Abstract
Background
Exoskeleton technology supports gait rehabilitation in individuals with spinal cord injury (SCI), promoting walking independence. However, current powered exoskeletons often face challenges in adaptability, particularly skin problems caused by inadequate fit to the lower limbs.
Objective
This study aimed to develop a powered exoskeleton for SCI featuring a unique mechanism to adjust the left-right joint angle, accommodating diverse lower limb alignments. Functional evaluation included fitting (device-to-body clearance, subjective pain, discomfort, and fit) and kinematic (hip and knee joint angles) parameters using markerless motion capture.
Methods
A comparative study was conducted to compare walking with and without a prototype.
Results
Ten healthy adults (seven males, three females; average height, 167 ± 10 cm; weight, 58 ± 9 kg) participated in the study. Clearance values were acceptable, pain and discomfort were minimal, and fit satisfaction was high. Gait analysis revealed significant positive correlations between normal walking and using the prototype for both joint angles (hip: r = 0.70, p < 0.001; knee: r = 0.84, p < 0.001), indicating effective gait assistance.
Conclusions
A prototype exoskeleton with adjustable mechanism was developed, fitting well and effectively assisting with walking. Further refinement and testing with SCI patients are necessary for its clinical application.
Introduction
A powered exoskeleton, a robot that generates a walking motion, has been used for gait rehabilitation in individuals with spinal cord injuries (SCI). SCI is a traumatic event with a global incidence ranging from 3.6 to 195.4 cases per million. 1 In 2023, approximately 18,000 new cases of SCI were reported in the United States, 2 while Japan reported approximately 4600 cases in 2018. 3 Occupational accidents were responsible for 703 (28%) of the 2515 cases documented in the Japanese National Spinal Cord Injury Database between 1997 and 2007. 4 These accidents are most common in construction and civil engineering, often resulting from falls. Recent trends show an increasing prevalence of SCI related to falls on level ground among the aging population and those in tertiary industry activities. 5 Reported return-to-work rates are 13% 6 and 25%, 7 highlighting a critical need to improve treatment outcomes. Additionally, 70.6% of SCI survivors remain unemployed, underscoring the urgent need for vocational rehabilitation services. 8
SCI often results in paralysis, sometimes complete, making walking difficult. Consequently, assistive devices such as orthoses and wheelchairs are required for mobility. Recent advancements in exoskeleton technology offer a promising solution for SCI patients. According to the U.S. Food and Drug Administration (FDA), a powered exoskeleton is a prescription device with an external motorized orthosis placed over paralyzed or weakened lower extremities for rehabilitation. 9 These devices feature actuators at the hip and knee joints, controlled by a computer to perform walking, standing, and sitting programs. They can potentially restore locomotive function, enabling SCI patients to reintegrate into their homes, communities, and workplaces.
The use of powered exoskeletons in rehabilitation began in the early 1970s, 10 with advancements in digital technology leading to various exoskeleton models. Promising results have been reported for three commercial exoskeletons—ReWalk, Indego, and Ekso—each of which has undergone rigorous FDA certification testing and has helped patients with paraplegia regain walking independence.11–13 Systematic14–16 and scoping 17 reviews further support their clinical effectiveness. Additionally, exoskeletons have the potential to increase daily living activities at home and in the community, although the limited availability for community use following rehabilitation remains a concern.18,19
Nevertheless, the use of exoskeletons poses several challenges, including device malfunctions, skin injuries, device misalignment, user errors, and falls. 20 Device mass and means of powering/batteries are also known issues. Among these challenges, skin injuries are notably prevalent and a significant concern at our affiliated rehabilitation hospital in Japan. Clinicians at the hospital have observed that the foreign-made exoskeleton is often too large to fit the Japanese body size. Additionally, because many Japanese people tend to have O-legs, gaps or interference between the exoskeleton frame and the lower limbs can occur. The checkpoints for lower limb orthoses 21 emphasize that significant deviations between the orthosis joint axis and human joint axes can lead to poor connection, causing pressure and misalignment of the cuff edges, thereby adversely affecting the limb. Narrow clearance can cause skin injury, while too wide a clearance can cause friction with clothing. Although the use of pads or towels between the exoskeleton and the human body is a common clinical practice, further advancements in device design are required to better fit the human body. Although the current commercial exoskeleton design has size-adjustment functions for parameters such as length and width, it does not have the capability for lateral adjustments. Thus, current exoskeletons are incapable of accommodating diverse lower limb shapes.
This study aimed to develop a powered exoskeleton with an adjustable mechanism capable of fitting various heights and shapes of lower limbs. The exoskeleton features a unique mechanism for adjusting the left-right joint angle, accommodating variations in leg alignment such as Valgus and Varus (X-leg and O-leg)—a feature currently absent in existing exoskeletons for SCI. The development process includes a functional evaluation of the prototype, including evaluating whether the device can appropriately fit the lower limb and execute a walking motion program. Fit-related parameters, such as the clearance between the device and body, as well as subjective ratings for pain, discomfort, and fit while wearing the device, were assessed. Additionally, the study analyzed the correlation between lower limb joint angles during gait, both with and without the device. Successful fitting of the device could mitigate skin injuries, while demonstration of the walking program’s functionality could suggest potential for improving mobility in SCI individuals comparable to conventional exoskeletons. This research will lay the groundwork for future clinical applications.
Methods
Research design
A comparative study design was used to compare gait with and without the prototype in a small group of healthy adult participants. The participants were recruited by staff through research networks in a specific research facility. The recruitment process included the use of flyers and direct invitations. Eligibility criteria included healthy adults aged 20–60 years, no history of neurological disorders, and normal gait or no severe gait impairments. Recruitment considered a wide range of heights, weights, and ages.
Device
Overview of prototype exoskeleton
A prototype exoskeleton was used as an experimental device. Figure 1 shows the external features of the prototype exoskeleton, and Table 1 highlights its key specifications. The primary unit consists of a structure resembling a hip-knee-ankle-foot orthosis equipped with a gait trigger sensor, a control unit, a battery, and actuators. Additionally, it includes forearm-supported clutches (Lofstrand crutches) with an operational interface and a tablet PC equipped with adjustment software. The exoskeleton assists individuals in walking, maintaining posture, and transitioning between sitting and standing using Lofstrand crutches. Appearance of the prototype-powered exoskeleton for spinal cord injury. (a) Gait trigger sensor. (b) User interface with a remote control built into Lofstrand crutches. (c) Actuators attached to the hip and knee joints. (d) Control unit and battery. (e) Separable frame. (f) Adjustment software installed on a Tablet PC. Key design specifications of the prototype.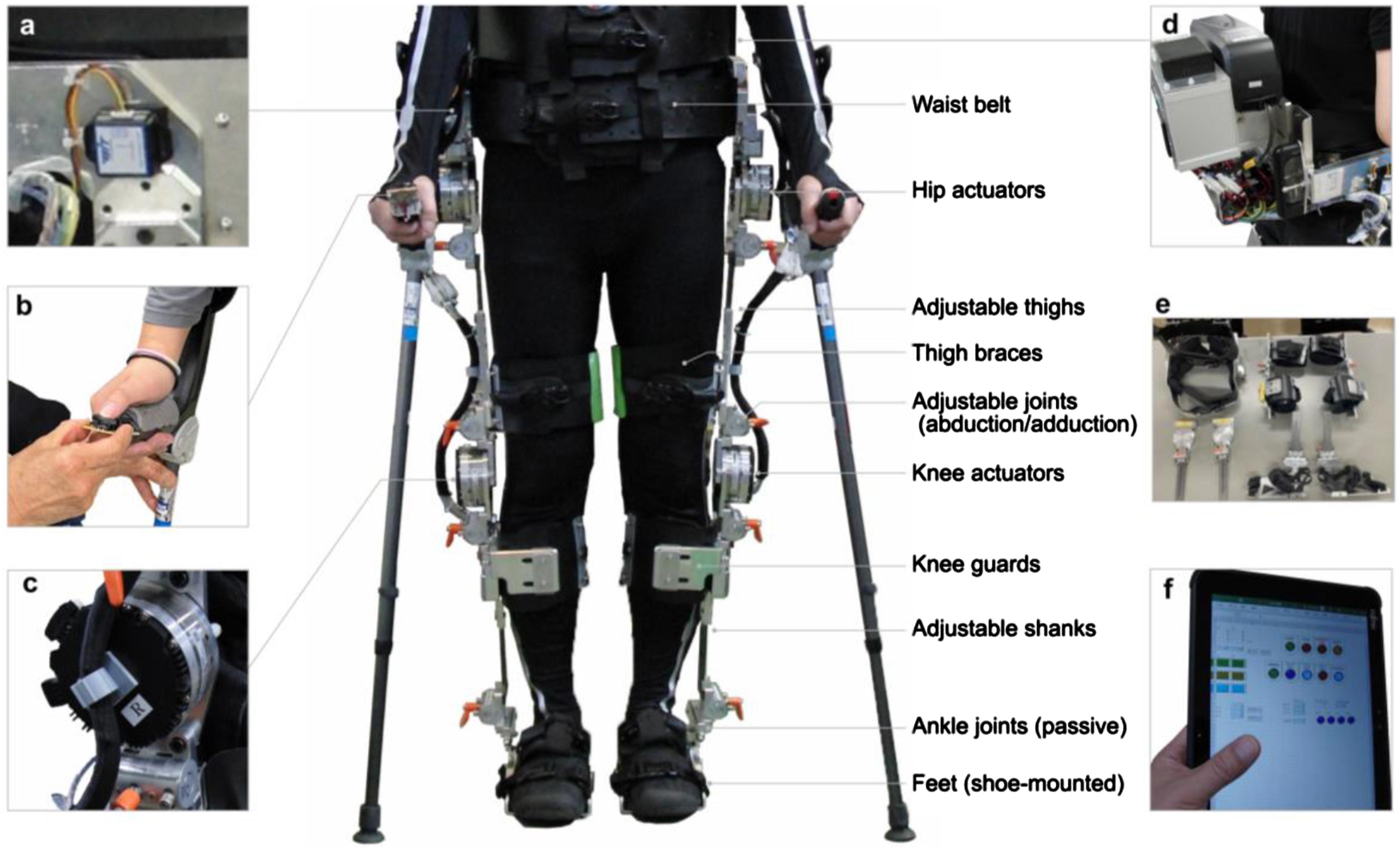
The design concept emphasizes safety and user-friendliness, aiming to restore mobility in individuals with SCI to levels comparable to those of healthy individuals. The design is inspired by the lower limb-powered orthosis developed by Miyamoto et al., 22 and it aligns with the FDA’s definition of a medical exoskeleton. 9
A structured schedule was followed to implement this design concept. In 2020, we conducted a comprehensive survey and performed a risk assessment of existing exoskeletons. 23 In 2021, we focused on engineering principles and user assistance to design an exoskeleton for individuals with SCI. We developed a design concept, established requirements for the design, and created an exoskeleton structure. In 2022, we integrated an actuator and a control system into the exoskeleton structure, resulting in a functional prototype. 24 In 2023, we tested the prototype exoskeleton.25,26 Throughout this period, we gathered input and feedback from medical doctors and physical therapists, which was incorporated into the design specifications. 24
The fundamental design requirements were set using the ReWalk exoskeleton (ReWalk Robotics, Inc., Yokneam, Israel) as a reference 27, focusing on safety and usability. Our initial criteria included height adjustments, accommodation of variations in leg alignment, ease of use, portability, overload protection, and user-friendly interface.
The target users of this product are individuals with SCI at levels T4–L5 in hospitals and rehabilitation centers, based on the target user of ReWalk. 27 These individuals experience complete paralysis of their lower extremities but have good upper limb functionality and balance.11,20,28 They should have a height of 145–180 cm (ranging from the 5th percentile for adult females to the 95th percentile for adult males) and a weight limit of 80 kg (the 95th percentile for adult males in Japan) according to Japanese anthropometric data. 29
This study specifically examines orthosis-based (overground) exoskeletons, rather than treadmill-based models. Fall prevention is a key priority; however, developing effective fall prevention technologies presents challenges, including predictive fall detection and control. A trunk-mounted harness can also reduce falls; however, practical limitations in clinical settings have led to its exclusion. Hence, this study assumed that the prevention of falls depends on traditional human support, such as contact assistance and monitoring by physical therapists.
Design elements
Structure
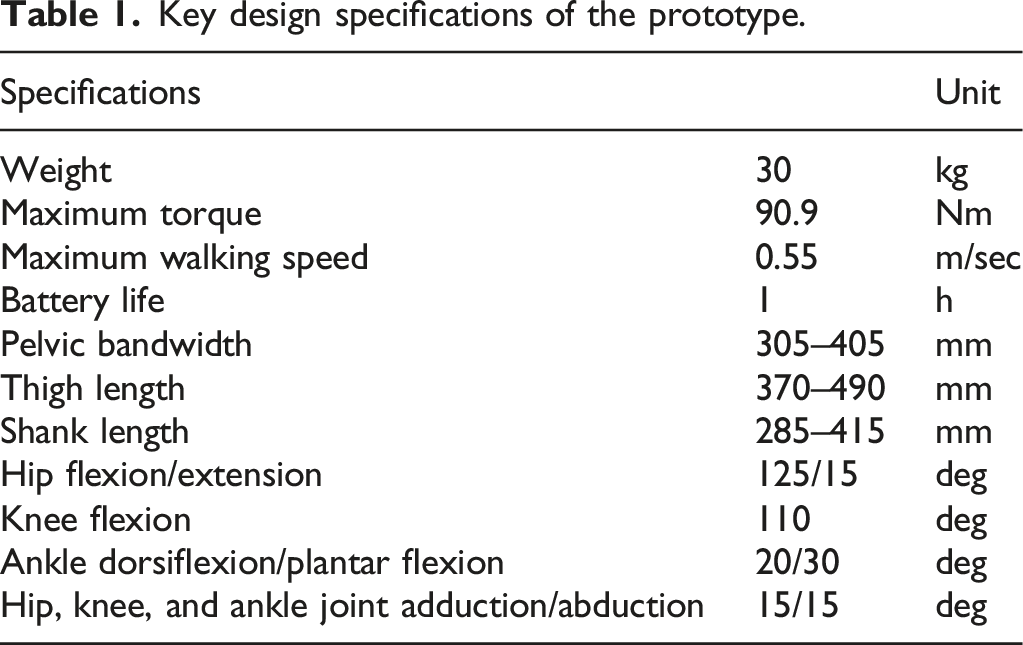
The structure is required to firmly support the body while fitting the length and shape of the lower limbs. Figure 2 illustrates the structural dimensions and functionalities of the prototype. The design incorporates an ergonomic structure with adjustable components, braces, and soft padding materials to minimize the risk of skin injuries and ensure proper alignment. The dimensions were customized to accommodate a wide variety of individuals, ranging from the 5th percentile for adult females to the 95th percentile for adult males, based on Japanese anthropometric data.
29
Each segment included a sliding mechanism that could be easily secured with pin-type screws, thus simplifying the adjustments without the need for additional tools. Adjustable dimensions of the prototype-powered exoskeleton.
The joint range of motion was set according to the guidelines of the Japanese Orthopaedic Association, the Japanese Society of Rehabilitation Medicine, and the Japanese Society for Surgery of the Foot. 30 This configuration ensured the necessary ranges of motion for standing and walking. The prototype features a unique mechanism that allows angle adjustments of up to 15° in the directions of adduction and abduction of the hip, knee, and ankle joints. This adjustment function accommodates variations in lower limb shapes, such as the X-leg or O-leg. The design was inspired by Kardofaki’s 31 scalable exoskeleton. The ankle joints include a passive mechanism inspired by the double Klenzak design to effectively limit dorsiflexion during walking. This mechanism is particularly beneficial for patients with paraplegia.
The prototype also included foldable and detachable knee guards to prevent knee flexion while standing. A gel-like material (EXGEL; Kaji Corp., Shimane, Japan) was used for the inner surfaces. The thigh braces were equipped with a soft, 10-mm-thick urethane padding. Additionally, a three-dimensional molded resin component, called the “hip shell,” was attached to the pelvic band to conform to the contours of the pelvis. This lowered the pressure on the sacral area, reducing its susceptibility to pressure ulcers.
The foot components were designed to be shoe-mounted, allowing users to wear them over their existing footwear. Attachment to the body was done using strap belts on the waist, thighs, shanks, and feet. These belts featured ratchet-type buckles for added convenience, unlike traditional velcro closures. Additionally, a detachable handle was strategically positioned on the rear side of the pelvic band to assist physical therapists in providing support. The structural frame was primarily composed of duralumin and weighed 12.8 kg. It could be dismantled into the pelvic, thigh, shank, and foot components for easy transport and storage.
Actuator
The actuator must be capable of generating the torque required for walking and sitting/standing with crutches, while also ensuring safety. During typical walking, the hip joint requires a maximum normalized torque of approximately 1.1 Nm/kg, 32 whereas rising with arm support requires roughly 0.72 Nm/kg at the knee joint. 33 When walking with crutches, approximately 47% of the body weight is supported. 34 Assuming a maximum patient weight of 80 kg (95th percentile adult males in Japan) and an exoskeleton weight of 30 kg, the required hip joint torque for walking with crutches is approximately 64 Nm, and the knee joint torque for standing movements with crutches is approximately 79 Nm. Therefore, the motor torque required for the exoskeleton exceeded 79 Nm.
To satisfy this requirement, an ultra-flat actuator (model number: WPMZ-50-100-SN-3958; NIDEC Corp., Kyoto, Japan) was selected for the hip and knee joints. These actuators had a reduction gear with a 1:101 ratio. They were equipped with a brushless DC motor (rated voltage: DC48V, rated capacity: 220 W, rated rotation speed: 29.7 rpm) capable of producing a rated torque of 47 Nm and a maximum torque of 90.9 Nm. Each actuator weighed 1.1 kg, with an outer diameter of 90 mm and a thickness of 50.5 mm. This made them the slimmest and lightest actuators in their category. Despite a high reduction ratio of 1:101, they maintained excellent backdrivability (approximately 11 Nm), allowing for a manual axis drive after disengagement of the servo.
The actuator was equipped with safety features, including an integrated encoder and a dedicated motor driver (model number: FWPB4338120-48; NIDEC Corp., Kyoto, Japan) that continuously monitors and regulates the position, speed, and torque. These protective mechanisms are designed to address potential anomalies such as overcurrent, overload, and excessive speed. They also help to automatically halt the actuator whenever an overload is detected. In addition, the actuator includes software limiters and mechanical stoppers as hierarchical safety mechanisms to prevent unintended movements.
If the emergency stoppage, including automatic stoppage, is activated while walking, the hip and knee joints are locked in their respective positions, and the servos remain engaged. The servos could be deactivated by releasing the emergency stop button, allowing the exoskeleton to return to a standing position under its weight while receiving the frictional force due to backdrivability. However, the assistance of therapists is essential in such situations.
Gait trigger sensor
Considering the possibility of neuroplasticity induced by voluntary movement effort, which is necessary for motor function recovery in patients, the prototype was designed to detect the user’s movements and respond by moving accordingly. The motion sensor (BWT61 Gyroscope sensor; WitMotion Co., Ltd., Shenzhen, China) was used as a gait trigger sensor and attached to the side of the pelvic band. The walking mode is activated when the user tilts their trunk forward and surpasses a predefined angle, initiating the walking sequence. The trigger angle was preset to 10° from the vertical and could be freely adjusted within the range of 0° to 30°.
Control unit
The control system utilizes a programmable logic controller (PLC) (KV-8000; KEYENCE Corp., Osaka, Japan) to manage functions, such as standing, walking, or sitting modes, gait triggers, walking cessation, voice-guided instructions, and abnormalities with handling. The PLC, motor drivers, and a DC48 V lithium-ion battery (eBike Battery; Guangdong Greenway Technology Co., Ltd., Dongguan, China) were enclosed in an aluminum housing on the back of the pelvic band, weighing approximately 16 kg.
Figure 3 outlines the operational program flowchart: (1) The user attaches the exoskeleton while in a servo-stopped state at the origin. (2) Standing mode is selected and confirmed to initiate standing movement. (3) After a successful standing movement, each axis actuator maintains its standing position in the servo-stopped state. (4) To start walking, the user should select walking mode and then wait for the walking-start trigger. Walking is automatically initiated when the motion sensors detect a forward inclination beyond a preset angle. Pressing the walking-stop switch causes the swinging leg to return to a standing position via the shortest route in the servo-stopped state. If the sitting mode is selected, the system returns to the servo-stopped state upon completion of the sitting movement from the standing position. Flowchart of the operating program to control the prototype-powered exoskeleton.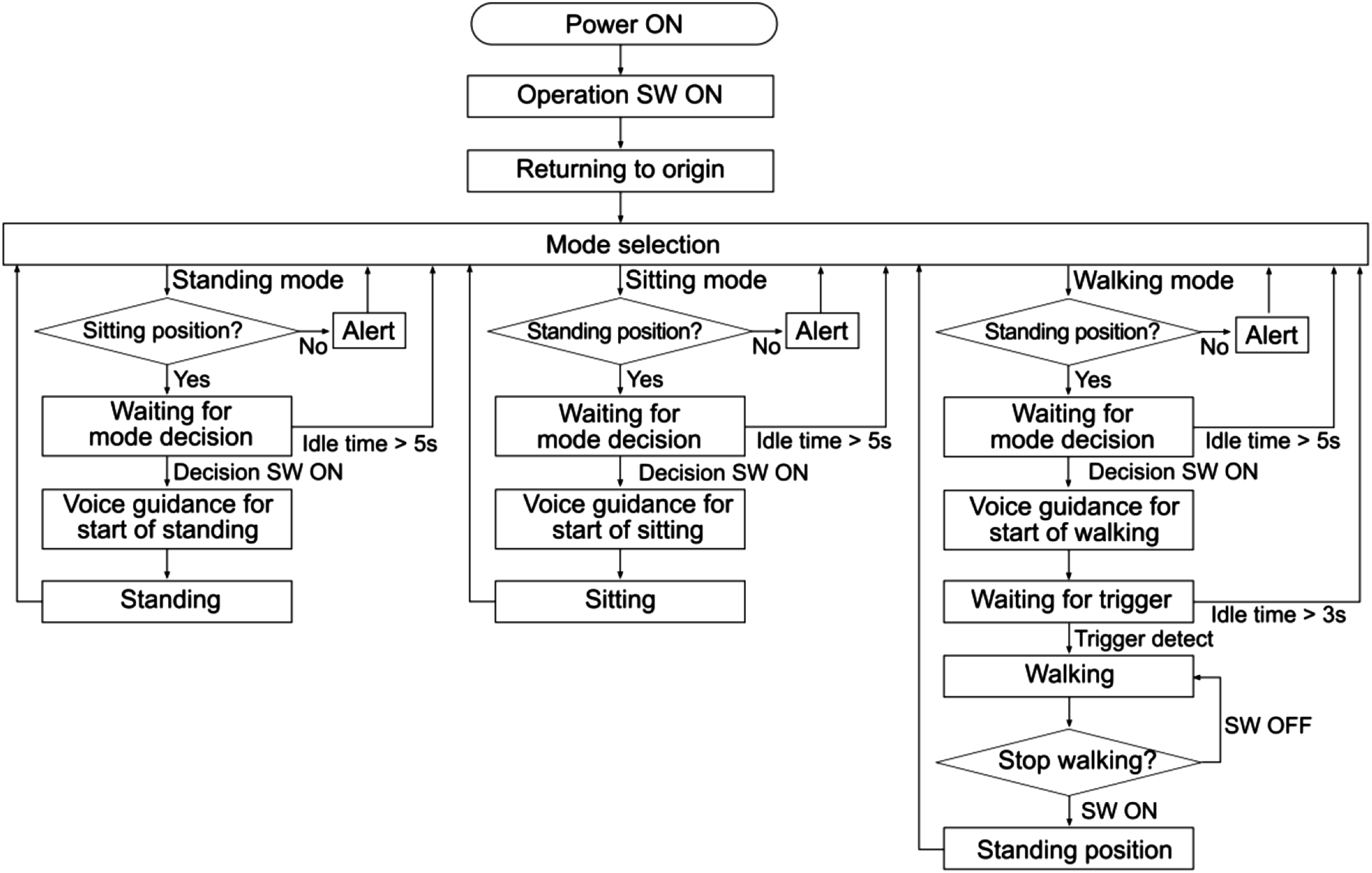
Gait patterns were created using dedicated PLC software (KV COM + for Excel, Ver. 1.4; KEYENCE Corp., Osaka, Japan). The basic gait pattern was set based on the eight gait phases set by the Rancho Los Amigos Hospital,
35
with the knee and hip joint angles serving as references for typical gait in a healthy individual. Figure 4 shows the variations in hip and knee joint angles during the gait cycle. The vertical axis represents joint angles, and adjustments to the flexion or extension angles can be made by modifying the values. Meanwhile, the horizontal axis represents the timeline, and the timing of flexion or extension is adjustable through modifications to velocity, acceleration, and deceleration. Gait patterns and parameters. The horizontal axis labels in the figure indicate the following phases: IC, initial contact; LR, loading response; MSt, mid-stance; TSt, terminal stance; PSw, pre-swing; ISw, initial swing; MSw, mid-swing; TSw, terminal swing.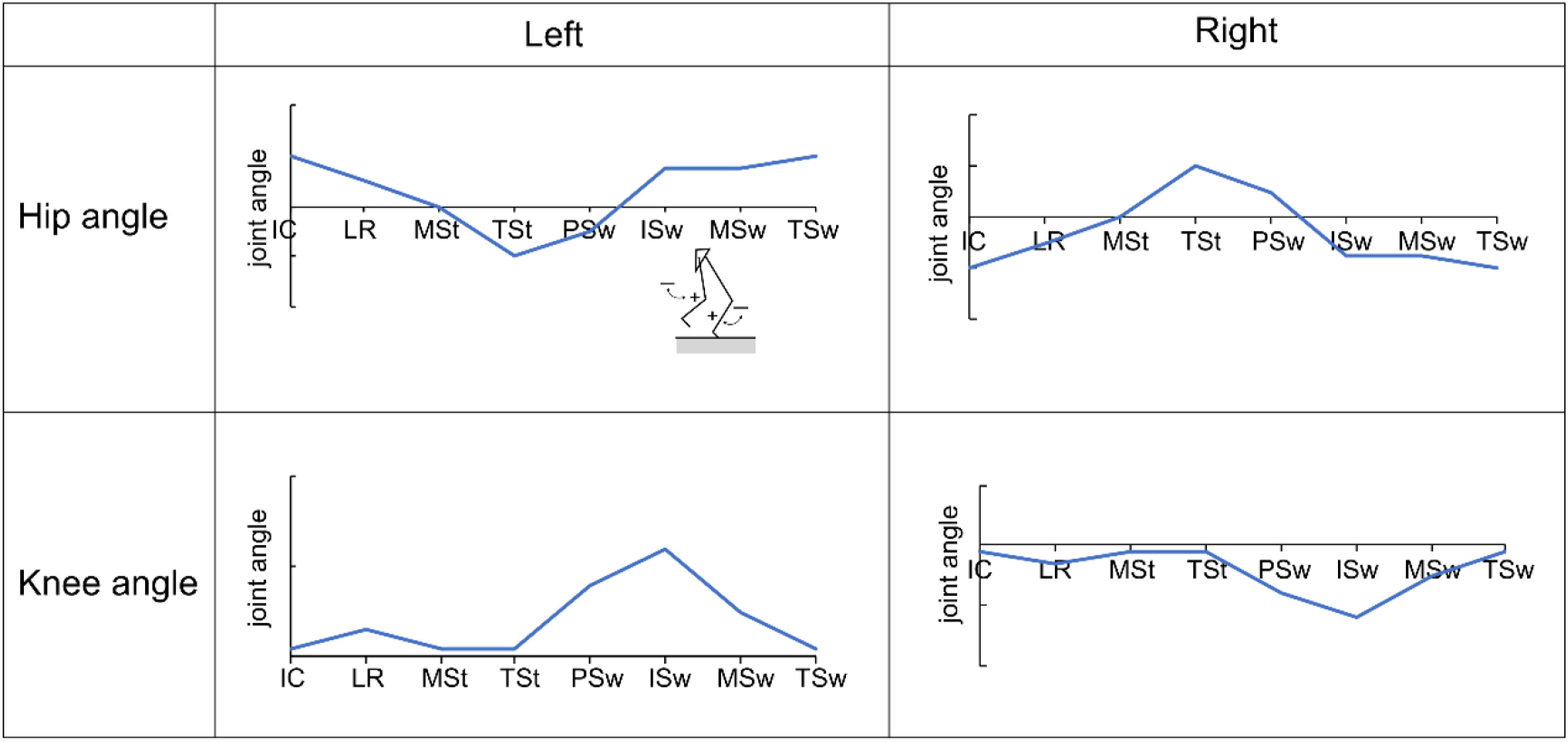
The PLC memory stores a predefined basic gait pattern, allowing the autonomous operation of the exoskeleton. The predefined gait parameters were assumed to be adjusted based on the patient’s condition. User-friendly graphical user interface software was developed to facilitate this process. The program can be installed on a tablet or laptop with a Windows OS. This feature allows physical therapists to adjust the gait parameters to meet the specific clinical needs of their patients.
User interface
The user interface was designed considering the user’s ability to operate independently. This design differs from the ReWalk reference model because it allows patients to control the exoskeleton independently, eliminating the need for specialized training. This was achieved by converting a commercially available clutch grip into a versatile switch that users can operate directly. Intentional designs were incorporated to minimize the risk of user error. As highlighted in a previous study, 36 the mode-selection switch was located on the right grip, whereas the stop switch was located on the left.
To enhance safety, an algorithm-based timed-timer function was incorporated to automatically reset the system to its initial state after a specified elapsed time period from activation of the mode-selection switch. The idle time was set to a minimum of 5 s, as shown in Figure 3. This feature guarantees that patients can revert to their original state even if the switch is accidentally or incorrectly activated.
The device featured MP3 playback alarms and Bluetooth technology connected to the PLC, allowing for synchronized voice-guided instructions that correspond to the operational mode. Emergency stop can be easily accessed through switches on both the clutch grip (by long-pressing the stop button) and control unit. Exiting the emergency stop mode deactivates the servo, allowing manual adjustments of the joint angle through the therapist’s assistance without significant resistance from the motor.
Outcome measured
Fitting-related parameters
Clearances between the thigh and lower leg and the thigh and shank segments of the prototype were measured in millimeters using calipers. This measurement evaluated compliance with the checkout criteria for lower limb orthotics, where clearance was set at approximately 5–7 mm for nonarticulated regions. 21 A narrow clearance means an increased risk of skin injuries, whereas an excessively wide clearance may lead to increased friction with clothing.
Subjective sensations of pain, discomfort, and fit were evaluated through a questionnaire administered at three specific sites: the pelvic area, thighs, and shanks. Pain and discomfort levels were evaluated using an 11-point numerical rating scale (NRS), where 0 indicated no pain or discomfort and 10 indicated very intense pain or discomfort. Fit was also assessed using the NRS, with 0 indicating not fit and 10 indicating a very good fit.
The time taken to put on and remove the device was measured using a stopwatch. Donning time refers to the time required to attach the strap belts to the pelvic area, thighs, shanks, and feet in a seated position, while doffing time refers to the time to remove them.
Kinematic parameters
Gait was analyzed using a markerless motion capture system (e-skin MEVA; Xenoma Inc. Tokyo, Japan) (Figure 5). A markerless system was chosen since conventional optical motion capture systems are markers are hidden by the exoskeleton frame, thus making them unsuited for this study. This system tracked hip and knee joint angles during gait, with and without the prototype. Mechanical joint goniometers may also face interference issues from the exoskeleton frame, which could compromise the measurement accuracy. Consequently, the sensors are either integrated into the exoskeleton rotor or attached externally to the exoskeleton.37,38 Markerless motion capture system (e-skin MEVA).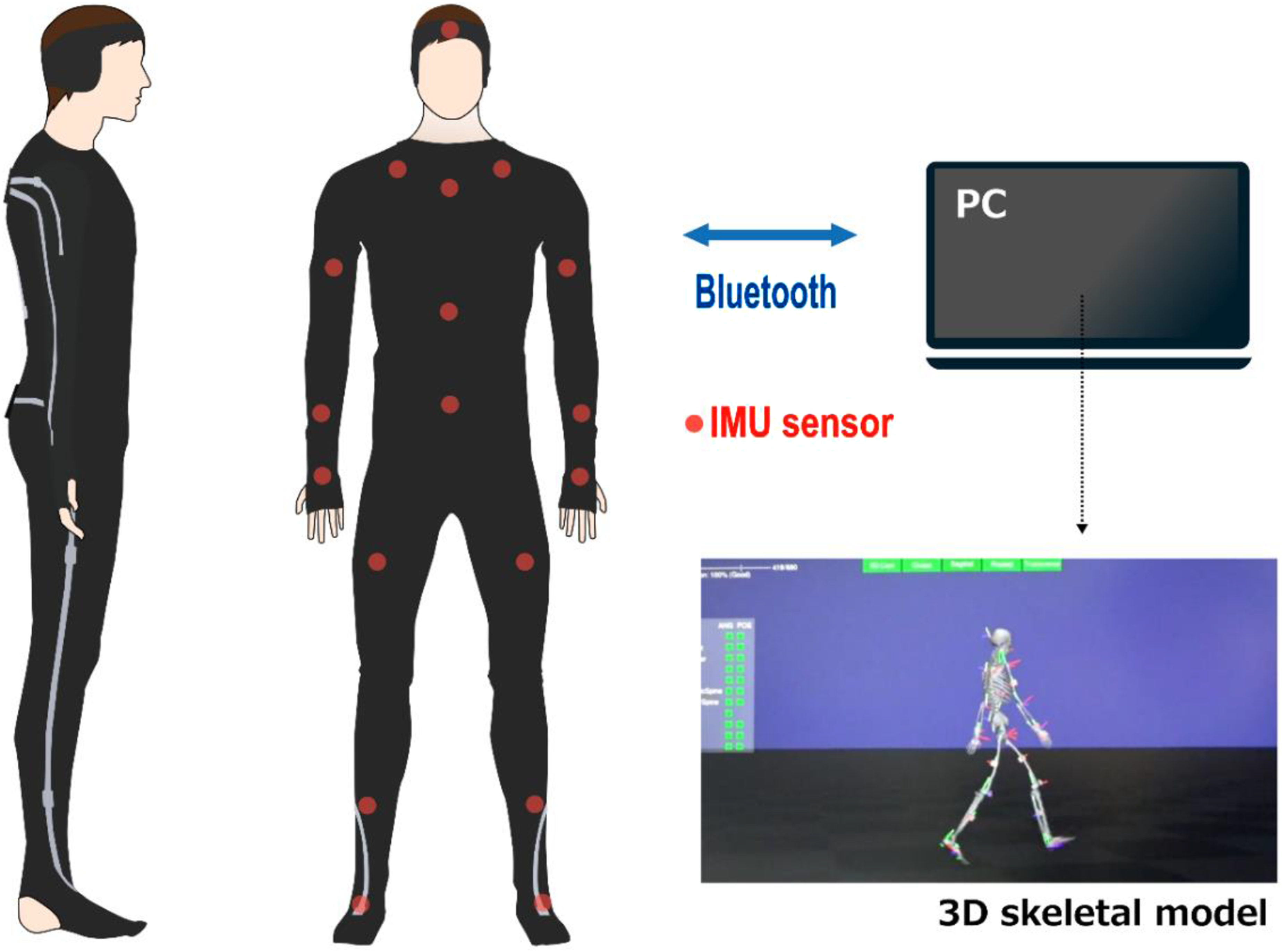
In contrast, the e-skin system features a wearable design, 39 ensuring minimal interference with the exoskeleton frame. The system uses an inertial measurement unit (IMU) sensor that comprises 3-axis acceleration and gyro sensors. The IMU sensors were positioned at 18 locations: one on the headband, ten on the upper-body shirt, and seven on the lower-body pants. The sensors were slim and lightweight. The difference in joint angles between this method and the conventional optical method is only 2°, with a correlation coefficient of 0.98, for both the hip and knee joint angles in the sagittal plane. 40 Data were recorded at a sampling frequency of 100 Hz and transferred to a PC via Bluetooth.
Data from the second trial were used for gait analysis. To account for the variability in walking time among the individual participants, the joint angle data within a single gait cycle were normalized, including both the swing and stance phases, and defined as 100%. We then used the maximum (peak flexion angle) and minimum (peak extension angle) hip and knee joint angles to represent and compare the differences attributed to the exoskeleton usage.
The gait cycle was divided into eight phases, each with a specific percentage of the cycle according to the Rancho Los Amigos Hospital method: initial contact (IC) at 0%, loading response (LR) at 0%–12%, mid-stance (MSt) at 12%–31%, terminal stance (TSt) at 31%–50%, pre-swing (PSw) at 50%–62%, initial swing (ISw) at 62%–75%, mid-swing (MSw) at 75%–87%, and terminal swing (TSw) at 87%–100%. 35 At each of these phases, stable hip and knee joint angles were extracted. The correlations between normal gait and gait with the prototype were then analyzed for these datasets. The correlation coefficient, r, ranges from −1 to 1. As a general guide for interpreting the strength of the correlation, ∣r∣ = 0 to 0.2 indicates little to no correlation, 0.2 to 0.4 indicates a weak correlation, 0.4 to 0.7 indicates a moderate correlation, and 0.7 or higher indicates a strong correlation.
Gait velocity was calculated using the walking distance and time. The stride length was calculated using the walking distance and number of steps taken during walking with and without the prototype.
Procedure
As part of the experimental preparation, the participants were given detailed instructions on how to operate the prototype. A Martin anthropometer (Tsutsumi Work Inc., Chiba, Japan) was used to measure their thigh lengths, lower leg lengths, and waist widths. The length and width of the exoskeleton frame were adjusted based on these measurements. Participants donned the prototype while seated, performing a standing motion to evaluate and adjust the tightness of the straps or belts as necessary. During this process, the joint angles of the left and right lower limbs were adjusted, and clearance measurements were taken. After completing this stage, participants returned to their seats, and the prototype was removed.
The participants wore calibrated markerless motion capture suits, and each completed two 6-meter walks at their usual pace. Joint angles were recorded using motion capture technology, walking time was measured with a stopwatch, and frontal and side views were captured using video cameras (Model HDR-CX680; SONY Corp., Tokyo, Japan).
The participants then donned the prototype again while seated. The time taken to don the prototype while seated was recorded using a stopwatch. Prior to the experiment, the participants were made to familiarize themselves with the prototype using a custom-made walker equipped with casters. The participants were allowed to choose between simultaneous bilateral or alternating thrusting motions with crutches. They were instructed not to exert force on their lower limbs while walking with the prototype. A caregiver stood behind each participant to ensure safety and prevent falls. Furthermore, air mats were placed on both sides of the walking path.
The participants completed a 6-meter walk with the prototype twice. Joint angles were recorded using motion capture technology, walking time was measured with a stopwatch, and frontal and side views were captured with video cameras. After the activities, the prototype was removed and the time taken for doffing was measured using a stopwatch. Finally, the participants completed the NRS questionnaires.
Statistical analysis
The data are presented as mean ± standard deviation. Participant characteristics [height, weight, and body mass index (BMI)] and fitting-related outcomes (clearance, subjective pain, discomfort, and fit and the time required for donning and doffing) were summarized using basic descriptive statistics (mean ± standard deviation). For the kinematic outcomes, Pearson’s correlation coefficients were calculated to assess the hip and knee joints during a single gait cycle with and without the prototype. In addition, paired t-tests were used to compare gait velocity, stride length, and peak flexion/extension angles of the hip and knee joints with and without the prototype. All statistical analyses were conducted using IBM SPSS Statistics version 28 (IBM Corp., Armonk, N.Y., USA). Statistical significance was set at p < 0.05.
Results
Participants
Ten healthy adults (seven males and three females), aged between 20 and 60 years, participated in this study and provided written informed consent. Their characteristics were: mean height 167 ± 10 cm (range: 152–179 cm), mean weight 58 ± 9 kg (range: 41–70 kg), and mean body mass index 21 ± 2 (range: 17–25).
Fitting-related outcomes
The mean clearance between the device and the human body was 10.2 ± 4.6 mm for the thigh and 10.9 ± 5.2 mm for the shank.
The mean pain and discomfort ratings were recorded as follows, respectively: pelvis area = 0.0 ± 0.0, 0.2 ± 0.7; thigh area = 0.1 ± 0.3, 0.3 ± 0.7; and lower leg area = 0.7 ± 1.1, 0.4 ± 0.7. No pain or discomfort ratings exceeded 3 during the sessions. The subjective ratings for the feeling of fit were as follows: around the pelvis = 6.2 ± 3.0, in the thigh region = 7.0 ± 3.0, and in the lower leg region = 7.1 ± 3.0.
The mean time required for donning the device was 3.8 ± 0.6 min, while the mean doffing time was 0.6 ± 0.2 min.
Kinematic analysis outcomes
The results of the kinematic parameters of the hip and knee joints throughout the gait cycle are shown in Figure 6. Significant positive correlations were observed between normal walking and gait with prototype in both the hip joint angle (r = 0.70, p < 0.001) and the knee joint angle (r = 0.84, p < 0.001). The results of the peak hip and knee angles with and without prototype are shown in Table 2. The mean peak flexion angles of the hip joint were 30 ± 3.5° for normal walking and 30 ± 6.1° when using the prototype. The mean peak extension angles of the hip joint were −13 ± 3.7° for normal walking and −6 ± 5.1° when using the prototype. Significant differences in these parameters were observed between the two scenarios (p = 0.002). The peak flexion angles of the knee joint were 74 ± 4.5° with normal walking and 44 ± 5.2° when using the prototype, with significant differences observed between the two scenarios (p < 0.001). Changes in hip and knee joint angles during one gait cycle with and without the prototype. The vertical axis in the figure represents joint angles, with (+) indicating flexion and (−) indicating extension. The horizontal axis is normalized to represent one gait cycle, including the swing and stance phases, at 100%. The solid line represents the subject’s average, while the dashed line represents the standard deviation. Peak hip and knee angles with and without the prototype.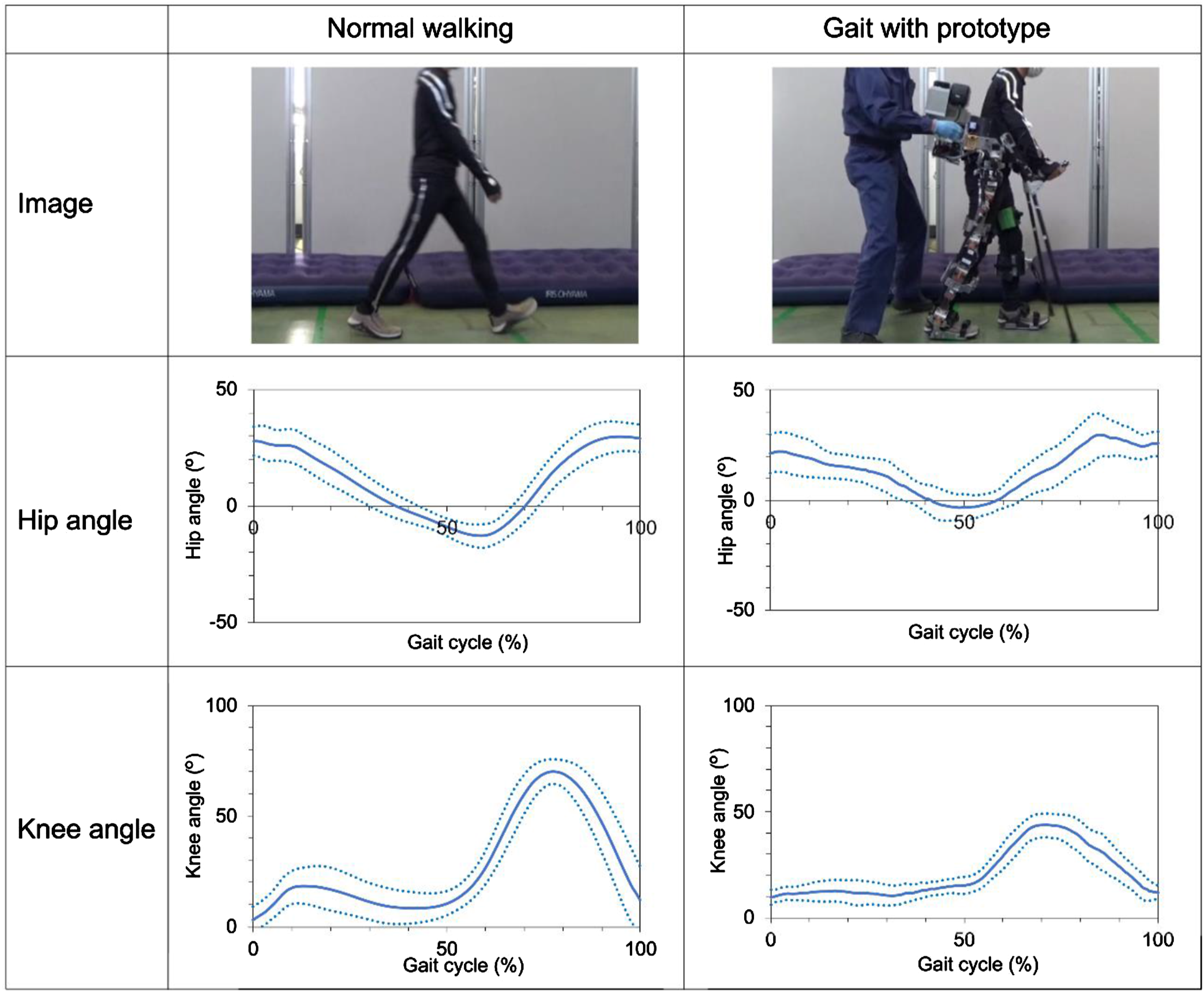

Participants achieved a mean gait velocity of 1.0 ± 0.1 m/s during normal walking, which decreased to 0.1 ± 0.1 m/s when using the prototype. Similarly, stride lengths were 1.2 ± 0.1 m during normal walking but decreased to 0.8 ± 0.3 m when using the prototype. Statistical analysis revealed significant differences in gait velocity (p < 0.001) and stride length (p = 0.0025) with and without the prototype.
Discussion
Fitting the exoskeleton to the human
This paper presented an adjustable exoskeleton. The experiment aimed to verify whether the device could fit the lower limb properly and execute a walking motion program effectively. The mean clearance between the device and the human body was 10 mm. Compared with the criteria for lower limb orthotics, the clearance values of the prototype are slightly larger but still within a reasonable range. Subjective fit ratings consistently indicated high levels of comfort in all areas. The ergonomic approach of combining adjustable structures and soft materials within a brace effectively would minimize risk of skin injuries, as evidenced by the low pain and discomfort ratings. In addition, the proper fit of the exoskeleton frame to the human lower limbs eliminated the need for traditional tasks such as attaching and removing pads. This suggests that physical therapists would benefit from reduced preparation time in clinical settings as well.
Potential for improving walking ability
The prototype successfully executed the intended walking motion program. The kinematic results indicate the correlation coefficients between the conditions of with and without the prototype were 0.70 for the hip and 0.84 for the knee. Since correlation coefficients of 0.7 or higher are generally considered to represent a strong correlation, both our results can be considered to represent strong correlations. Thus, changes in the hip and knee joint angles during the gait cycle have a similar trend with and without the prototype, proving that the prototype provides gait assistance. This performance suggests that the prototype has properties similar to conventional exoskeletons and can potentially improve the walking ability of individuals with SCI.
However, significant differences were observed in the peak values of the knee joint flexion angle and hip joint extension angle. During the stance phase of walking with the prototype, the knee was inadequately flexed, particularly during the weight response phase. In addition, the maximum angle of knee flexion during the swing phase was smaller than that during normal walking. Furthermore, the maximum hip extension angle with the prototype was smaller than that during normal walking. These deviations may result from the slow gait speed and forward-leaning posture associated with the use of crutches. Observations suggest that in the prototype, the slower gait speed compared with normal walking causes the foot to make ground contact before the knee fully extends (reaching the target value in the walking program). Additionally, the use of crutches may have induced a forward-leaning posture, making it more difficult to swing the leg forward. Thus, the smaller knee joint angle observed in the prototype may have contributed to its shorter stride length. Therefore, adjusting the settings of the prototype’s walking program will be necessary in the future.
The mean gait velocity achieved with the prototype was only 10% of that in normal walking. This was because the participants were using the exoskeleton for the first time, and some expressed apprehension about going faster. Hence, the speed of the exoskeleton was intentionally set to a slow pace to improve safety. As the prototype’s specifications allow it to reach speeds of up to 0.5 m/s, equivalent to that of a conventional exoskeleton, it will be necessary to gradually adjust the speed according to the user’s condition and proficiency level.
Study strengths and limitations
A major strength of this study is that we developed a novel exoskeleton with adjustment mechanism and quantitatively measured subtle body movements within the exoskeleton frame using a markerless motion capture system, which was previously considered difficult. Our results provide strong evidence that the prototype can successfully perform walking program when worn by healthy participants. Additionally, the dimension and left-right joint angle adjustment mechanism allowed the exoskeleton to fit the human lower limbs. This suggests the possibility of effectively addressing common problems associated with exoskeleton use, such as skin injuries.
However, this study has some limitations. We used a small sample size, gender selection bias, and relatively short experimental duration, which could have introduced some bias into our findings. This could be addressed by expanding the study to include more participants and longer-term investigations. Another limitation included conducting only two walking trials and using the data from a single gait cycle in one of those trials as representative values. In addition, there were technical challenges, particularly with the weight of the prototype, which exceeds that of conventional devices. Using lightweight materials such as carbon fibers, optimizing battery capacity adjustments, and developing a specially designed control system instead of a general-purpose PLC could be explored to achieve the desired weight target. For feasibility, the target weight is under 20 kg. Adjusting gait parameters based on normal gait data and reevaluating the basic gait pattern are also necessary. Specifically, assuming that adjustments will be made to suit the user, it would be advisable to preset the peak knee flexion angle to approximately 70° and the peak hip extension angle to approximately 10°. Given these limitations, it is imperative to continue improving and adapting the prototype.
The prototype must be tested on individuals with SCI. Additional usability tests, such as the System Usability Scale (SUS) 41 and the Quebec User Evaluation of Satisfaction with Assistive Technology (QUEST 2.0) 42 , should be considered alongside traditional outcome measures such as ambulation, balance, and physiological improvements. 28 The SUS is a simple scale consisting of ten questions used to evaluate the usability of a system or interface, with subjective ratings on a 5-point scale. The QUEST 2.0 is a tool designed to assess user satisfaction with assistive technologies. It evaluates various aspects such as the effectiveness, ease of use, and overall satisfaction of users with different assistive technologies. Furthermore, including a mental workload assessment, such as the National Aeronautics and Space Administration Task Load Index (NASA-TLX) 43 , would provide a more comprehensive understanding of user experience. Moreover, since SCI patients often have lost sensation in their lower limbs, subjective evaluations of pain and discomfort used in this study may be challenging. Therefore, alternative objective methods, such as detecting contact pressure between the device and the lower limbs, should be considered. A comprehensive evaluation of SCI patients in clinical settings is essential for a better understanding of the exoskeleton and its potential applications.
Conclusion
A prototype exoskeleton equipped with an adjustable mechanism to fit human lower limbs was designed for individuals with SCI. The prototype features a unique mechanism for adjusting the joint angle in the left-right direction, enabling it to accommodate diverse lower limb alignments. Experimental evaluations with healthy participants indicated that the clearance (averaging 10 mm) was within a reasonable range, with minimal pain or discomfort at contact points (averaging below 1 on an 11-point scale) and high satisfaction regarding the fit (averaging above 6 on the scale). These results suggest that the adjustment mechanism may contribute to reducing skin injuries caused by inadequate lower limb fitting. Additionally, the markerless motion capture system enabled the quantitative measurement of lower limb movements within the exoskeletal frame. The results revealed strong positive correlations between normal walking and using the prototype for both the hip and knee joint angles (hip: r = 0.70, p < 0.001; knee: r = 0.84, p < 0.001). This indicates that the prototype provides effective gait assistance, potentially improving mobility for individuals with SCI. However, further improvement of the device, adjustment of gait parameters, and testing of the prototype in individuals with both complete and incomplete SCI are essential for its future clinical applications.
Statements and declarations
Footnotes
Acknowledgments
The authors thank Dr. Kazunari Furusawa and Mr. Yoshinori Yamada for their valuable advice and the medical staff of Kibi Kogen Rehabilitation Center for their cooperation. They also thank Arcelis Inc. for their cooperation in manufacturing the structural frame for prototype development, and Altech Corp. and Digital Spice Corp. for their cooperation in manufacturing the control system.
Author contributions
Conception: Hideki Oyama and Hiroyasu Ikeda. Performance of work: Hideki Oyama and Hiroyasu Ikeda. Interpretation or analysis of data: Hideki Oyama. Preparation of the manuscript: Hideki Oyama. Revision for important intellectual content: Hideki Oyama and Hiroyasu Ikeda. Supervision: Hiroyasu Ikeda.
Conflicting interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical considerations
The study was conducted in accordance with the Declaration of Helsinki and approved by the Ethics Committee of the Japan Organization of Occupational Health and Safety (approval number: 2022-17). All participants provided their written informed consent to participate in this study.
