Abstract
Background:
Through actions of calcium channel trafficking inhibition and sodium/water retention, pregabalin may increase the risk of acute heart failure (AHF).
Objective:
The objective of this study was to determine the prevalence of heart failure (HF) acute exacerbations, measured by a composite of emergency department (ED) visits, per-patient per-year (PPPY) hospitalizations, time-to first ED admission, and time-to hospitalizations in pre-existing HF patients taking pregabalin compared with those who were pregabalin-naive.
Methods:
A retrospective cohort study of pregabalin users with HF were propensity score–matched to pregabalin-naïve patients with HF to evaluate the composite of ED admissions or PPPY hospitalizations, time-to first ED admission, and time-to hospitalizations during the 365 days post-index. Doubly robust generalized linear regression and Cox-proportional hazard regression modeling were undertaken for analysis of differences between groups.
Results:
The matched cohort of 385 pregabalin users and 3460 pregabalin nonusers were principally middle-aged, equally gender distributed, and primary Caucasian. Most patients were on guideline-directed HF medical therapy. The estimated cumulative incidence of the primary outcome was a hazard ratio of 1.099 (95% CI: 0.789-1.530; P = 0.58).
Conclusion and Relevance:
This large, single-center, cohort study shows pregabalin is not associated with an increased risk of AHF events in patients with pre-existing HF.
Introduction
Heart failure (HF) is a leading cause of hospitalizations, mortality, and economic burden to the US health care system. Heart failure affects 37.7 million individuals worldwide, including 6.2 million Americans.1-3 A significant portion of the US HF population have multiple comorbidities that exacerbate pre-existing HF, including coronary heart disease, diabetes, hypertension, depression, and anxiety. 4
Treating multiple HF comorbidities contributes to poly-pharmacy, thereby complicating therapy. Heart failure patients take an average of 6.8 prescription medications per day. 5 Many drugs can exacerbate stable HF, such as nonsteroidal anti-inflammatory drugs (NSAIDs), anti-arrhythmic agents, and anti-diabetic agents. 5
Pregabalin is a common medication often prescribed in HF to treat anxiety, insomnia, depression, and pain, despite the caution by the US Food and Drug Administration regarding weight gain and fluid retention, possibly exacerbating or leading to HF. 6 It is a γ-aminobutyric acid derivative used for the treatment of partial-onset seizures, diabetic neuropathy, and other neurological disorders. 7 The agent decreases the release of substance P, norepinephrine, and glutamate by antagonizing the α2δ1,2 units of the L-type calcium channel found in the myocardium. 8 A systematic review of randomized clinical trials found that up to 20% of patients receiving pregabalin developed peripheral edema. 9 Pregabalin’s inhibition of the heart’s calcium channel trafficking and its potential to cause fluid retention give rise to concerns that it may be capable of worsening HF. 10
In 2011 and 2019, respectively, 2 separate case reports were published linking γ-aminobutyric acid derivatives, gabapentin and pregabalin, to HF. One describes a patient with no cardiac history being prescribed pregabalin 300 mg/kg for neuropathic pain who developed peripheral and central edema. After discontinuing pregabalin, the edema resolved. 11 Gabapentin was substituted for pregabalin and the edema returned. The second one describes a 44-year old woman with spinal stenosis prescribed gabapentin for pain. 12 Two months after initiating therapy, the patient was diagnosed with nonischemic cardiomyopathy.
Two population-based studies investigating the link between pregabalin and cardiomyopathy have been published. Propensity score matching was used in both studies, with one study comparing pregabalin with an active control. However, both studies limited follow-up to 90 days post-index and, despite large national and regional databases, enrolled relatively small numbers of pregabalin users.13,14 In addition, only one of these studies examined risk specifically related to pre-existing HF. 13 Neither study statistically demonstrated pregabalin increased the risk of HF exacerbation requiring hospitalization or emergency department (ED) visits.
Considering the limitations of existing data, the aim of this large population-based cohort, single-center study was to determine the prevalence of HF acute exacerbations, measured by a composite of ED visits, per-patient per-year (PPPY) hospitalizations, time-to first ED admission, and time-to hospitalizations in pre-existing HF patients taking pregabalin compared with those who were pregabalin-naive.
Methods
This was a population-based real-world retrospective cohort study. All data were collected from the University of Utah Enterprise Data Warehouse (EDW). The EDW is a repository of historical and comprehensive medical records for more than 2.5 million University of Utah Healthcare system patients. The study was conducted in accordance with the 1964 Declaration of Helsinki, including its later amendments, and received approval from the University of Utah Institutional Review Board.
We reviewed the structured field of EDW from January 1, 2015, to December 31, 2021. Patients were included if they have received their first HF diagnosis between January 1, 2016, and December 31, 2020. Diagnosis was identified using ICD-9 (428.x) and ICD-10 (150.x) diagnosis codes. Patients were excluded if they had an HF diagnosis 12 months prior to the enrollment period, a pregabalin prescription in the previous 12 months prior to their first HF diagnosis, missing key data such as background characteristics or outcome data, been enrolled in a clinical trial during the study period, a history of peripartum cardiomyopathy or cardiac transplant, and/or a left ventricular assist device.
Patients were sorted into the exposure group if they were newly prescribed pregabalin within 1 year following their first HF diagnosis code. The index date for the exposure group was the patient’s first pregabalin prescription. For our primary analysis, HF patients without a pregabalin prescription were the control group. Their index date was considered the date of their first HF diagnosis.
The outcome of analysis was a composite of HF-related hospitalizations, HF-related emergency room (ER) visits, and all-cause mortality. Encounters were determined to be HF-related by diagnosis codes (ICD-10, I50.x) listed within the first 3 diagnosis positions for that encounter.
As a secondary composite outcome, we analyzed all-cause admissions, ER visits, and death. Selected patients were followed up until their last encounter available from the electronic health record, outcome of interest, or 365 days after the index event, whichever happened first.
Statistical Analysis
Summary statistics of the baseline characteristics include mean and standard deviation (SD) and percentage of all other categorical variables. Student t test was performed to compare age distribution at the index date between the exposure and control groups. For categorical variables, we used χ2 tests to assess any significant difference in patient characteristics.
We accounted for the patient characteristics in the pregabalin-outcome associations using propensity scores of being assigned to pregabalin versus non-pregabalin cohort. The logistic regression model generating the propensity scores was finalized upon a stepwise forward variable selection process in which P values for model entry and retention were 0.20 and 0.15, respectively. Each pregabalin patient was matched using a 1:10 ratio with replacement and unrestricted random sampling with a range of 0.2 times of the pooled standard deviation of the logit of propensity score.
We calculated product-limit cumulative incidence from Kaplan-Meier approach. The HR of the outcomes for pregabalin was calculated using Cox-proportional hazard regression model in which a multivariable adjustment controlled for any residual confounder after propensity matching (P < 0.1). A P < 0.05 was considered statistically significant. All statistical analyses were performed using SAS v.9.4 (SAS Institute, Cary, North Carolina).
Results
A total of 22 113 patients meeting the inclusion study criteria were identified. After applying the exclusion criteria, 19 573 total patients were included: 385 pregabalin users and 19 188 pregabalin nonusers. Before matching, pregabalin cohort were younger (mean age of 61.5 vs. 65.7 years), had a lower proportion of men (47.8% vs. 55.4%), and carried more cardiovascular and metabolic risk factors (obesity 35.8% vs 12.0%, P < 0.01; hypertension 60% vs 38.1%, P < 0.01; diabetes 39.5% vs 19.2%, P < 0.01).
Of the potential confounders, age, year of index date, race, ethnicity, hypertension, hypercholesterolemia, obesity, diabetes, alcohol use, obstructive sleep apnea, chronic nonsteroidal anti-inflammatory drug use, end-stage renal disease, history of peripheral vascular disorder, and the presence of common pregabalin indications (neuralgia, neuritis, fibromyalgia) were selected to calculate the propensity scores. The confounders were selected based on listed concomitant diagnoses in the original data set. The matched cohort consisted of 346 pregabalin users and 3460 pregabalin nonusers (Table 1). Some residual confounding remained due to differences in the use of serotonin and norepinephrine reuptake inhibitors (SNRIs) (P < 0.01). A multivariable analysis was subsequently conducted to control for residual confounding.
Baseline Characteristics Across Pregabalin Users Compared With Pregabalin Nonusers Before and After Propensity Score Matching.
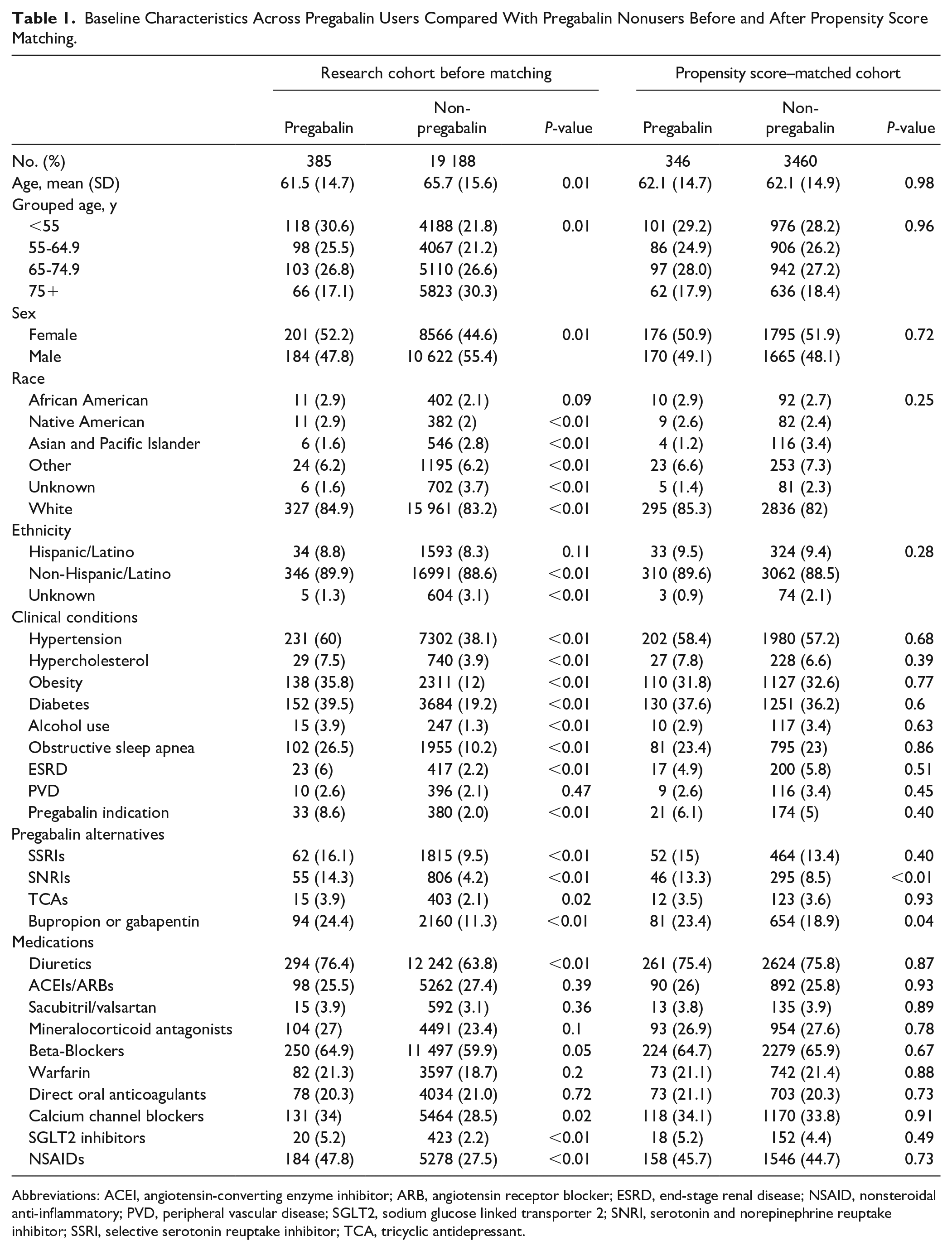
Abbreviations: ACEI, angiotensin-converting enzyme inhibitor; ARB, angiotensin receptor blocker; ESRD, end-stage renal disease; NSAID, nonsteroidal anti-inflammatory; PVD, peripheral vascular disease; SGLT2, sodium glucose linked transporter 2; SNRI, serotonin and norepinephrine reuptake inhibitor; SSRI, selective serotonin reuptake inhibitor; TCA, tricyclic antidepressant.
After matching, the analytic cohort was middle-aged, equally gender distributed, and primarily Caucasian. Frequent concomitant disorders included hypertension, diabetes, obesity, and obstructive sleep apnea. Most patients were prescribed guideline-directed medical therapy (GDMT), including loop diuretics (75%), β-adrenergic blockers (65%), renin-angiotensin-aldosterone inhibition (57%), and sodium-glucose-cotransporter-2 inhibitors (4%-5%). Of note, NSAIDs were prescribed in approximately 45% of patients. Alternatives to pregabalin were recorded in 32% to 38% of patients (P = 0.04).
The primary outcomes of pregabalin users compared with nonusers were not statistically different.
Figure 1 shows pregabalin users were slightly more likely to record a composite of HF admission, HF ED visits, or death compared with pregabalin nonusers. The crude (unadjusted) HR was 1.120 (95% CI: 0.805-1.559; P = 0.50). After adjustment, the HR was 1.099 (95% CI: 0.789-1.530; P = 0.57). The adjusted HR of the secondary outcome was 0.835 (0.692-1.007; P = 0.06) (Figure 2).
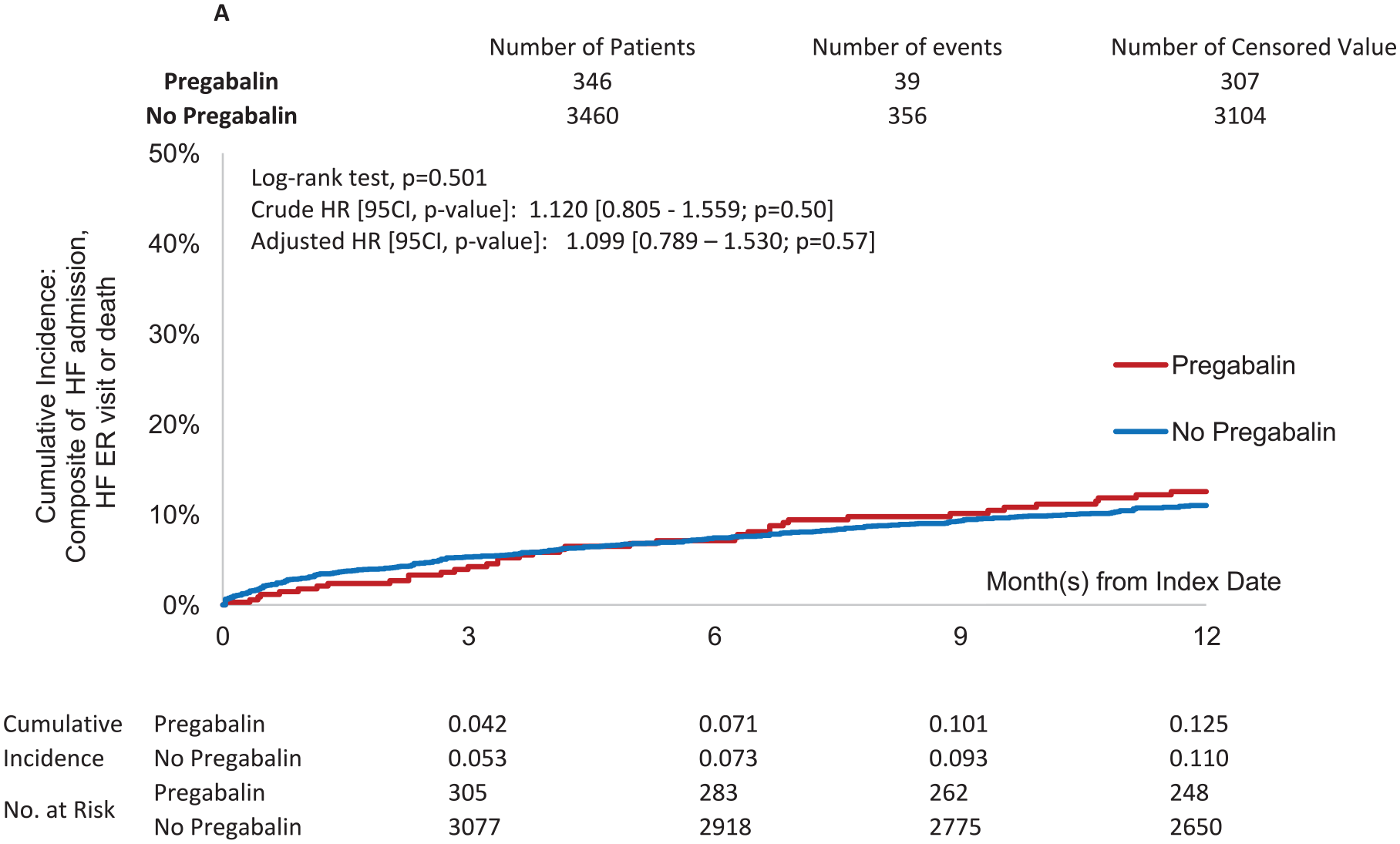
Composite outcome of HF-related emergency room visits, HF-related hospitalizations, and all-cause mortality of pregabalin users compared with pregabalin nonusers over time.
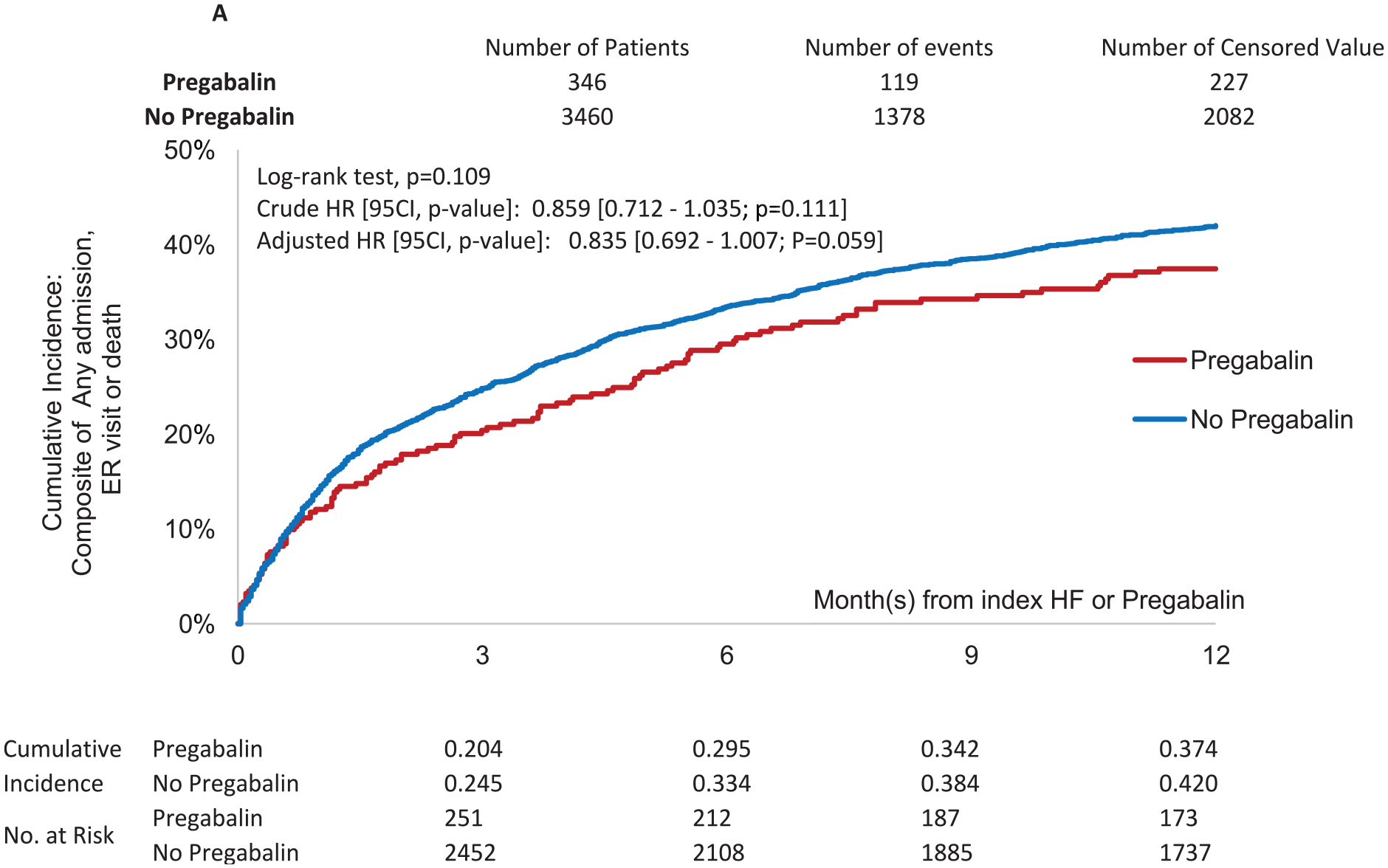
Composite outcome of overall ER visits, hospitalizations, and all-cause mortality of pregabalin users compared with pregabalin nonusers over time.
Discussion
This large single-center cohort study demonstrated no significant HF-related safety profiles associated with pregabalin. The HR estimate for HF-related admissions or death falls within the generally acceptable noninferiority HR margin of 10% to 15% and was insignificant.
Although the statistical inference on the point estimate and 95% CI would be “inconclusive,” the absolute difference in the cumulative incidence of 1.5% suggests that pregabalin appears to be safe for prevalent HF patients. The lack of significant secondary HF risk after pregabalin is consistent with existing literatures that found no increased risk of new-onset HF or exacerbated pre-existing HF in patients taking pregabalin.8,9 Besides the HF-related admissions, the HR of all-cause admission or death pregabalin was 0.835 with an absolute incidence reduction of 4.6%. Although statistically insignificant, this secondary outcome analysis is generally supportive of pregabalin safety in patients with HF.
Although pregabalin pharmacology suggests the agent’s actions may exacerbate HF, these data combined with previous research support the conclusion that pregabalin most often does not result in acute heart failure (AHF). Pregabalin-induced peripheral edema is not associated with laboratory changes suggestive of deterioration in renal or hepatic function. 6 The agent does not have a negative inotropic action nor result in excessive fluid accumulation seen with other agents that cause AHF such as anti-arrhythmic agents or thiazolidinediones. 5
This observational study was subject to confounding factors. This study’s data source is limited to a single center that may limit widespread applicability. However, the outcomes were not different from previously published cohorts that contained greater number of institutions. Assessment of patient adherence to their prescribed medications was limited based on the evaluation of initial prescription orders without long-term prescription adherence data.
Conclusion and Relevance
This large, single-center, cohort study shows pregabalin is not associated with an increased risk of AHF events in patients with pre-existing HF.
Footnotes
Author Contributions
All authors fully contributed to the content of this manuscript, including meeting the 4 criteria of the Internal Committee of Medical Journal Editors. All authors had full access to all the data in the study and take full responsibility for integrity of the work and accuracy of the data analysis, from inception to published article.
Author Disclosures
The authors have no disclosures with the content of this manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the University of Utah Cardiovascular Clinical Pharmacology Research Fund.
Data for Trial
Available with Mark A. Munger, PharmD, Department of Pharmacotherapy, College of Pharmacy, University of Utah Health.
