Abstract
Background:
The optimal loading dose of digoxin in patients with reduced kidney function is unknown. Tertiary references recommend reduced loading doses; however, these recommendations are based on immunoassays that are falsely elevated by the presence of digoxin-like immunoreactive substances, a problem that is minimized in modern assays.
Objective:
To determine whether chronic kidney disease (CKD) or acute kidney injury (AKI) is associated with supratherapeutic digoxin concentrations after a digoxin loading dose.
Methods:
A retrospective analysis on patients who received an intravenous loading dose of digoxin with a digoxin concentration collected 6 to 24 hours after the end of the dose. Patients were stratified into 3 groups: AKI, CKD, and non-AKI/CKD (NKI) based on glomerular filtration rate and serum creatinine. The primary outcome was frequency of supratherapeutic digoxin concentrations (>2 ng/mL) and secondary outcomes included frequency of adverse events.
Results:
A total of 146 digoxin concentrations were included (AKI = 59, CKD = 16, NKI = 71). Frequencies of supratherapeutic concentrations were similar between groups (AKI: 10.2%, CKD: 18.8%, NKI: 11.3%; P = 0.61). Pre-planned logistic regression demonstrated no significant relationship between kidney function group and the development of a supratherapeutic concentration (AKI: odds ratio [OR]: 1.3, 95% confidence interval [CI]: 0.4-4.5; CKD: OR 4.3, 95% CI: 0.7-23).
Conclusion and Relevance:
This is the first study in routine clinical practice evaluating the relationship between kidney function and digoxin peak concentrations that differentiates AKI from CKD. We did not find a relationship between kidney function and peak concentrations; however, the group with CKD was underpowered.
Background
Digoxin, a Na+/K+ ATPase inhibitor, is used in heart failure, atrial fibrillation, and supraventricular tachycardia. Goal therapeutic concentrations vary by indication; however, there is a correlation between increased digoxin concentrations and the risk of adverse events including digitalis toxicity. 1 Signs of toxicity associated with digoxin include nausea, vomiting, visual disturbances, hyperkalemia, and cardiac arrhythmias. 2
Loading doses of digoxin can be administered intravenously for rapid achievement of therapeutic concentrations and are typically administered for acute rate control in atrial fibrillation. The labeled loading dose in the manufacturer’s package insert is 8 to 12 µg/kg of ideal body weight (IBW) with roughly half the total given as the first dose and additional fractions at 6- to 8-hour intervals. 2 Some tertiary drug information sources recommend to decrease the digoxin loading dose by 50% when administered to a patient with reduced kidney function while others do not.3,4 This recommendation is not in the manufacturer prescribing information, but is a result of studies using older immunoassays to determine serum digoxin concentrations. 5 However, these immunoassays are influenced by the presence of digoxin-like immunoreactive substances (DLISs). 6 DLIS are present in patients with kidney disease, hepatic impairment, pregnancy, and neonates leading to falsely elevated digoxin concentrations. 6 It may have been these falsely elevated digoxin concentrations that resulted in recommendations to decrease loading doses. Updated assays may be less affected by the presence of DLIS, and is it unknown if there is any true relationship between kidney function and peak concentrations after digoxin loading doses.7-11
Objectives
The purpose of this study is to determine whether chronic kidney disease (CKD) or acute kidney injury (AKI) is associated with supratherapeutic digoxin concentrations in patients that receive a digoxin loading dose.
Methods
Design
This was a retrospective cohort study of patients receiving an intravenous (IV) loading dose of digoxin at an 886-bed academic medical center between January 2013 and September 2019. The study was deemed exempt by the institutional review board.
Participants
Patients at least 18 years of age were identified via the electronic medical record as having received an IV loading dose of digoxin. As digoxin loading doses are administered in a partitioned manner, patients were included if they had a serum digoxin concentration within 48 hours of the first and within 6-24 hours of the last partitioned loading dose. Patients were excluded if they had previously received digoxin within 14 days of the loading dose, a serum creatinine was not recorded within 72 hours of the loading dose, the medical record was missing height, weight, or baseline serum creatinine within the past year, weight was not between 40 and 120 kg or were pregnant. Finally, patients were excluded that met both the criteria for acute and chronic kidney disease; this decision was made to avoid contamination between the renal function groups.
Patients were categorized based on renal function into 1 of 3 groups, AKI, CKD, or non-AKI/CKD (NKI). Patients were included in the AKI group based on Acute Kidney Injury Network (AKIN) classification accounting for percent change in serum creatinine from baseline. 12 Baseline serum creatinine was the lowest measurement within 12 months to 24 hours prior to admission and was compared to the closest serum creatinine recorded prior to the start of the digoxin loading dose. Those with an estimated glomerular filtration rate (eGFR), as calculated by the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) equation, of less than 30 mL/min/1.73 m2 at baseline and digoxin administration were included in the CKD group along with those with pre-admission dialysis. 13 Those who did not meet criteria for AKI or CKD were included in the NKI group.
Digoxin Assay
The serum digoxin assay was performed on the Roche cobas ® 8000 modular analyzer with an institutional reference range of 0.8 to 2.0 ng/mL. The COBAS® INTEGRA Digoxin test is a kinetic interaction of microparticles in solution (KIMS) homogeneous immunoassay based on the principle of measuring changes in scattered light or absorbance which result when activated microparticles aggregate. 10
Data Collection
Data collection included patient demographics such as age, sex, weight, baseline kidney function, baseline potassium and magnesium concentrations, digoxin dose, total digoxin loading dose (including number of doses), and timing of first and last loading dose and digoxin concentration. Other medications administered (eg, beta blockers, calcium channel blockers, antiarrhythmics, vasopressors/inotropes) within 24 hours before and after the digoxin concentration, the occurrence a supratherapeutic digoxin concentration between 6 and 24 hours after the last dose of the digoxin loading dose, and minimum and maximum heart rate within 24 hours from the digoxin concentration were captured. In patients with a digoxin concentration greater than or equal to 1 ng/mL, documented signs of symptomatic toxicity such as nausea/vomiting, blurred vision, headache, or confusion within 24 hours of the digoxin concentration were collected.
Two investigators collected the data using standardized electronic collection tool (REDCap [research electronic data capture]) and data dictionary. One investigator collected a portion of the data and the other investigator a separate portion of the data for each patient instead of dividing up full patient data collection to reduce variability between patients. The senior investigator was available to clarify any questions related to the data collection points to ensure standardized data collection.
Outcomes
The primary outcome was to compare the frequency of supratherapeutic digoxin concentrations (concentration greater than 2 ng/mL) across groups. Secondary outcomes included evaluating risk factors for the development of supratherapeutic digoxin concentrations and rates of adverse events such as bradycardia and digitalis toxicity. In order to capture clinically relevant bradycardia, it was defined in this study as a minimum heart rate of less than or equal to 50 beats per minute. In those patients with a digoxin concentration greater than or equal to 1 ng/mL, possible signs and symptoms of digitalis toxicity were obtained from chart review for key words including: “nausea,” “vomiting,” “blurred vision,” “headache” and/or “confusion” within 24 hours of the digoxin concentration.
Outcome Analysis
All descriptive statistics are reported as medians with interquartile ranges (IQRs) or number (percent). Comparisons between groups were made using the Kruskal-Wallis test and Fisher exact test. A logistic regression model was planned with occurrence of supratherapeutic digoxin concentration as the outcome of interest. Variables included in the regression analysis were pre-specified and included the number of hours between last partitioned loading dose and serum digoxin concentration, the dose of digoxin per kilogram IBW, and the kidney function grouping. Two pre-planned sensitivity analyses were conducted, the first included only patients who received a loading dose of 8 to 12 µg/kg of IBW (the labeled loading dose), and the second reclassified all patients with eGFR less than 60 mL/min/1.73 m2 in the CKD group. All analyses were performed using R 3.6.2 (R Core Team (2019). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/.)
Results
A total of 637 patient encounters were identified, 238 met inclusion criteria and 146 were included in the final analysis (NKI = 71, AKI = 59, CKD = 16) (Figure 1). Baseline demographics are displayed in Table 1. Baseline eGFR and serum creatinine across the AKI and NKI groups were similar. Most patients in the CKD group were dialysis dependent at pre-admission baseline. There were no differences among groups in digoxin dosing or timing of digoxin concentrations. The CKD group was younger and had less vasopressor/inopressor use at baseline compared to the other groups (Table 1).
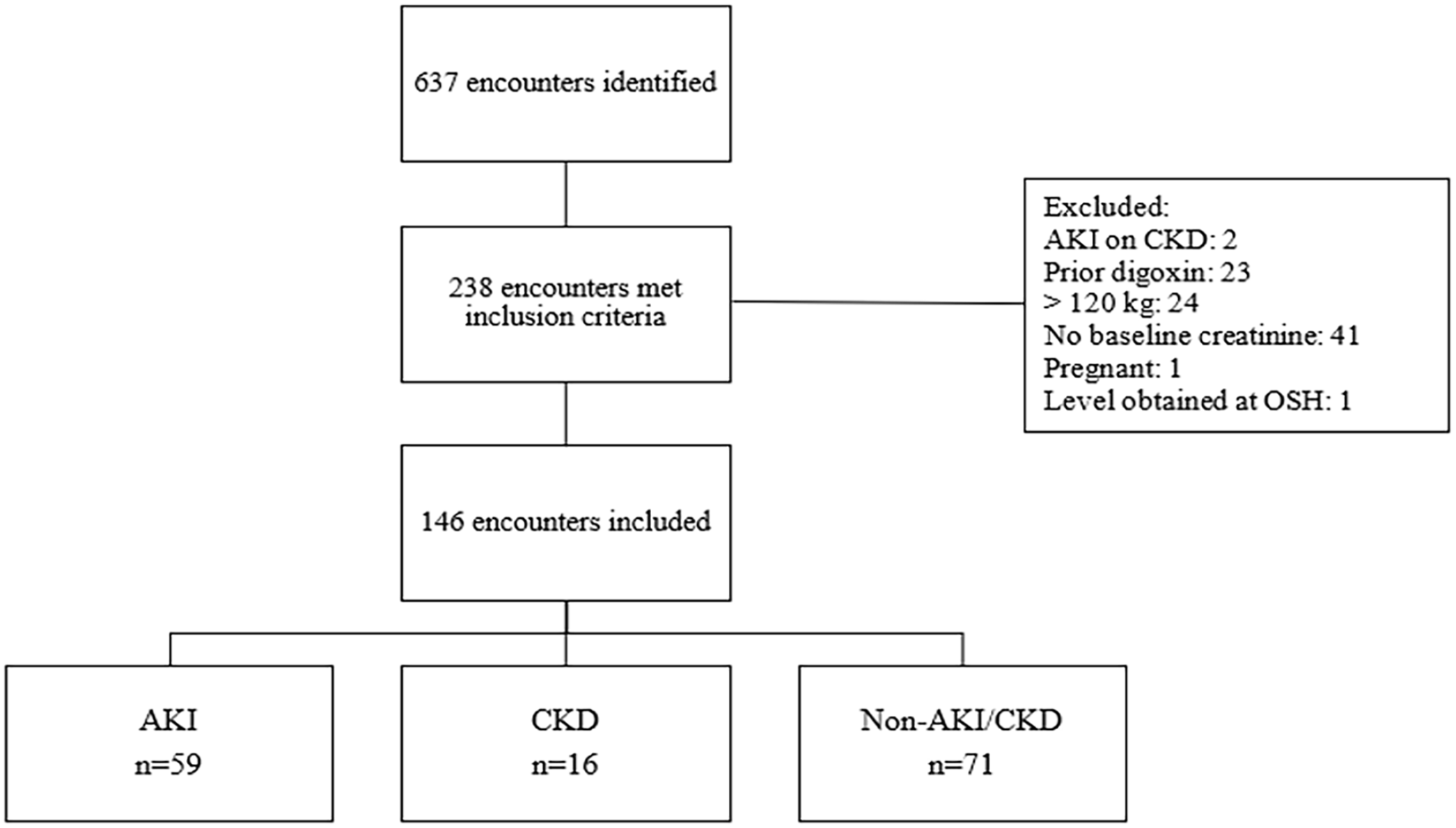
Included and excluded encounters.
Baseline Demographics and Treatment Characteristics.
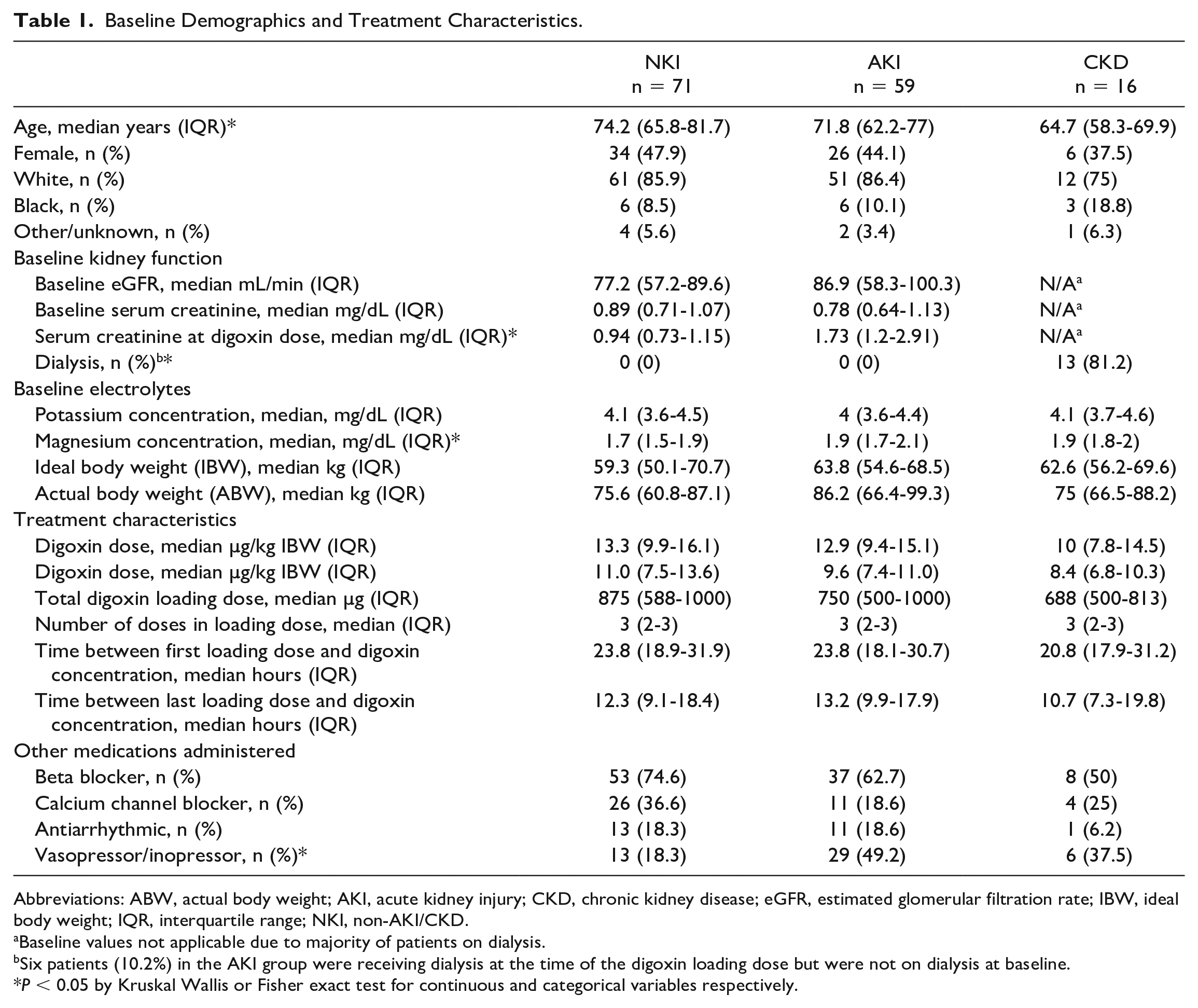
Abbreviations: ABW, actual body weight; AKI, acute kidney injury; CKD, chronic kidney disease; eGFR, estimated glomerular filtration rate; IBW, ideal body weight; IQR, interquartile range; NKI, non-AKI/CKD.
Baseline values not applicable due to majority of patients on dialysis.
Six patients (10.2%) in the AKI group were receiving dialysis at the time of the digoxin loading dose but were not on dialysis at baseline.
P < 0.05 by Kruskal Wallis or Fisher exact test for continuous and categorical variables respectively.
For the primary outcome, there was no difference in the frequency of supratherapeutic concentrations among the 3 groups (Table 2). Adverse events were rare and the incidences were similar across all groups. The most common adverse events were central nervous system disturbances/vision changes and nausea/vomiting. No doses of digoxin immune Fab were administered during the study period (Table 2).
Primary and Safety Outcomes.
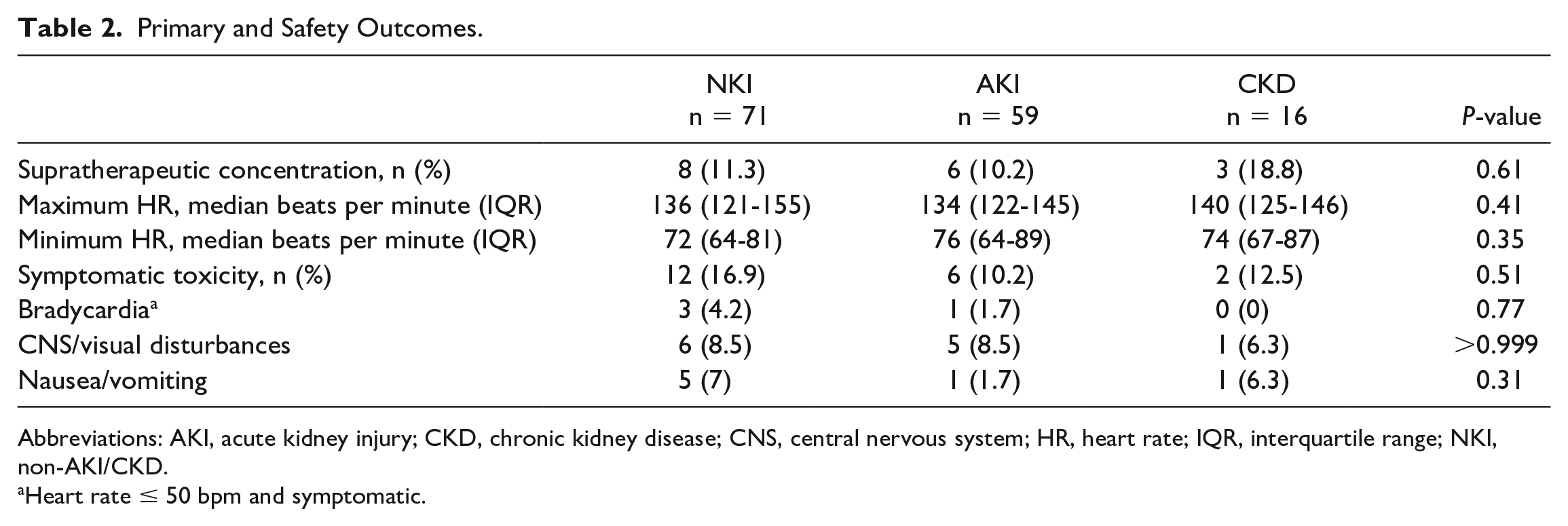
Abbreviations: AKI, acute kidney injury; CKD, chronic kidney disease; CNS, central nervous system; HR, heart rate; IQR, interquartile range; NKI, non-AKI/CKD.
Heart rate ≤ 50 bpm and symptomatic.
A univariate analysis was conducted on patients who developed a supratherapeutic concentration versus those who did not (Table 3). Dose in µg/kg of both IBW and actual body weight were associated with a supratherapeutic level. Patients who developed a supratherapeutic level had lower IBW, but this difference was not found to be statistically significant. There were no differences between groups in occurrence of bradycardia or vasopressor use.
Characteristics Based on Digoxin Concentration.
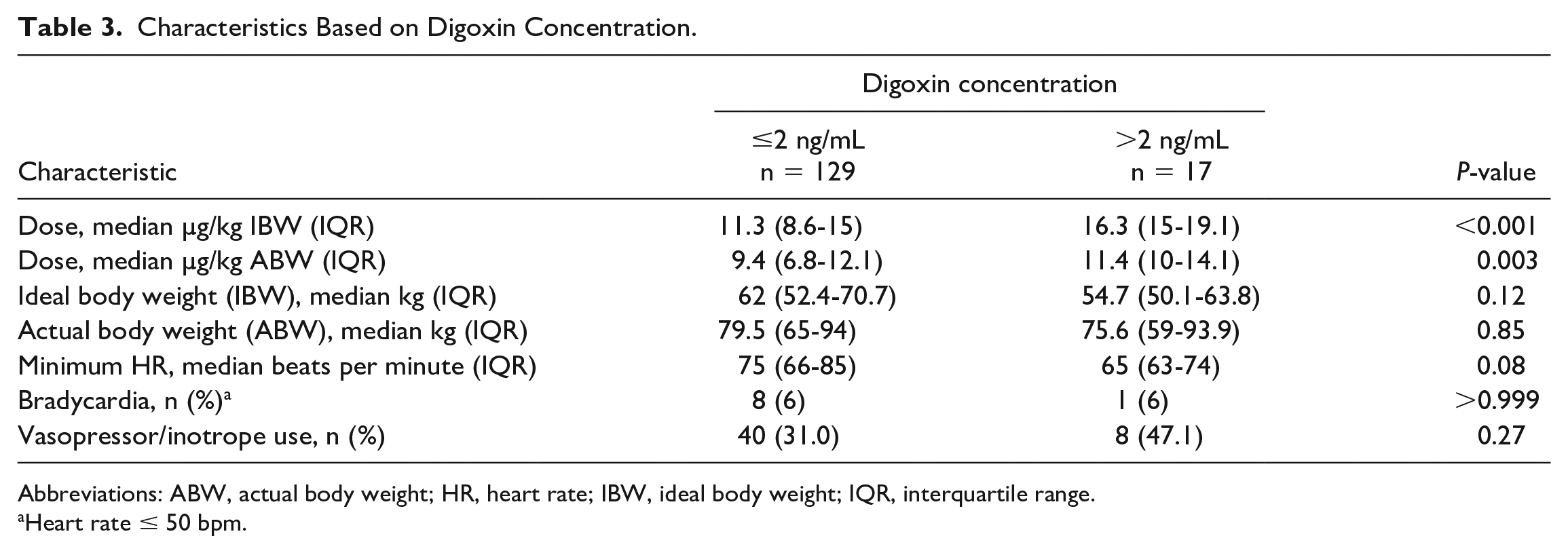
Abbreviations: ABW, actual body weight; HR, heart rate; IBW, ideal body weight; IQR, interquartile range.
Heart rate ≤ 50 bpm.
A logistic regression was performed evaluating the outcome of supratherapeutic digoxin concentration (Table 4). Covariates were pre-selected and included kidney function group and digoxin dose in µg/kg of IBW. We planned to include time from end of loading dose to concentration measurement; however, this had minimal impact on the model and in the interest of parsimony, this covariate was not included. The odds of developing a supratherapeutic concentration were not different between groups (AKI: odds ratio [OR]: 1.3 [95% confidence interval [CI]: 0.4-4.5], CKD: OR 4.3 [95% CI: 0.7-23.0], NKI: referent). There was a significant relationship found between dose (µg/kg of IBW) and development of a supratherapeutic level (OR 1.3 [95% CI: 1.1-1.5]).
Logistic Regression.
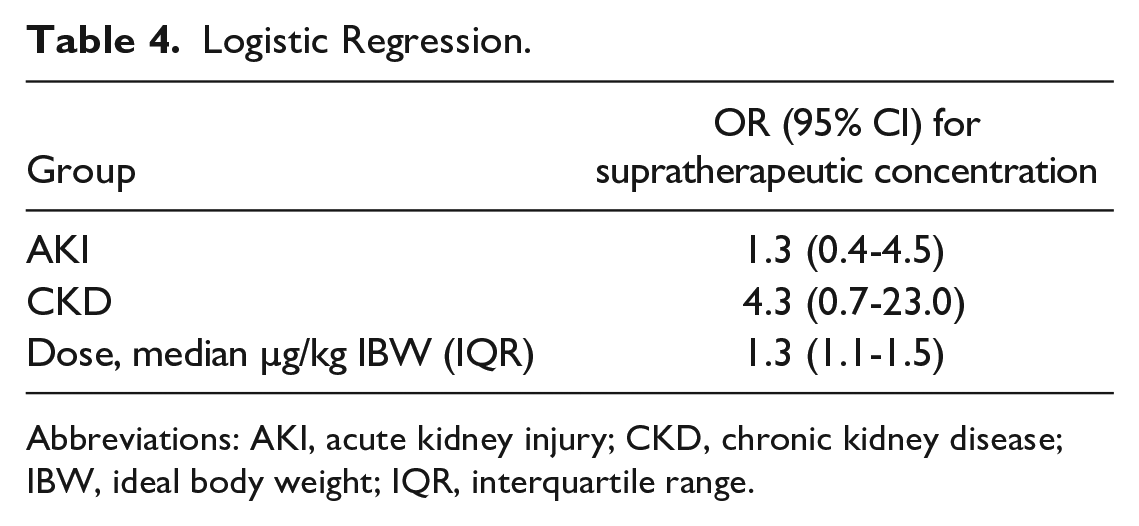
Abbreviations: AKI, acute kidney injury; CKD, chronic kidney disease; IBW, ideal body weight; IQR, interquartile range.
The pre-planned sensitivity analysis included only those who received digoxin 8 to 12 µg/kg of IBW resulted in no supratherapeutic levels and no further evaluation could be performed. The second sensitivity analysis changing the eGFR cutoff in the CKD group to 60 mL/min/1.73 m2 increased the number of patients in that group from 16 to 36; however, there remained no difference in the primary outcome. Repeating the logistic regression demonstrated a significant difference in the odds of developing a supratherapeutic level in the CKD group when accounting for dose (µg/kg IBW), (OR: 4.95 [95% CI: 1.19-24.15]); however, there was no difference in the AKI group (OR: 2.10 [95% CI: 0.51-9.79]).
Discussion
We found no relationship between kidney function and digoxin concentrations after a loading dose in a broad range of patients, including those with AKI and CKD. Prior to this evaluation, most studies exploring the optimal loading dose of digoxin in reduced kidney function originated in the 1970s. 5 A limitation of these studies was use of immunoassays that were later found to be influenced by the presence of DLIS, resulting in falsely elevated serum digoxin levels. 6 The most recent study exploring this question was conducted by Pawlosky et al 14 who concluded a reduced loading dose of digoxin is necessary in patients with reduced kidney function. This study used a creatinine clearance cutoff of less than 60 mL/min to define each group but did not separate patients with CKD from those with AKI, which may have resulted in differing conclusions.
We had a low incidence of adverse events with no differences between renal function groups. Fifty three percent of patients with a supratherapeutic concentration developed symptomatic toxicity, similar to what is reported in the prescribing information. 2 Concomitant medication use was also similar between groups, with the exception of a higher rate of vasopressor/inopressor use in the AKI group. This likely reflects an increase in baseline acuity of illness.
Our initial sensitivity analysis demonstrated that regardless of kidney function, no patients who received 8 to 12 µg/kg IBW of digoxin for a loading dose developed a supratherapeutic concentration. However, the median loading dose of digoxin was outside this recommended range in 2 of our 3 groups. It is a common institutional practice to prescribe a digoxin loading dose of 500, 750, or 1000 µg based on symptoms with little consideration of IBW or renal function. This practice of non-weight-based dosing and the safety of 8 to 12 µg/kg IBW is corroborated by a recent study conducted by Clark et al 15 who determined a significant decrease in supratherapeutic concentrations when dosed by IBW using the labeled range.
The second sensitivity analysis increased the size of the CKD group by including patients with eGFR < 60 mL/min/1.73 m2. Although the primary outcome was not different in this analysis, logistic regression showed an increase in the risk of a supratherapeutic digoxin concentration in the CKD group when adjusting for dose. While these pre-specified analyses demonstrate that there may be an increase in risk of supratherapeutic concentrations in an adequately powered CKD group, the labeled dose of 8 to 12 µg/kg IBW appears safe. Dosing digoxin based on lean (or ideal) body weight is a recommendation found in the primary literature, manufacturer’s prescribing information and tertiary drug references.2-5,8,11,16 Our results call into question the necessity of a 50% reduction in the labeled loading dose in patients with kidney dysfunction, especially those with AKI.
One strength of our study is the specific inclusion criteria in regard to timing of digoxin concentrations. There was no significant difference in the number of hours between dose and timing of concentration between groups. In the secondary analysis, when timing of concentrations was incorporated into the logistic regression, there was no improvement in model fit, likely due to the similarities in timing across groups. Another strength of our study was that those with AKI were separated from those with CKD. In clinical practice, these are 2 different patient populations. Some tertiary references suggest dosage adjustment may not be necessary in acute renal failure; however, this statement is based only on expert opinion. 3
The limitations of this study relate mostly to its retrospective design. The analysis was conducted through chart review of laboratory values, notes and vital signs by multiple study personnel; however, it lacked a formal second verification of data consistency. Timing and modality of dialysis were not collected nor use of medications known to interfere with tubular secretion of creatinine. Additionally, baseline serum creatinine was estimated from the previous 12 months of data which may have classified new CKD patients into the AKI group. Additionally, is it likely our study is underpowered and at risk for Type II error. For example, as the number of patients classified as CKD was increased in the sensitivity analysis, there were significant differences between groups in our logistic regression model. The reason this group was underpowered is likely due to prescribing practices, as digoxin is not typically administered de novo to patients with CKD. Lastly, our definition of CKD used an eGFR cutoff of 30 mL/min/1.73 m2 which is not a universal definition of CKD and race was included in the calculation which has been shown to overestimate GFR up to 15% among non-white patients. 17 This definition contributed to the limited number of patients in this group. If a higher eGFR cutoff was utilized, similar to what was used in the sensitivity analysis, a clinically significant difference may have been detected. Similarly, there is not a universal definition for a supratherapeutic digoxin concentration. There is a well-described relationship between digoxin concentrations and toxicity; however, patients may be adequately digitalized without toxicity at concentrations above 2 ng/mL and may demonstrate toxicity at concentrations below 2 ng/mL.
Conclusion and Relevance
This is the first study in routine clinical practice evaluating the relationship between kidney function and digoxin peak concentrations that differentiates AKI from CKD. We did not find a relationship between kidney function and peak concentrations; however, the group with CKD was underpowered. No supratherapeutic concentrations developed in any patient who received less than or equal to 12 µg/kg of IBW demonstrating the safety of the labeled dose in patients with variable kidney function, especially AKI.
Footnotes
Acknowledgements
Nicole M. Acquisto, Pharm.D., FASHP, FCCM, FCCP, BCCCP
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
