Abstract
Complications after craniosynostosis surgery occur in 11% to 36% of cases and may be precipitated by poor soft tissue coverage and concomitant exposure of non-sterile regions; sequelae may result in infection, osteomyelitis, and bone loss requiring complex reconstruction. In the pediatric population, autologous cranioplasty remains the gold standard due to growth potential and a more favorable complication profile than synthetic cranioplasty. Virtual surgery planning (VSP) and computer-assisted design (CAD)/computer-assisted manufacturing (CAM) technology can be utilized to create innovative, patient-specific autologous solutions, similar to the approach with synthetic cranioplasty. A novel surgical approach using VSP was used for an 18-month-old female with near total bifrontal bone loss. Surface area measurements were used to determine the amount of bone available to replace the infected frontal bone. VSP was utilized to determine the most efficient construct configuration possible to achieve maximal coverage via calculation of cranial bone surface area measurements. Surgical reconstruction of the defect was planned as a Modified Visor Bone Flap with Posterior Brain Cage. A construct was fashioned from available cranial bone struts to obtain widespread coverage. 3D Recon images from before and after surgery demonstrate almost complete re-ossification of the cranial vault with significant resulting clinical improvement. Reconstruction of total frontal bone loss is possible by utilizing this technique. VSP can improve the safety and efficiency of complex autologous cranial bone reconstructions. We propose a treatment algorithm to address the problem of near total frontal bone loss in young children for whom alloplastic implants are not suitable.
Background
Major and minor complications after surgery for craniosynostosis are estimated to occur in between 11% and 36% of cases and are often due to poor soft tissue coverage and the concomitant exposure of non-sterile regions.1‐5 These complications can result in challenging scenarios such as infection, osteomyelitis, and bone loss which may require complex surgical reconstruction. Cranial bone infections create challenging problems for the craniofacial surgeon to reconstruct and the decision to remove a bone flap is not straightforward, requiring a full understanding of the extent of infection, likelihood of possible salvage, and the morbidity associated with flap removal. 3 While many synthetic options are available for cranial reconstruction, in the pediatric population (less than age of 3 years) where implant-based reconstruction is impractical due to complication profile and potential growth disturbance, there are few options for management of large cranial defects.
Historically, free flaps have been used for soft tissue coverage of cranial defects with Buncke and McLean first describing coverage of a skull defect with a free omental flap in 1972. 6 While free flap reconstruction with vascularized bone is possible (fibula, iliac crest, scapula, etc.), it is often insufficient for large bony defects. Exchange cranioplasty with particulate bone grafting has also been described although reports indicate an upper limit for defect size close to 85 cm2 with larger defects resulting in persistent bony gaps.7,8 Furthermore, non-vascularized calvarial grafts are usually taken as split-thickness grafts whereby the bone is split in the diploic space. In children younger than 3 years, this is not an option as the skull is not yet bicortical and diploe may not be present although this assertion has been challenged for small bone defects.7,9,10 Thus in children under the age of 3 years, a real challenge exists as autologous options are limited. Presently, in the literature to date, management of total frontal bone loss in children is not well defined. In this patient population, the ideal solution would be an autologous one without the need for an extra-cranial donor site, and one that would allow for adequate protection of the brain while providing an idealized contour.
With the advent of virtual surgical planning (VSP), new solutions to complex reconstructive problems can be generated to maximize the use of autologous bone. Advantages of autologous bone reconstruction from the calvarium include lack of a donor site, incorporation of bone, revascularization, and capacity for growth which is essential in young children. In young patients in which alloplastic reconstruction is suboptimal, and autologous bony reconstruction is preferred, novel solutions to address significant bone loss are needed.
While significant frontal bone loss is rare, this problem can result in devastating consequences especially in young children in whom reconstructive options are limited. Here we demonstrate a novel approach and treatment algorithm to address total bifrontal bone loss in young children.
Methods
A surgical approach using VSP was used to address near total bifrontal loss in an 18-month-old female. This patient was diagnosed with right coronal synostosis at 5 months of age and treated with frontal orbital advancement at 11 months of age. She experienced postoperative infection 5 days after her index surgery requiring multiple wound washouts without resolution and ultimately required hardware and bone explant at 8 months of age after which she was continued on rifampin and vancomycin for 3 weeks postoperatively with normalization of her ESR and CRP. She presented for reconstruction at 17 months of age with a large bifrontal cranial defect.
To specifically address this problem, we loosely adapted the “visor flap” technique, a method which has been described for soft tissue reconstruction of the scalp. 11 The maximal advancement of skin flaps afforded by the visor flap is predicated on wide undermining and elevation of a bi-pedicled flap with disinsertion of soft tissue from its posterior attachments. This concept, applied to bony reconstruction, involves transposition of bone from the vertex of the cranium and more posterior portions, moving it forward like a motorcycle visor to replace missing frontal bone (Figure 1). However, in our case, the traditional visor flap concept was found to be inadequate to reconstruct the large defect. Therefore, we chose to transpose bone from the occiput to cover the frontal defect and utilized the remaining bone available for reconstruction as bone struts to obtain widespread coverage of the posterior calvarium. Surface area measurements were used to determine the amount of bone available to replace the infected frontal bone, as well as the resultant secondary defect. VSP was used to calculate the most efficient configuration of bone, which we have termed a “brain cage”. Given the osteogenic potential of dura in patients less than 2 years, this patient population is especially amenable to autologous reconstruction and in this case, we were able to establish a total cranial construct from limited remaining bone. 7
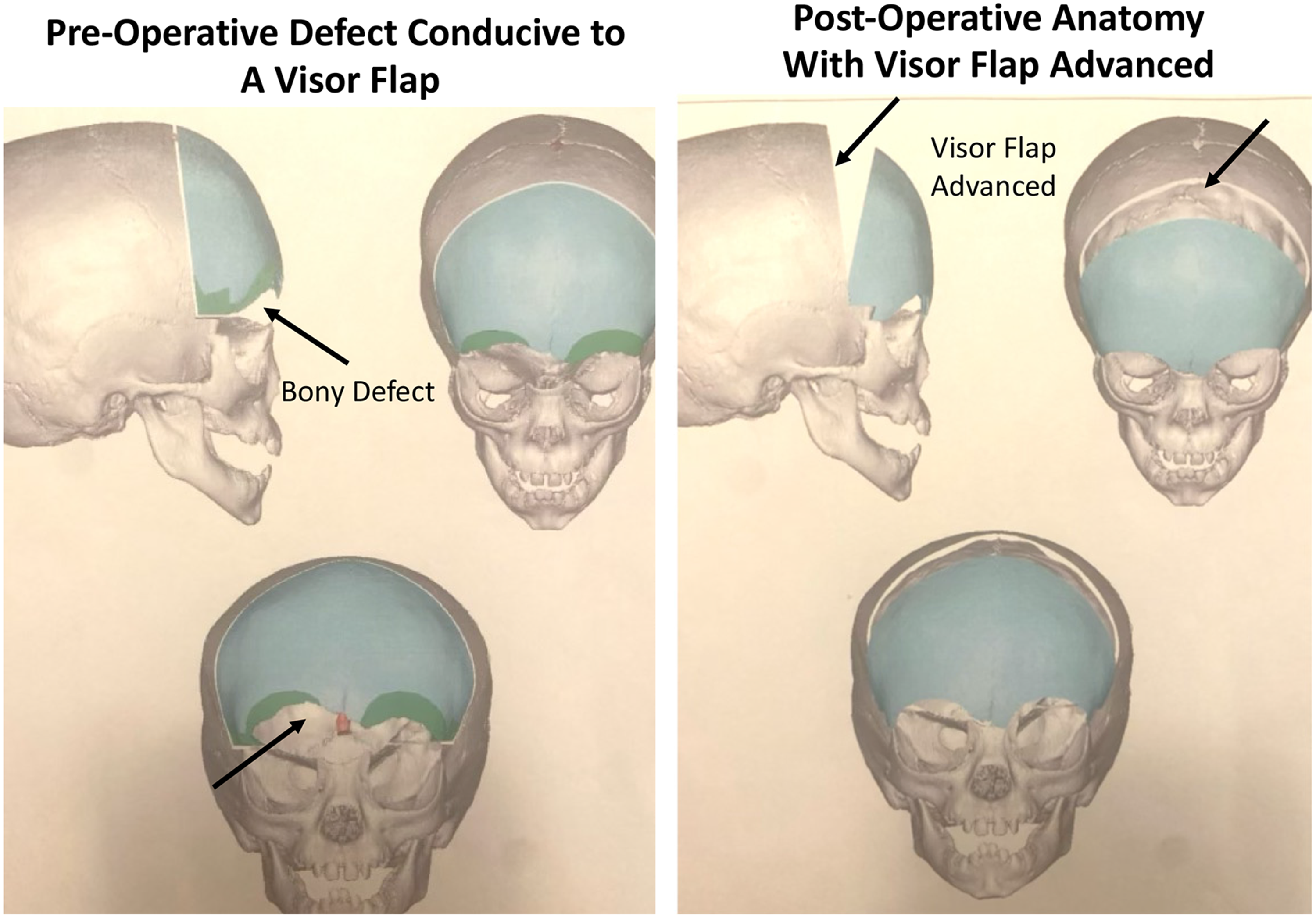
Schematic of visor flap for frontal bone reconstruction.
Results
Using VSP, surgical reconstruction of the bifrontal bone defect was planned as a Modified Visor Bone Flap with Posterior Brain Cage. The patient underwent posterior cranial vault remodeling with a construct fashioned from available cranial bone struts to obtain widespread coverage over the parietal-occipital bone area. VSP was utilized to determine the most efficient construct configuration possible to achieve maximal coverage via calculation of cranial bone surface area measurements (Figure 2). The occipital bone was then remodeled to replace her missing frontal bone bilaterally. In this fashion, both the posterior and anterior vaults were remodeled to address significant frontal bone loss. In the table, the pieces were fashioned together to create a “brain cage” (Figures 2 and 3). The 3D recon images from before and 9 months after surgery demonstrate almost complete re-ossification of the cranial vault with significant resulting clinical improvement (Figures 4 and 5)
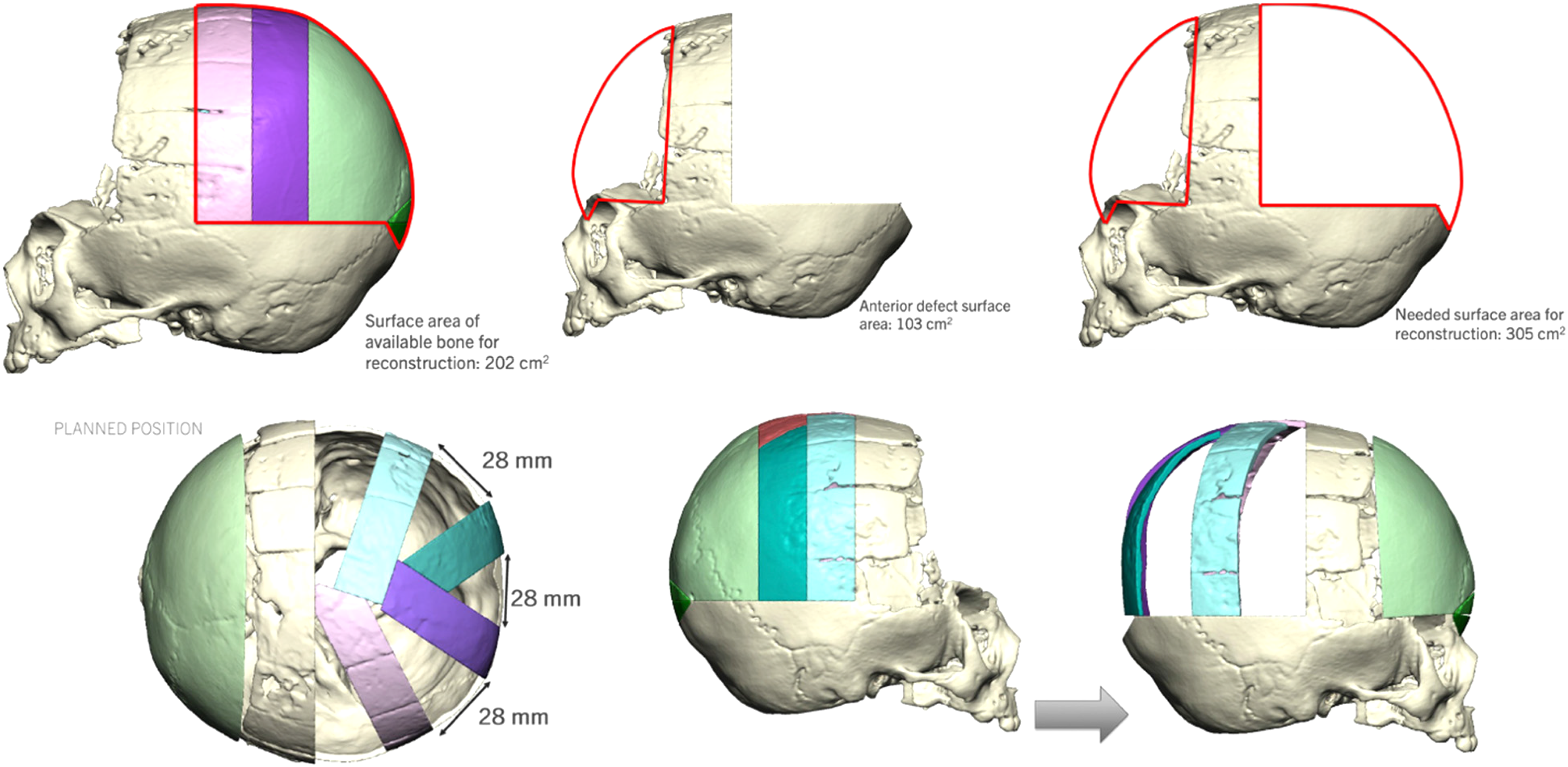
Virtual surgical planning for pre-operative planning.
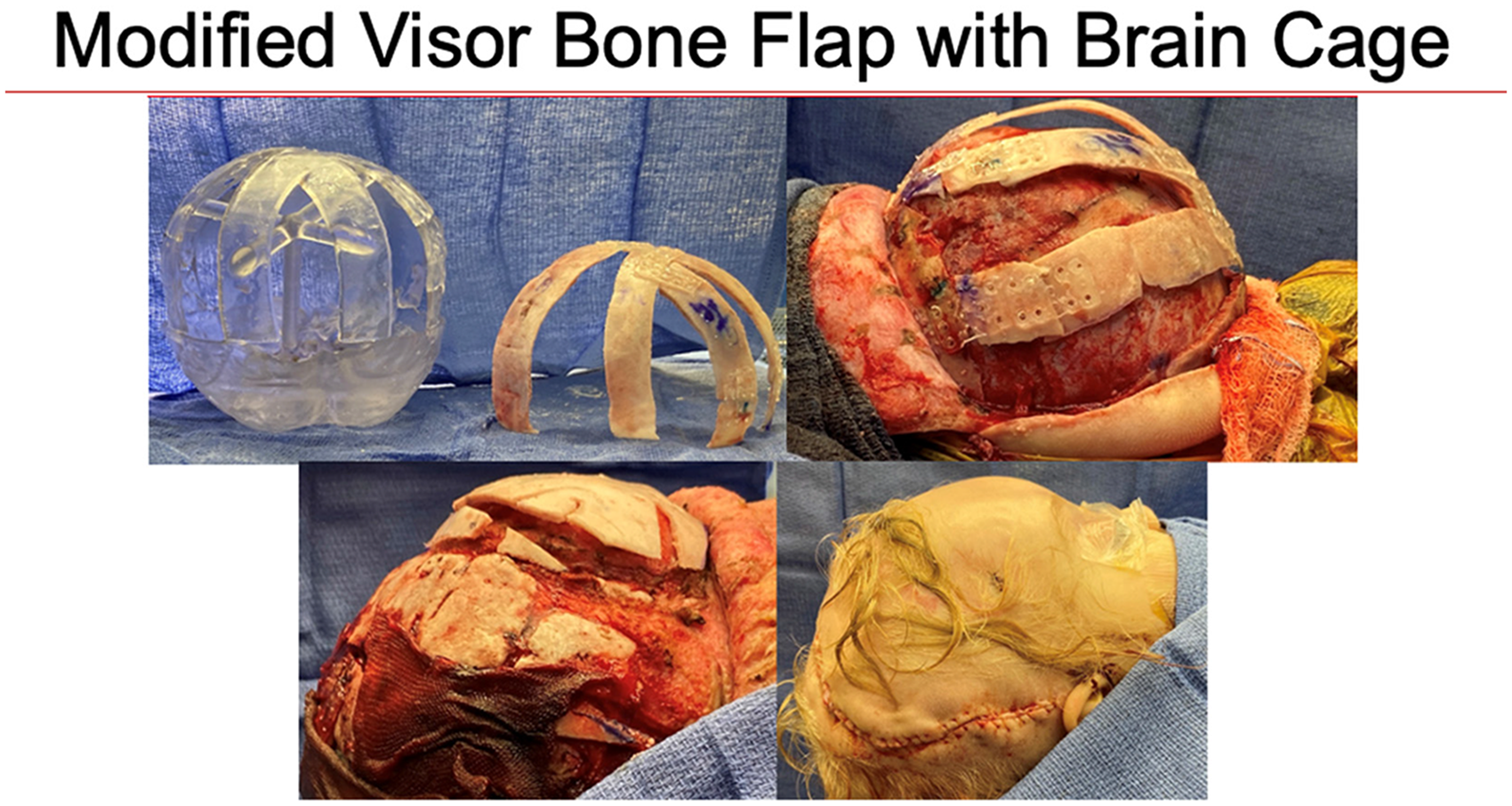
Intraoperative illustration of the brain cage construct.
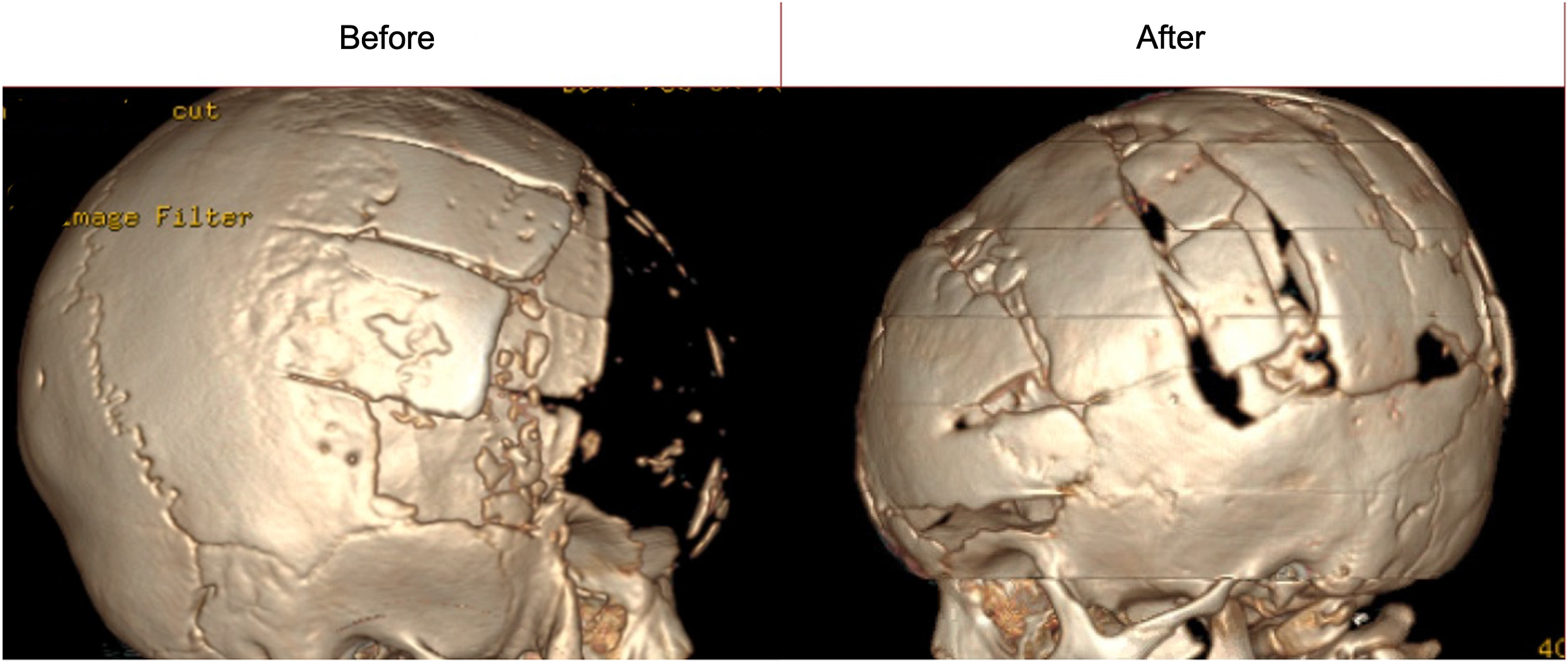
Before and after CT images demonstrating bony regeneration.
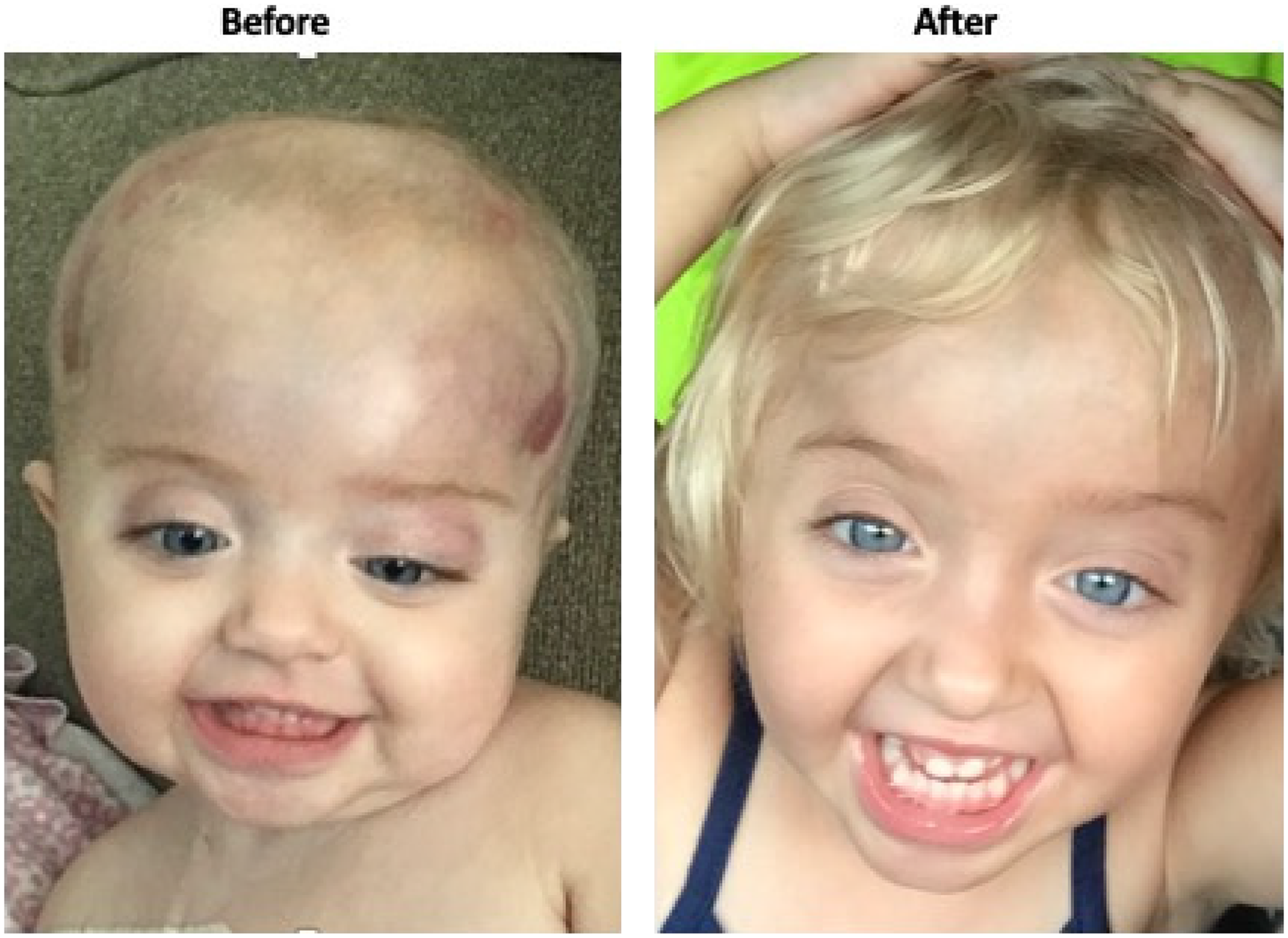
Before and after clinical images.
Discussion
The incidence of total frontal bone loss following surgery for craniosynostosis has not been adequately described in the literature. Though uncommon, this type of complication presents with potentially devastating clinical consequences for the patient while also posing a significant reconstructive challenge for the craniofacial surgeon. Salvage of native bone, when possible, may produce optimal results. In instances where salvage is not possible, little is written about significant calvarial loss in children and the subsequent options for reconstruction. The current reconstructive options most frequently discussed involve the use of implants, an option that is less than ideal in very young children where skull growth is likely to be significant. 12 Autologous options which have been described include the utilization of bone grafts from multiple sites (ie, rib, calvarium) or particulate bone grafting, however, these options are likely to prove insufficient for large-scale reconstruction. 7
To address this issue, we propose a novel treatment algorithm for children with near total frontal bone loss. The algorithm is based on patient age and acknowledges that in children over the age of 3 years, large-scale split calvarial bone grafting and alloplastic reconstruction may be reasonable options. In patients less than 3 in whom alloplastic reconstruction is contraindicated, a unique reconstructive problem arises. In our algorithm, we indicate that in instances where the surface area of the defect is greater than half of the surface area available for reconstruction, then the patient is a candidate for a modified visor flap with brain cage. Alternatively, if the surface area of the defect is less than half of the surface area available for reconstruction, a visor bone flap alone is recommended (Figure 6).
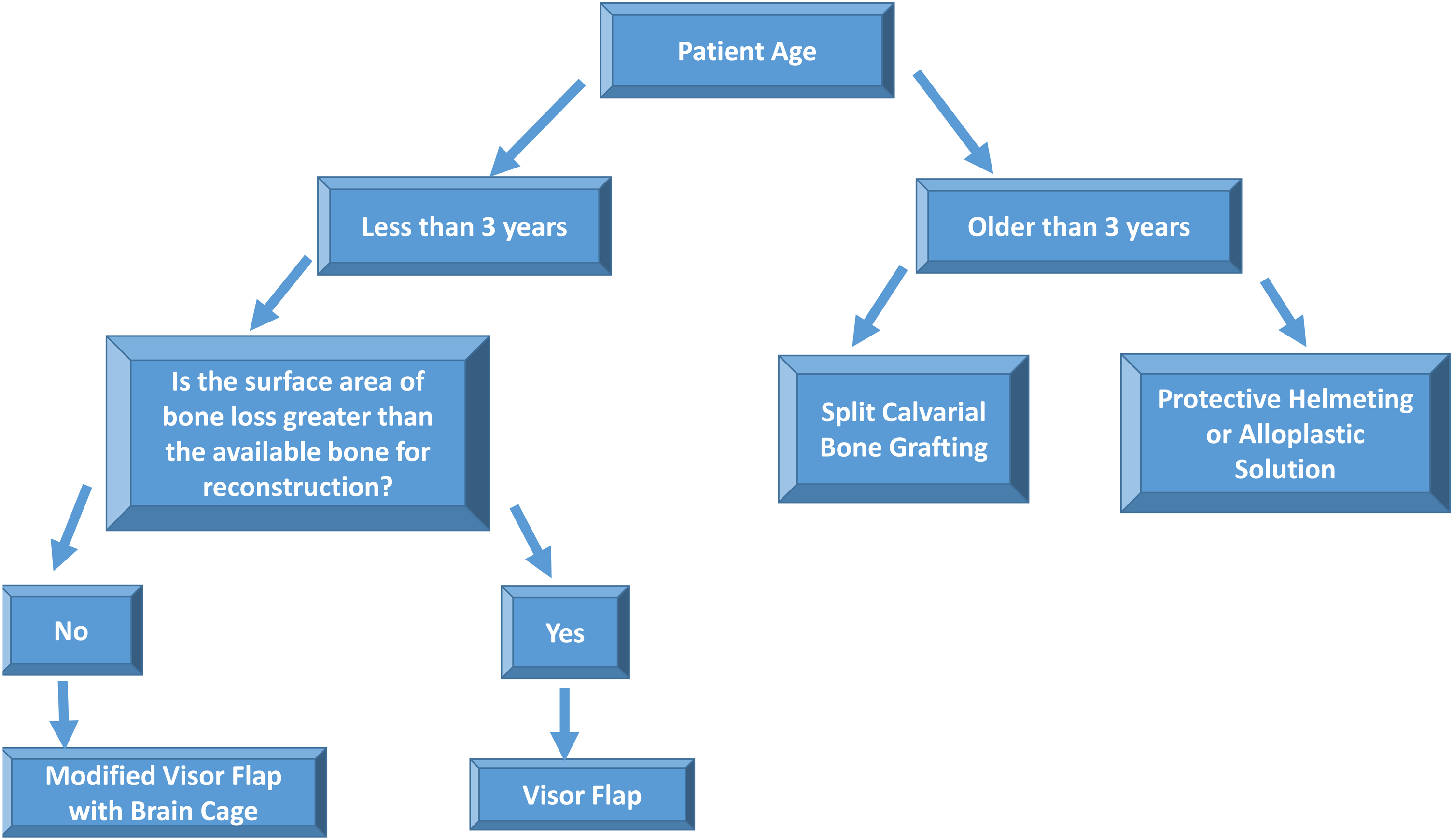
Treatment algorithm for near total frontal bone loss after cranial vault remodeling.
In the case described, we used VSP to create a Modified Visor Flap with Posterior Brain Cage. As cranioplasty is rarely an emergent procedure, use of VSP is ideal and can be carefully executed while preparing the patient for surgery by eradicating infection and optimizing soft tissues and comorbidities. VSP is a useful adjunct to complex craniofacial surgery, requiring input from a number of craniofacial practitioners such as pediatric neurosurgeons, plastic surgeons, and clinical software engineers. 13 Use of this technology involves obtaining contour data from computed tomography (CT) scans of the patient‘s head that are packaged as DICOM images and subsequently transformed into 3D renderings via a software platform that is virtually manipulated by the software engineers. Patient head shape data such as biparietal diameter, fronto-occipital diameter, and cranial length are tabulated and conformed to help with surgical planning, namely osteotomy placement and alignment of bone segments.14,15
This tool is typically used in conjunction with computer-assisted design (CAD) and manufacturing (CAM) to establish stereolithographic templates and precise cutting guides which dictate osteotomy placement and bone segmentation ahead of the procedure date, allowing the surgeon ample time to review and plan the subsequent surgery. To assist with long-term aesthetic outcomes, this tool typically uses CT data of age-matched controls, overlapping them with the patient‘s skull dysmorphology to guide operative manipulation.16‐18 Moreover, in the realm of pediatric craniofacial surgery, VSP serves an important pedagogical role for trainees and families who are often encouraged to join preoperative interdisciplinary VSP meetings.19,20
Here, VSP was used to establish a novel construct for a unique surgical problem. It precisely tabulated cranial bone surface area volumes to establish an optimal framework whereby remaining bone pieces were fashioned into their best new anatomic configurations maximizing the available surface area. While this could in theory be accomplished without VSP, this novel technology allows for far more precise tabulation of surface areas than would be reasonably performed manually. Further, this technology has been shown to potentially decreases operative time as much of the surgical decision making is completed in the preoperative period. Additionally, the combination of VSP with CAD/CAM technology to create high-fidelity cutting guides and positioning models has been indicated as a mechanism to improve aesthetic outcomes for craniofacial surgery. In this case of near total frontal bone loss in a young child, we believe our chances of success would have been diminished without this technological assistance.
There are several limitations to this technique including lack of long-term follow-up. Given that native bone is used, we presume proportional growth of the bone graft however long-term data is lacking. Similarly, more study is needed to determine rates of secondary synostosis in children undergoing this procedure. As in any autologous-based craniofacial reconstruction, this technique relies on the revascularization of large bony grafts. More data is needed to determine the long-term viability of this. Furthermore, the authors recognize that this technique may be equally applicable to other pathology involving large frontal bone loss including encephaloceles and pediatric craniofacial trauma. Further follow-up is needed to identify additional clinical applications.
Conclusions
Given the result of this case, we propose a novel treatment algorithm to address total frontal bone loss in pediatric patients. The key to our success in this instance was the use of VSP to establish a construct design and to calculate and optimize surface area coverage.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article
