Abstract
Background:
Tyrosine kinase inhibitors (TKIs) are the front-line therapy for chronic myeloid leukemia (CML), where phase 3 clinical trials have demonstrated their safety and efficacy. However, trial patients may not be representative of real-world patients (RWPs).
Objective:
To evaluate RWP clinical factors associated with effectiveness and safety in CML patients treated with TKIs.
Methods:
Patients with CML treated with at least 30 days of imatinib, dasatinib, nilotinib, or bosutinib between 2014 and 2018 were included. Patients were stratified into categories based on the number of factors that would have precluded enrollment into pivotal TKI phase 3 trials (0, 1, ≥2). End points included complete hematologic response (CHR), early molecular response (EMR), major molecular response (MMR), adverse event (AE)-induced dose decreases, treatment interruptions, and treatment discontinuations.
Results:
Final analyses included 174 patients. Patients with ≥2 factors had a higher risk of dose decreases (relative risk = 1.54; 95% CI = 1.02-2.34; P = 0.02) and a shorter time to dose decrease (hazard ratio = 2.43; 95% CI = 1.23-4.97; P = 0.006) compared with patients with 0 factors. Significant differences were observed in CHR at 1 month and MMR at 3 months between patients with 0 and ≥2 factors (P = 0.03 and P = 0.04, respectively).
Conclusion and Relevance:
Approximately 60% of our RWPs would have been excluded from the pivotal phase 3 TKI trials. These data suggest that RWPs require more precise dosing to achieve CML clinical milestones and to mitigate AEs, but findings should be validated prospectively.
Keywords
Introduction
Chronic myeloid leukemia (CML) accounts for 15% of all leukemias, and in 2021, it is estimated that there will be approximately 9110 new CML diagnoses and 1220 CML-related deaths. 1 Oral tyrosine kinase inhibitors (TKIs) have revolutionized CML from a fatal malignancy into a chronic disease with dramatically reduced mortality.2,3 Major molecular response (MMR), defined as at least a 3-log reduction of BCR-ABL1 mRNA transcript levels (ie, BCR-ABL1 levels ≤0.1% of baseline) at 12 months, is the gold standard measurement of treatment response in patients with CML because it is a significant predictor of treatment-free remission.4,5 Other clinical milestones include a complete hematologic response (CHR) at 1 month and an early molecular response (EMR) at 3 months (ie, BCR-ABL1 mRNA transcripts ≤10% of baseline). 4
Despite the efficacy of CML TKIs in phase 3 clinical trials, studies examining outcomes in real-world patients (RWPs) with CML remain scarce. Pivotal phase 3 clinical trials for CML TKIs included patients who were younger, were healthier, and had fewer concomitant medications compared with RWPs with CML.6-10 Although clinical trial data have suggested that patients with CML treated with TKIs may achieve a near-normal life expectancy, real-world registry studies have shown a 2-fold increased risk of death among patients with CML in the United States. 11 In addition, a recent study revealed that RWPs experienced a higher rate of TKI dose interruptions or treatment discontinuations (≥45%) compared with phase 3 clinical trial patients (≤25%), and only 54% of the imatinib-treated patients with CML remained on therapy after 3 years. The authors surmised that in RWPs the threshold for switching treatment, secondary to treatment-emergent adverse events (AEs), was likely lower than in clinical trials. Notably, the median age of patients enrolled in phase 3 trials was approximately a decade younger (46-49 years) than the median age of RWPs included in this study (57-58 years).7,8,12 Also, RWPs were more likely to have comorbidities, with three-quarters of patients having an age-adjusted Charlson Comorbidity index of at least 1 or higher. 12 In contrast, phase 3 clinical trial patients were significantly healthier, as evidenced by trial exclusion criteria, which included uncontrolled hypertension, diabetes, organ dysfunction, past medical history of stroke, myocardial infarction, congestive heart failure, HIV/AIDS, malabsorptive disease, or use of potentially interacting medications.6-8,10,12
Thus, there is an unmet clinical need to determine effectiveness and tolerability of TKI therapy in patients with CML who would not have been included in clinical trials (ie, RWPs) compared with patients who present with similar demographics from phase 3 clinical trials. The objective of this single-center retrospective study was to evaluate and describe RWP clinical and demographic factors that were associated with effectiveness and safety in patients with CML treated with TKIs.
Patients and Methods
Study Design
This was a single-institution, retrospective study of chronic phase patients with CML treated with TKIs at the University of North Carolina (UNC) Medical Center (UNC IRB #18-0905). To determine whether specific patient factors may affect TKI effectiveness and safety, this study evaluated differences in clinical milestones and TKI-induced AEs between patients who would have been eligible for the pivotal CML TKI phase 3 clinical trials6-10 and those who would not have been eligible for phase 3 trial enrollment (Table 1). The primary end point of the study was MMR at 12 months. The secondary end points included CHR at 1 month, EMR at 3 months, MMR at 3 months, and TKI-induced AEs. These AEs include incidence of AE-related dose decreases, incidence and duration of treatment interruptions, or permanent treatment discontinuation.
Exclusion Factors Used for Study Group Stratification. a
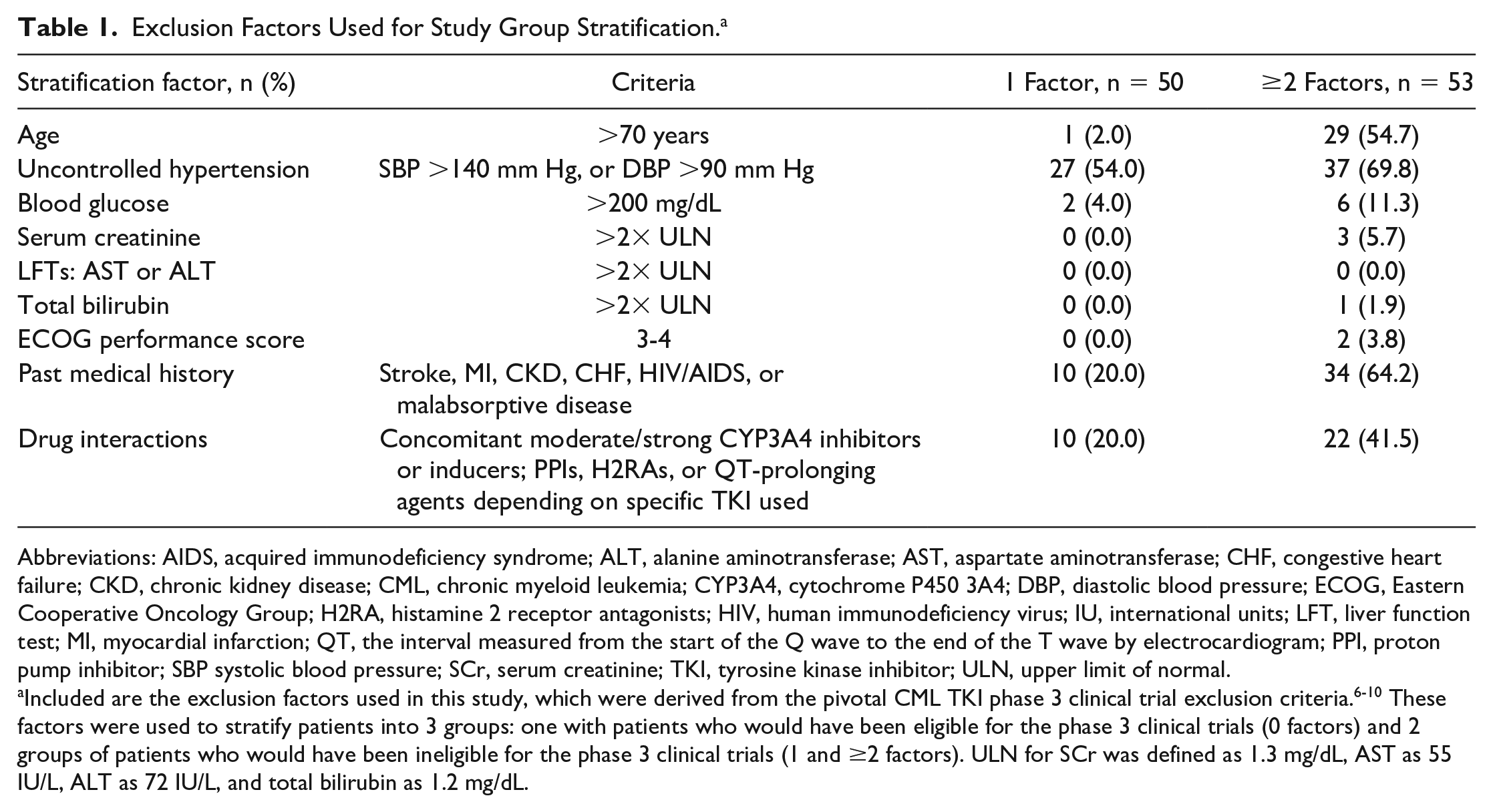
Abbreviations: AIDS, acquired immunodeficiency syndrome; ALT, alanine aminotransferase; AST, aspartate aminotransferase; CHF, congestive heart failure; CKD, chronic kidney disease; CML, chronic myeloid leukemia; CYP3A4, cytochrome P450 3A4; DBP, diastolic blood pressure; ECOG, Eastern Cooperative Oncology Group; H2RA, histamine 2 receptor antagonists; HIV, human immunodeficiency virus; IU, international units; LFT, liver function test; MI, myocardial infarction; QT, the interval measured from the start of the Q wave to the end of the T wave by electrocardiogram; PPI, proton pump inhibitor; SBP systolic blood pressure; SCr, serum creatinine; TKI, tyrosine kinase inhibitor; ULN, upper limit of normal.
Included are the exclusion factors used in this study, which were derived from the pivotal CML TKI phase 3 clinical trial exclusion criteria.6-10 These factors were used to stratify patients into 3 groups: one with patients who would have been eligible for the phase 3 clinical trials (0 factors) and 2 groups of patients who would have been ineligible for the phase 3 clinical trials (1 and ≥2 factors). ULN for SCr was defined as 1.3 mg/dL, AST as 55 IU/L, ALT as 72 IU/L, and total bilirubin as 1.2 mg/dL.
Study Population Identification
The Carolina Data Warehouse for Health (CDW-H), a central data repository containing clinical and research data sourced from the UNC Medical Center and UNC Health Care System, was queried to identify potentially eligible patients for this study. Patients were included if they were ≥18 years old, treated at UNC between April 2014 and May 2018, and treated with imatinib, dasatinib, nilotinib, or bosutinib for at least 30 days within that same time frame. Patients were excluded if they were prescribed a TKI for a diagnosis other than CML, prescribed ponatinib (given its limited use as a last option or only in patients with T315I mutation), missing baseline hematopathology results, or treated at an oncology practice external to UNC.
Clinical Data Extraction
Patient clinical and demographic data were collected from the UNC electronic medical record. All data were extracted from patient visits and labs at the time of TKI initiation, and standard-of-care follow-up visits. CML diagnosis was confirmed via ICD-9/10 coding, clinic documentation, and presence of elevated BCR-ABL1 mRNA transcript levels at baseline. Drug dosing information was collected at initiation and 1, 3, 6, 9, and 12 months thereafter, and included the name of the TKI, line of therapy, dose, and frequency of administration. BCR-ABL1 mRNA transcript values were collected at baseline and 3, 6, 9, and 12 months after TKI initiation. Complete blood count with differential results were collected at baseline, 1, and 3 months to evaluate CHR. The first incidence of an AE-induced dose decrease, TKI interruption, or TKI discontinuation were recorded throughout the 12-month period after TKI initiation.
Exclusion criteria from the pivotal phase 3 clinical trials6-10 were compiled a priori, and patients were evaluated for the presence or absence of these factors. Given minor differences in exclusion criteria in phase 3 trials, a composite list of exclusion factors were designed (Supplementary Table 1, available online). Patients were then stratified into groups based on the presence of 0, 1, or ≥2 factors. Factors that would have excluded patients included age >70 years, uncontrolled hypertension, baseline blood glucose >200 mg/dL, baseline labs >2× the upper limit of normal (serum creatinine, total bilirubin, or additional liver function tests), Eastern Cooperative Oncology Group performance status 3 to 4, concomitant medications that could alter TKI exposure-response relationships, or a past medical history significant for stroke, myocardial infarction, chronic kidney disease, congestive heart failure, HIV/AIDS, or malabsorptive disease (Table 1).
Statistical Analyses
Descriptive statistics for clinical and demographic data were reported as means with standard deviations, or as medians with ranges and percentages. A significance level of 0.05 (P < 0.05) was selected a priori, and all analyses were 2-sided. Generalized linear models with binomial distribution and log link function were used to compute and compare the unadjusted binary outcomes across factor groups, as well as adjusted relative risks (RRs) that controlled for TKI treatment effect. The Kaplan-Meier method was used to compare the time to AE-induced dose decrease across factor groups (including patients who did not experience a decrease as censored events at 12 months). Cox proportional hazards models were fit to compute and compare the unadjusted hazard ratios for time to AE-induced dose decrease across factor groups. Hazard ratios were also adjusted to control for TKI treatment effect. Because of the exploratory nature of this study, results were not corrected for multiple comparisons. All statistical analyses were conducted using SAS JMP v14.0.0 (SAS, Cary, NC), and figures were created using GraphPad Prism v8.2 (GraphPad Software, La Jolla, CA).
Results
Demographic and Clinical Characteristics
A total of 409 adult patients with CML were identified from the CDW-H, and 235 patients were deemed ineligible for final analyses (Figure 1). The most common reasons for exclusion were the absence of initiating clinical and lab data in the medical record (n = 119), as well as patient identification discrepancies where patients were incorrectly identified by CDW-H as having a CML diagnosis (n = 55). In all, 174 patients with CML treated at UNC were stratified into 3 groups based on factors derived from the exclusion criteria in the pivotal phase 3 clinical trials (Supplementary Table 1).6-10 Whereas all patients were eligible to be evaluated for AE outcomes, only 144 were eligible to be evaluated for effectiveness outcomes because 30 patients had BCR-ABL1 mRNA transcript levels <10% at the time of initiation of second-line (and beyond) TKI treatment, or were treated for less than 30 days before discontinuing a TKI (Figure 1). Among the final study cohort, the median patient age was 55 years (interquartile range = 45-68), approximately 55% of the patients were female, 72% of the patients were White, and imatinib was the most commonly prescribed TKI (n = 62; 35.6%). The mean total daily maintenance doses for imatinib, dasatinib, nilotinib, and bosutinib were 357, 86, 546, and 403 mg, respectively (Table 2).
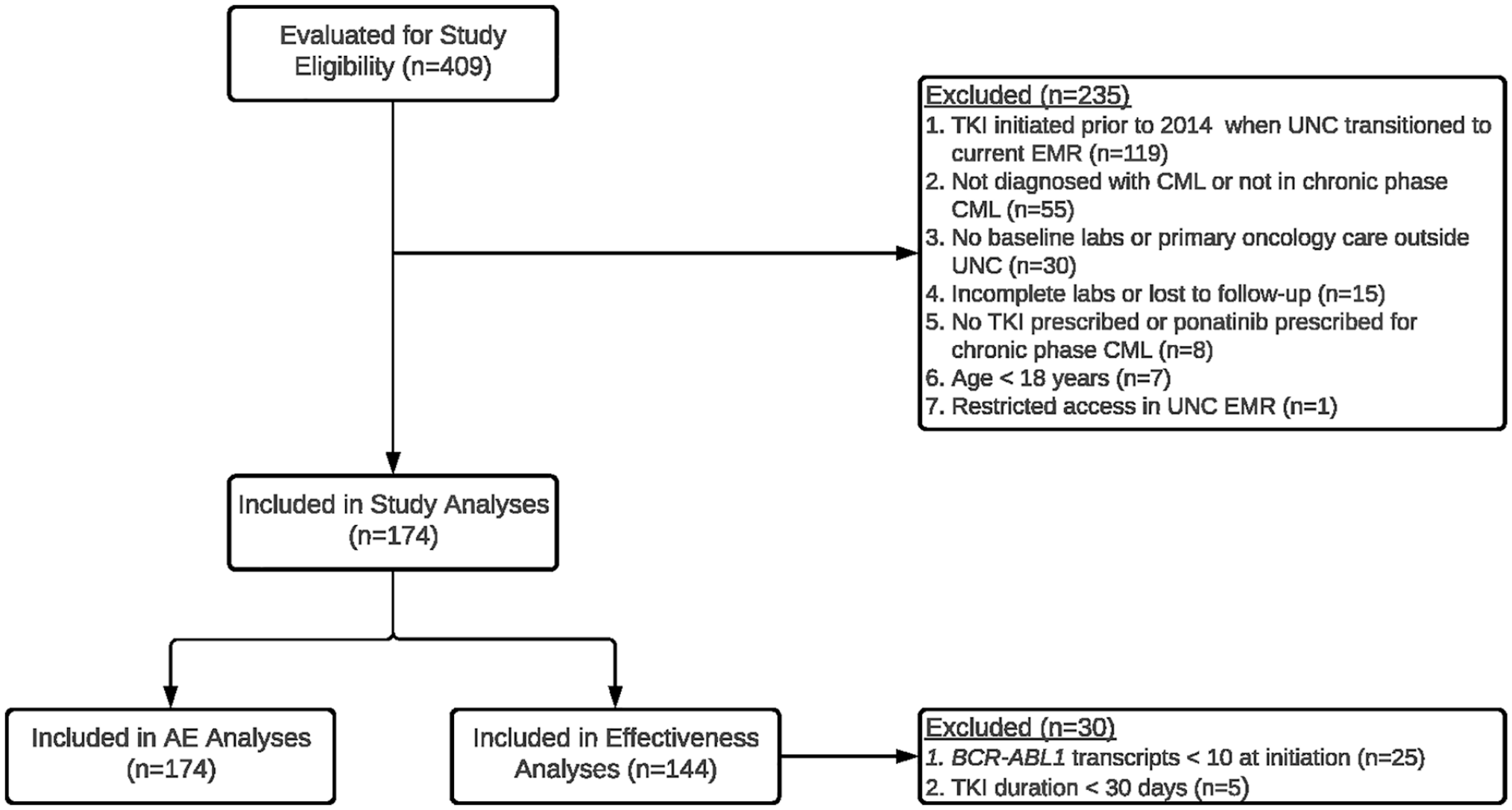
Study schematic.
Patient Baseline Clinical and Demographic Characteristics. a
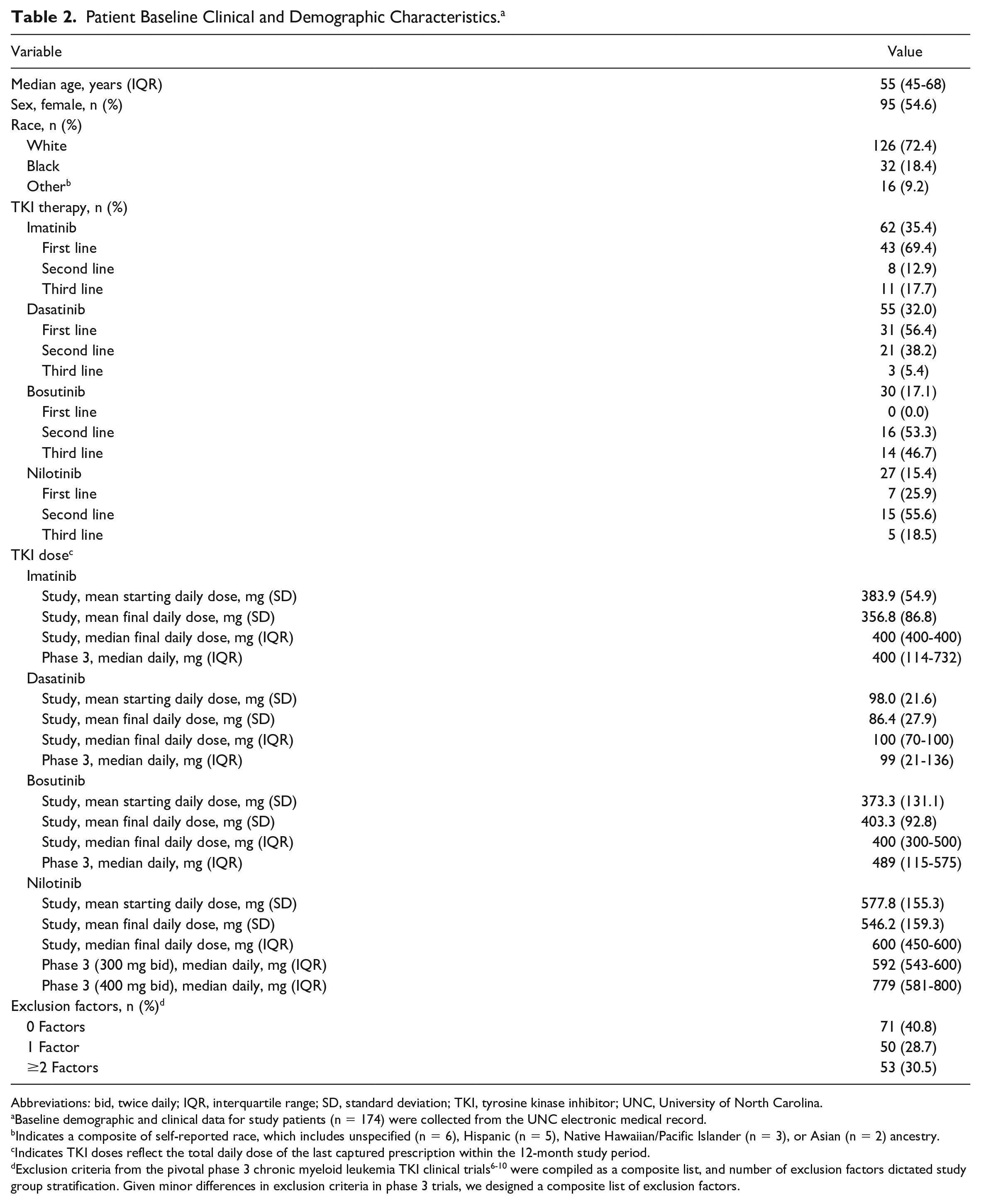
Abbreviations: bid, twice daily; IQR, interquartile range; SD, standard deviation; TKI, tyrosine kinase inhibitor; UNC, University of North Carolina.
Baseline demographic and clinical data for study patients (n = 174) were collected from the UNC electronic medical record.
Indicates a composite of self-reported race, which includes unspecified (n = 6), Hispanic (n = 5), Native Hawaiian/Pacific Islander (n = 3), or Asian (n = 2) ancestry.
Indicates TKI doses reflect the total daily dose of the last captured prescription within the 12-month study period. dExclusion criteria from the pivotal phase 3 chronic myeloid leukemia TKI clinical trials6-10 were compiled as a composite list, and number of exclusion factors dictated study group stratification. Given minor differences in exclusion criteria in phase 3 trials, we designed a composite list of exclusion factors.
Patients were stratified into 1 of 3 groups (0, 1, or ≥2 factors) based on the presence of a composite list of factors developed from phase 3 clinical trial exclusion criteria (Table 1 and Supplementary Table 1, available online). Among the study cohort, approximately 60% of the patients presented with ≥1 factor that would have excluded their enrollment into a phase 3 clinical trial (n = 103). A total of 71 patients (40.8%) presented with 0 factors at baseline, 50 (28.7%) presented with only 1 factor, and 53 (30.5%) presented with ≥2 factors. Among those with only 1 factor, the most common were uncontrolled hypertension (n = 27, 54.0%) as well as drug-drug interactions (DDIs) between concomitant medications and the prescribed TKI and comorbidities in their past medical history (n = 10, 20.0% each) (Supplementary Table 2, available online). Importantly, among the patients prescribed concomitant medications with potential for DDIs, all had TKI doses that were appropriately adjusted according to US Food and Drug Administration (FDA) package labeling instructions.13-16 For those with ≥2 factors, the most prevalent factors were uncontrolled hypertension (n = 37, 69.8%), past medical history (n = 34, 64.2%), and age >70 years (n = 29, 54.7%; Table 1).
Effectiveness Outcomes
A total of 144 patients were eligible for effectiveness evaluations, which included 59 patients with 0 factors, 39 patients with 1 factor, and 46 patients with ≥2 factors. After 12 months of treatment, 72.2% of the evaluable patients (n = 104) had achieved MMR. However, the study failed to achieve its primary end point because no significant difference between patients with 0 factors versus 1 factor and 0 versus ≥2 factors were observed for MMR at 12 months, even when controlling for the effects of TKIs (P = 0.61 and P = 0.19, respectively; Table 3). Furthermore, 37 (25.7%) patients achieved early MMR at 3 months; of those, 13 (22%) had 0 factors, 5 (12.8%) had 1 factor, and 19 (41.3%) had ≥2 factors. Patients with ≥2 factors were nearly twice as likely to achieve an early MMR at 3 months when compared with patients with 0 factors (RR = 1.87; 95% CI = 1.04-3.38; P = 0.04); however, significant differences in MMR at 3 months were not observed between patients with 0 or 1 factor (P = 0.26). After controlling for the TKI treatment effect, patients with ≥2 factors were still about twice as likely to achieve early MMR at 3 months, when compared with patients with 0 factors (RR = 1.89, 95% CI = 1.05-3.32, P = 0.03; Table 3 and Figure 2A).
Evaluation of TKI Effectiveness End Points. a
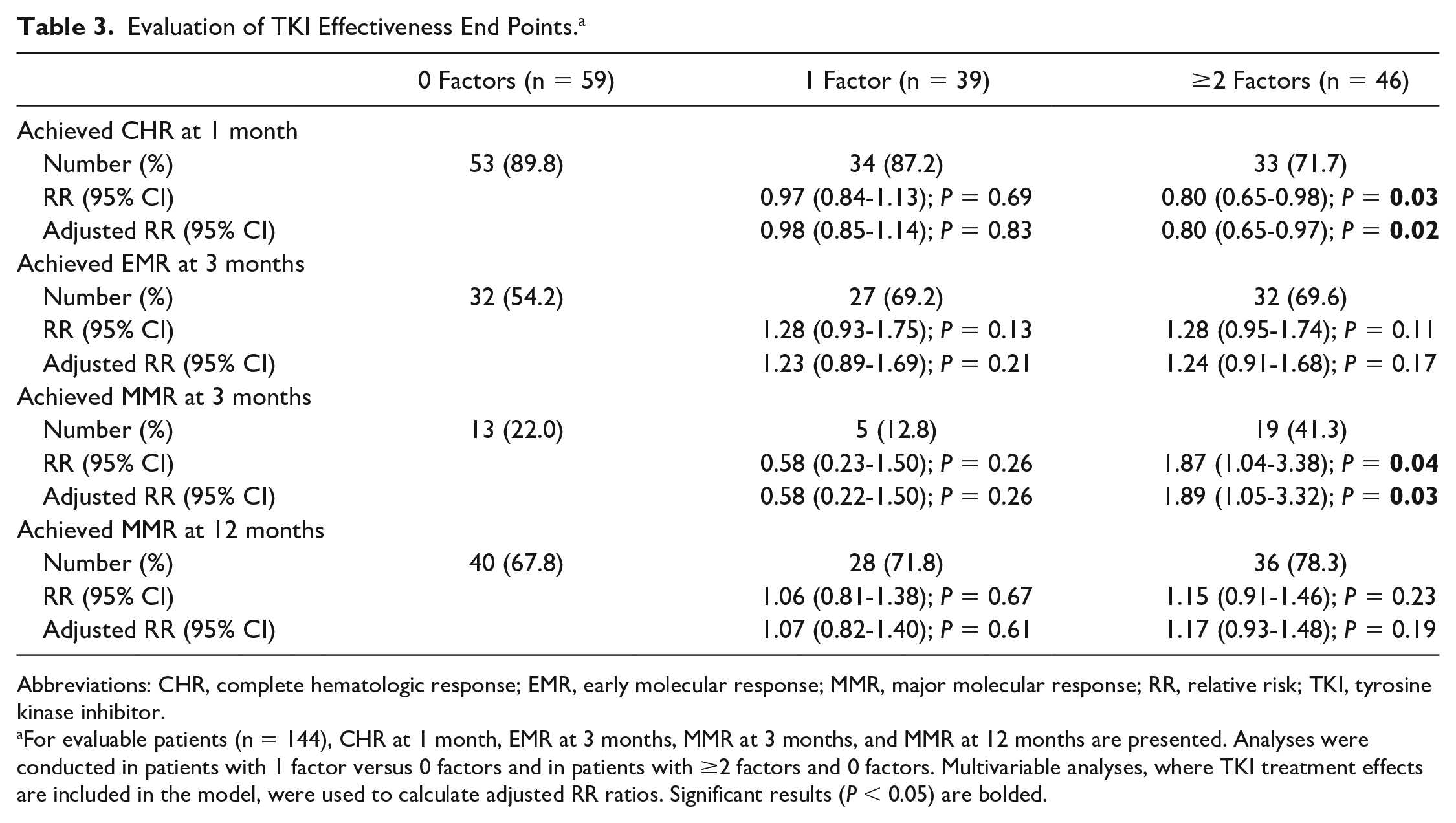
Abbreviations: CHR, complete hematologic response; EMR, early molecular response; MMR, major molecular response; RR, relative risk; TKI, tyrosine kinase inhibitor.
For evaluable patients (n = 144), CHR at 1 month, EMR at 3 months, MMR at 3 months, and MMR at 12 months are presented. Analyses were conducted in patients with 1 factor versus 0 factors and in patients with ≥2 factors and 0 factors. Multivariable analyses, where TKI treatment effects are included in the model, were used to calculate adjusted RR ratios. Significant results (P < 0.05) are bolded.
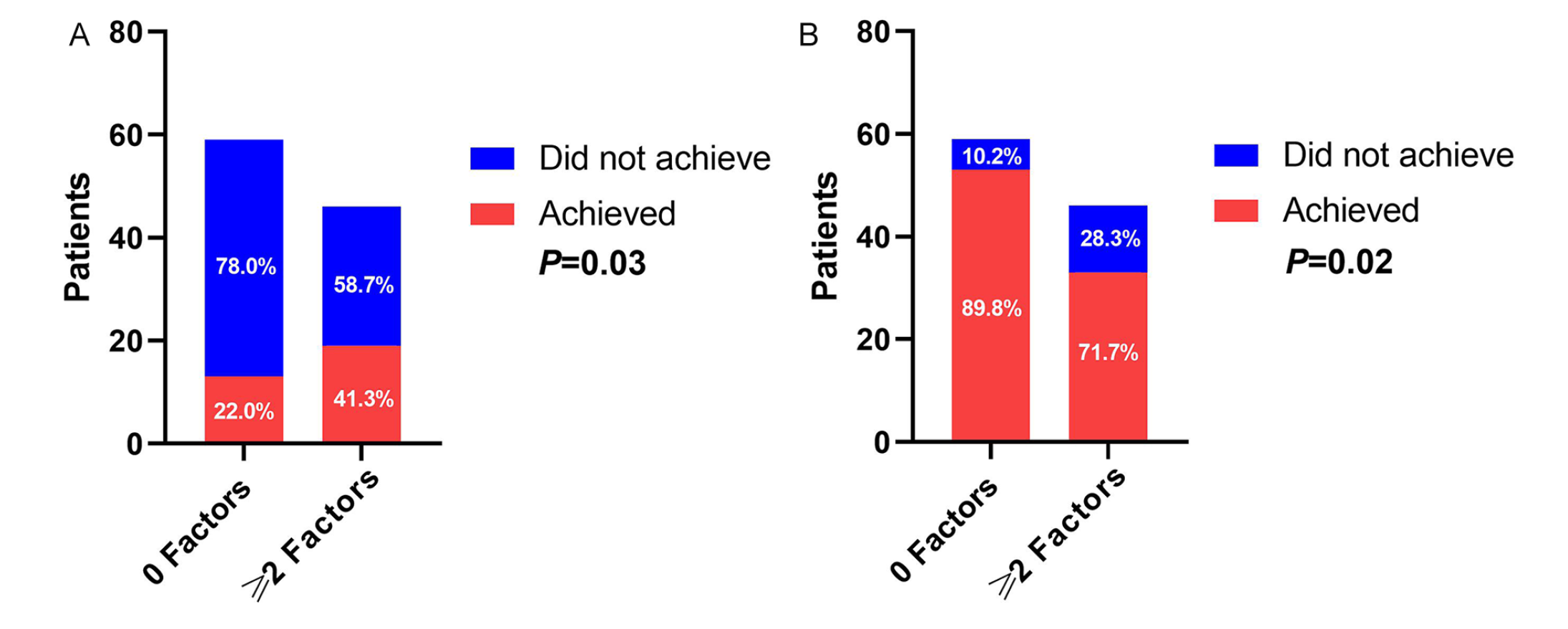
CHR at 1 month and early MMR at 3 months. Panel A depicts differences in relative risk of achieving early MMR by 3 months among study patients with 0 factors versus ≥2 factors (n = 59 and n = 46, respectively). Panel B depicts differences in relative risk of achieving CHR by 1 month among study patients with 0 factors versus ≥2 factors.
Next, after 1 month of treatment, 83.3% of the evaluable patients (n = 120) had achieved CHR. Only 71.7% (n = 33) of the patients with ≥2 factors achieved CHR at 1 month, whereas 89.8% (n = 53) and 87.2% (n = 34) of patients with 0 or 1 factor achieved CHR, respectively. Patients with ≥2 factors were 20% less likely to achieve CHR at 1 month when compared with patients with 0 factors (RR = 0.80; 95% CI = 0.65-0.98; P = 0.03); however, a significant difference in CHR at 1 month was not observed between patients with 0 factors or 1 factor (P = 0.69). Moreover, when controlling for TKI treatment effect, patients with ≥2 factors remained 20% less likely to achieve CHR at 1 month when compared with patients with 0 factors (RR = 0.80, 95% CI = 0.65-0.97, P = 0.02; Figure 2B). A significant difference in CHR at 1 month was not observed between patients with 0 factors or 1 factor, even after controlling for TKI effect (P = 0.83; Table 3).
After 3 months of TKI treatment, 91 (63.2%) patients had achieved EMR. Of these, 54.2% of patients with 0 factors, 69.2% of patients with 1 factor, and 69.6% of patients with ≥2 factors achieved EMR at 3 months. When comparing the 3 groups, no significant differences were observed for EMR at 3 months (P > 0.05).
Adverse Event Outcomes
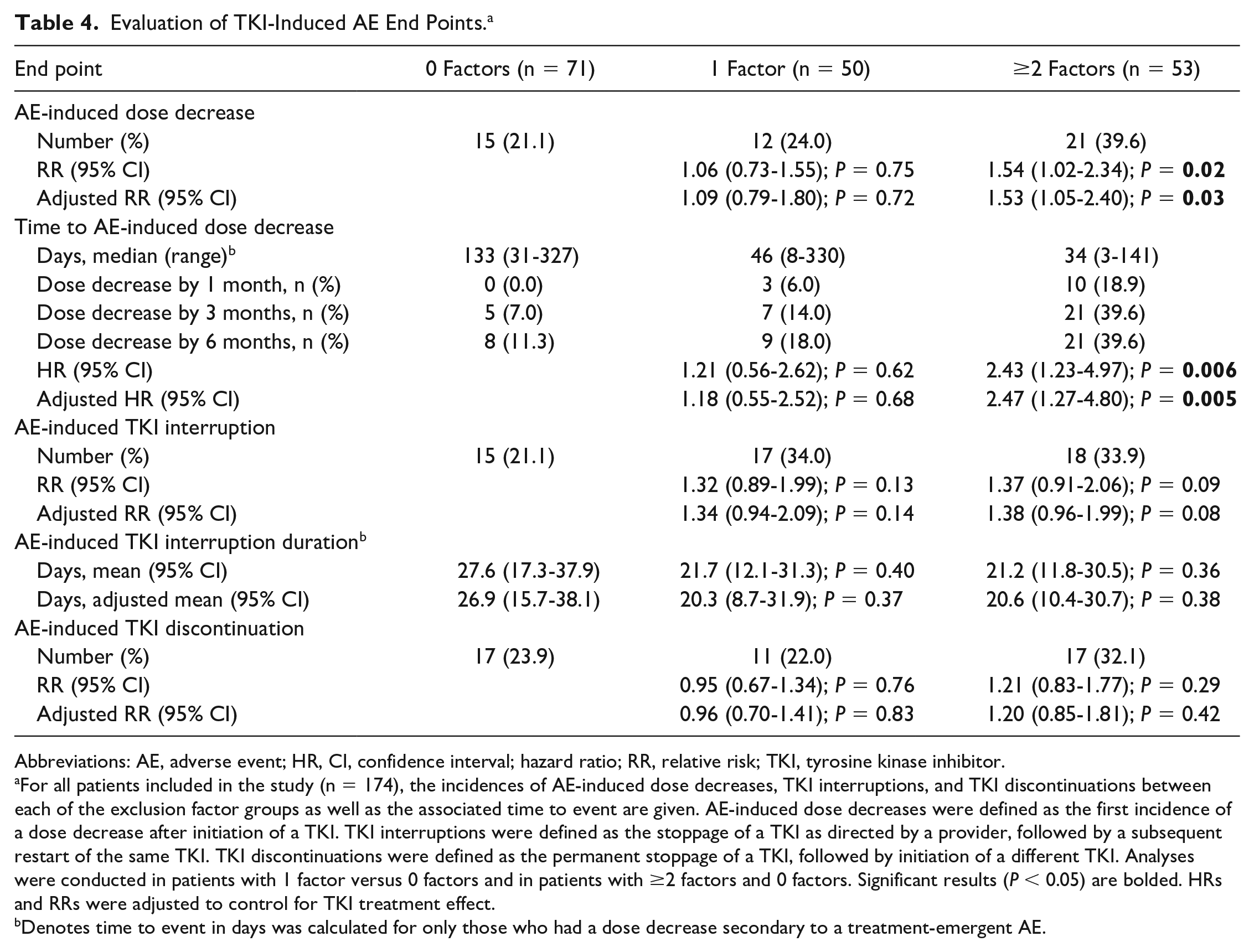
All 174 patients were evaluable for AE analyses. A total of 48 (27.6%) patients required a dose reduction as a result of a TKI-induced AE. Among those who experienced a toxicity, 39.6% of patients with ≥2 factors required a dose decrease by 6 months on TKI therapy compared with 11.3% and 18.0% of patients with 0 factors and 1 factor, respectively. The median times to a TKI-induced AE were 133, 46, and 34 days for patients with 0, 1, and ≥2 factors, respectively (Table 4). Patients with ≥2 factors were at higher risk for an AE-induced dose decrease of their TKI when compared with patients with 0 factors (RR = 1.54; 95% CI = 1.02-2.34; P = 0.02), which remained significant after controlling for TKI treatment effect. Patients with ≥2 factors were also at a nearly 2.5-fold increased cumulative incidence of a dose decrease resulting from TKI-induced AEs when compared with patients with 0 factors (hazard ratio = 2.43; 95% CI = 1.23-4.97; P = 0.006), and this also remained significant after controlling for TKI treatment effect (Table 4, Figure 3). However, an increased risk of AE-induced dose decrease was not observed between patients with 0 factors or 1 factor (P = 0.75), or between patients with 1 factor and ≥2 factors (P = 0.06).
Evaluation of TKI-Induced AE End Points. a
Abbreviations: AE, adverse event; HR, CI, confidence interval; hazard ratio; RR, relative risk; TKI, tyrosine kinase inhibitor.
For all patients included in the study (n = 174), the incidences of AE-induced dose decreases, TKI interruptions, and TKI discontinuations between each of the exclusion factor groups as well as the associated time to event are given. AE-induced dose decreases were defined as the first incidence of a dose decrease after initiation of a TKI. TKI interruptions were defined as the stoppage of a TKI as directed by a provider, followed by a subsequent restart of the same TKI. TKI discontinuations were defined as the permanent stoppage of a TKI, followed by initiation of a different TKI. Analyses were conducted in patients with 1 factor versus 0 factors and in patients with ≥2 factors and 0 factors. Significant results (P < 0.05) are bolded. HRs and RRs were adjusted to control for TKI treatment effect.
Denotes time to event in days was calculated for only those who had a dose decrease secondary to a treatment-emergent AE.
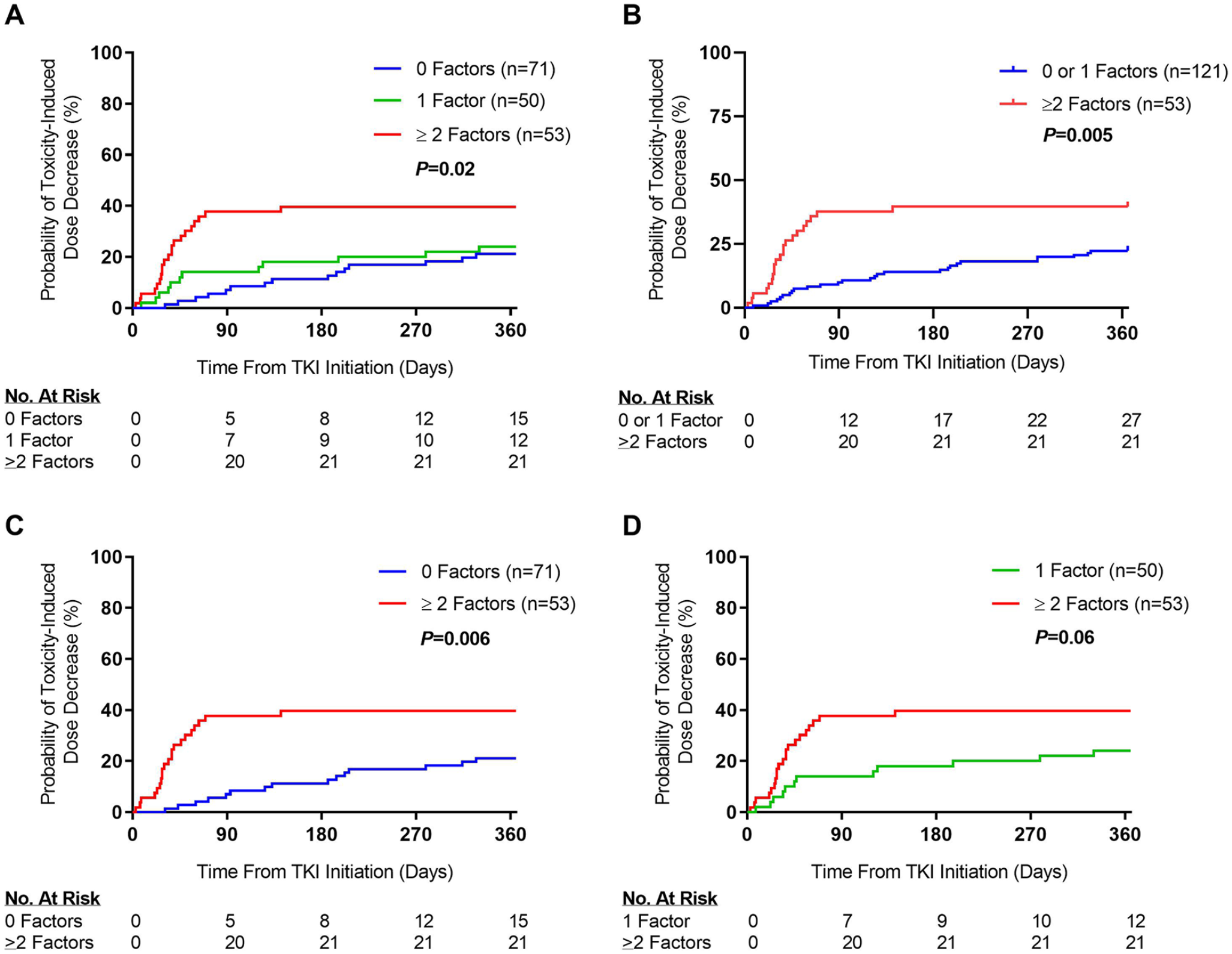
Cumulative incidence plots for time to dose decrease as a result of TKI-induced AEs by number of factors. Panel A depicts differences in the cumulative incidence of AE-induced TKI dose decreases among study patients with 0, 1, and ≥2 factors. Panel B depicts differences among study patients with 0 and 1 versus ≥2 factors. Panel C depicts differences among study patients with 0 versus ≥2 factors. Panel D depicts differences among study patients with 1 versus ≥2 factors.
There were no significant differences (P > 0.05) for the total number of TKI interruptions or discontinuations observed among patients with 0, 1, and ≥2 factors (Table 4).
Discussion
Despite their routine use in CML, the efficacy-effectiveness and toxicity profiles of TKIs differs between RWPs and phase 3 clinical trials because of the heterogeneity among the RWP population. This study evaluated RWPs who would have been eligible for phase 3 clinical trials against those who would have been precluded from enrollment. Approximately 60% of patients in this study had at least 1 factor that would have excluded enrollment,6-10 suggesting that a significant proportion of RWPs with CML are not represented in the pivotal trials.
In the overall study population, 1 in 5 RWPs with CML required a dose interruption in the first month of treatment if they had ≥1 factor. During the first several weeks immediately following TKI initiation, patients often experience early-onset AEs such as nausea, vomiting, diarrhea, or edema, which are managed with dose reductions and interruptions until a sustainable maintenance dose can be achieved. 17 Patients who experience a higher number of AEs warrant more frequent therapy adjustments, which may inevitably delay early treatment milestones, such as CHR. Similarly, a higher incidence of AEs may suggest that RWP factors cause increased TKI exposure or a higher sensitivity to drug effects. Although early responses can suggest a more promising response in some patients, these results may not be sustainable because of the higher incidence and prolonged duration of AEs. Accordingly, it is important to recognize that standard TKI dosing may not be appropriate for all patients. TKI AEs are also known to contribute to significantly increased health care expenditures at the patient and health-system levels. 18
Although there was not an increased risk of AEs observed among RWPs with only 1 factor that would have precluded enrollment in a clinical trial, those with ≥2 factors experienced significantly more AE-induced dose reductions, and presented with AEs earlier, compared with those with 0 factors. Among patients with ≥2 factors, an increased incidence of TKI-induced AEs and a quicker time to AEs were observed. Interestingly, patients with ≥2 factors were less likely to achieve CHR at 1 month but more likely to achieve early MMR at 3 months. Observing such an early MMR was surprising considering MMR by 12 months is the primary treatment goal in CML.4,5 Among the 19 patients with ≥2 factors who achieved an MMR by 3 months, nearly 50% (n = 8) also experienced TKI-induced AEs that required a dose reduction. Moreover, patients with ≥2 factors who achieved MMR by 3 months were at a significantly higher risk for a dose decrease as a result of TKI-induced AEs (P < 0.0001). A potential explanation for these effectiveness and toxicity findings could be related to TKI exposure-response relationship differences.
In patients with CML, frequent treatment interruptions have been shown to increase the risk of BCR-ABL kinase domain point mutations, and subsequent resistance to treatment. 19 Treatment-emergent AEs are the most common barrier that leads to nonadherence and compromises dose intensity of TKIs.20,21 It has been observed that near-perfect medication adherence (>90% of doses) is a major predictor of TKI response in patients with CML achieving MMR, and thus, dose interruptions resulting from toxicity can lead to a subtherapeutic treatment response.20,22 Although 12-month rates of MMR were similar between the patients in the 3 study groups, prior published data suggest that frequent dose interruptions would lead to loss of molecular response over a longer period (>18 months). 19 Future prospective studies should be designed to evaluate MMR for a longer follow-up period (eg, 18 or 24 months) to ascertain if RWPs have less MMR as a result of more frequent AEs and dose interruptions, and ultimately, compromised medication adherence. Unfortunately, because of challenges inherent to retrospective research, adherence could not be included as a covariate in this study’s analyses but should be an important factor that is included in future prospective research. In addition, rare but severe treatment-emergent AEs can occur with these TKIs, and compromise MMR and other clinical milestones. 23 In the future, prospective research could also integrate germline genotyping for functional pharmacogenetic variants and pharmacokinetic sampling, with the goal of identifying patients most at risk for serious toxicities and who could benefit from personalized TKI dosing or another TKI altogether.
Limitations to this study include an insufficient sample size to comprehensively evaluate clinical outcomes with individual TKIs and their sequencing, the lack of an external validation cohort, and inability to detect the effect of each clinical or demographic characteristic. It should also be acknowledged that exclusion criteria varied slightly across phase 3 clinical trials, and a composite list of criteria for the exclusion factors was designed for this study. Additionally, only the first incidence of AEs was captured, and outcomes were not evaluated beyond 12 months after initiation of a TKI, limiting the ability to extrapolate these results to long-term outcomes. When considering issues with concomitant medications, individual DDIs and DDI severity were not evaluated in this study. In future prospective studies, grading the DDI severity may be prudent to estimate how individual concomitant medications may affect TKI effectiveness and safety.
A common pattern has emerged for medications approved by the FDA, where the benefit-to-risk relationship of a drug post-approval differs from what was reported in phase 3 clinical trials. This pattern is often a result of the greater diversity among RWP populations, relative to the clinical trial population. 24 Factors that can significantly differ between RWPs and those enrolled in phase 3 clinical trials can include age, sex, socioeconomic status, weight, hepatic or renal function, genotypic variation, comorbidities, concomitant medications, and medication adherence. These differences may affect the drug effectiveness and safety profile, and subsequently require drug dose adjustments beyond those recognized in the FDA label. The difference in outcomes between phase 3 clinical trial patients and RWPs is known as the efficacy-effectiveness gap.25,26 Accounting for individual factors that affect dose response can maximize effectiveness and minimize AEs in RWPs. 27 Given our study findings, we designed and executed a prospective study (NCT03885830) that seeks to associate effectiveness and toxicity outcomes with TKI plasma concentrations, with the long-term goal of guiding TKI dose selection based on RWP factors.
Conclusion and Relevance
This study supports the hypothesis that a “one size fits all” TKI dosing regimen is not optimal for all patients. Although effectiveness was similar between groups, RWPs were more intolerant of TKIs and required dose modifications. It is imperative to identify and validate RWP characteristics that associate with supratherapeutic and subtherapeutic TKI concentrations that ultimately influence effectiveness and AEs. Ideally, our future prospective work will lead to the implementation of TKI individualized dosing to improve clinical outcomes that enhance health-related quality of life and reduce unnecessary health care resource utilization.
Supplemental Material
sj-docx-1-aop-10.1177_10600280211044160 – Supplemental material for Response to Tyrosine Kinase Inhibitors in Real-World Patients With Chronic Myeloid Leukemia
Supplemental material, sj-docx-1-aop-10.1177_10600280211044160 for Response to Tyrosine Kinase Inhibitors in Real-World Patients With Chronic Myeloid Leukemia by Andy H. Szeto, Tyler Bucci, Allison Deal, Anqi Zhu, Majd Ahmad, Amanda S. Cass, Margaret R. Sketch, Ryan Kemper, Joshua F. Zeidner, Matthew C. Foster, Benyam Muluneh and Daniel J. Crona in Annals of Pharmacotherapy
Supplemental Material
sj-docx-2-aop-10.1177_10600280211044160 – Supplemental material for Response to Tyrosine Kinase Inhibitors in Real-World Patients With Chronic Myeloid Leukemia
Supplemental material, sj-docx-2-aop-10.1177_10600280211044160 for Response to Tyrosine Kinase Inhibitors in Real-World Patients With Chronic Myeloid Leukemia by Andy H. Szeto, Tyler Bucci, Allison Deal, Anqi Zhu, Majd Ahmad, Amanda S. Cass, Margaret R. Sketch, Ryan Kemper, Joshua F. Zeidner, Matthew C. Foster, Benyam Muluneh and Daniel J. Crona in Annals of Pharmacotherapy
Supplemental Material
sj-docx-3-aop-10.1177_10600280211044160 – Supplemental material for Response to Tyrosine Kinase Inhibitors in Real-World Patients With Chronic Myeloid Leukemia
Supplemental material, sj-docx-3-aop-10.1177_10600280211044160 for Response to Tyrosine Kinase Inhibitors in Real-World Patients With Chronic Myeloid Leukemia by Andy H. Szeto, Tyler Bucci, Allison Deal, Anqi Zhu, Majd Ahmad, Amanda S. Cass, Margaret R. Sketch, Ryan Kemper, Joshua F. Zeidner, Matthew C. Foster, Benyam Muluneh and Daniel J. Crona in Annals of Pharmacotherapy
Footnotes
Acknowledgements
The authors would like to thank the UNC Pharmacy Analytics team and the Carolina Data Warehouse for Health for their assistance obtaining the patient data evaluated in this study. The authors would also like to acknowledge Dr Jessica Auten, Dr Jill Bates, and Dr Katherine Morgan for their valuable advice during the study conception and project development phase. Finally, the authors would like to thank Dr Lana Crona for providing expertise in medical writing and manuscript editing.
Authors’ Note
AHS and TB contributed equally to this work. BM and DJC contributed equally to this work. Joshua F. Zeidner and Matthew C. Foster are now affiliated with Division of Hematology, Department of Medicine, School of Medicine, University of North Carolina, Chapel Hill, NC, USA.
Author Contributions
Conceptualization, AHS, TB, AD, ASC, MRS, JFZ, MCF, BM, and DJC; data collection, AHS, TB, MA, ASC, MRS, and RK; data curation, AHS, TB, AD, AZ, and DJC; formal analysis, AD, AZ, and DJC; resources, BM and DJC; writing—original draft preparation, AHS, TB, RK, BM, and DJC; writing—review and editing, AHS, TB, AD, AZ, MA, ASC, MRS, RK, JFZ, MCF, BM, and DJC; supervision, BM and DJC; project administration, BM and DJC. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: ASC has received an honorarium from Genentech. MCF reports having received research funding from Bellicum Pharmaceuticals, Celgene, and Macrogenics and has served as a consultant for Daiichi Sankyo. JFZ reports having received research funding from Celgene, Merck, Takeda, and Tolero Pharmaceuticals, honoraria from Agios, Celgene, Daiichi Sankyo, Pfizer, and Tolero Pharmaceuticals, and has served as a consultant for AsystBio Laboratories, Celgene, and Covance. All remaining authors have no financial interests or conflicts of interest to disclose.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: DJC, BM, and MCF acknowledge the generous support provided by a Tier 1 grant the Eshelman Institute for Innovation at the UNC Eshelman School of Pharmacy.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
