Abstract
Background:
Chemotherapy, the only treatment option for metastatic triple-negative breast cancer (mTNBC), showed decreased survival rates. Trophoblast cell surface antigen-2 (Trop-2) could be a possible target for antibody-drug conjugates (ADCs).
Objective:
Sacituzumab govitecan (SG), an anti-Trop-2 ADC for pretreating relapsed/refractory mTNBC patients, was studied to know the efficacy and safety profile of the drug in mTNBC.
Methods:
The present review searched MEDLINE (via PubMed), WHO Clinical Trial Registry, Clinical Trials.gov, and Cochrane Central Register of Controlled Trials until December 25, 2022. The studies searched comprised randomized trials and observational studies (retrospective [case-control, cross-sectional] and prospective [cohort designs]). Efficacy assessment was performed in terms of complete response (CR), partial response (PR), objective response rate (ORR), stable disease (SD), progressive disease (PD), and clinical benefit rate (CBR), and safety in terms of adverse events.
Results:
The overall random-effects pooled prevalence of CR was 4.9 (95% CI: 3.2-7.1), PR was 35.6 (95% CI: 31.5-39.9), ORR was 6.8 (95% CI: 5.9-7.8), SD was 8.0 (95% CI: 6.7-9.4), PD was 5.1 (95% CI: 4.1-6.3), and CBR was 13.4 (95% CI: 11.8-15.1). Adverse events associated with the drug were neutropenia, fatigue, anemia, nausea, and others.
Conclusion and Relevance:
This is the first meta-analysis conducted in relapsed/refractory mTNBC patients and found that SG is efficacious but associated with some adverse effects that are related to exposure to the drug. The application of these results will allow clinicians to use SG in the management of patients with mTNBC.
Introduction
A total of 2 261 419 new cases of breast cancer (BC), the second leading cause and often diagnosed cancer, were reported worldwide in 2020. 1 One of the subtypes of BC is triple-negative breast cancer (TNBC), characterized by the absence of estrogen and progesterone receptors as well as a lack of human epidermal growth factor receptor 2 (HER2) gene amplification, 2 exhibiting worst clinical response 3 and a paucity of therapy-targetable molecular targets. 4 It represents 15% of all BC cases and is more common in women older than 40 years, 5 those with a breast cancer susceptibility gene (BRCA) mutation, premenopausal status, obesity, and maternal-related variables such as parity and age at first pregnancy. 6 Due to the lack of hormone and HER2 receptors, targeted therapies are inefficient for treating metastatic TNBC (mTNBC); thus, chemotherapy remains the standard of care. 7 Introduction of immunotherapy in patients with a metastatic disease expressing programmed death ligand 1 (PD-L1) has been approved for use as a neoadjuvant. A combination of immunotherapy and chemotherapy is considered the new treatment for PD-L1 and mTNBC patients. Several cancers are currently being studied using a combination of immunotherapy (immune checkpoint inhibitors) and targeted therapies (poly-ADP-ribose polymerase-1 [PARP] inhibitors). 8 Addition of immunotherapy has led to the development of new therapeutic agents such as PARP inhibitors, tyrosine kinase inhibitors, checkpoint inhibitors, antiandrogens, antibody-drug conjugates (ADCs), and other targeted therapies. 9 However, only about 30% of TNBC patients achieve a complete response (CR), and recurrence and mortality rates remain higher than those for non-TNBC subtypes.10,11 In addition to poor response and survival consequences, patients with localized TNBC have a greater chance of relapse and death during the first 3 years than those with other types of BC associated with substantial toxicity.12-15 As a result, determining the molecular characteristics of TNBC, targeting specific changes in the internal and external tumor environments, and developing new treatment regimens are all immediate concerns that need to be addressed. Protein kinase B (Pan-AKT) inhibitors, bromodomain inhibitors, aurora kinase inhibitors, and checkpoint kinase 1 (CHK1) inhibitors have shown promising results as chemotherapy adjuvants, but these trials are still in the early stages. The treatment for TNBC may have a promising future due to vaccination and hormonal blockade. Vaccinating patients with tumor neoantigens may increase their immunogenicity, making them more responsive to proven immunotherapies.
Trophoblast cell-surface antigen 2 (Trop-2) is overexpressed in 95% of TNBCs but is absent from normal tissue. It is an effective therapeutic target due to its continuous overexpression in TNBC.16,17 Sacituzumab-hziy (aka IMMU-132, Trodelvy) was authorized as a monotherapy in the European Union on November 22, 2021, for the treatment of adult patients with mTNBC who had had 2 or more prior systemic regimens. 6 It is an ADC that targets the Trop-2 protein and is coupled to the topoisomerase-1 inhibitor SN-38. 18 When -hziy (free or conjugated form) binds to Trop-2, it is internalized and distributes SN-38 – 7-ethyl-10-hydroxy camptothecin (SN-38) into the tumor cell, both intracellularly and in the tumor microenvironment. Sacituzumab govitecan (SG)-bound tumor cells are killed via intracellular SN-38 uptake, while nearby tumor cells are killed by extracellular SN-38 release.19,20 Topoisomerase inhibitors-1 have shown effectiveness in the treatment of mTNBC, but their use has been limited due to inadequate intratumor exposure and dose-limiting toxicities. 21
A significant clinical response was reported after the administration of SG in clinical studies with mTNBC patients who had received a median of 3 previous regimens. The response rate was 33.3% (complete vs partial [3 vs 33]), the median duration of response was 7.7 months, and the OS was 13 months. However, the drug’s adverse effects include anemia and neutropenia. 17 A phase 1/2 trial with SG in mTNBC patients found an increase in objective response rate (ORR) of 33%, median progression-free survival (PFS) of 5.5 months, and median overall survival (OS) of 13 months. Nausea, anemia, neutropenia, fatigue, and other common adverse effects were noted.22,23 Sacituzumab govitecan also demonstrated significant activity in pretreated mTNBC patients in a phase 1/2 basket study, which was later verified in a randomized phase 3 trial versus single-agent chemotherapy of physician’s choice. 17 Based on these encouraging findings, SG was considered an impressive drug in patients with mTNBC; however, significant concerns were raised about the drug’s role in causing adverse effects, as this drug has been linked with toxicities such as neutropenia, nausea, anemia, and some others. However, the utility of topoisomerase-1 inhibitors as a treatment for mTNBC is limited due to inadequate intratumor exposure and dose-limiting toxicities.
In this systematic review, we discuss the efficacy and safety of SG in patients with refractory mTNBC. We hypothesized that SG has a good response rate in relapsed/refractory mTNBC patients with a better safety profile in relation to other options available for the treatment of mTNBC.
Methods
Pointers from Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) for systematic reviews 24 and meta-evaluation of observational research in epidemiology (MOOSE) guidelines 25 were used for developing, conducting, and reporting this systematic literature review. PROSPERO registration number CRD42022370858 was issued to the study.
Data Sources and Searches
We reviewed through Medline (using PubMed), the WHO Clinical Trial Registry, Clinical Trials.gov, and the Cochrane Central Register of Controlled Trials until December 25, 2022. We looked for “Safety and Efficacy with Sacituzumab govitecan and Triple Negative Breast Cancer”, “Efficacy and Sacituzumab govitecan”, and “Safety and Sacituzumab govitecan” keywords. We also examined gray literature using Google Scholar and a reference list of relevant articles. The data search was carried out by the principal author (M.K.), who verified the publications for eligibility. All the listed articles were double-checked, and any differences were arbitrarily settled by senior reviewers (N.K. and A.S.).
Inclusion and Exclusion Criteria
This review covered studies that looked at the efficacy and safety of SG in patients with TNBC. The studies comprised randomized trials as well as observational studies such as retrospective (case-control, cross-sectional) and prospective (cohort designs). Reviews, editorials, case studies, letters, meta-analyses, consensus reports, research written in languages other than English, full-text articles that were unavailable, and research that lacked the necessary information were all eliminated.
Quality Assessment
To assess the quality of data in the included studies, 2 authors adopted the US National Institutes of Health (NIH) Quality Assessment Tool 26 developed by the National Heart, Lung, and Blood Institute from the National Institute of Health. It was chosen because it is comprehensive and widely used for a thorough examination of data quality. The tools were designed to assist reviewers in focusing on topics relevant to the critical evaluation of a study’s internal validity, rather than to provide a list of items that go into generating a numerical score. The instruments for analyzing potential problems in study techniques or applications included things such as bias sources (eg, patient selection, performance, attrition, and detection), confounding, study power, the strength of causality in the link between interventions and outcomes, and other considerations. Quality reviewers may answer each question on the tool with “yes,” “no,” or “cannot determine/not reported/not applicable.”
Outcomes
The main aim of the study was to assess the efficacy and safety profile of SG; used as a second-line treatment. Efficacy assessment was performed in terms of CR, partial response (PR), ORR, stable disease (SD), progressive disease (PD), and clinical benefit rate (CBR) in patients with metastatic TNBC after administration of the drug, and a safety assessment was performed in terms of severity of adverse events.
Data Extraction
A primary author (M.K.) input data into a standardized data-extraction table (Microsoft Excel), which was then double-checked for accuracy by a senior author (N.K. and A.S.). The first author’s name, year of publication, study design, age, gender, presence or absence of a BRCA mutation, prior regimens, adverse events (≥grade 3), and response rate (CR, PR, OS, and so on) were all collected from the articles.
Data Synthesis
All eligible studies that met the inclusion criteria will be included for quantitative synthesis of efficacy and safety measures. If uniformity is found in providing the result estimates among the trials, a meta-analysis will be done. Individually calculated rates from observational studies will be determined using pooled random-effect models. Cochran Q-test with a P value < 0.1 and I2 greater than 50% will be used to investigate the potential source of heterogeneity related to both methodological and clinical aspects of the studies. Publication bias will be addressed if there are more than 10 articles for each outcome. For the effect of study-level variables, a P value of <0.05 will be regarded as statistically significant. Stata v16.1 will be used for statistical analysis (Stata Corp, College Station, Texas). A P value <0.05 will be regarded as statistically significant. Medcalc Version 23.0 will be used for statistical analysis.
Results
Study Characteristics
After a comprehensive search using the keywords “Safety and Efficacy and Sacituzumab govitecan and Triple Negative Breast Cancer”, “Safety and Sacituzumab govitecan”, and “Efficacy and Sacituzumab govitecan”, a total of 85 articles were retrieved. After filtering the data, 67 records were screened based on title/abstract, and 46 articles were selected as possible publications for full-text screening. Thirty-one publications were rejected due to the following reasons: wrong drug (n = 2), wrong result (n = 4), wrong population (n = 5), review (n = 16), editorial (n = 1), and wrong study design (n = 3). In consideration of predefined inclusion and exclusion criteria, a total of 5 studies were included in this systematic review (Figure 1). The study included 633 individuals, and 628 of them were female. The baseline parameters of the studies included in our analysis are listed in Table 1.
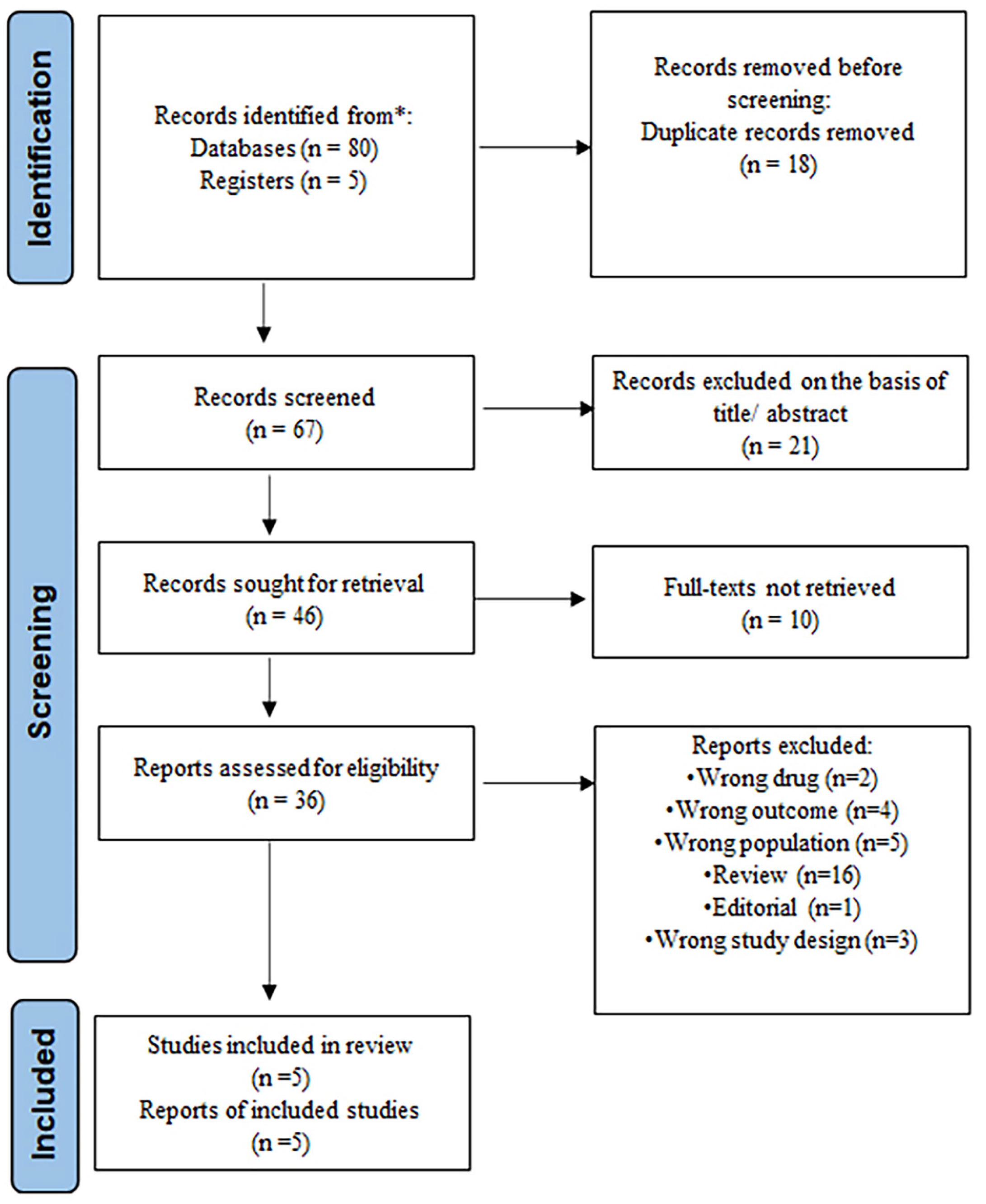
PRISMA flow diagram of the number of studies screened and included in the systematic review.
Characteristics of the Studies Included in the Meta-analysis.
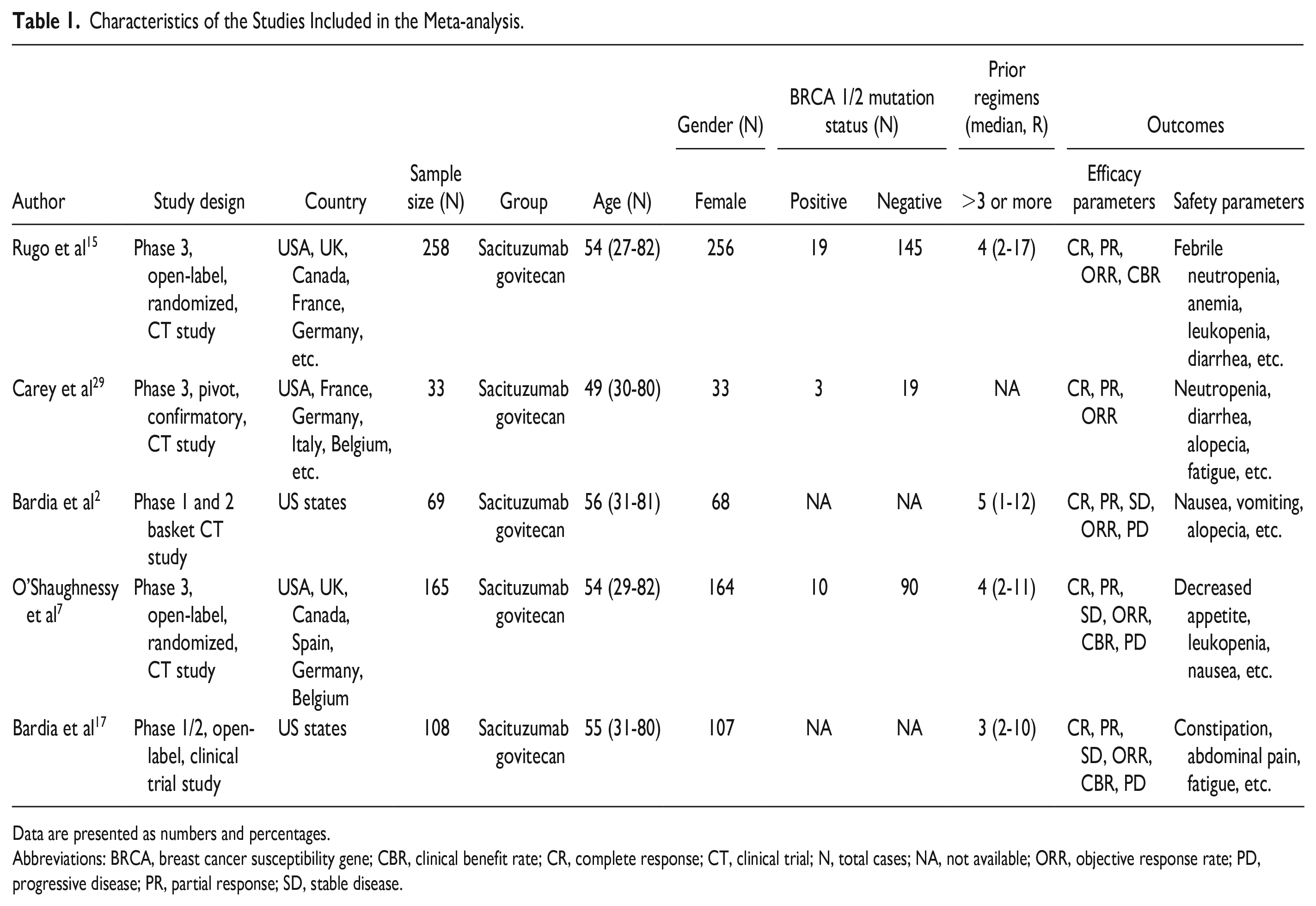
Data are presented as numbers and percentages.
Abbreviations: BRCA, breast cancer susceptibility gene; CBR, clinical benefit rate; CR, complete response; CT, clinical trial; N, total cases; NA, not available; ORR, objective response rate; PD, progressive disease; PR, partial response; SD, stable disease.
Quality Assessment
We assessed the data quality in the included studies using NIH quality assessment methods (Supplementary Table S1). According to the quality assessment, most of the included studies (5 in total) were of acceptable quality. In 91% of the studies, the research subject or purpose was explicitly stated. All research used inclusion/exclusion criteria and explicitly specified the population. In all the research, the subjects were chosen from the same or similar demographics.
Efficacy Assessment of SG
We assessed the efficacy of SG in terms of CR, PR, ORR, SD, PD, and CBR. Five studies reported CR rates in 25 subjects. The overall random-effects pooled prevalence of CR was 4.9 (95% CI: 3.2-7.1). Cochran Q test showed that there is no difference in the effect size of individual studies included in this systematic review, and the heterogeneity (I2 = 48.54%) is moderate (Figure 2). The overall random-effects pooled prevalence of PR was 35.6 (95% CI: 31.5-39.9). Cochran Q test showed that there is a difference in the effect size of individual studies included in this systematic review, and the heterogeneity (I2 = 91.59%) is substantial. Similarly, the overall random-effects pooled prevalence of ORR, SD, PD, and CBR were 6.8 (95% CI: 5.9-7.8), 8.0 (95% CI: 6.7-9.4), 5.1 (95% CI: 4.1-6.3), and 13.4 (95% CI: 11.8-15.1), respectively. Cochran Q test showed that there is a difference in the effect size of individual studies included in this systematic review, and the heterogeneity is moderate to substantial in the abovementioned parameters (Figure 2) (Supplementary Table S2).
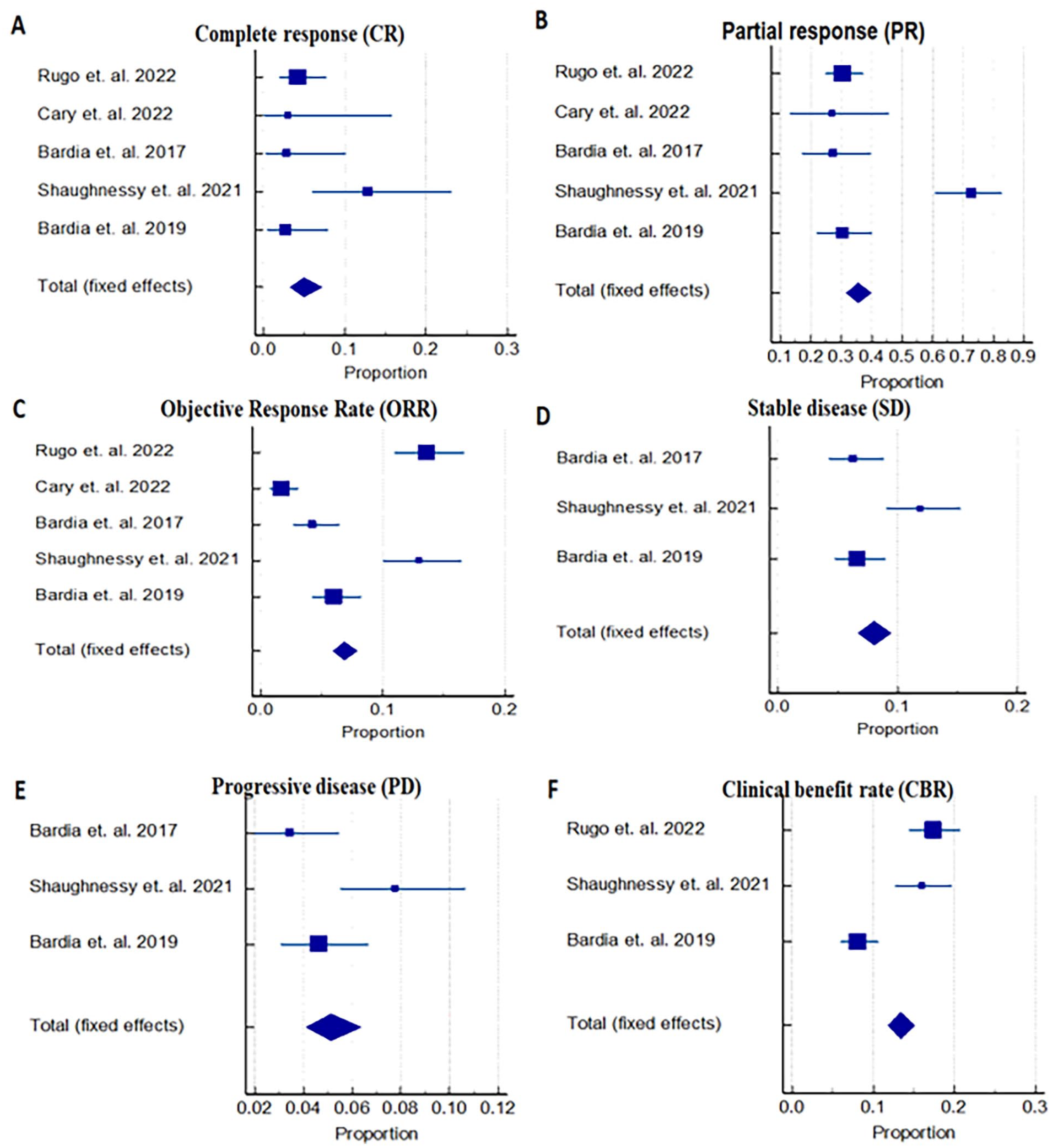
Forest plots represent the pooled prevalence of efficacy in terms of A: complete response; B: partial response; C: objective response rate; D: stable disease; E: progressive disease. F: clinical benefit rate with the administration of Sacituzumab govitecan.
Safety Assessment of SG
The safety of SG was assessed in 5 studies which enrolled a total of 633 subjects. The overall random-effects pooled prevalence of safety parameters is listed in Table 2. Neutropenia, anemia, fatigue, nausea, and so on were among the commonly observed toxicities associated with SG. Cochran Q test showed that there is a difference in the effect size of individual studies included in this systematic review, and the heterogeneity is moderate (I2 = 98.85%) to substantial.
Pooled Prevalence of Adverse Events Associated With Sacituzumab Govitecan Administration in the Study Subjects.
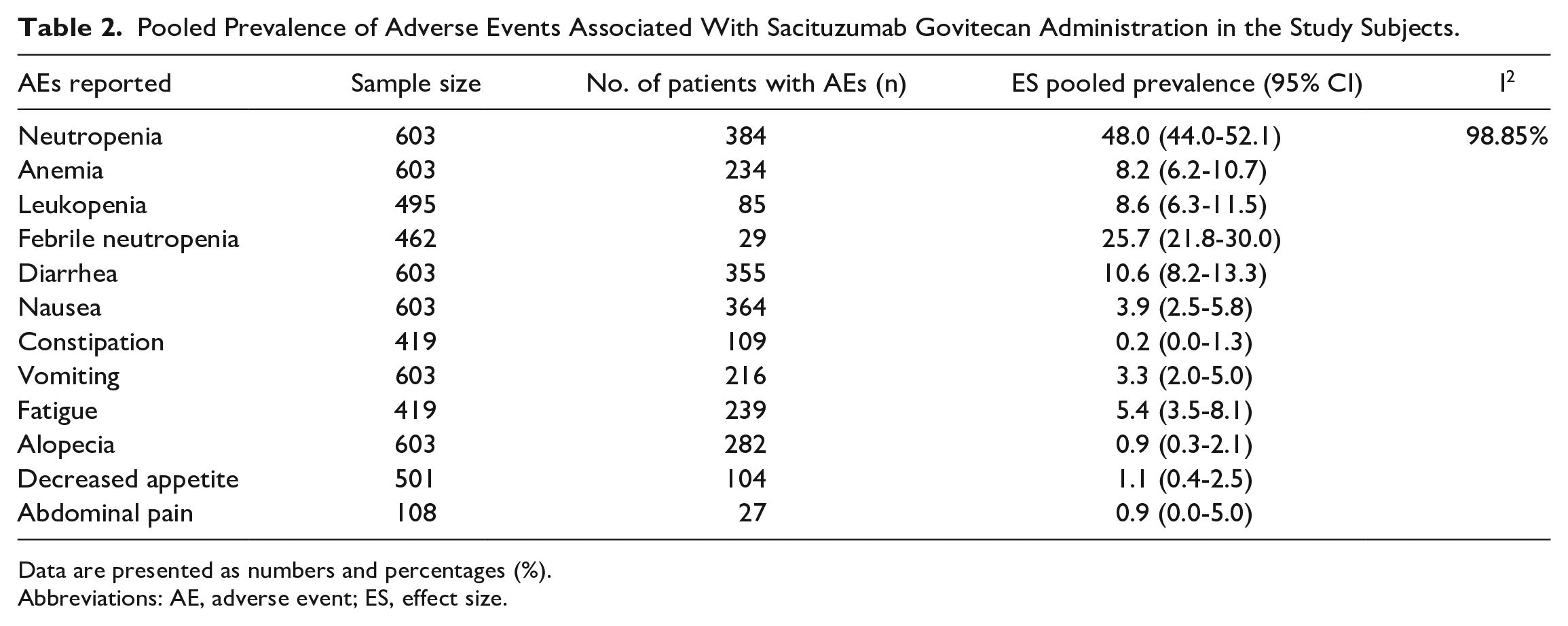
Data are presented as numbers and percentages (%).
Abbreviations: AE, adverse event; ES, effect size.
Discussion
The present systematic review and meta-analysis included 5 studies with 633 relapsed/refractory mTNBC patients to assert SG as a second-line treatment therapy and make a good clinical decision by evaluating its efficacy and safety characteristics. When assessing adverse episodes throughout studies, the heterogeneity (I 2 ) was greater than 95%; thus, no definitive conclusion could be drawn. Our findings suggest that SG is effective in patients with relapsed or refractory mTNBC; however, there are a few toxic adverse effects associated with the drug, such as neutropenia, anemia, diarrhea, and so on: Nevertheless, these toxicities are linked to the duration of drug exposure.
Sacituzumab govitecan, an ADC consists of SN-38, a topoisomerase-1 inhibitor and anti-Trop 2 humanized monoclonal antibody conjugated with it via a specific hydrolyzable linker. This interaction enables the release of cytotoxic SN-38 in the acidic tumor microenvironment without the necessity for internalization and subsequent enzymatic cleavage, facilitating “bystander effect” tumor cell killing. Its drug antibody allows the release of high localized SN-38 concentrations from SG after binding.27,28
In terms of efficacy, we studied the included trials’ CR, PR, SD, ORR, CBR, and PD parameters and found that 4.9% of patients attained CR, 8% attained SD, and 35.6% attained PR out of the entire population. Similarly, 6.8% of patients achieved ORR, 5.1% achieved PD, and 13.4% achieved CBR after receiving SG. A pivotal, phase 3 ASCENT clinical trial found a significant improvement in the SG therapy group of severely pretreated mTNBC patients, specifically with ORR (35%), CBR (45%), and median duration of response (6.3 months). 17 The ASCENT trial’s subgroup analyses revealed that patients treated with SG had a greater ORR (30%), 1 patient had CR, 9 patients had PR, and the CBR was 42%. 29 A phase 3 randomized, open-label clinical trial in 529 patients with mTNBC who had previously undergone at least 2 conventional chemotherapies reported higher ORR (31%) and median PFS (4.6 months) (hazard ratio 0.48; 95% CI: 0.32-0.72) in the SG treatment arm. 7 The present research found similar results for the efficacy measures following SG administration. ASCENT clinical trial (Trial of Sacituzumab Govitecan in Participants With Refractory/Relapsed Metastatic Triple-Negative Breast Cancer).
In terms of safety, we evaluated the adverse events related to the drug commonly reported in the included studies. Most SG patients experienced treatment-related adverse effects (TRAEs) of grade 3, including neutropenia, diarrhea, anemia, and febrile neutropenia. The toxicities associated with irinotecan are neutropenia and diarrhea, which are caused by its active metabolite, SN-38, which is the cytotoxic load of SG.30-32 Six hundred sixty patients participating in a pivotal, randomized, open-label, phase 3 study, IMMU-132-05 (ASCENT), who had received a single dose of SG at 10 mg/kg reported TRAEs including diarrhea (65.1%) and neutropenia (64.0%), followed by nausea (62.4%), fatigue (51.6%), alopecia (46.9%), anemia (39.5%), and vomiting (33.3%). 17 Another clinical trial found that the most common TRAEs in subgroups of ASCENT trial patients who had received 1 line of therapy prior included neutropenia (76%), alopecia (70%), diarrhea (58%), nausea (52%), anemia (39%), and vomiting (36%) during their treatment. 29 A phase 3 randomized, open-label clinical trial in 529 patients with mTNBC who had previously undergone treatments found the most common grade 3 TRAEs were neutropenia (8%), leucopenia (12%), anemia (8%), and diarrhea (7%). 7 Our data showed that neutropenia, anemia, diarrhea, fatigue, and some other adverse effects were the most common ones in patients who are taking SG, which is consistent with other studies’ findings and may be a risk factor for the development of TNBC, but it is linked to the duration of drug exposure.
A single-arm, multicentric trial in 69 patients with relapsed/refractory mTNBC who had received a starting dose of 10 mg/kg on days 1 and 8 of 21-day repeated cycles found a 30% confirmed ORR (PR, n = 19; CR, n = 2) and a 46% CBR (CR + PR + SD ≥ 6 months). Adverse events reported in those patients were predominantly of grade 3 or higher, including neutropenia (39%), leukopenia (16%), anemia (14%), and diarrhea (13%); the prevalence of febrile neutropenia was 7%. 2 Another phase 1/2 single-arm, multicenter clinical trial with 108 female patients having mTNBC and undergone at least 2 regimens before administration of the test drug was conducted. There were 3 CRs and 33 PRs noted, with an ORR of 33.3% (95% CI: 24.6-43.1) and clinical benefit (95% CI: 35.8-55.2). Nausea (67%), neutropenia (64%), diarrhea (62%), fatigue (55%), anemia (50%), and vomiting (49%) were the most common adverse effects observed. However, only 8% of patients had febrile neutropenia. No treatment-related deaths were reported. 17
Although toxicity is associated with the drug, it can be managed with dose alterations. The advantage of SG treatment outweighed the toxicity given the significant improvement in OS and CBR. As a result, we can conclude that SG is a successful and safe medication that can be used as a second-line treatment for mTNBC. Our findings show that SG is a novel therapeutic that can be utilized as a second-line treatment for patients with relapsed or refractory mTNBC.
Limitations
This study, however, has some shortcomings. To begin with, there was no comparator group in any of the trials chosen. Second, the heterogeneity revealed by the studies was substantial in the majority of the included research variables. Third, due to the differences in sample size among the studies, an acceptable inference regarding efficacy and safety could not be reached. And it is likely that we might have overlooked important publications by omitting studies written in languages other than English while searching data.
Conclusion and Relevance
This study highlighted the efficacy and safety profile of the SG drug as a second-line treatment for relapsed/refractory mTNBC. From the results, we can infer that SG is efficacious when compared to other treatment options available but is associated with some of the adverse events that are related to drug exposure. The effect size of all included studies in a meta-analysis along with the heterogeneity index (I2) had been used to determine the significant impact of the individual articles.
Overall, our study provides more statistical data about SG that would allow clinicians to use SG in the management of patients with mTNBC along with other therapies in order to have a better prognosis and not to burden the patient with discomfort, pain, and high cost.
It is the first meta-analysis on the efficacy and safety of SG analyzed in relapsed/refractory mTNBC patients. Thus, large-scale prevalence trials demonstrating SG’s efficacy and safety are required to address important gaps in understanding the disease better.
All authors have approved the final manuscript.
Supplemental Material
sj-docx-1-aop-10.1177_10600280231164110 – Supplemental material for Sacituzumab Govitecan as a Second-Line Treatment in Relapsed/Refractory Metastatic Triple-Negative Breast Cancer Patients: A Systematic Review and Meta-analysis
Supplemental material, sj-docx-1-aop-10.1177_10600280231164110 for Sacituzumab Govitecan as a Second-Line Treatment in Relapsed/Refractory Metastatic Triple-Negative Breast Cancer Patients: A Systematic Review and Meta-analysis by Meghavi Kathpalia, Anurag Sharma and Navkiran Kaur in Annals of Pharmacotherapy
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported in part by the Indian Council of Medical Research (ICMR) under the project titled “Biological significance of disease specific sialoglycosylation in triple negative Breast Cancer” (project no. 5/13/47/2020-NCD-III).
Ethics Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Data Availability Statement
The author confirms that the data supporting the findings of the study are available within the article (and/ or its supplementary material).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
