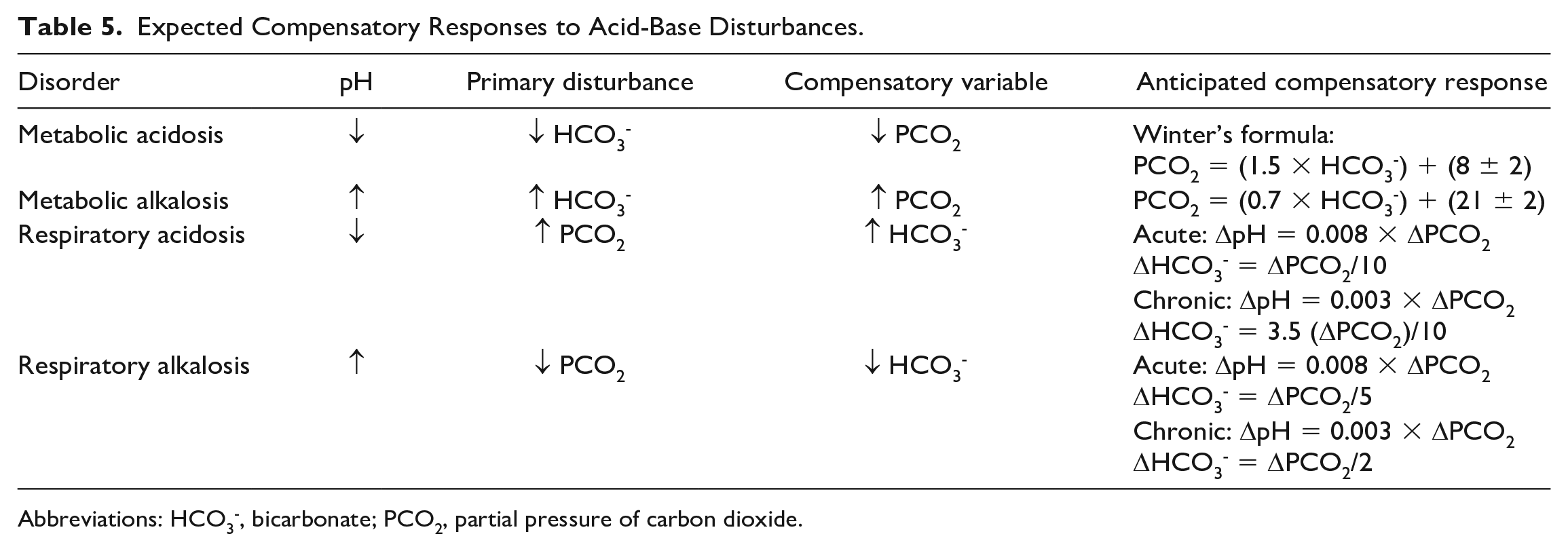
Abbreviations: HCO3-, bicarbonate; PCO2, partial pressure of carbon dioxide.
While AG forms the basis for assessment of the metabolic component within the physiologic approach, other anions may also influence the equation. Serum albumin is an anion that accounts for a large portion of the AG. Patients with hypoalbuminemia, which is frequently present in the critically ill, may require adjustments to the calculated AG. 10 Failure to adjust for albumin may lead to misinterpretation of the metabolic component when using the physiologic approach. A corrected AG formula has been proposed that accounts for an expected decrease of 2.5 units per each 1 g/dL decrease in albumin concentration. 9
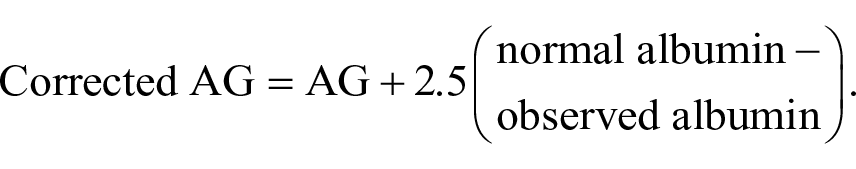
The physiologic approach remains advantageous for clinical interpretation of acid-base status due to its simplicity in data components and application. This approach is limited because changes in bicarbonate concentration are not able to quantify how much acid or base has been added to the system unless the single buffer, PCO2, is held constant. 13 In addition, the physiologic approach does not accurately account for non-bicarbonate buffer systems.
The Base Excess/Deficit Model
To address limitations with the physiologic approach, Singer and Hastings introduced an alternative method which is based on the amount of acid or base that must be added to a sample of blood in vitro to obtain a pH of 7.4. 13 This approach is referred to as the base excess model. Base excess (BE) measures the metabolic component (using hemoglobin as a buffer), independent from the respiratory component of acid-base status; BE should be zero under normal circumstances. 9 Inaccuracies with using this method in vivo have led to modifications in the equations used to determine BE resulting in the term standard base excess (SBE). SBE is reported by most commercially available ABG analyzers. Generally, a SBE greater than +5 mmol/L represents a metabolic alkalosis (base excess), whereas less than –5 mmol/L represents a metabolic acidosis. 14 In practice, a negative base excess is often referred to as a base deficit and could indicate hypoperfusion.
A limitation of the SBE is that it does not provide insight into the mechanism of the metabolic disorder. This can be improved by combining SBE with the AG. Furthermore, equations for SBE assume normal weak acid concentrations (eg, albumin); therefore, conditions such as hypoalbuminemia can lead to inaccuracies. 15 SBE, though, is often used to determine severity of illness following an acute traumatic injury and can be used to help guide resuscitation. In fact, SBE may be superior to vital signs or shock index for predicting the risk of death in this population. 16
The Physicochemical/Strong Ion Approach (Stewart)
An additional approach to acid-base analysis was developed by Peter Stewart based on the concept of strong ion difference (SID). Stewart defined strong ions as those which fully dissociate at the pH of body fluids and whose concentrations could be used to calculate the hydrogen ion concentration of the blood. 10 Strong ions include sodium, potassium, calcium, magnesium, chloride, and lactate.10,13 Based on the laws of electroneutrality, the difference between the sum of strong anions and strong cations (strong ion difference or SID) when combined with weak acids and bases within the blood should yield a neutral result. 13 As opposed to a strict buffer system, the physicochemical or strong ion approach proposed by Stewart was based on the classification of each variable as either dependent or independent in determining H+ concentration in the blood. 9 Using the Stewart approach, there are 3 variables independently responsible for determining H+ concentration: SID, total concentration of weak acids (ATOT), and PCO2. Therefore, SID and ATOT, not bicarbonate concentration, are the main determinants of metabolic disorders, and PCO2 is the main determinant of respiratory disorders. 9 Stewart proposed that bicarbonate contributed to the gap between strong cations and strong anions, and the concentration of bicarbonate was dependent on the amount of strong cations and anions within the blood. 10 To accurately account for all strong and weak cations and anions, the physicochemical approach is based on the measurements of apparent SID (SIDa) which represents the difference between measured strong cations and anions [SIDa = (Na+ + K+ + Ca2+ + Mg2+) – (Cl- + lactate-)], and effective SID (SIDe), which accounts for weak acids and bases (including bicarbonate, albumin, and inorganic phosphate). 9 In a healthy individual, a normal SIDa is approximately +40 to 42. 14 Based on the laws of electroneutrality, SIDa and SIDe should be equal, and any difference would yield a gap of unmeasured strong ions (due to a pathophysiologic process), or strong ion gap (SIG). The SIG represents the pure representation of unmeasured ions, similar to the AG, but theoretically more robust.
While the 3 approaches vary in independent and dependent variables used to assess acid-base status, no approach has been proven to be more efficacious during clinical diagnosis and assessment. The physicochemical approach was proposed as an alternative to better quantify acid-base status; however, when examined in critically ill patients, quantitative analysis did not offer diagnostic or prognostic advantages over more traditional approaches originally described by the Henderson-Hasselbalch equation. 17 The physiologic approach that relies on descriptive analysis of acid-base status may be considered more useful given its simplicity for the bedside clinician, without sacrificing diagnostic or clinical efficacy.18,19
A Systematic Method to Interpretation of Acid-Base Disorders
Accurate interpretation and diagnosis of acid-base disorders can be a lifesaving intervention in the critically ill patient. Although multiple approaches may exist to evaluate acid-base status, a systematic method for interpretation (Figure 3) of clinical data, diagnostic evaluation, and treatment is useful to easily identify simple and mixed disorders.2,12 The first step in the process is to assess the situation and the patient’s clinical presentation. In some cases, the expected acid-base abnormality can be predicted which could then be compared with the results from the stepwise approach. Results that differ should be reevaluated for accuracy or an alternative explanation. After a thorough review of the patient’s history, the primary disorder should be determined along with the presence of a compensatory response. For some disorders, such as metabolic acidosis, there are additional steps. It is important to go through this systematic approach, even when pH may be in the normal range because a complex or unidentified acid-based disorder may exist (ie, a concomitant acidosis with an alkalosis). In fact, it is possible for up to 3 acid-base disorders to exist at any one time. A delta-delta calculation or delta ratio may help identify these scenarios. While it is possible to have 2 separate metabolic abnormalities at once, only 1 respiratory disorder can exist. In reality, mixed acid-base disorders are quite common in critically ill patients because of the complex disease states encountered coupled with chronic diseases that may be present and the effects of underlying therapies.
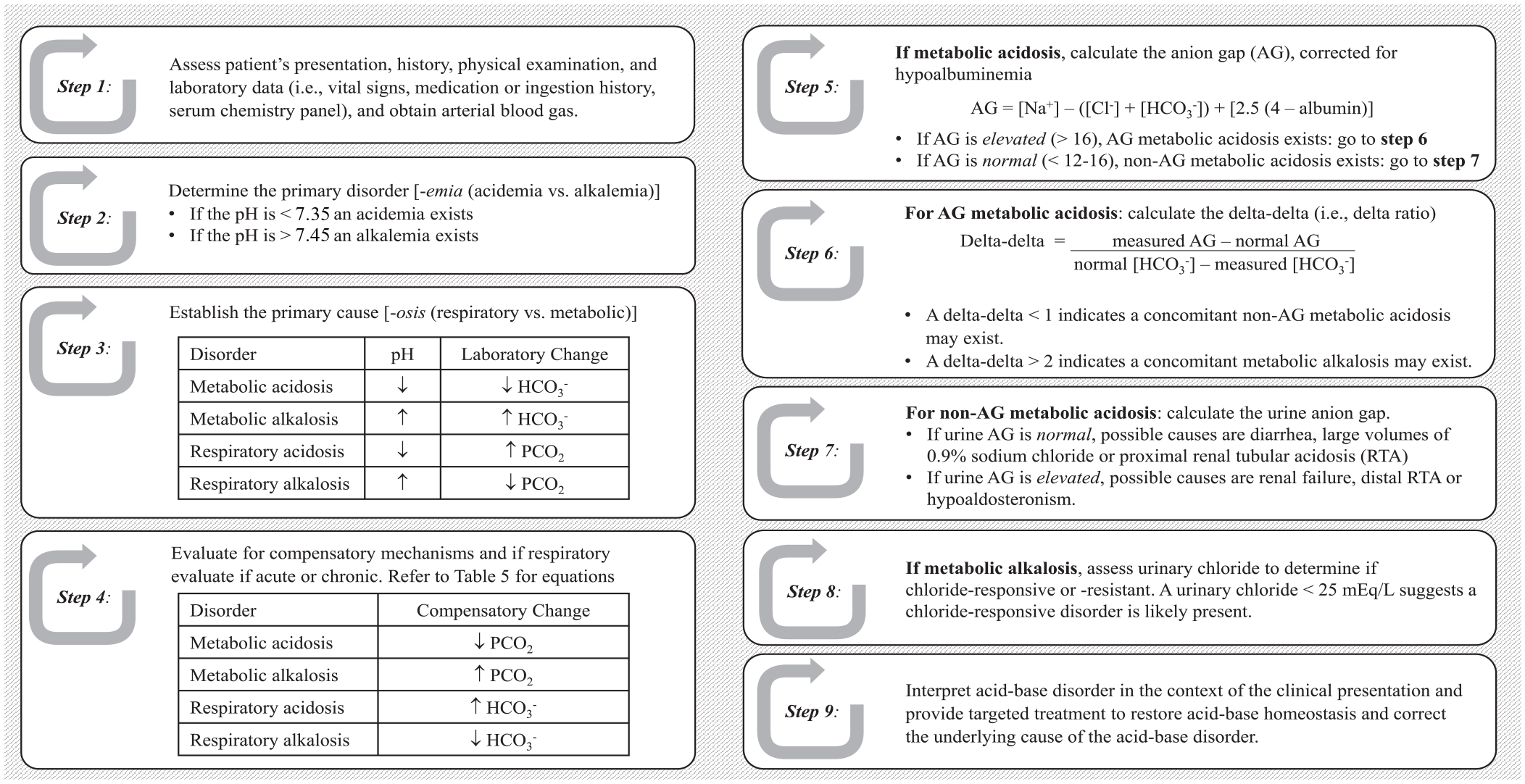
Stepwise approach to acid-base analysis.
Metabolic Disorders
In the context of acid-base abnormalities, metabolic disorders relate to an imbalance between H+ ion and bicarbonate in the body. These can broadly be divided into metabolic acidosis and metabolic alkalosis. Metabolic acidosis can further be differentiated as anion gap metabolic acidosis (AGMA) or non-anion gap metabolic acidosis (NAGMA). Metabolic alkalosis can be viewed as chloride responsive or non-responsive.
Metabolic Acidosis
Metabolic acidosis derives from the overproduction or reduced elimination of acid, exogenous exposure to acid or acid precursors, or the loss of base. As mentioned above, the AG reflects the concept that usual measured cations are present in a larger quantity than usual measured anions. The presence of a metabolic acidosis with an elevated AG reflects the addition of unmeasured anions, especially organic acids. Various unmeasured anions, accompanied by protons, can accumulate in the body, including lactate, keto acids, and many toxins, some of which can be assessed separately (eg, lactate level). Conversely, the presence of a metabolic acidosis without an elevated AG indicates a derangement with the ions found in the AG equation, such as the loss of bicarbonate or the gain of chloride ions. The presence or absence of an AG can help discern the cause of the metabolic acidosis recognizing there are exceptions. In fact, a specific cause for an increased AG can be found in 29% of patients who have an AG between 17 and 19, 65% when between 20 and 24 and >80% when greater than 25. 20 Several mnemonics have been suggested to recall the common etiologies of AGMA (Table 6).
Causes of Metabolic Acidosis and Alkalosis.
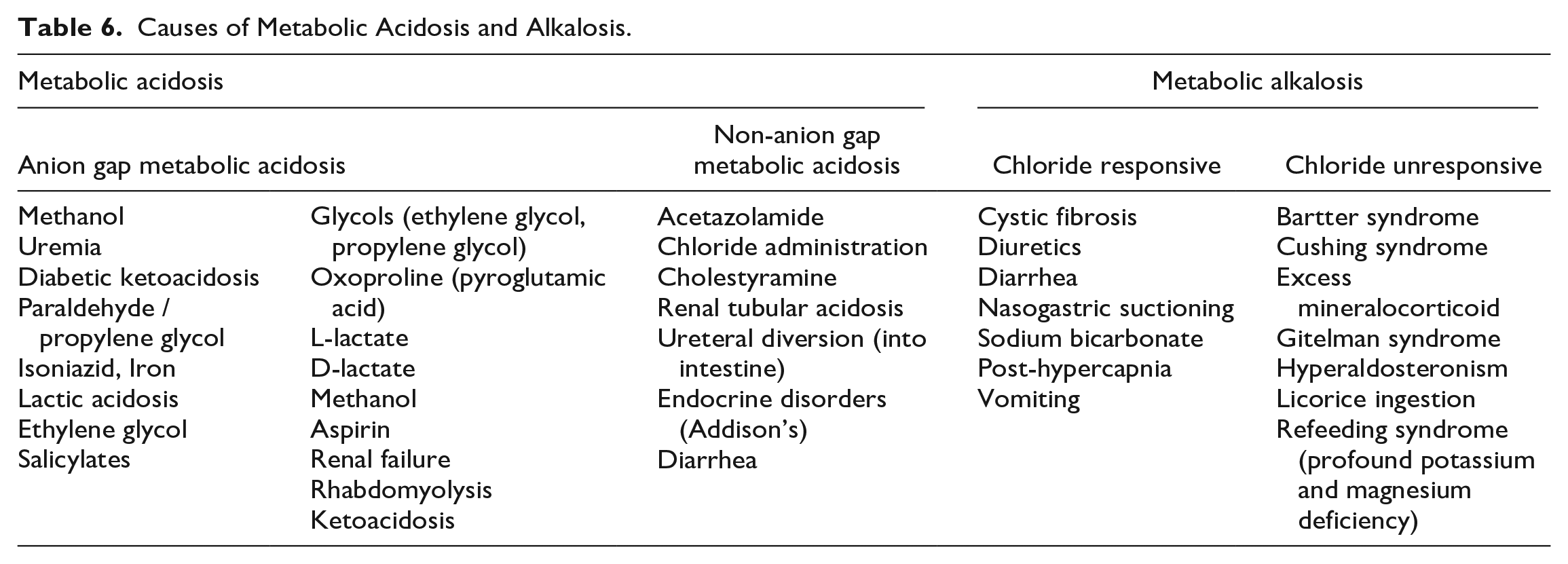
A common cause of AGMA in critically ill patients is lactic acidosis. Lactic acidosis, defined as a lactate concentration > 4 mmol/L, is commonly seen in the setting of shock where cellular and tissue hypoxia shifts metabolism to anaerobic glycolysis. 12 This is also known as a type A or hypoxic lactic acidosis. Other causes include mesenteric ischemia, carbon monoxide poisoning, and cyanide. 2 Lactate can also accumulate for nonhypoxic reasons (type B lactic acidosis) such as thiamine deficiency and medications (Table 7).12,21 In critical illness and specifically shock, lactate levels may be elevated for a variety of reasons, including overproduction (hypoxic or nonhypoxic) as well as reduced clearance or mitochondrial dysfunction. 22 However, lactic acidosis may not always be identified accurately with AG, even after correction for hypoalbuminemia. 23
Common Drug-Induced Metabolic Acidosis and Alkalosis.
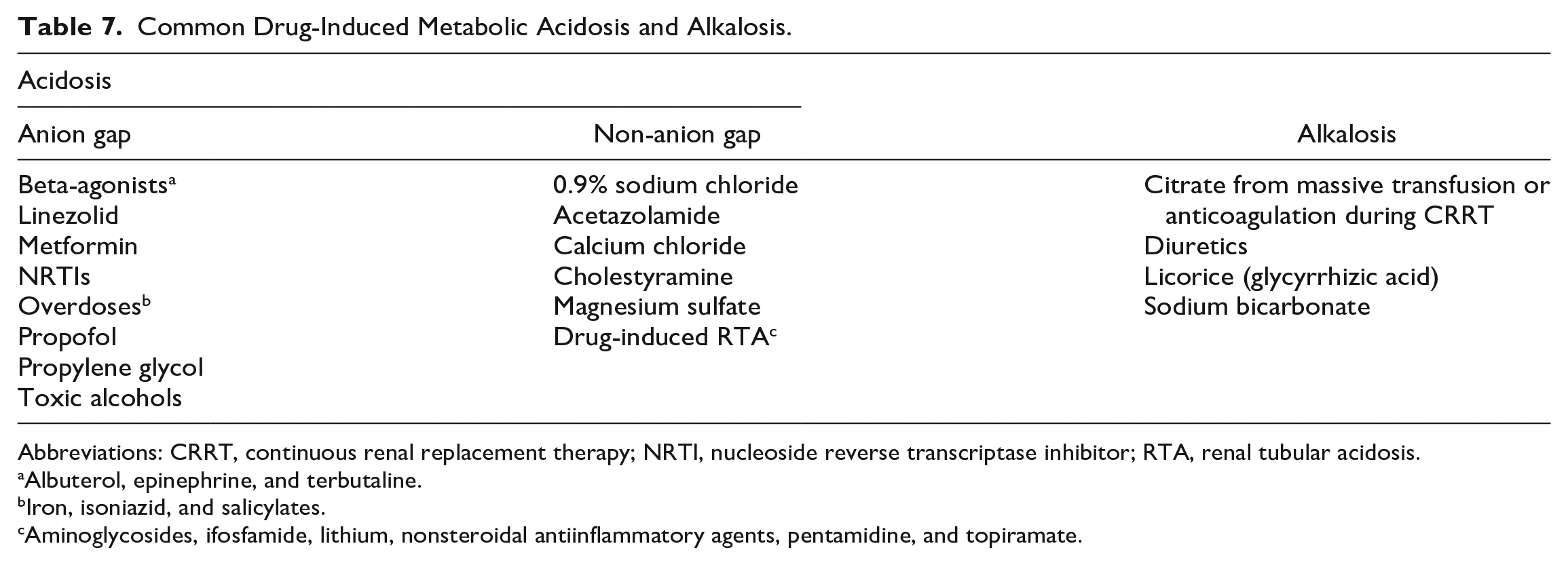
Abbreviations: CRRT, continuous renal replacement therapy; NRTI, nucleoside reverse transcriptase inhibitor; RTA, renal tubular acidosis.
Albuterol, epinephrine, and terbutaline.
Iron, isoniazid, and salicylates.
Aminoglycosides, ifosfamide, lithium, nonsteroidal antiinflammatory agents, pentamidine, and topiramate.
Non-AG metabolic acidosis results from several etiologies where derangements occur specifically with the measured ions accounted for in the AG equation: sodium, chloride, and/or bicarbonate. One common mnemonic for NAGMA is presented in Table 6. Many of these disorders lead to metabolic acidosis via loss of bicarbonate through the urine or gastrointestinal tract. Critically ill patients may be particularly susceptible to NAGMA when large volumes of 0.9% sodium chloride are used for resuscitation (0.9% sodium chloride contains 154 mEq of sodium and 154 mEq of chloride; SID = 0 mEq/L). Notably, a NAGMA may be present without a significantly elevated chloride level, in the context of hyponatremia. 24 Balanced solutions such as Ringer’s lactate, Plasma-Lyte, and Normosol are more physiologic in nature (SID is approximately 28 mEq/L) and contain buffers (lactate or acetate) which are metabolized to bicarbonate and mitigate the acidifying effect. At usual volumes and administration rates, the lactate contained in Ringer’s lactate does not appreciably contribute to lactate concentrations. 22 Renal tubular acidosis (RTA) is also a common cause of NAGMA and has 4 major forms depending on the portion of the nephron that is no longer able to maintain normal acid-base and potassium homeostasis. A related disorder, Fanconi’s syndrome, features RTA along with other findings, and can rarely be caused by several drugs (Table 7). 25
The routine use of sodium bicarbonate therapy for the treatment of acidosis may lead to deleterious effects without improvements in patient outcomes. 26 For example, sodium bicarbonate therapy may lead to sodium and volume overload leading to hypernatremia and pulmonary edema, respectively, worsening of tissue acidosis from CO2 diffusion into myocardial and cerebral cells, and impairment of oxygen release form hemoglobin. 27 Sodium bicarbonate should be considered in critically ill patients with severe NAGMA when caused by gastrointestinal or renal disturbances or in the setting of specific overdoses such as salicylate and tricyclic antidepressants. In addition, sodium bicarbonate may have a role in patients with severe metabolic acidosis and stage 2 or 3 acute kidney injury. 28
Metabolic Alkalosis
Metabolic alkalosis results from an excess of base or a loss of acid, mainly observed through an elevated bicarbonate level. Metabolic alkalosis can be viewed along a continuum with NAGMA as a disorder characterized by an imbalance between sodium, chloride, and bicarbonate. However, in alkalosis the bicarbonate is elevated above normal. This is why an AGMA can exist alongside NAGMA or metabolic alkalosis, but NAGMA and metabolic alkalosis cannot both exist at the same time. It is therefore always important to evaluate for a concomitant NAGMA or metabolic alkalosis (ie, the “delta-delta” or “delta gap” approach) when an AGMA is present.
Urine chloride concentrations may facilitate diagnosis of the etiology of metabolic alkalosis. Typically a problem originating outside the kidney will result in a low urine chloride (< 25 mEq/L) as the kidneys are trying to retain acid via ammonium chloride. A problem originating within the kidney will have a high urine chloride signifying the kidney’s inability to retain acid. 29 These are also referred to, respectively, as “chloride responsive” and “chloride unresponsive” metabolic alkalosis (Table 6). This test, however is not routinely performed in the ICU as the clinical picture, and patient history are often enough to discern the most likely etiology (eg, excessive loop diuretic use in the setting of de-resuscitation).
Chloride depletion is a common etiology and can result from various mechanisms including loss from the gut, urine, and skin. Other causes of metabolic alkalosis result from potassium depletion which usually is accompanied by an excess in mineralocorticoid activity. Aldosterone has a strong influence on acid-base status by facilitating acid excretion through the α-intercalated cells of the late distal convoluted tubule and collecting duct, and potassium loss independently contributes to alkalosis by inducing bicarbonate reabsorption in the proximal tubule. 30 Loop diuretics, commonly used in the critically ill, are a common cause of metabolic alkalosis. Chloride and potassium loss are the principal mechanisms, though this is sometimes described as “contraction alkalosis”; most accurately, this refers to contraction of the extracellular fluid volume around a fixed bicarbonate amount, rather than implying a hypovolemic state. 30 Acetazolamide, a carbonic anhydrase inhibitor, may occasionally be prescribed to counteract this effect. Hypokalemia though should be addressed as it can directly perpetuate the alkalosis.
Respiratory Disorders
Primary respiratory acid-base disorders are propelled by an alteration in CO2 elimination regulated by alveolar ventilation. An increase in PCO2 results in a fall in pH (respiratory acidosis), whereas a decrease in PCO2 results in a rise in pH (respiratory alkalosis). Determining an acute versus chronic process will be determined by the severity of change and degree of renal compensation (Table 5).
Respiratory Acidosis
Respiratory acidosis occurs from hypoventilation, defined as the failure to excrete CO2 (hypercapnia). There are several acute and chronic conditions that arise from decreased respiratory rate or tidal volume leading to respiratory acidosis (Table 8). There are no direct drug treatments for respiratory acidosis, rather appropriate treatment will rely on alleviating the underlying condition considering the severity and chronicity of the acid-base derangement. Hypoxemia is the primary life-threatening consequence of respiratory acidosis which may result from acute, severe hypercapnia at room air (PCO2 > 80 mm Hg). In addition, in severe respiratory acidosis, hypotension from reduced cardiac output and decreased vascular resistance may ensue. 31 Patients with severe hypoxemia (PaO2 < 40 mm Hg) should receive supplemental oxygen via non-invasive or invasive ventilatory support while treating the underlying cause. Common therapies include bronchodilators for the treatment of bronchospasm, naloxone for opioid intoxication, or thrombolytics for massive pulmonary embolism. The use of supplemental oxygen in spontaneously breathing patients with chronic hypercapnia (eg, patients with chronic obstructive pulmonary disease) should be used with caution as this may blunt a patient’s respiratory effort and worsen alveolar ventilation. 32 Furthermore, gradual reductions in PCO2, to a patient’s estimated baseline (if unknown a reasonable PCO2 target is 60 mm Hg), will avoid cerebral vasoconstriction and ischemia and facilitate liberation from mechanical ventilation. 33 In addition, reducing PCO2 beyond the patient’s baseline value will lead to loss of compensatory bicarbonate and will result in an uncompensated respiratory acidosis when mechanical ventilation is removed. Exogenous administration of sodium bicarbonate should not be used to correct respiratory acidosis as it may precipitate metabolic alkalosis or worsen the respiratory acidosis.
Causes of Respiratory Acidosis and Alkalosis.
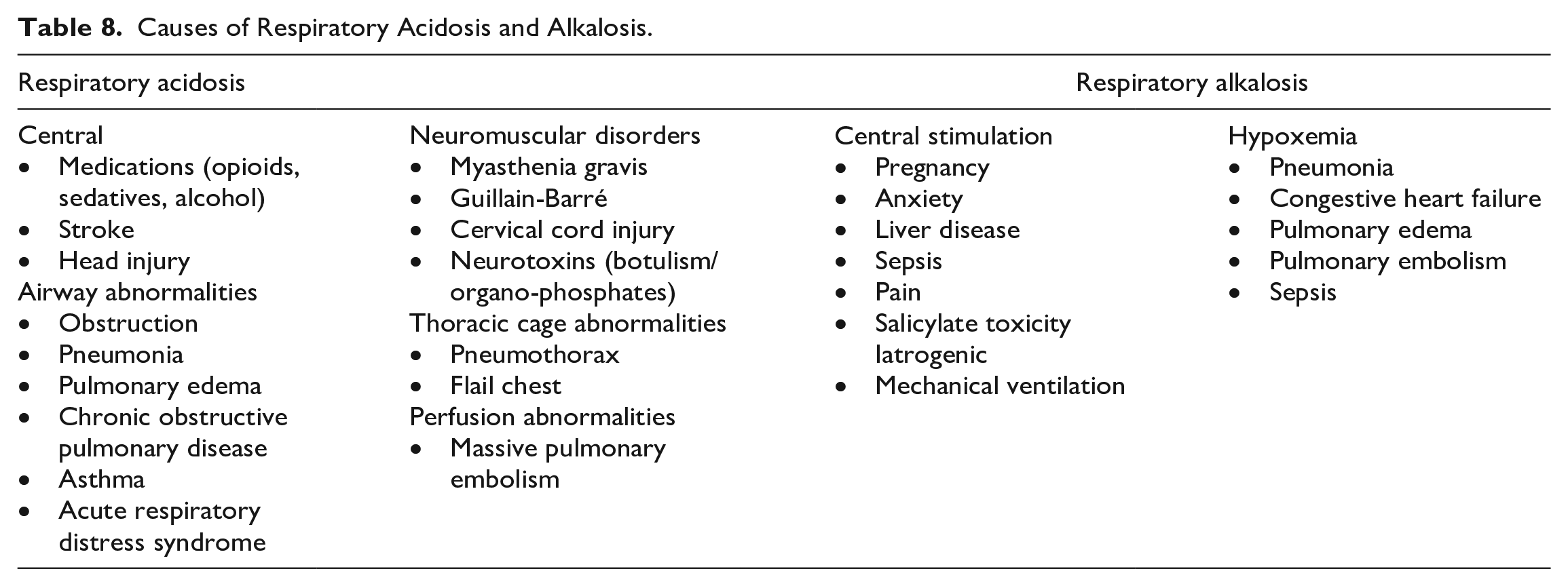
Respiratory Alkalosis
Respiratory alkalosis results from hyperventilation or an excessive elimination of CO2. As with respiratory acidosis, there are a variety of acute and chronic causes (Table 8). In severe respiratory alkalosis, patients may experience cardiac arrhythmias from increased sensitization to circulating or exogenously administered catecholamines, electrolyte aberrations such as hypokalemia, hypocalcemia, and hypophosphatemia, and reductions in cerebral blood flow in the presence of elevated intracranial pressure. As with respiratory acidosis, supplemental oxygen therapy should be considered in all patients presenting with hypoxia. Treating the underlying cause such as sedatives for anxiety or analgesics for pain in the critically ill is the mainstay of this acid-base abnormality.
Relevance to Patient Care and Clinical Practice
Acid-base disorders are often caused by complex disease processes but can also stem from drug therapy, electrolyte management or fluid administration. Pharmacists are uniquely positioned as the medication expert on the multidisciplinary team and can play a pivotal role in the evaluation of acid-base abnormalities. Furthermore, many acid-base disorders are managed with medications for which the pharmacist must discern their appropriateness.
Conclusion
Acid-base aberrations are common in critically ill patients. A systematic approach should be applied to every patient to facilitate identification of acid-base abnormalities. Consideration of the physiological mechanisms contributing to metabolic and respiratory disorders coupled with the clinical settings in which they commonly occur will assist in prompt treatment and the necessary monitoring. To evaluate the understanding of common acid-base abnormalities in critically ill patients, Supplemental Appendix A highlights common scenarios observed in the ICU.
Supplemental Material
sj-docx-1-aop-10.1177_10600280231165787 – Supplemental material for A Systematic Approach to Understanding Acid-Base Disorders in the Critically Ill
Supplemental material, sj-docx-1-aop-10.1177_10600280231165787 for A Systematic Approach to Understanding Acid-Base Disorders in the Critically Ill by Jeffrey F. Barletta, Justin Muir, Judah Brown and Amy Dzierba in Annals of Pharmacotherapy
Footnotes
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: JFB, consultant for Wolters Kluwer. The remaining authors have no conflicts of interest to report.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
