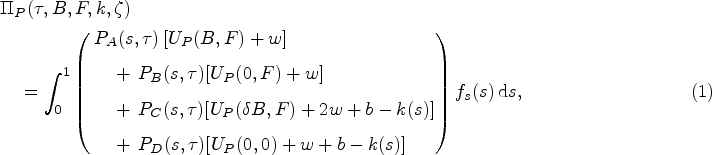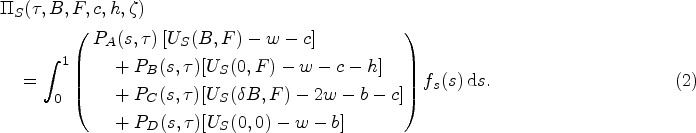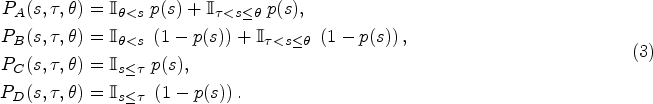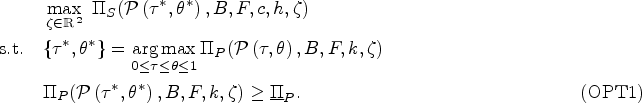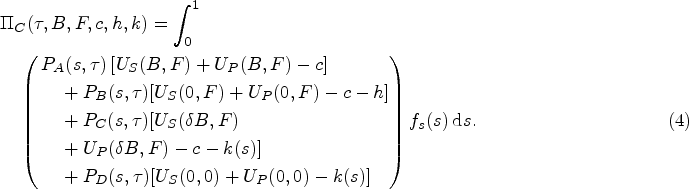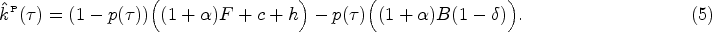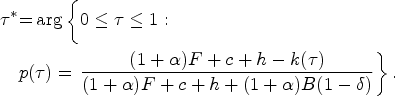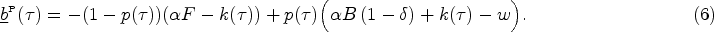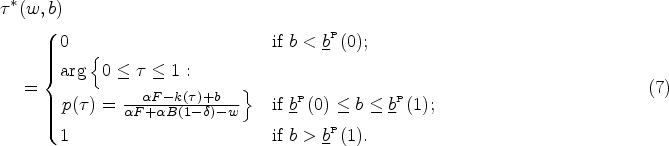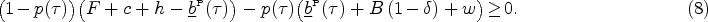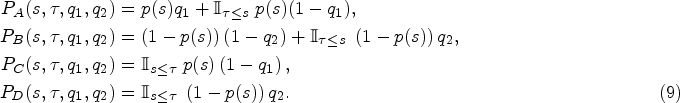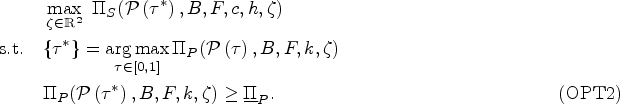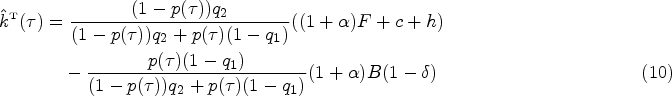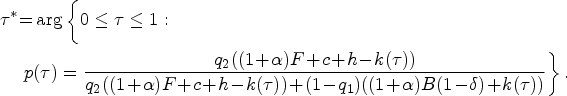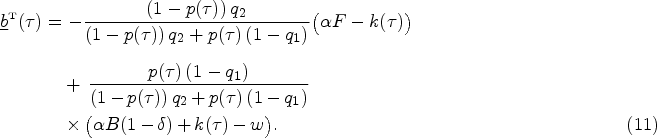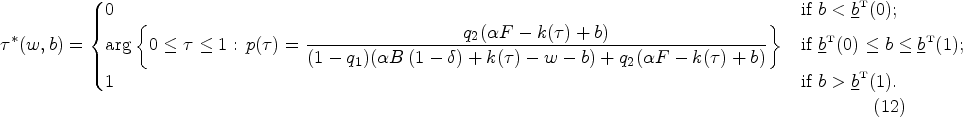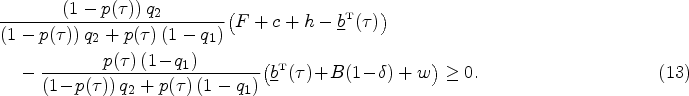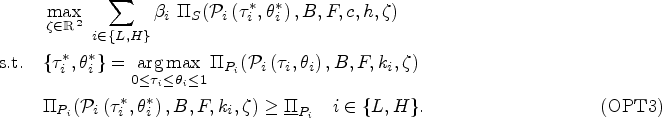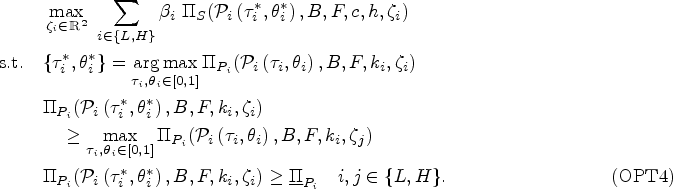Abstract
Antibiotic resistance is an ongoing public health crisis fueled by overuse and misuse of antibiotics. The goal of this article is to examine the impact of action-based incentive payments on reducing inappropriate antibiotic prescriptions in primary care, where 30%–50% of antibiotic prescriptions are inappropriate. Various financial incentive programs to reduce the rate of inappropriate antibiotic prescriptions have been implemented and studied empirically. However, there have not been analytical studies to evaluate payment model contract design features and the potential for payment models to impact diagnosis decision making. We develop a stylized physician compensation model to study the interaction between a payer and a provider. The payer offers a payment contract, with a bonus tied to the prescription, to maximize social welfare, considering total costs of providing care and social harm from antibiotic resistance. Given the contract offered and their own opportunity cost associated with factors such as fear of misdiagnosis and time spent explaining to patients why antibiotics are not indicated, the provider chooses whether or not to prescribe antibiotics to patients for whom antibiotics are not clinically indicated. We consider four cases: when diagnostic accuracy relies on symptom presentation versus additional diagnostic testing and when the opportunity cost of not prescribing antibiotics is public versus private information of the provider. When there is no information asymmetry, an action-based incentive payment can co-ordinate care and achieve the first-best policy, decreasing the rate of inappropriate prescribing, even when incentive payments can affect the diagnosis behavior. However, when the diagnosis depends on additional testing, the first-best policy results in fewer inappropriate antibiotic prescriptions, when the test has high specificity. Therefore, when an accurate technical diagnostic is available, a simple to implement action-based incentive payment can be effective in reducing inappropriate antibiotic prescribing. In the realistic setting where the provider’s opportunity cost is private information, an action-based incentive payment cannot eliminate inappropriate antibiotic prescribing. In these settings, the introduction of point of care diagnostics to aid in objective diagnostic criteria will reduce the unintended consequences of the contract.
Keywords
Introduction
Antibiotic resistance is an ongoing public health crisis threatening the ability to treat common infectious diseases (WHO, 2021). Antibiotic-resistant infections are associated with prolonged illnesses, worse clinical outcomes, increased healthcare costs, and higher mortality compared to antibiotic-susceptible infections (CDC, 2019). Annually, antibiotic-resistant infections are estimated to be responsible for more than 35,000 deaths in the United States, 33,000 deaths in Europe, and many more in the developing world (Cassini et al., 2019; CDC, 2019).
Antibiotic resistance develops naturally in bacteria through spontaneous mutation and bacterial conjugation (Mazel and Davies, 1999). Very high levels of antibiotic use, including misuse and overuse, creates a selective pressure that increases the prevalence of resistant strains (Spellberg et al., 2008). Global sales of antibiotics for human consumption increased by 39% between 2000 and 2015 with the highest rates of growth in the developing world where previously limited access to antibiotics is improving (Klein et al., 2021). However, even in high-income countries with active stewardship programs, antibiotic consumption continues to rise (Klein et al., 2021). The U.S. Centers for Disease Control and Prevention (CDC) describes an inappropriate antibiotic prescribing the “most important modifiable risk factor for antibiotic resistence” (Sanchez et al., 2016).
Approximately 60% of U.S. antibiotic expenditures for human health are associated with outpatient settings where 30%–50% of antibiotic prescriptions are inappropriate (Sanchez et al., 2016; Silverman et al., 2017). Unnecessary antibiotic prescription rates are particularly high for nonbacterial acute upper respiratory infections (AURIs) presenting to primary care practitioners such as viral bronchitis, rhinosinusitis, and the common cold (Palms et al., 2018). For example, in one study of 185,014 patients diagnosed with nonbacterial AURI during a visit to a primary care practitioner (the most common documented diagnosis was a common cold), 46% of patients were inappropriately prescribed an antibiotic (Silverman et al., 2017). Sore throat represents 3%–6% of all family physician office visits, and, among sore throats in adults, 85%–90% are caused by viral infections where antibiotics are not indicated (Linder and Stafford, 2001; Worrall et al., 2007). Despite the personal harms associated with side effects of antibiotics and the social harm of propagating antibiotic resistance, inappropriate use of antibiotics for the treatment of sore throat is widespread, with antibiotics prescribed in over 70% of visits (Linder and Stafford, 2001).
There are many predictors and potential explanations for inappropriate antibiotic prescribing (reviewed by McKay et al. (2016)). Perceived patient expectations, a lack of accountability related to inappropriate prescribing, and the additional time needed with a patient to explain that antibiotics are not appropriate were identified as the three leading explanations physicians provided for inappropriate antibiotic prescribing to patients they had diagnosed with (viral) acute bronchitis (Dempsey et al., 2014). Observations in pay-per-encounter systems support the hypothesis that pressure to reduce patient visit length and to increase patient throughput increases the frequency of antibiotic over-prescribing (Neprash et al., 2023; Silverman et al., 2017). For example, in a population wide study, primary care physicians seeing less than 25 patients per day were less likely to prescribe antibiotics for nonbacterial upper respiratory infections than were those seeing 25–44 patients/day; those seeing more than 44 patients/day were most likely to over-prescribe (Silverman et al., 2017). Recency of physician training (Degnan et al., 2021), knowledge gap (McCleary et al., 2021), decision fatigue (Linder et al., 2014), a desire to maintain positive relationships with patients who providers perceive to expect or demand antibiotics (Butler et al., 1998; Fletcher-Lartey et al., 2016; Ong et al., 2007), and a fear of misdiagnosis (Sanchez et al., 2014) also contribute to unnecessary prescription of antibiotics. Accounting for practice, physician, and patient features, there remains substantial heterogeneity in rates of inappropriate prescribing across physicians indicating that individual physicians respond to perceived patient expectations, challenges communicating with patients, decision uncertainty, and time pressures influencing inappropriate prescribing differently (Kitano et al., 2021).
Through patient and provider education, barriers to prescribing, standardized infection control practices, and promotion of tests to improve diagnostic accuracy, stewardship programs seek to reduce the inappropriate prescription of antibiotics and extend the useful life of existing antibiotics (reviewed by McDonagh et al., 2018). In addition, financial incentives can be used to influence the antibiotic prescription behavior of physicians (Martens et al., 2007; NHS, 2010). Financial incentives have improved drug prescription practices in many countries including the United States, but variation exists in the effectiveness of these interventions across physician type and by clinical indication (Anyanwu et al., 2020; Ellegård et al., 2018; Kilaru et al., 2021). Analytical studies of financial incentives targeting inappropriate prescribing may help to explain how variation in payment model design features, physician characteristics, and clinical indications may influence their effectiveness in practice.
Our study extends the analysis of incentive payment models to a mechanism seeking to reduce inappropriate antibiotic prescription. In particular, we develop a stylized physician compensation model to study the interaction between a healthcare payer (payer), representing a publicly funded health insurer seeking to maximize health in the population and reduce the social harm of antibiotic resistance, and a care provider (provider) who makes the antibiotic prescription decision for patients presenting for care. Patients present with symptoms corresponding to a probability of having a bacterial infection for which antibiotics are clinically indicated. We assume that only patients with bacterial infection for which antibiotics are clinically indicated benefit from antibiotics; patients with viral infections or bacterial infections for which antibiotics are not clinically indicated do not benefit from antibiotics but do suffer from potential harms associated with adverse drug reactions (Shehab et al., 2008). The payer offers a payment contract to providers to maximize social welfare, defined as the monetary value of health outcomes less the total costs of providing care. Given the contract payment options and their own expected opportunity cost of additional time spent with the patient to explain appropriate non-pharmaceutical treatment and why antibiotics are not indicated, the provider chooses whether or not to prescribe antibiotics to patients diagnosed with an infection for which antibiotics are not clinically indicated. Note that the expected opportunity cost does not need to be restricted to the time spent with the patient; it can include other potential drivers of inappropriate antibiotic prescriptions such as fear of misdiagnosis and the cost of damaging positive relationships with patients expecting antibiotics.
Our article yields several interesting and policy-relevant findings pertinent to fee-for-service (FFS) physician reimbursement systems. We first consider the case when diagnosis is based largely on clinical interpretation of symptom presentation, as occurs for many common conditions presenting to primary care providers. For example, classification of an infection of the middle ear (Segal et al., 2005), infection of the sinuses (Aring and Chan, 2011), or conjunctivitis (commonly called “pink eye”) (Yeu and Hauswirth, 2020) as bacterial or viral is based on the clinical interpretation of symptom presentation. In such cases, diagnostic accuracy relies on a provider’s own clinical interpretation of symptom presentation, meaning that a provider may change their diagnostic criteria in response to the incentive payments. As such, we will discuss how clinically and professionally acceptable diagnoses, within the bounds of what is reasonable, responsible, and ethical for physicians, can limit the unintended impacts of incentive payments. For this scenario, we find that the socially optimal level of antibiotic prescription (first-best policy) is defined by a threshold policy on providers’ diagnosis along the symptom distribution domain. In particular, this threshold is a point along the symptom distribution where the probability of antibiotics being clinically indicated is equal to the ratio of the cost of a false diagnosis of a bacterial infection to the total cost of any false diagnosis. We show that an action-based payment comprised of a fixed visit fee and a bonus payment, paid when no antibiotic is prescribed to a patient for whom it is not clinically appropriate, can co-ordinate the system when the opportunity cost of the provider is public information. A similar action-based payment system in Japan, where medical facilities collect an approximately $7 bonus when physicians do not prescribe antibiotics for outpatients diagnosed with acute upper respiratory infections, demonstrated a 17.8% reduction in antibiotic prescribing (Okubo et al., 2022). Next, we consider the case when a provider uses technical investigations to inform diagnosis (e.g., rapid antigen detection or throat culture for the evaluation of group A streptococcus infection in patients presenting with a sore throat (Cohen et al., 2016)). In this case, we find that the socially optimal level of antibiotic prescription is defined by a threshold policy along the symptom distribution such that the provider prescribes antibiotics to patients diagnosed with viral infection only if their symptoms are higher than a threshold. We show that adding an action-based bonus payment can coordinate antibiotic prescription decisions to socially optimal levels. Importantly, while action-based bonus payments can coordinate antibiotic prescription regardless of the diagnostic method, we demonstrate that the use of technical diagnostic testing with high specificity can lead to a reduction in inappropriate antibiotic prescriptions compared to cases where the diagnosis relies solely on a physician’s interpretation of symptoms.
In the realistic case when the opportunity cost is private information, financial incentive imposes a moral hazard on the provider’s prescription decision, increasing the likelihood of inappropriate antibiotic prescription to a patient with viral infection. Shifting the physician’s prescription characteristics reduces the effectiveness of coordinating contracts and the first-best antibiotic prescription policy is no longer achievable. In this case, incentive payments still reduce the rate of inappropriate prescriptions; however, providers with relatively higher opportunity cost continue to over-prescribe compared to the first-best policy and providers with relatively lower opportunity cost may shift their prescription criteria in order to increase the frequency with which they earn the bonus. Through extensive numerical studies, we show that a menu of contracts does not significantly improve the outcome achieved with a single contract.
Finally, we apply our framework to patients presenting to primary care providers with a sore throat. While 85%–90% of sore throats are caused by viral infections, the most common bacterial cause of sore throat is group A
Literature Review
Programs to Reduce Antibiotic Prescribing Behavior
The use, even appropriate use, of antibiotics creates a selection pressure on bacterial populations increasing the prevalence of antibiotic resistant strains such that, ultimately, antibiotics are a limited resource. High levels of antibiotic resistance affect the ability to treat common pathogens including S. aureus, S. pneumoniae, E. coli, group A Streptococcus, and C. difficile (CDC, 2019; Spellberg et al., 2008; WHO, 2021). However, individual patients, doctors, and antibiotic manufacturers have little incentive to moderate consumption. Numerous antibiotic stewardship programs have been implemented to limit the overuse and misuse of antibiotics (Davey et al., 2013; Johnson et al., 2015; McDonagh et al., 2018). For example, some programs implement antibiotic oversight committees to enforce clinical guidelines (e.g., Nathwani et al., 2011), restrict prescribing (e.g., Wang et al., 2020), aim to improve communication between patients and clinicians (e.g., Cals et al., 2013), inform clinicians about their own prescribing rates relative to their peers (e.g., Milani et al., 2019), leverage the value of public commitments to the judicious use of antibiotics (e.g., Meeker et al., 2014), and provide clinical education for healthcare professionals (e.g., Weiss et al., 2011). Multifaceted strategies have been shown to be most effective at reducing inappropriate antibiotic prescriptions (Llor and Bjerrum, 2014).
Empirical studies evaluating financial incentive programs have shown reductions in overall antibiotic prescribing and, in some cases, specific reductions in inappropriate prescribing. Yip et al. (2014) find that salary capitation combined with a pay-for-performance (P4P) payment model decreased antibiotic prescription by 15% in rural northwest China. Gong et al. (2016) showed that a regular review of prescription practices, feedback, and financial penalty for inappropriate prescribing dramatically reduced total inpatient and outpatient prescriptions. Financial incentives introduced to target inappropriate antibiotic prescribing by primary care physicians in the United Kingdom had the greatest impact on practices with a high baseline rate of prescribing (Anyanwu et al., 2020). Martens et al. (2007) showed that a one-time financial incentive that is independent of behavior leads to a small and short term change in prescribing practices. Ellegård et al. (2018) found that, in Sweden, a P4P model focused on shifting prescription patterns towards narrow-spectrum antibiotics (vs. broad-spectrum antibiotics) significantly increased the proportion of prescriptions that were narrow-spectrum. In contrast to these empirical studies, in this article, we design co-ordinating contracts that, in addition to considering healthcare spending, also attempt to improve the health outcome of patients by reducing the inappropriate use of antibiotics.
Coordination Mechanisms in Healthcare and Supply Chain
Our research is connected to the stream of literature in healthcare operations and supply chain management that uses applied game-theory to design coordination mechanisms (Betcheva et al., 2021; Keskinocak and Savva, 2020; Tsay and Agrawal, 2004). These studies mainly focus on outcome or performance-based coordination mechanisms. A stream of healthcare operations management studies focuses on payment contracts between payers and care providers, such as gainsharing agreements between a hospital and physicians (Gupta et al., 2021), bonus compensation for competitive providers (Jiang et al., 2020), and reference pricing (Nassiri et al., 2022). Bravo et al. (2023) and Ghamat et al. (2021) proposed performance-based payments to incentivize care coordination between post-acute care providers and hospitals, encouraging cost reduction and high-quality care as required by Medicare’s bundled payment models. Unlike these studies, we investigate an action-based payment model, where contract payments depend on the treatment choice of the physician. This distinct modeling feature captures a unique challenge of applying incentive payments in the primary care setting: the payer cannot evaluate provider decisions by monitoring the health outcome of all patients seeking care for upper respiratory symptoms.
Another stream of work evaluates payment models to incentivize providers to adopt the optimal treatment choice or intensity. Yaesoubi and Roberts (2011) studied payment models that achieve the optimal use of cancer screening tests when the provider is strictly concerned about the cost of treatment. Dai et al. (2017) evaluated the effect of a provider’s misdiagnosis concerns and a reimbursement ceiling on diagnostic test-ordering behavior. Ghamat et al. (2018) evaluated contracts that incentivize providers to use a genetic test for optimal chemotherapy prescription. Similar to our approach to modeling the provider’s altruistic behavior when patients receive inappropriate treatment, Dai et al. (2017) and Ghamat et al. (2018) explicitly modeled the provider’s concern about misdiagnosis and show that both over-testing and under-testing are possible outcomes of these concerns. Andritsos and Tang (2018) considered a co-managed situation where both the provider and the patient can exert effort to reduce hospital readmission rates. They show that bundled payment outperforms FFS by causing the provider and the patient to exert more effort to reduce readmission. Adida et al. (2017) study the impact FFS and bundled payment models have on patient selection and treatment intensity decisions finding that FFS results in over-treatment of patients. In another study, Adida and Dai (2023) examined the impact of physician compensation on their diagnostic effort and use of confirmatory tests. Unlike our study, these studies do not incorporate the misuse of treatment and the social harm of over-testing/over-treatment. Moreover, in addition to studying the impact of payment models on treatment choice, we evaluate potential unintended consequences of incentive payment models on the diagnosis behavior of a provider.
Model
Overview
We consider a system consisting of a payer, a healthcare provider, and a sequence of patients experiencing symptoms which could be due to either a bacterial infection for which antibiotics are clinically indicated (“bacterial”) or an infection (viral or bacterial) for which antibiotics are not clinically indicated (denoted for succinctness in the analysis sections as “viral”). We explore the existence and features of payer–provider contracts designed to reduce antibiotic prescriptions for viral infections and maximize social welfare. Because it is not possible for the payer to observe the true health state of all patients prescribed antibiotics nor to observe the social harm of increased antibiotic resistance from each inappropriate prescription, we focus on alignment of diagnosis and prescription decisions. We investigate a simple action-based payment contract in which the payer offers a contract
Our mathematical model captures trade-offs faced by physicians. Physicians are motivated to diagnose patients correctly and not to prescribe antibiotics inappropriately through an altruistic utility they obtain from a positive change in their patients’ health. However, factors such as fear of misdiagnosis, challenges communicating with patients, desire to maintain positive relationships with patients, and the additional time it can take to explain why antibiotics are not indicated and to recommend a course of non-pharmaceutical treatment when patients are expecting an antibiotic imposes an opportunity cost that incentivizes physicians to prescribe antibiotics to patients diagnosed with viral infection (Butler et al., 1998; Dempsey et al., 2014; Silverman et al., 2017).
Patient Encounter and Patient Outcomes
Consider a sequence of patient–provider encounters where, at each encounter, a patient presents with symptoms that are denoted by a random variable
We present health benefits, which naturally occur in terms of alleviated symptoms and severe outcomes averted but may also include monetary gains from averting lost days of paid work, solely into monetary units. In practice this unit-conversion can be done using the marginal willingness-to-pay threshold of the decision maker for health benefits (Neumann et al., 2016). Treatment with antibiotics provides an expected monetary health benefit
The provider may also prescribe antibiotics for patients diagnosed with viral infection because of uncertainty in the diagnosis and/or the opportunity cost of the provider rising from factors such as challenges communicating with patients, desire to maintain positive relationships with patients, and time spent explaining to a patient who expects antibiotics why they are not indicated. Therefore, the provider makes a decision
Since the likelihood of a bacterial infection increases with
The provider’s decision
Lemma 1 suggests that under the optimal prescription policy, the provider prescribe antibiotics to a patient diagnosed with a viral infection if and only if they exhibit symptoms more consistent with a bacterial infection over a specific threshold in
Factors beyond patients’ symptoms (e.g., communication challenges, understanding of viruses compared to bacteria, or their relationship with the provider) may also affect the opportunity cost of the provider. Our model assumes that these personal characteristics are evenly distributed along the symptom levels, so they do not affect the provider’s prescription decision on threshold
A schematic of sequence of events leading to possible patient health outcomes, with their corresponding probabilities for symptom profile
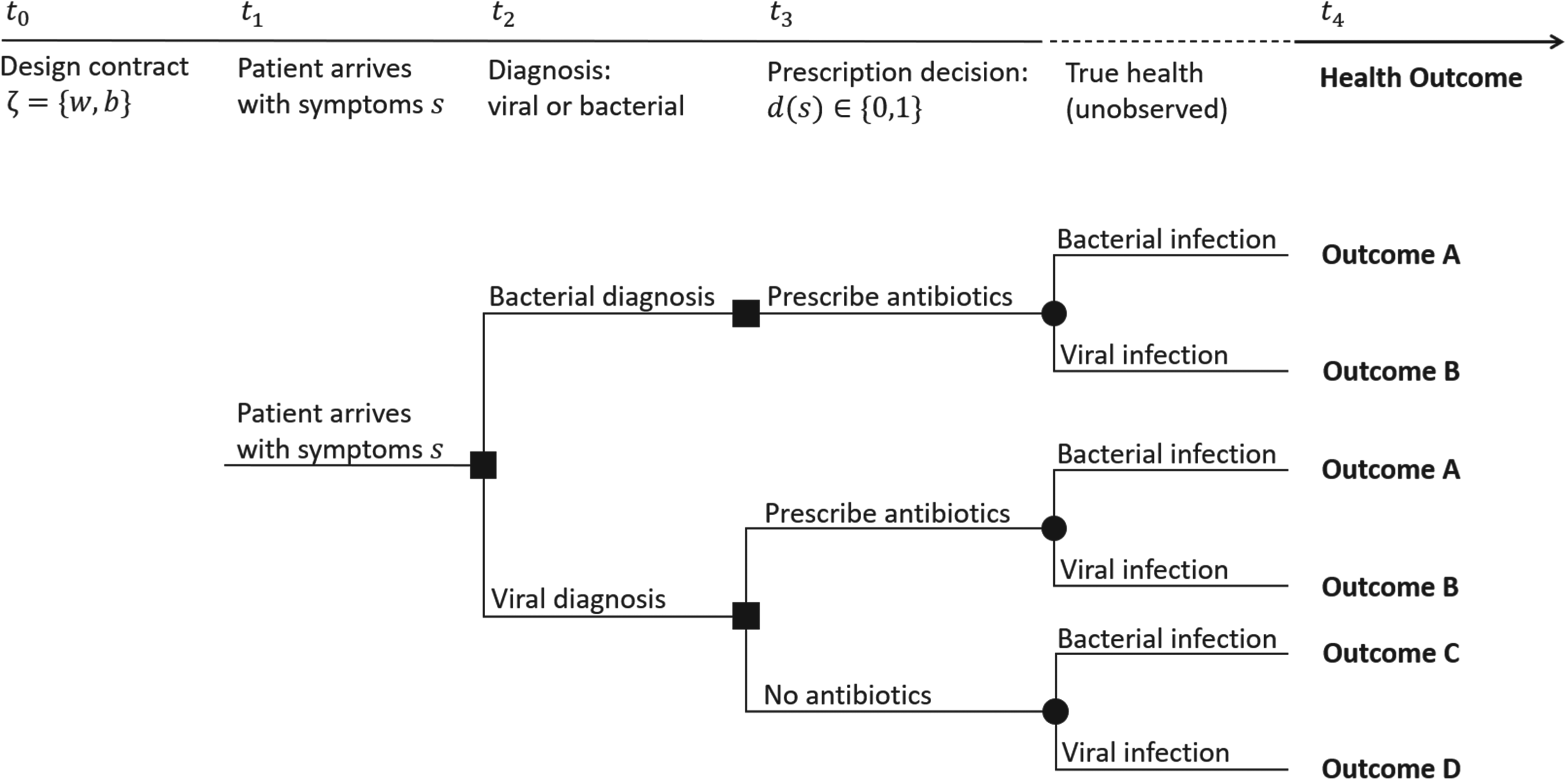
Sequence of events. The provider diagnoses the patient as having a bacterial or viral infection and then decides
Patients who truly have bacterial infection and receive antibiotics (Outcome A) receive health benefit
Providers obtain three types of rewards. First, we assume that the provider is altruistic, in that they care about the health outcome of their patients, and receives a utility of
Given the contract
Providers (and payers) do not observe the true health state of the patient. Because outcomes are probabilistic at the individual level, a patient who feels better after a few days may (falsely) attribute this to treatment with an antibiotic and may attribute adverse events from the antibiotic to the original illness. An outcome-based payment is therefore not practical or feasible which is why we propose a fully observable action-based payment where the payer receives bonus
We assume that the payer’s objective is to maximize social welfare, which is the monetary value of patient health outcomes minus the cost of providing care and the social harm of prescribing antibiotics (increased levels of antibiotic resistance). The payer obtains a reward
A contract structure where the payer penalizes the provider for prescribing antibiotics to a patient they have diagnosed with a viral infection leads to a parallel mechanism design and the same optimal solutions. This is because when deciding on antibiotic prescription for patients with viral diagnosis, the provider compares her profit from prescribing antibiotics (minus any applicable penalty) with her profit from not prescribing antibiotics (plus any applicable bonus). Therefore, the penalty and bonus can be captured with a single incentive payment either for prescribing or not prescribing antibiotics.
When diagnosis is established by clinical interpretation of symptom presentation, providers may vary in their diagnostic accuracy depending on how closely they adhere to clinical guidelines, variation in patient physiology and ability to describe symptoms, and potential confounding or masking of symptoms due to concomitant conditions or their treatment. In this situation, it is possible that bonus payments may not only affect physician prescribing decisions, but also unintentionally affect diagnosis decisions. If this occurs, it could harm the health of patients with bacterial infections, who would have, under current reimbursement policy, received a correct diagnosis of bacterial infection and, appropriately, treatment with an antibiotic. If physicians shift their diagnosis criteria in an effort to secure the bonus payment more often, patients with bacterial infections may incorrectly be diagnosed as viral infections delaying correct diagnosis and appropriate treatment.
Since we assume that symptom profiles are arbitrarily sorted based on the increasing likelihood of bacterial infection (i.e.,
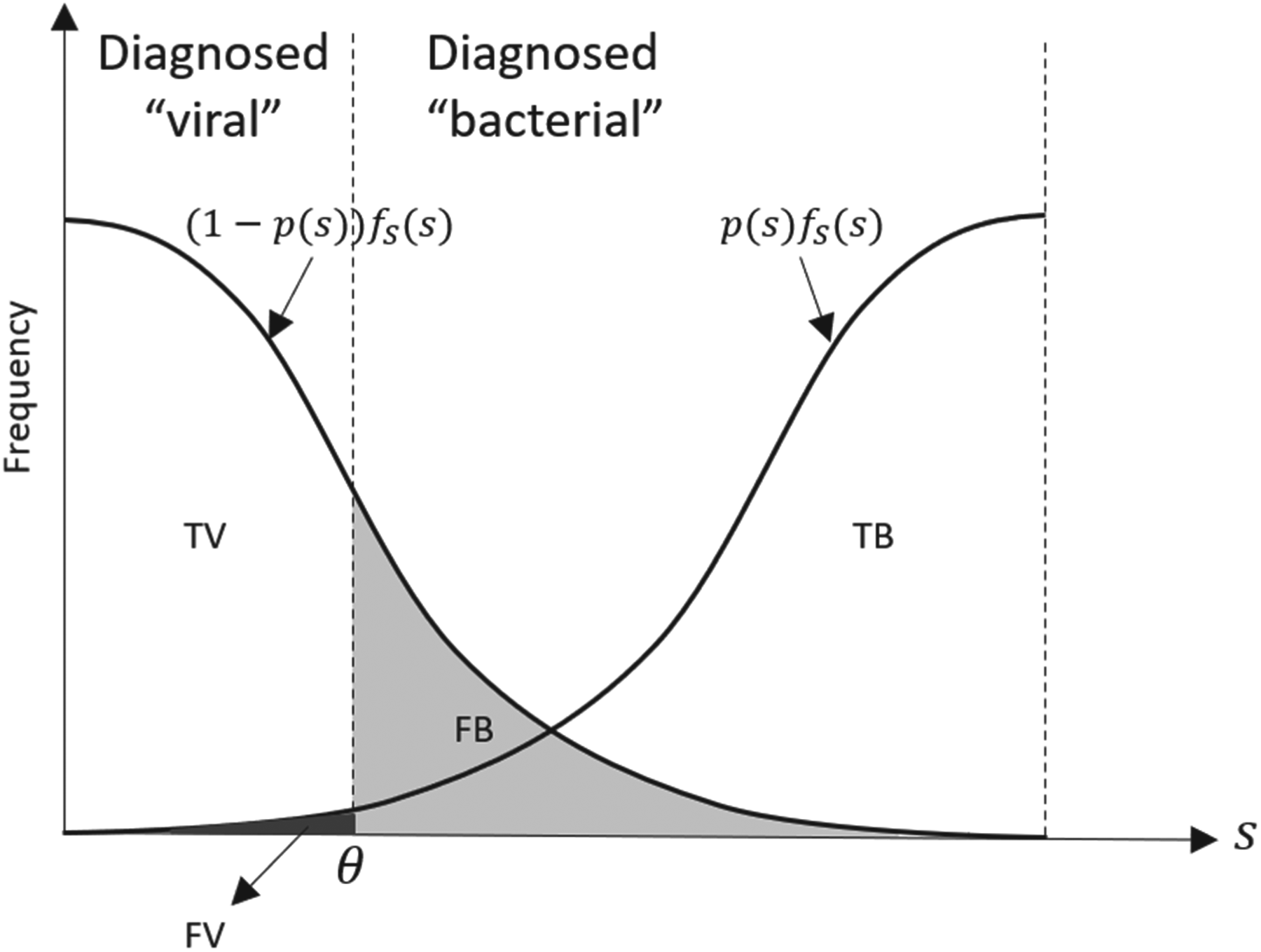
Illustration of the possible diagnostic outcomes over the range of presenting symptoms, using normal distribution for patient symptoms and
After diagnosing patients with either a viral or bacterial infection, the provider chooses the prescription threshold
In this optimization problem, the health outcome of a patient depends on two interacting decisions by the provider: the diagnostic threshold
In practice,
We first analyse optimal antibiotic prescription behavior in an integrated care system as a benchmark (Section 4.1) and then we investigate if an action-based payment model can coordinate care and achieve antibiotic prescription levels similar to the integrated care system (Section 4.2).
To identify the socially optimal use of antibiotics, we define an integrated care system such that the central planner’s objective function is the sum of the payer and the provider’s payoff functions. We define the socially optimal level of antibiotic prescription as the first-best antibiotic prescription policy. Note that in the integrated care system there is no monetary transfer between the two parties. The central planner chooses the prescription (and thus the diagnosis) threshold
Therefore, when choosing the prescription threshold
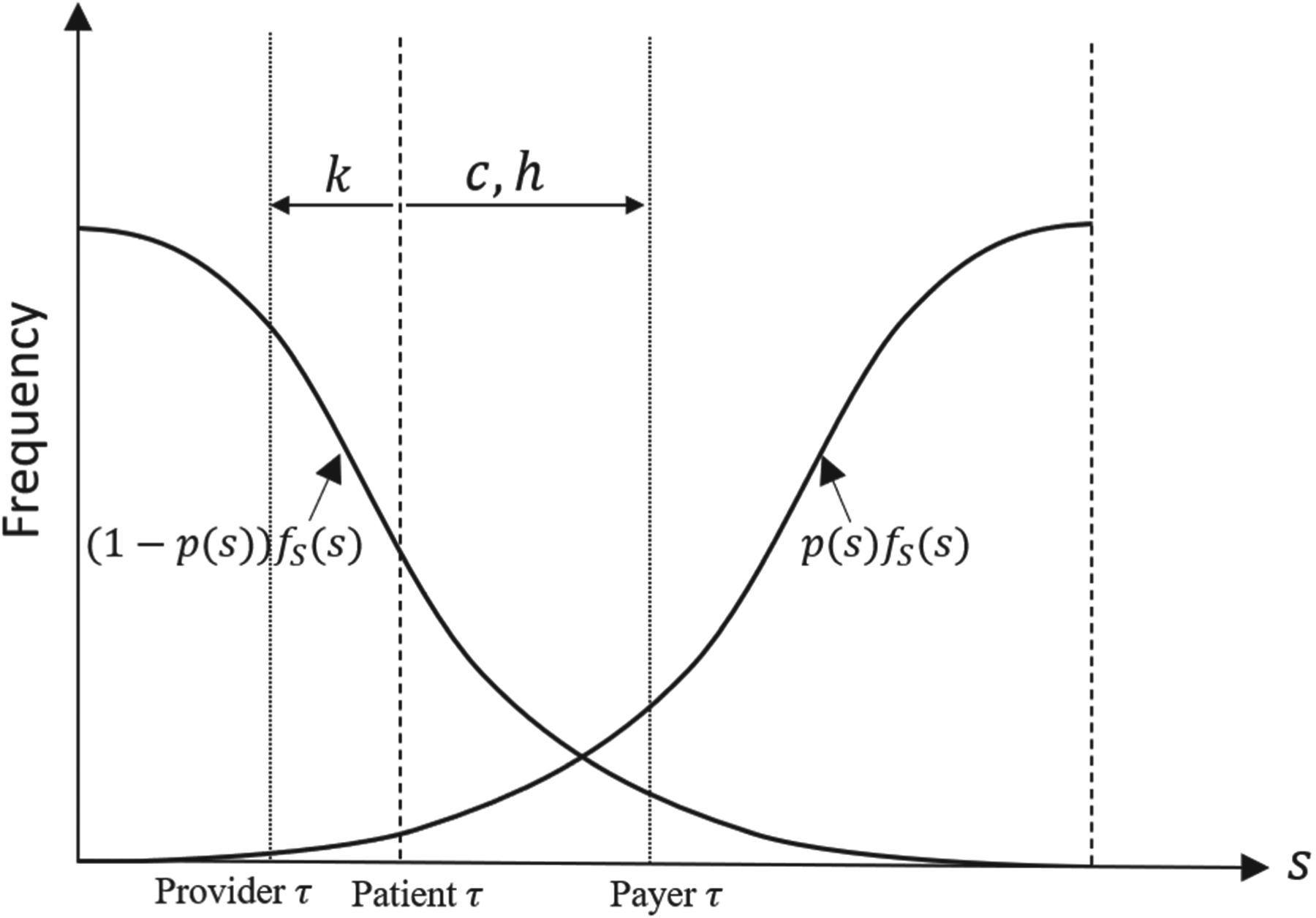
Impact of the provider’s opportunity cost (
Because the true health status of a patient is unknown, the diagnosis and prescription decisions are made given symptom presentation of a patient. Therefore, the central planner should diagnose a patient with symptom level
Since the provider’s opportunity cost threshold
When diagnosis relies on clinical interpretation of symptom presentation, in an integrated care system,
if if
As can be seen in Figure 2, shifting the prescription threshold to the right decreases the chance of FB (decreasing the risk of over-prescribing) and increases the chance of FV (increasing the risk of under-prescribing). Therefore, following the same logic as the critical fractile in the classic Newsvendor problem (Silver et al., 1998), at the optimal threshold
To focus on the realistic cases, throughout the article, we present our propositions assuming that our parameter values (i.e., the prevalence of the bacterial infection, sensitivity and specificity of the diagnosis, and the cost of over- and under-prescribing) guarantee the optimality of prescribing antibiotics to patients diagnosed with bacterial infection, regardless of the opportunity cost of the provider (i.e.,
In this section, we study the decentralized system where the provider’s antibiotic prescription behavior depends on the contract payment terms offered by the payer. Figure 4 illustrates how the forces of the payer’s contract offer balance the provider’s opportunity cost, leading to the selection of a prescription threshold at the socially optimal level (i.e.,
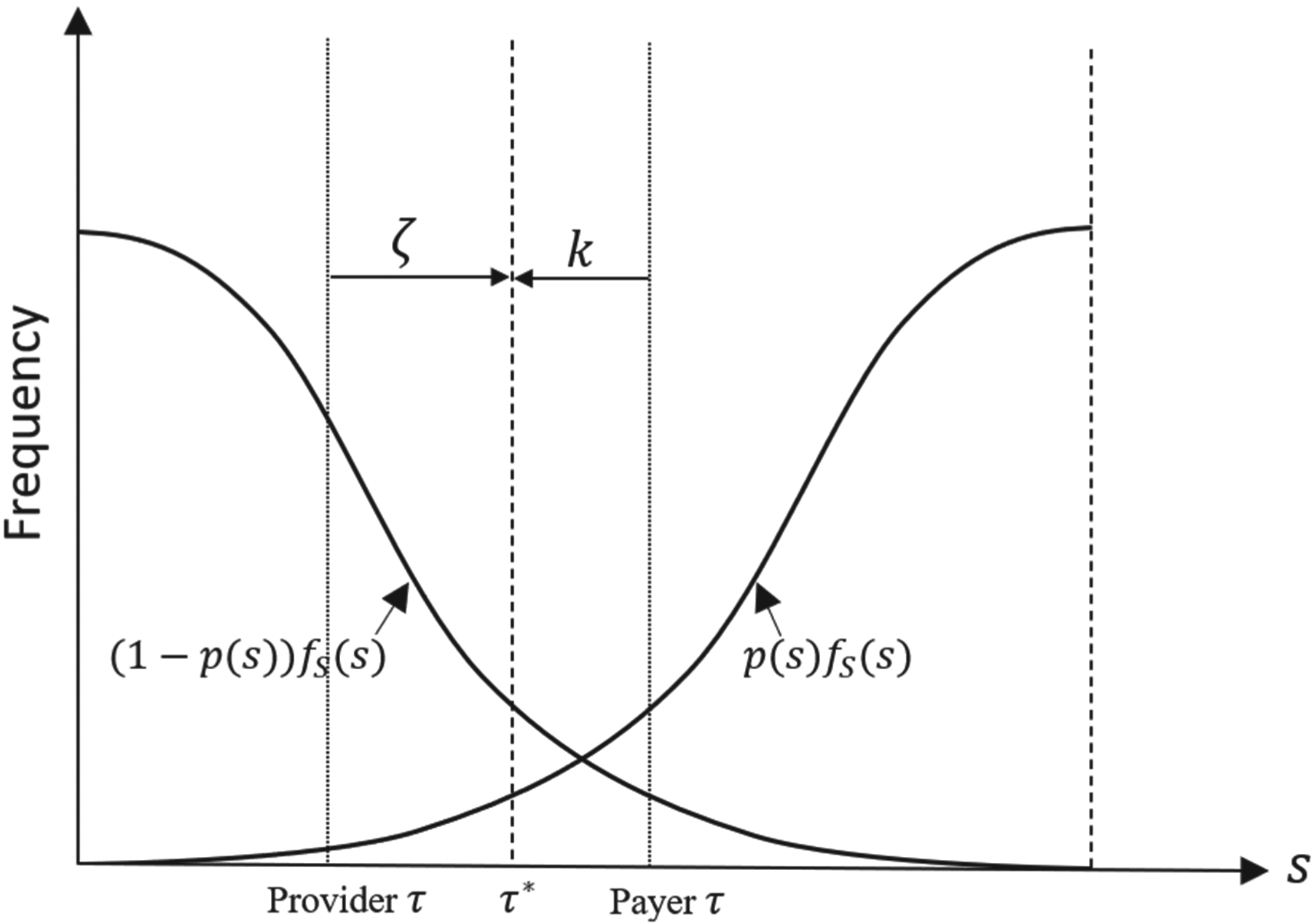
Impact of payment contract (
Similar to case of the central planner, diagnosing a patient with symptom level
When diagnosis relies on clinical interpretation of symptom presentation, given a payment contract
Lemma 2 shows that the provider sets the optimal prescription threshold
Finally, the payer is also looking to balance their cost of over- and under-prescribing, which are
When diagnosis relies on clinical interpretation of symptom presentation, the payer sets the visit fee if if
When the opportunity cost the provider incurs by not prescribing antibiotics to a patient with the lowest bacterial-type symptoms,
Corollary 1 confirms that a payment contract with only a per-visit fee (
When diagnosis relies on clinical interpretation of symptom presentation,
when when fewer inappropriate antibiotic prescriptions, or a lower cost to the payer.
With an action-based payment contract (i.e.,
Note that, in our base model, we do not include the social harm of antibiotic resistance in the provider’s utility function. In a sensitivity analysis, we included the social harm of antibiotics in the provider’s reward function to evaluate the robustness of our findings (details of such analysis are presented in the E-companion). If provider’s do weigh the social harm of antibiotic resistance in their decision making, fewer patients are prescribed antibiotics and a smaller bonus is necessary to further reduce antibiotic prescribing. Despite these changes, our structural findings remain robust.
In this section, we consider the scenario when the diagnosis is informed by the use of a technical diagnostic test, for example, rapid antigen detection or throat culture for the evaluation of Group A streptococcus infection in patients presenting with a sore throat. We assume the diagnostic accuracy of the technical diagnostic is exogenously fixed and characterized by sensitivity, denoted
The central planner may decide not to prescribe antibiotics to a viral diagnosis to avoid over-prescribing (in the case of TV), at the cost of increasing the risk of under-prescribing (in the case of FV). When using a technical diagnostic, the chance of a TV is the posterior probability of a viral infection, conditioned on the test indicating a viral infection, that is,
When diagnosis relies on technical diagnostic test, in an integrated care system,
if if
Threshold
In this section, we present the optimal solution for a decentralized care system where the provider’s optimal antibiotic prescription behavior is in response to the contract payment terms offered by the payer, as well as the diagnostic accuracy. Following the same logic as in Section 4.2, the provider’s response to a payment contract
When diagnosis relies on a technical diagnostic test, given a payment contract
Lemma 3 defines
Knowing the provider’s response, the payer balances their cost of over- and under-prescribing by providing the minimum bonus
When diagnosis relies on a technical diagnostic test, the payer sets the visit fee if if
Similar to the case when diagnosis relies on clinical interpretation of symptom presentation, when the provider’s opportunity cost for the patient at lowest symptom level,
To understand the impact of using a technical diagnostic test on antibiotic prescription decisions, Corollary 2 compares the proportion of true viral patients receiving antibiotics with or without bacterial testing.
All else equal, the use of a technical diagnostic test reduces the rate of inappropriate antibiotic prescription when the test has high specificity (i.e.,
The action-based incentive payment model ensures socially optimal antibiotic prescription levels, both with or without the use of a technical diagnostic test (Propositions 2 and 4). However, Corollary 2 shows that an accurate test can further reduce inappropriate antibiotic prescription by addressing misdiagnosis concerns and the associated costs of misdiagnosis for the payer and provider. We show that compared to the case where diagnosis relies on clinical interpretation of symptoms, the antibiotic prescription threshold is higher when diagnosis relies on a technical diagnostic test, because in this case the provider is unable to alter the diagnosis. Therefore, to reduce inappropriate antibiotic prescription it is sufficient for the test to accurately identify patients who have viral infection (i.e.,
Corollary 3 confirms that a payment contract with only a per-visit fee (
When diagnosis relies on a technical diagnostic test,
if if fewer inappropriate antibiotic prescriptions, or lower cost to the payer.
We apply our framework to patients presenting to primary care providers with a sore throat. Antibiotics are indicated in approximately 10%–15% of cases, when patients have a GABHS infection (“strep throat”), but antibiotics are prescribed in
A detailed exposition of all assumptions and inputs for the case study are presented in E-companion. As stated earlier, we assume the overall prevalence of bacterial infection is 10%, that is,
Diagnosis Relies on Symptom Presentation
Without the rapid test, diagnosis of GABHS relies on an assessment of clinical symptoms. Consistent with high observed rates of antibiotic prescriptions to patients presenting with sore throat in practice (Linder and Stafford, 2001; Humair et al., 2006; McIsaac et al., 2004), the opportunity cost associated with not prescribing antibiotics incentivizes physicians to prescribe antibiotics to a larger group of patients than are clinically indicated. Based on the rewards and probabilities of each outcome, the patient, the payer, and society strictly prefers a policy of no antibiotics to patients diagnosed with viral infections, but the provider prefers a policy of prescribing antibiotics to patients diagnosed with a viral infection.
We illustrate the provider’s expected reward for various
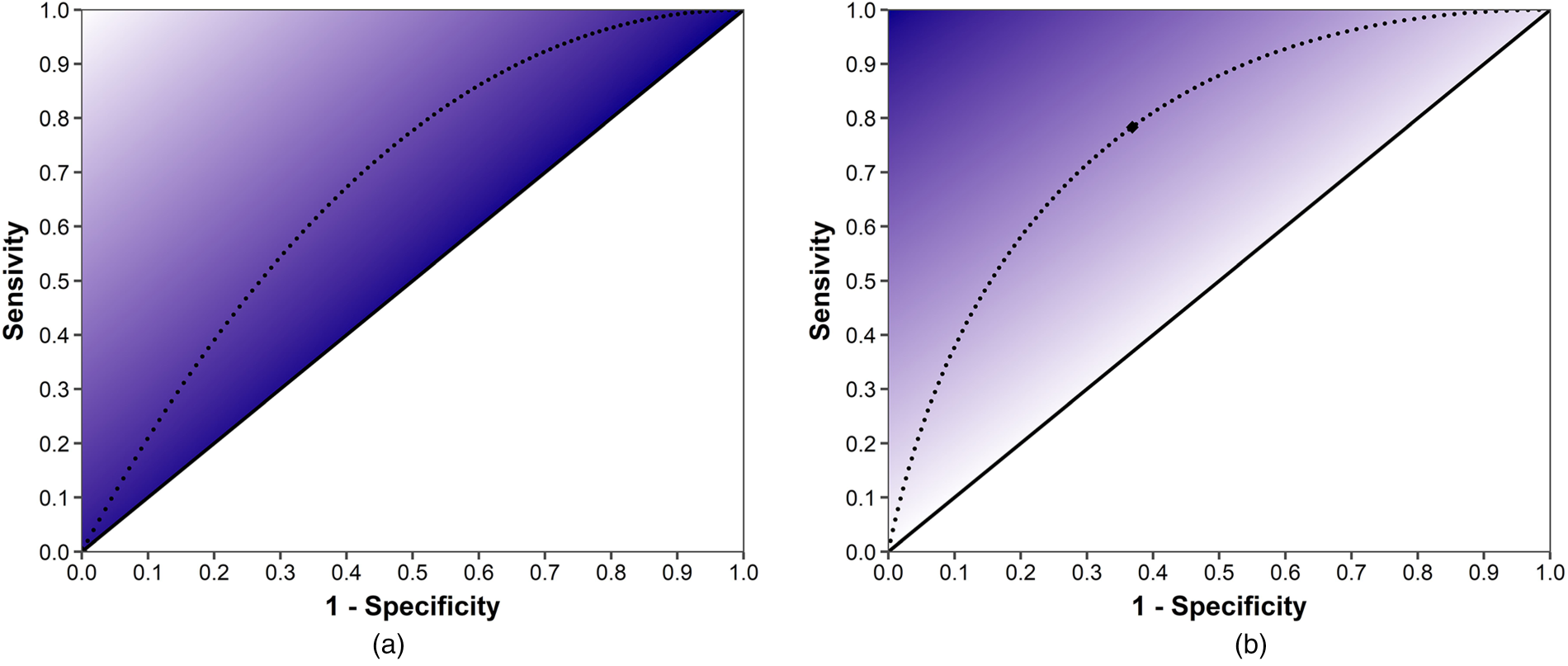
Provider’s relative reward at various diagnostic thresholds in response to bonus contract. Solid line represents where sensitivity equals specificity; diagnostic accuracy below this line would indicate a mislabeled diagnostic. Dotted line represents the receiver–operator curve. Providers can move their diagnostic accuracy along this curve;
We present the case of
A rapid antigen test is recommended for patients presenting with a sore throat who have an intermediate modified Centor scores of 1, 2, or 3 (Choby, 2009). With the rapid antigen test, more patients receive an accurate diagnosis. However, providers are still inclined to prescribe antibiotics to patients diagnosed with a viral infection to avoid the opportunity cost incurred when taking the time to explain that antibiotics are not indicated for viral infections and patient disappointment when their provider does not provide expected treatment. This inclination to prescribe antibiotics to patients diagnosed with viral infections remains true as long as the provider’s opportunity cost exceeds
Sensitivity analysis demonstrates that the optimal bonus payment is robust to the prevalence of bacterial infection and to the provider’s level of altruism (Figure 6(a)). However, the optimal bonus payment is highly sensitive to the provider’s opportunity cost (Figure 6(b)). Using equation (10), we find that there is a coordinating contract acceptable to both the payer and the provider as long as the provider’s opportunity cost is < $94.55.
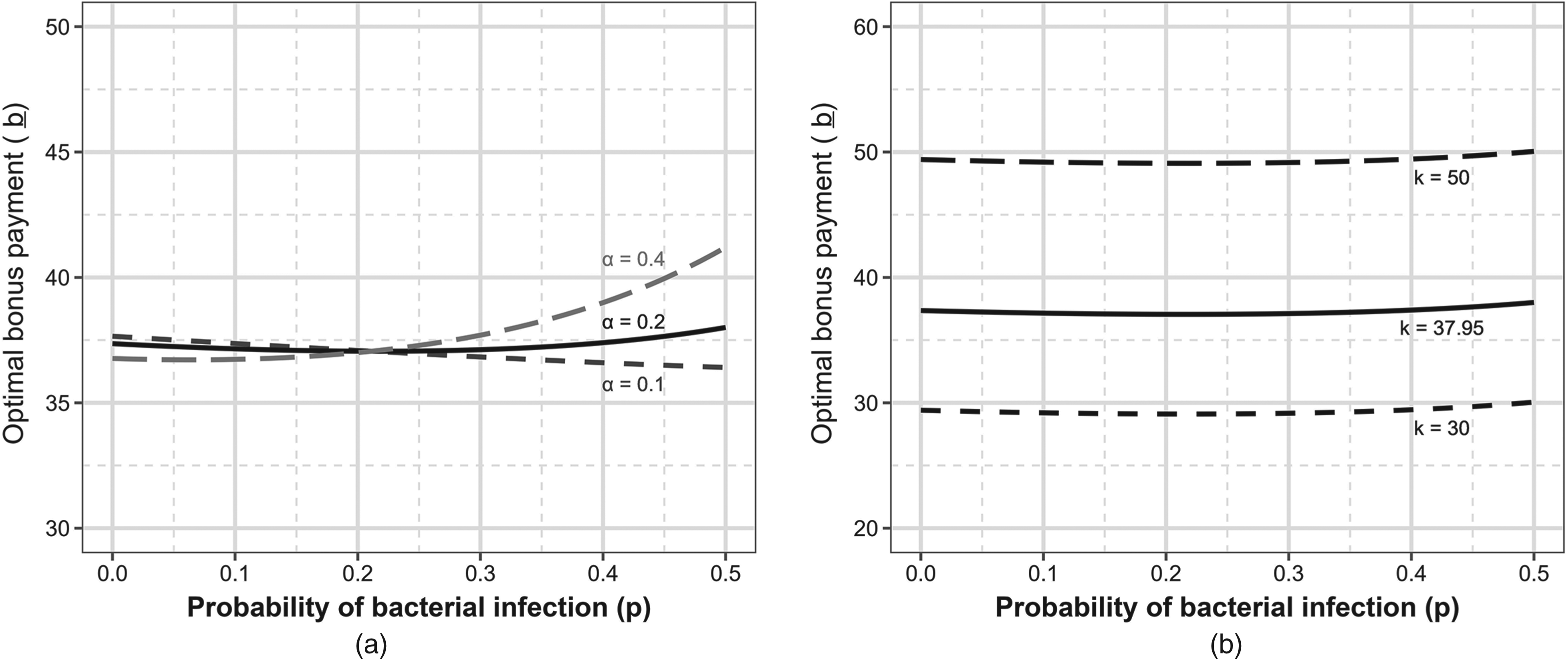
Sensitivity analysis on the optimal bonus payment when diagnostic accuracy relies on a technical diagnostic test, varying the probability of bacterial infection (
Rapid antigen tests are a low-cost diagnostic already recommended for use in patients presenting with sore throat who, based on their symptom profile, have a moderate probability (10%–40%) of GABHS infection (Kalra et al., 2016). Rapid tests have greater sensitivity and specificity compared to clinical assessment, remove substantial subjective judgement from the clinical diagnosis process, and do not require waiting 24-hours for the results of a throat culture. Bonus payments incentivize physicians to not prescribe antibiotics to patients they diagnose with a viral infection. However, when diagnosis relies on the clinical interpretation of symptom presentation, bonus payments also incentivize providers to shift their diagnostic accuracy, increasing the number of patients diagnosed with a viral infection, potentially to the clinical detriment of patients who truly have bacterial infections. A high-quality point-of-care diagnostic takes time to use which may extend the duration of a patient’s visit, but the results of the diagnostic may support the provider’s efforts to explain why they are not prescribing antibiotics. Ultimately, use of a high-quality diagnostic tool protects the health of patients with bacterial infections when implementing an incentive payment to reduce inappropriate antibiotic prescribing.
Specific to this example, a rapid strep test costs the payer $5.70 (Ontario Ministry of Health and Long Term Care, 2023) and an antibiotic prescription costs $12.55. We calculated the value of the test to the payer by comparing the expected value of outcomes, using the optimal bonus, with and without the rapid strep test. We identify that the test value exceeds the current cost of providing the test ($5.75), for patients with expected pre-test probability of bacterial infection between 9% and 65% (Figure 7), which is a broader range than is currently recommended. Therefore, it is cost-effective to insist providers use this low-cost point of care test for the majority of patients presenting with sore throat with a contract optimized for the diagnostic accuracy of the test, rather than offer a contract optimized for the poorer diagnostic accuracy of physician assessment alone.
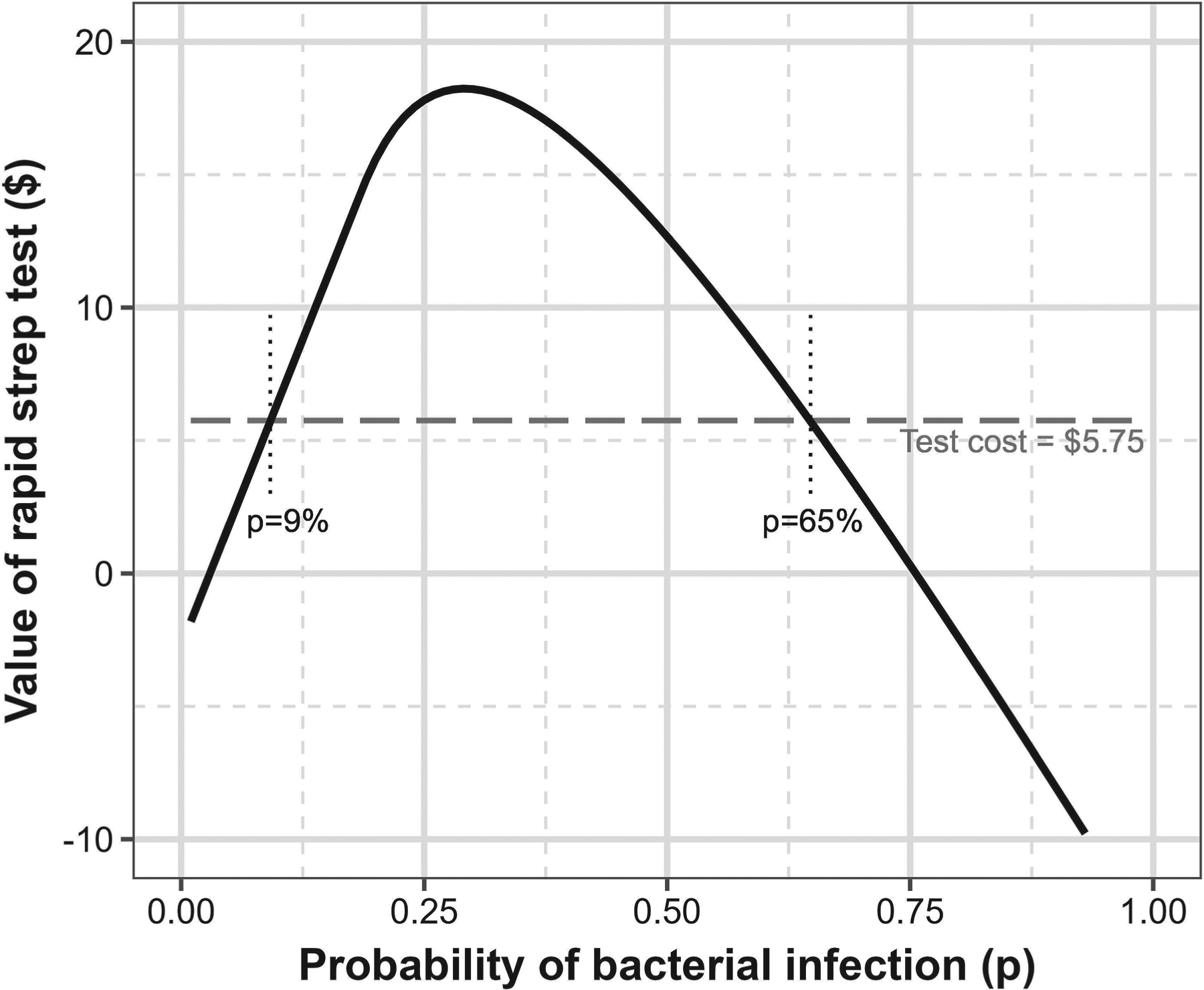
Value of a rapid strep test, varying the probability of bacterial infection (
So far, we assumed that the opportunity cost is public information. However, as discussed in Section 3.3, there might be heterogeneity in the expected opportunity cost of providers because of differences in factors such as physician training, fear of misdiagnosis, challenges communicating with patients, and time pressure. This results in different perceived opportunity cost by providers that cannot be known or observed by the payer. Therefore, in this section, we study the impact of private information of the providers about their opportunity cost on the application of incentive payments in achieving the socially optimal levels of antibiotic prescription defined in Sections 4.1 and 5.1.
We assume that the provider can be one of two types: low opportunity cost (type-
First, in Section 7.1, we extend our analysis to situations where the payer offers a single contract to both provider types. Then, in Section 7.2, we consider offering menu of contracts to providers.
Single Contract for Both Provider Types
To incorporate the two types of providers, we update the payer’s problem when diagnosis relies on clinical interpretation of symptoms, defined in (OPT1), as shown in (OPT3).
We illustrate the behavior induced by the optimal contract for the case where the diagnosis relies on additional diagnostic testing in Figure 8 (using the parameters of the case study in Section 6, and assuming both opportunity costs
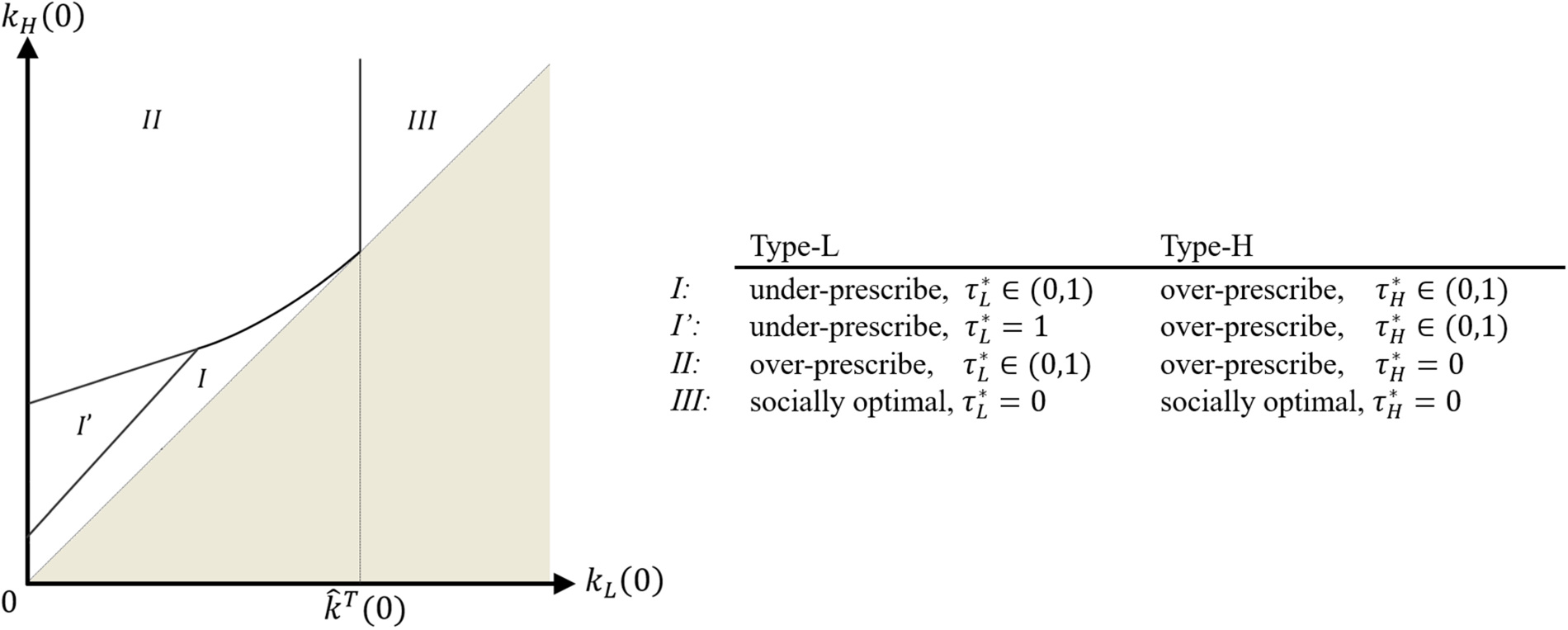
Antibiotic prescription behavior of provider when opportunity cost is private information.
When
A single contract needs to balance the incentive of type-
Throughout our analysis in this section, we assumed that there are only two types of providers which is a common assumption in the literature (e.g., Yaesoubi and Roberts, 2011; Cakanyıldırım et al., 2012; Xiao and Xu, 2012). However, extending the analysis to multiple types follows directly. The unique opportunity costs of providers can be ordered such that
The payer may consider offering a menu of contracts to achieve coordinated care while learning the provider’s true type (i.e., opportunity cost). In this section, we extend our analysis to evaluate how menu of contracts impact outcomes when bonus payments can affect the diagnosis behavior of the provider. In particular, the payer’s problem becomes determining a menu of contracts
This problem is not mathematically tractable because the provider can change the proportion of the patients receiving antibiotics by changing the prescription threshold that is their private information (moral hazard). Therefore, when applying the revelation principle, a menu of contracts may not guarantee IC. Specifically, when switching between the contracts, the provider can always maintain the outcome from the other contract by adjusting their prescription threshold. Through extensive numerical studies, we observe that when the optimal diagnostic threshold is an interior solution (i.e.,
Overuse and misuse of antibiotics significantly contributes to antibiotic resistance and adverse patient outcomes. In this study, we examine the implications of incentive payments on primary care physician antibiotic prescription behavior. In particular, we develop a physician compensation model to study the interaction between a healthcare payer and a care provider who makes the antibiotic prescription decision for patients presenting with symptoms potentially representing a viral or bacterial infection. To maximize social welfare, the payer offers an action-based payment contract to providers, which includes a bonus payment for not prescribing antibiotics to patients diagnosed with a viral infection. In doing so, the payer is seeking to compensate physicians for the time spent explaining to patients with a viral diagnosis why antibiotics are not indicated. The provider decides whether to prescribe antibiotics to patients they have diagnosed with viral infection. We consider both interpretation of symptom presentation and technical diagnostic testing for diagnostic accuracy and so we explore the possibility that incentive payments may affect the diagnosis behavior of the provider.
Our research provides practical guidance for payers regarding incentive payments to manage antibiotic use and reduce the frequency of inappropriate prescriptions. Objective diagnostic information is not available in all circumstances and so diagnostic classification relies on physician assessment of clinical symptoms. In this case, diagnostic accuracy becomes endogenous and it is possible that incentive payments will manipulate a provider’s diagnostic assessments. In this setting, we show that a simple action-based payment contract with bonus payment can coordinate provider behavior to socially optimal antibiotic prescription levels. We demonstrate that the bonus payments establish a prescription threshold within the symptom distribution domain. According to this threshold policy, only patients with symptoms above this threshold should be diagnosed with bacterial infection and prescribed antibiotics. Further, we show that, in a scenario where diagnosis relies on additional technical point-of-care testing, there is a socially optimal antibiotic prescription threshold along the symptom distribution domain. Consequently, patients diagnosed with a viral infection who exhibit symptom levels below this threshold will not receive antibiotics. Importantly, the rate of inappropriate antibiotic prescriptions is reduced when diagnoses are based on an additional technical diagnostics. Our results show that adding a bonus payment to the contract terms of providers can only co-ordinate the system when the opportunity cost of the provider is public information.
In realistic situations, a provider’s opportunity cost is private information and a bonus payment creates a moral hazard where, in an effort to increase revenue from the bonus, the provider changes their prescription threshold decreasing the proportion of patients receiving antibiotics. Providers with lower opportunity cost may benefit from information rent by not prescribing antibiotics including to patients that they would have otherwise diagnosed with a bacterial infection, but now diagnose as having a viral infection in order to obtain the bonus. Therefore, while incentive payments would reduce the frequency of inappropriate antibiotic prescriptions, when compared to the first-best policy, providers with high opportunity cost would continue to over-prescribe antibiotics even with bonus payments and providers with a low opportunity cost may over-respond to the incentives adversely affecting patient care. Through extensive numerical studies, we show that when opportunity cost is private information, a menu of contracts does not significantly improve the outcome achieved with a single contract.
Our analysis focuses on one factor influencing inappropriate antibiotic prescribing behavior (avoiding opportunity cost) and one type of stewardship effort (pay-for-performance contracts). Inappropriate prescribing practices include selecting the wrong antibiotic, selection of a broad-spectrum antibiotic when a narrow-spectrum antibiotic would suffice, or prescribing antibiotics for the wrong duration. Opportunity cost is also not the only reason that providers prescribe antibiotics inappropriately. Providers may also be uncertain about which symptom combinations are clinically indicated for antibiotics under the most recent clinical guidelines, the frequency of side effects from antibiotics, or the social harm of inappropriate antibiotic prescriptions. An action-based contract will be most effective if partnered with other antibiotic stewardship efforts demonstrated to reduce inappropriate antibiotic prescribing.
That said, our model provides insight into the effectiveness of other types of stewardship programs. Many programs seek to reduce the opportunity costs for providers by educating patients about when to realistically expect antibiotics and educating physicians about how to engage in effective patient communication. Other stewardship programs target physicians’ diagnostic skills, reducing physicians’ fears of misdiagnosis, and providing physicians access to rapid on-site diagnostics. The availability of point-of-care diagnostic testing can transition some clinical assessments from relying on the interpretation of symptoms to an exogenous diagnostic setting, while increasing physician confidence in the diagnosis. This is important because we found that bonus payments are more effective at reducing inappropriate antibiotic prescribing when diagnostic tests have high specificity. Therefore, clinical diagnostic decisions in which point-of-care diagnostics are available to differentiate between viral and bacterial infections are potentially ideal settings in which the addition of a bonus payment can reduce the rate of inappropriate antibiotic prescribing. However, it is notable that even in these settings, diagnostic accuracy is not strictly exogenous as the provider must also make the decision to use the diagnostic test. While we have shown that it is possible to achieve the socially optimal use of antibiotics using a bonus without risk of manipulating patient diagnoses in settings in which there is a technical diagnostic, the question of whether it is valuable to add a technical diagnostic or incentivize the use of a technical diagnostic depends on features of that test including its cost.
Supplemental Material
sj-pdf-1-pao-10.1177_10591478241264022 - Supplemental material for Influencing Primary Care Antibiotic Prescription Behavior Using Financial Incentives
Supplemental material, sj-pdf-1-pao-10.1177_10591478241264022 for Influencing Primary Care Antibiotic Prescription Behavior Using Financial Incentives by Salar Ghamat, Mojtaba Araghi, Lauren E Cipriano and Michael Silverman in Production and Operations Management
Footnotes
Acknowledgements
The authors are grateful to department editor Sergei Savin, the senior editor, and the three anonymous referees for their many constructive comments and suggestions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
Canada Research Chair in Healthcare Analytics, Management, and Policy (Tier 2) (CRC-950-233070, Cipriano). Canada Research Chair in Business Analytics in Supply Chain (Tier 2) (CRC-2022-00130, Ghamat). Discovery Grants from the Natural Sciences and Engineering Research Council of Canada (RGPIN-2017-05297, Araghi; RGPIN-2020-06665, Cipriano). Insight Development Grants from the Social Sciences and Humanities Research Council of Canada (430-2021-00490, Ghamat).
How to cite this article
Ghamat S, Araghi M, Cipriano LE and Silverman M (2024) Influencing Primary Care Antibiotic Prescription Behavior Using Financial Incentives. Production and Operations Management 33(10): 2051–2072.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.