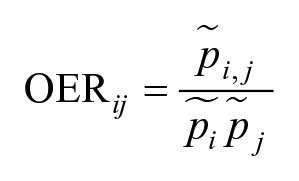Abstract
Comorbidity network analysis (CNA) is a technique in which mathematical graphs encode correlations (edges) among diseases (nodes) inferred from the disease co-occurrence data of a patient group. The present study applied this network-based approach to identifying comorbidity patterns in older patients undergoing hip fracture surgery. This was a retrospective observational cohort study using electronic health records (EHR). EHR data were extracted from the one University Health System in the southeast United States. The cohort included patients aged 65 and above who had a first-time low-energy traumatic hip fracture treated surgically between October 1, 2015 and December 31, 2018 (n = 1,171). Comorbidity includes 17 diagnoses classified by the Charlson Comorbidity Index. The CNA investigated the comorbid associations among 17 diagnoses. The association strength was quantified using the observed-to-expected ratio (OER). Several network centrality measures were used to examine the importance of nodes, namely degree, strength, closeness, and betweenness centrality. A cluster detection algorithm was employed to determine specific clusters of comorbidities. Twelve diseases were significantly interconnected in the network (OER > 1, p-value < .05). The most robust associations were between metastatic carcinoma and mild liver disease, myocardial infarction and congestive heart failure, and hemi/paraplegia and cerebrovascular disease (OER > 2.5). Cerebrovascular disease, congestive heart failure, and myocardial infarction were identified as the central diseases that co-occurred with numerous other diseases. Two distinct clusters were noted, and the largest cluster comprised 10 diseases, primarily encompassing cardiometabolic and cognitive disorders. The results highlight specific patient comorbidities that could be used to guide clinical assessment, management, and targeted interventions that improve hip fracture outcomes in this patient group.
Introduction
Hip fracture is an ongoing health issue among older adults. More than 340,000 hospitalizations for hip fractures occur annually among those ages 65 years or older in the United States (Wu et al., 2021). With the population aging, the cases are projected to double from 2.6 million by 2025, further increasing to 4.5 million by 2050, imposing even greater societal burdens (Penrod et al., 2008).
Hip fracture shares risk factors with several chronic conditions, such as chronic pulmonary disease, diabetes mellitus, dementia, and cancer, which explains why patients with hip fractures exhibit a high incidence of comorbidity (Holroyd et al., 2008; Jørgensen & Schwarz, 2008; Kanis et al., 2001; Sennerby et al., 2009). Especially for older patients undergoing hip fracture surgery, comorbidity has a critical role in the course of treatment because this is associated with postoperative complications, higher cost of hospitalization, longer length of hospital stay, decreased quality of life, and lower likelihood of survival (Hershkovitz et al., 2010; Öztürk et al., 2019; Nikkel et al., 2012; Peeters et al., 2016; Sheehan et al., 2018). Thus, examining comorbidity is of vital clinical importance.
Beyond evaluating the presence or severity of comorbidity with simple descriptive statistics or a composite score, identifying the specific comorbidity patterns—that is, understanding the integrated associations among different health conditions—has attracted many researchers’ attention (Fotouhi et al., 2018). Such an understanding could help elucidate high-impact comorbidities or patterns with meaningful clustering of disorders, which could guide specific screening or intervention procedures among inpatients. Furthermore, the comorbidity patterns may reveal clinically relevant differences in response to treatment or prognosis (Brunson et al., 2020).
Using network analysis to assess the associations among diseases is an emerging research strategy. In brief, network analysis is a graph theory-based methodology that can enhance our understanding of the complex relations among the entities investigated, where entities are represented as “nodes” and associations among entities are represented as “edges” between nodes in the network (Borsboom & Cramer, 2013). In recent decades, network analysis has become a powerful method for studying phenomena as diverse as interpersonal interaction, connections among genes, and individual symptom association, or biological mechanisms (Pavlopoulos et al., 2011). It is increasingly being used in clinical science and medical informatics to assess the “diseasome” characterized by comorbid associations among disorders (Brunson et al., 2020).
In particular, comorbidity network analysis (CNA) is an approach that assesses networks aggregated from measures of co-occurrence between pairs of disorders (Hidalgo et al., 2009; Fotouhi et al., 2018). In the comorbidity network, an individual disease is represented by a “node,” and the significant co-occurrence of two diseases is represented by an “edge.” Specifically, CNA can help identify influential disease associations and specific sets of comorbidities for further investigation by examining strongly connected individual nodes (i.e., diseases) and subnetworks of strongly connected nodes (i.e., clusters of comorbidities) in the network model (Brunson & Laubenbacher, 2018). In doing so, this approach may not only confirm existing knowledge on disease comorbidity but also reveal novel clinical comorbidity relationships that other approaches have not reported (Mu et al., 2020). Thus far, CNA has been applied to study comorbidity patterns among inpatient samples, including patients with depression (Qiu et al., 2022), ischemic heart disease (Zhou et al., 2022), and colorectal cancer (Ljubic et al., 2020). Nevertheless, to our knowledge, no studies have comprehensively analyzed the comorbidity patterns among older patients undergoing hip fracture surgery, nor have they employed CNA in such a context.
Objective
Using CNA, the present study aimed to investigate the comorbidity patterns among older patients undergoing hip fracture surgery. Specific aims were to (a) identify the complex associations among different diseases through network-based measures and (b) uncover the specific clusters of comorbidities through the cluster detection method. Particularly, we constructed comorbidity networks based on the standardized International Classification of Diseases (ICD) diagnosis codes included in the Charlson Comorbidity Index (CCI), which takes into consideration major medical comorbidities.
Methods
Design and Setting
This retrospective observational cohort study relied on electronic health records (EHR) data from Duke University Health System. EHR data offer rich and valuable insights into the nature of comorbidities. In particular, standardized ICD codes found in EHR comprise the principal method of recording diseases with the aid of national variants and exhibit significant promise for assessing, at the same time, the whole range of diagnoses, both physical and mental, in the process of evaluation of the patterns of comorbidity. Data were extracted through the Duke Enterprise Data Unified Content Explorer (DEDUCE), a web-based clinical research query tool that enables researchers to access administrative and clinical information such as diagnostic codes generated during patient care (Horvath et al., 2011). DEDUCE employs tools of business intelligence to provide insights into Duke Medicine’s enterprise data warehouse, offering a Guided Query functionality with the aid of a wizard-like interface enabling users to sift through millions of clinical records, delve into aggregate reports, and export extracts (Horvath et al., 2011).
Cohort Selection
The study comprised an initial cohort of older patients aged 65 and above (at the admission of the index hospitalization) who underwent surgery for the first time for a low-energy traumatic fracture of the hip between October 1, 2015 and December 30, 2018 (n = 1,171). We identified the records of patients with hip fractures through the ICD-10-CM diagnosis code of proximal femur fracture S72.0 (femoral neck fracture), S72.1 (pertrochanteric fracture), or S72.2 (subtrochanteric fracture) (see Supplemental Table S1). As ICD-10-CM replaced ICD-9-CM in 2015 and our period of interest relating to index hospitalization was after October 1, 2015, only ICD-10-CM codes were used. Surgery procedures were selected from ICD-10th procedure codes combined with the Current Procedural Terminology codes (see Supplemental Tables S2 and S3). Hip fracture surgery was defined as open reduction internal fixation, hemiarthroplasty, or total hip arthroplasty. The codes were selected based on consultations with two surgeons and an insurance coding expert. Two authors performed the cohort selection independently and discussed and resolved any discrepancies by going through the patients’ charts. Further details of cohort selection in this study are shown in Figure 1 and previously reported (Lee et al., 2022).
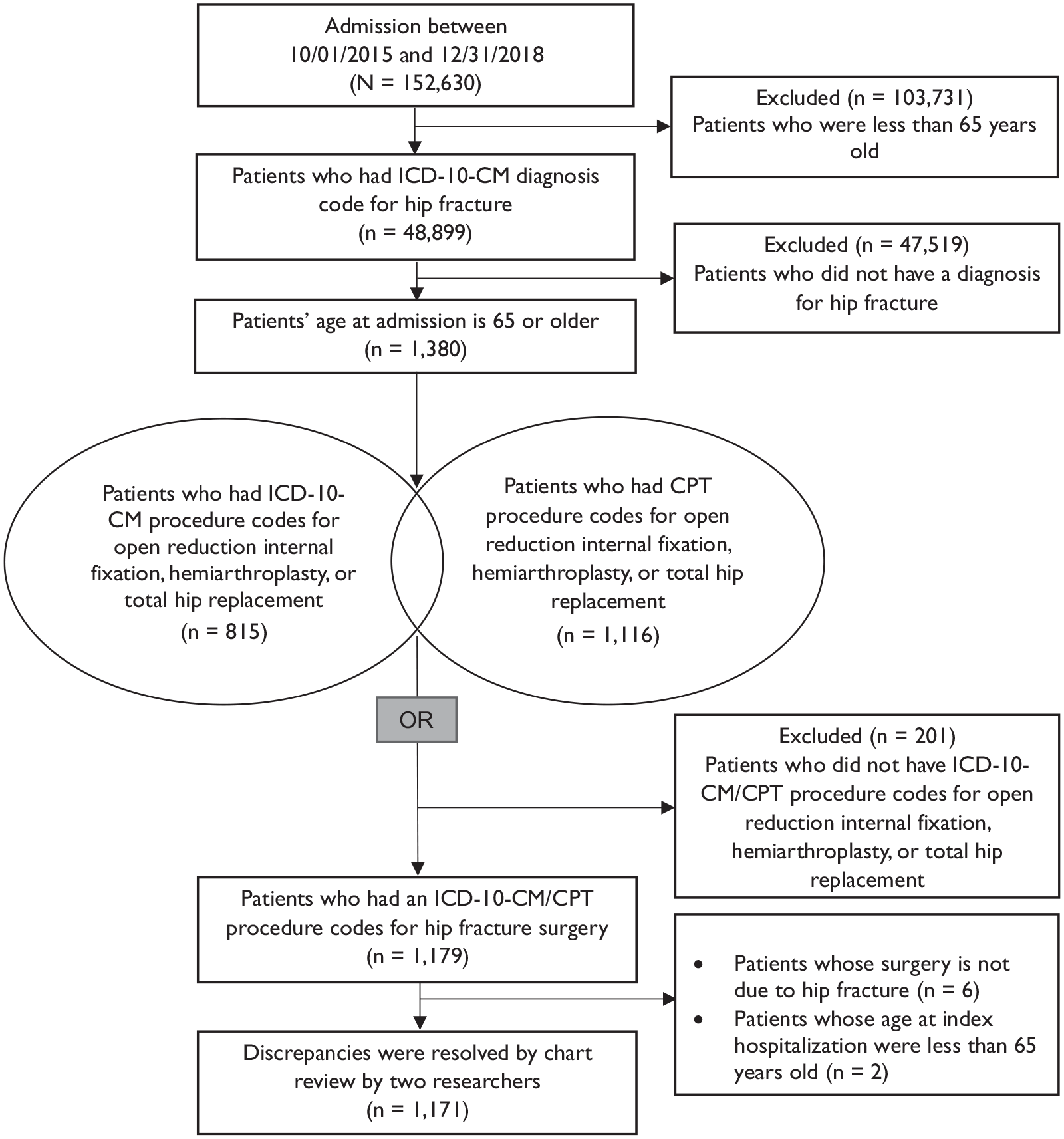
Process of cohort selection method.
Measures
In the current study, comorbidity includes 17 chronic conditions classified by the CCI. It includes myocardial infarction, congestive heart failure, peripheral vascular disease, cerebrovascular disease, dementia, chronic pulmonary disease, connective tissue disease/rheumatic disease, peptic ulcer disease, mild liver disease, diabetes without complications, diabetes with complications, hemi/paraplegia, renal disease, cancer, moderate or severe liver disease, metastatic carcinoma, and human immunodeficiency virus/acquired immunodeficiency syndrome. These conditions were identified based on the ICD diagnosis from encounters before and at the index hospitalization considering both ICD-9-CM and ICD-10-CM codes by converting ICD-9-CM to ICD-10-CM (Glasheen et al., 2019) (see Supplemental Table S4 for more details on diagnosis codes).
Statistical Analysis
All analyses were performed with R software 4.2.2 (The R Foundation, Vienna, Austria) and its associated packages. Descriptive and univariate statistics were used for the initial characterization of sample populations.
Comorbidity Identification (Aim 1)
A disease network model represents prevalent diseases as nodes in a graph, with pairwise co-occurrence relationships between diseases as edges. The purpose of the CNA is to assess whether diseases tend to co-occur more often than expected. To quantify the strength of such comorbid relationships, we calculated the observed-to-expected ratio (OER) of all possible disease pairs using the statistical approach described by Fotouhi et al. (2018).
OER ij , the comorbidity strength of the disease pair i and j, is defined as the ratio of the observed prevalence of disease pair i and j to the expected prevalence that is estimated as the product of the observed disease i prevalence and the observed disease j prevalence under the assumption that the diseases are independent:
OER is closely related to the relative risk in the previous studies of disease networks (Hidalgo et al. 2009; Park et al. 2009; Jeong et al. 2017).
Generally, OER greater than 1 signifies that pairwise diseases co-occurred more often in the same individual than expected, and OER less than 1 means that pairwise diseases tend to be mutually exclusive. Statistical tests were performed to examine the proportional differences between the denominator and the numerator, and p-values were obtained based on continuity correction. Only disease pairs with OERs > 1 and p-value < .05 were included in a network model, the associations were visualized using the R-package visNetwork. The diameter of each node reflects the prevalence of each disease, whereas the thickness of the edges reflects the OER value. The edges have no directionality due to the cross-sectional nature of the study design. For clarity, diseases with a prevalence of less than 1.0% of the total sample were excluded from the analysis (e.g., AIDS/HIV).
Several network centrality measures were used to evaluate the importance of nodes, namely degree, strength, closeness, and betweenness centrality (Hevey, 2018). Degree centrality is the simplest property of the nodes, estimated as the number of direct connections from each node in the network to other nodes. In other words, a disease with a higher degree centrality has more co-occurrence relationships with other diseases. Strength centrality is calculated by the sum of the strengths of the directly connected edges of a node, representing how strongly a disease is connected to other diseases. Closeness centrality gives the average shortest distance of a node to all other nodes, reflecting how fast a node can be approached from other nodes; a disease with a higher closeness centrality has a higher risk of being diagnosed with other diseases in fewer steps. Lastly, betweenness centrality denotes the number of times that the node acts as the bridge along the shortest paths, indicating how important a node is in the average path between other pairs of nodes.
Cluster Detection (Aim 2)
Various clustering algorithms implemented in the R-package igraph enable distinguishing the subsets of nodes in a network model that have a greater density of connections within their subset compared to across subsets. In the present study, following the study of Lee & Park (2021), we used the “infomap” algorithm available in igraph to distinguish those subsets of potential comorbidities that are more closely connected in a graph. The clusters and their respective nodes are illustrated in different colors.
Ethical Considerations
This was a retrospective observational cohort study using existing EHR data, and therefore it was exempted by the Duke university institutional review board (protocol number Pro0010528). The safety and security of all study data were ensured by storing, managing, and analyzing it within the highly protected virtual network space of our institution.
Results
Table 1 provides the sociodemographic and clinical characteristics of patients. The mean age of the patients (n = 1,171) was 81.5 years [standard deviation = 8.73], and the median was 82 years. Females accounted for 71.2% of this total, and the majority were White (82.7%). More than 60.0% of the patients had at least 2 of the 17 diagnoses. Furthermore, 22.8% of the patients had at least 5 of the 17 diagnoses. The most common were cerebrovascular (34.2%), chronic pulmonary (34.0%), and peripheral vascular (30.3%) diseases. More details are shown in Table 2.
Sociodemographic and Clinical Characteristics of Patients (n = 1,171).

Comorbidities Between 17 Diseases, Ranked by Frequency.
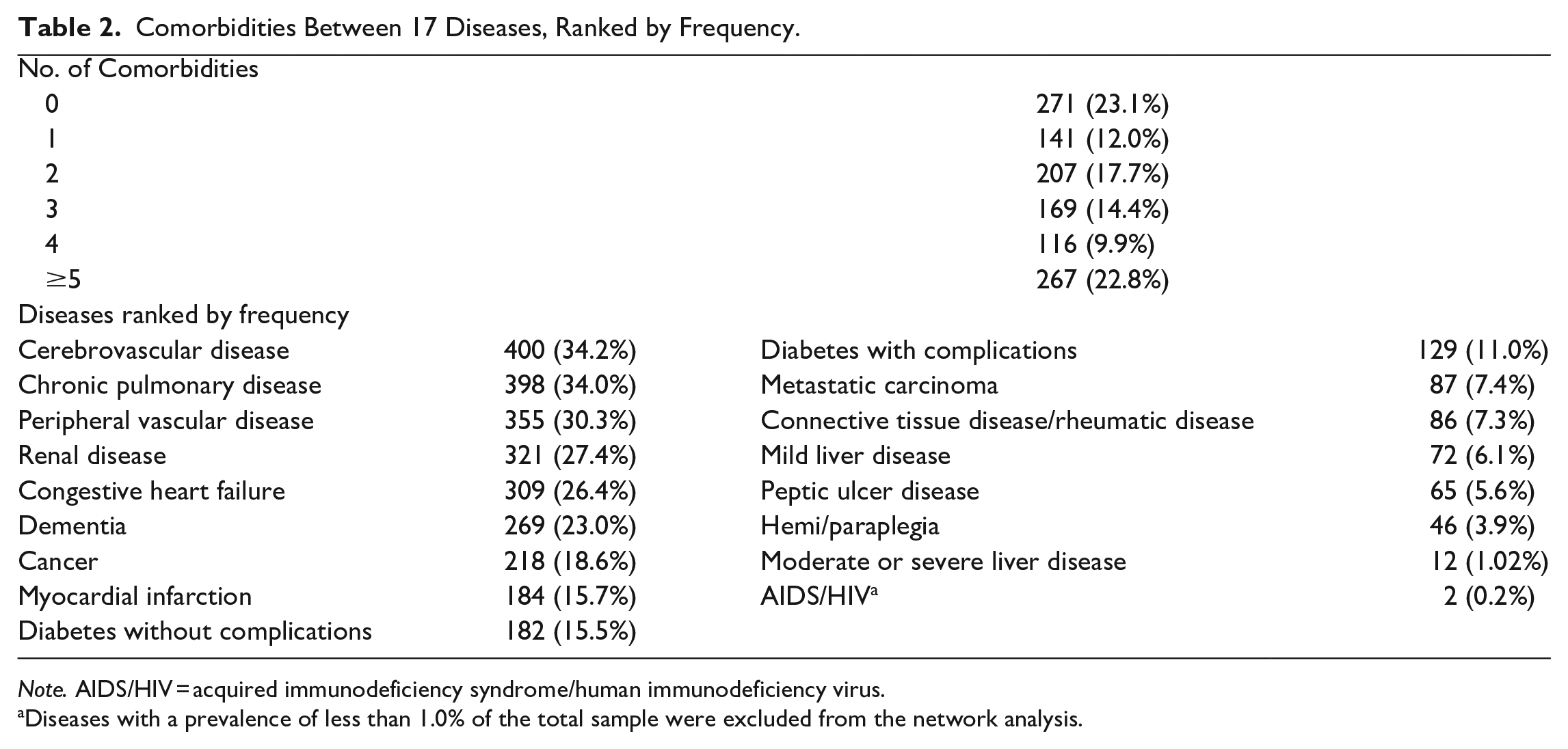
Note. AIDS/HIV = acquired immunodeficiency syndrome/human immunodeficiency virus.
Diseases with a prevalence of less than 1.0% of the total sample were excluded from the network analysis.
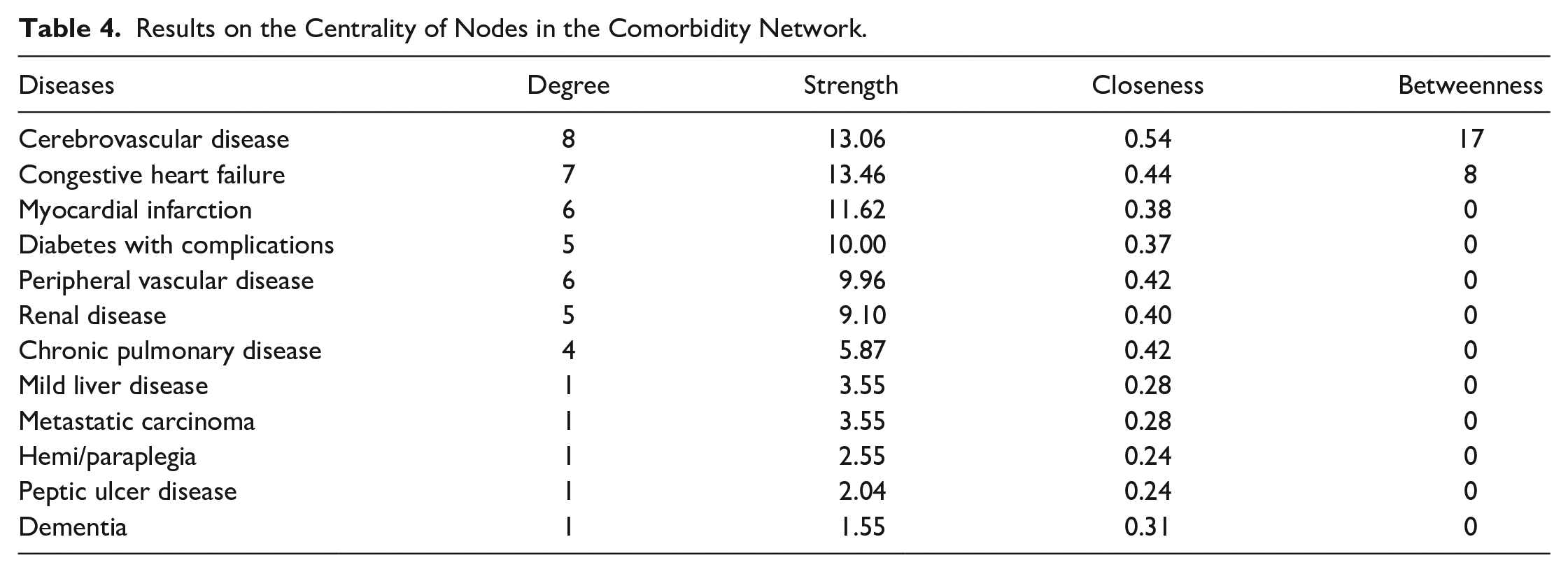
Twelve diseases were observed via visNetwork within the comorbidity network of the 17 diseases analyzed (OER > 1 with p-values < .05; Figure 2). Table 3 lists the strengths of the disease associations represented by OER values and the corresponding p-values. Specific examples indicate more robust associations than others, such as metastatic carcinoma and mild liver disease, myocardial infarction and congestive heart failure, and hemi/paraplegia and cerebrovascular disease. The probability of co-occurrence for each of these disease pairs was more than 2.5 times higher than expected (OER > 2.5). Table 4 details the results on the node centrality. Cerebrovascular disease and congestive heart failure were the most connected (central) diseases in the network based on the calculated node degree, strength, closeness, and betweenness values. Myocardial infarction also demonstrated higher strength and closeness values than other diseases in the network.
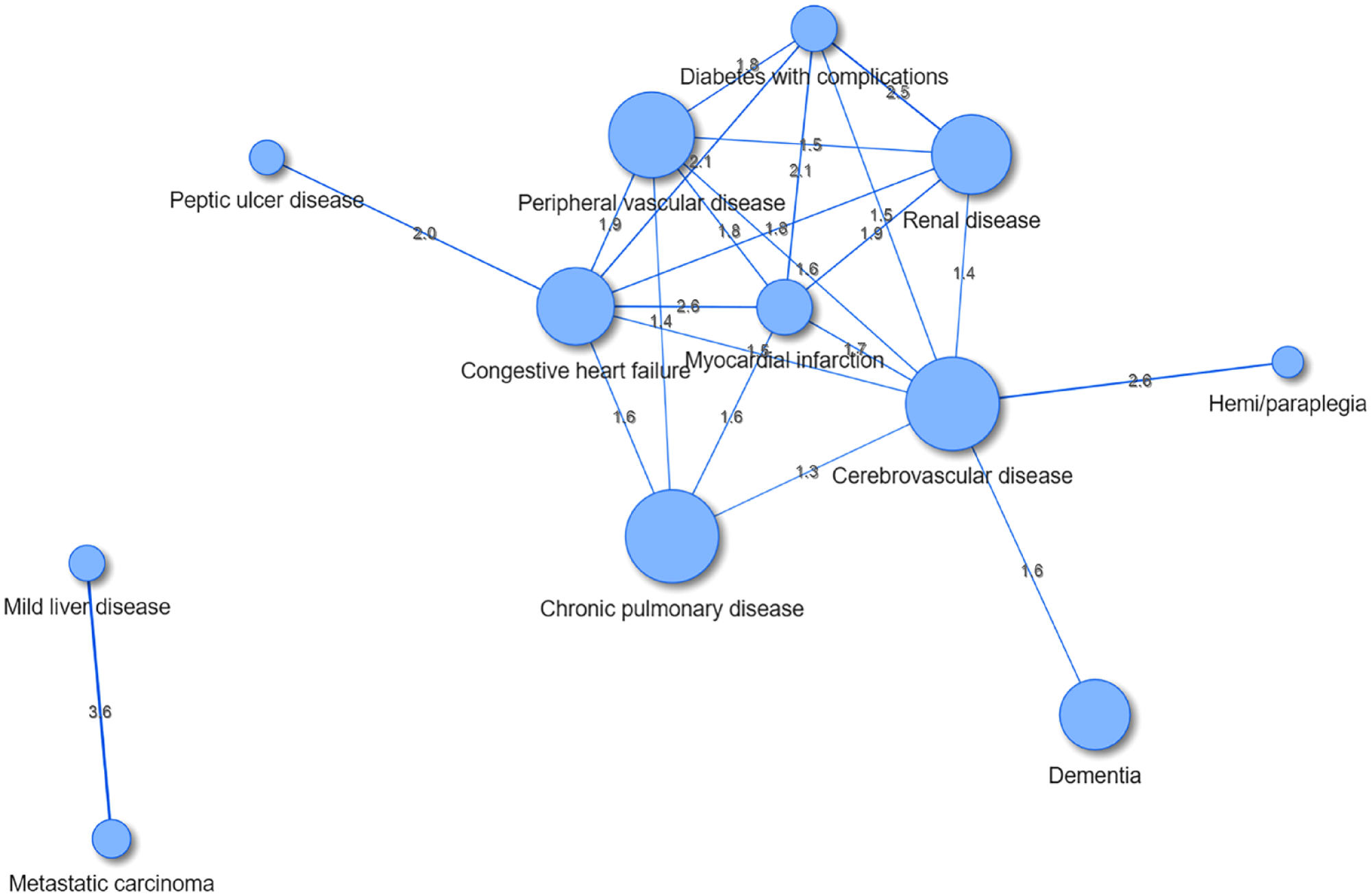
Comorbidity network among older patients undergoing hip fracture surgery.
Association Strength Between Diseases (largest to smallest).
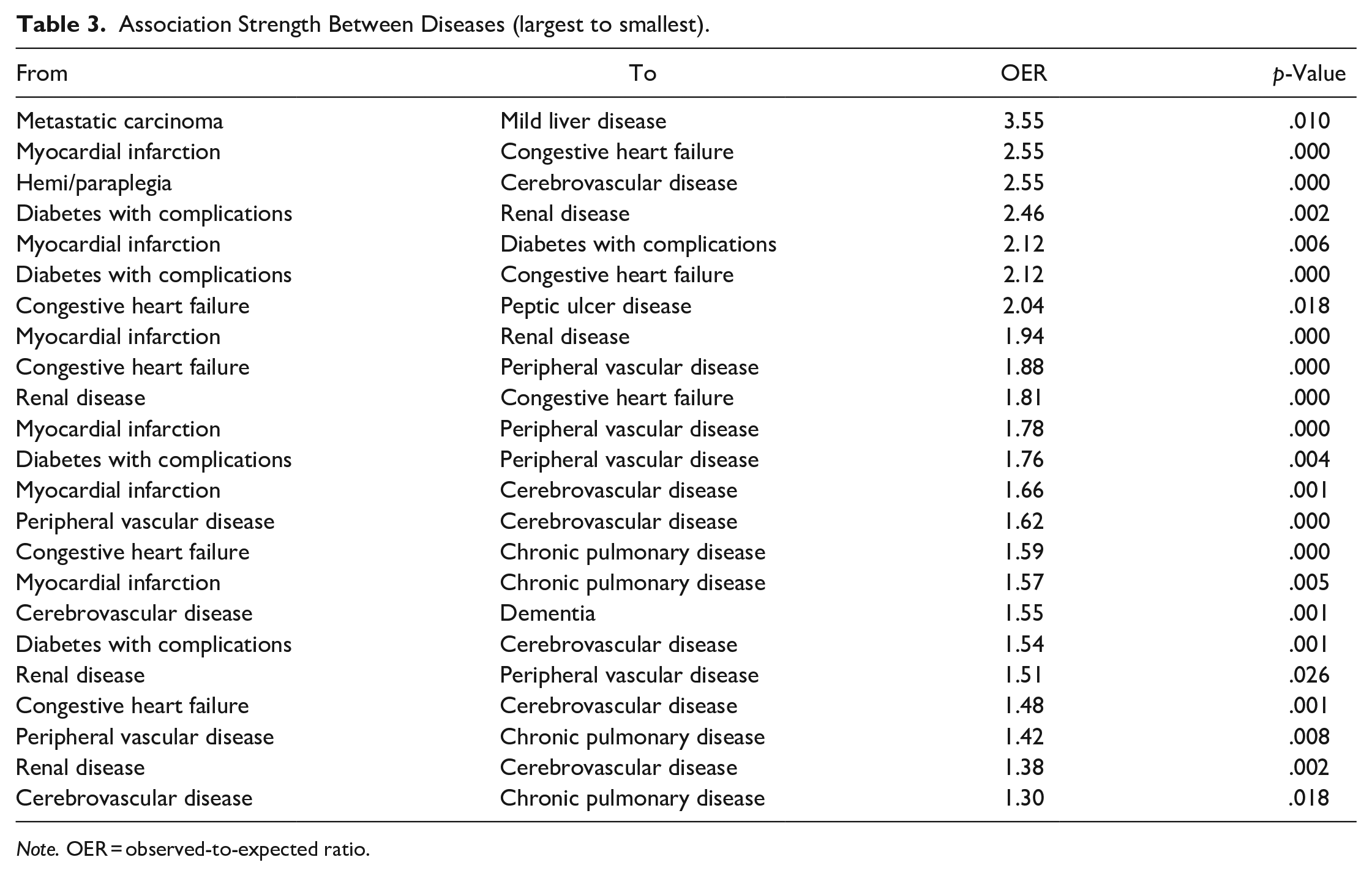
Note. OER = observed-to-expected ratio.
Results on the Centrality of Nodes in the Comorbidity Network.
Using igraph, the network was separated into two clusters. The largest cluster had 10 nodes (i.e., congestive heart failure, cerebrovascular disease, myocardial infarction, diabetes with complications, peripheral vascular disease, renal disease, chronic pulmonary disease, hemi/paraplegia, peptic ulcer disease, and dementia). Metastatic carcinoma and mild liver disease were also clustered into a group. The clusters and nodes are color-coded in Figure 3.
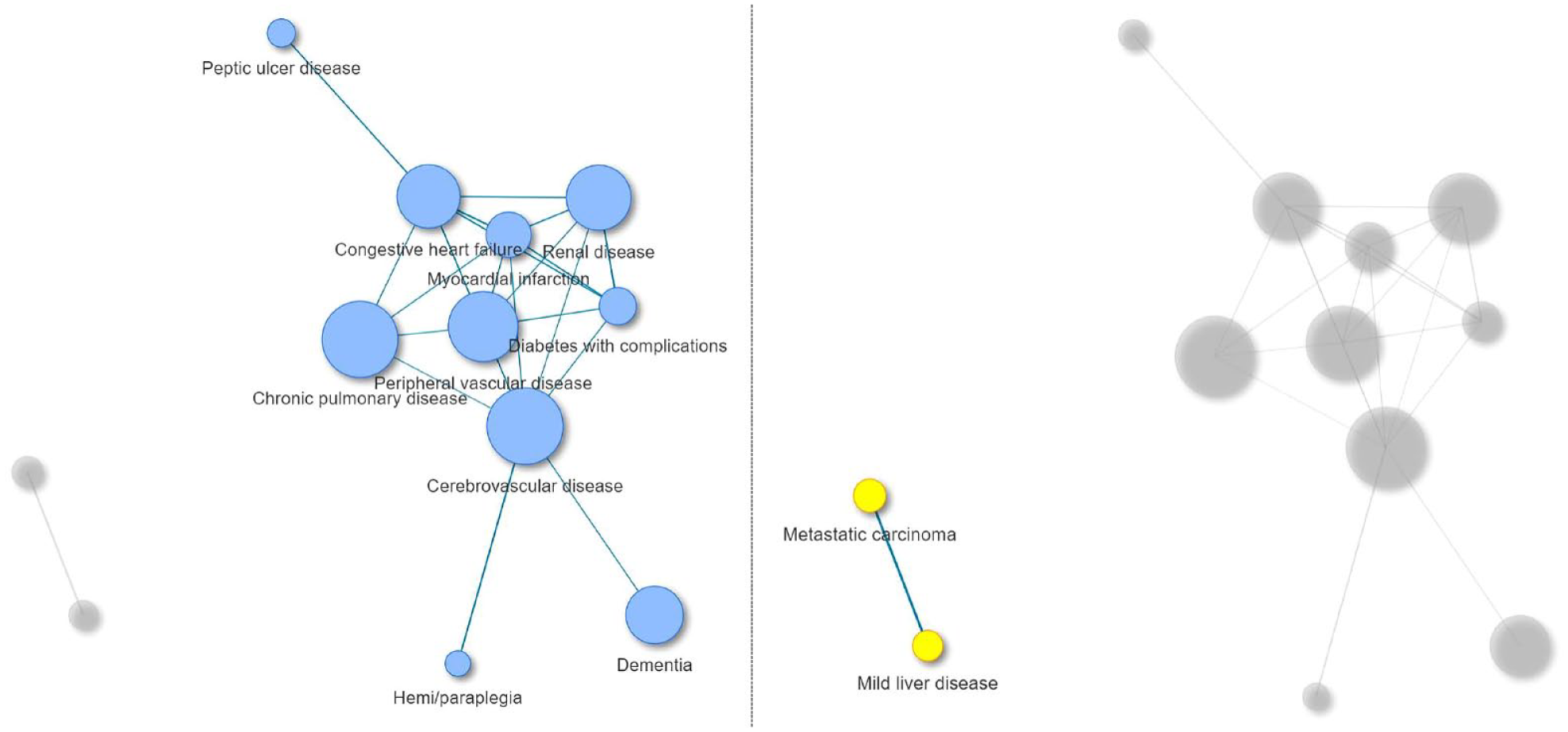
Results of a clustering algorithm.
Discussion
This study applied a network-based approach to analyzing and visualizing the comorbidity patterns of older patients undergoing hip fracture surgery. Most patients had more than two comorbidities, confirming observations from previous studies (Edelmuth et al., 2018; Nikkel et al., 2012). The diseases most prevalent among patients were cerebrovascular disease, chronic pulmonary disease, and peripheral vascular disease. Indeed, these diseases are the common risk factors for fractures. Mineralization and vascular calcification share common pathological pathways and medications, including cholesterol-lowering statins and bisphosphonates, which affect atherosclerosis and osteoporotic fractures (Holroyd et al., 2008; Kanis et al., 2001; Sennerby et al., 2009). Furthermore, patients with chronic pulmonary disease have an increased risk of osteoporosis and fractures; smoking, physical inactivity, malnutrition, and several pathophysiological explanations (e.g., a chronic inflammatory state) may be responsible for this association (Jørgensen & Schwarz, 2008). Chronic pulmonary disease has been consistently reported as one of the most prevalent diseases in older patients with hip fractures (Nikkel et al., 2012; Padron-Monedero et al., 2017; Wei et al., 2019).
Notably, cerebrovascular disease, congestive heart failure, and myocardial infarction were identified as central diseases that co-occur and significantly interlink with several diseases in the network. Cerebrovascular disease emerged as the most central disease in the patient group, possibly due to its high prevalence, shared pathogenic processes, and common risk factors with other diseases. Such findings support the necessity of a comprehensive approach to treating patients diagnosed with these central diseases to ensure optimal postoperative care and effectively manage the concurrent health conditions they may have. For optimal recovery and to prevent postoperative complications in patients with hip fractures, clinical guidelines or treatment strategies oriented to individual diseases might be ineffective and incomplete.
Critically, the strongest association involving cerebrovascular disease was with hemi/paraplegia. The literature supports that patients with hip fractures and post-stroke hemiplegia history benefit less from rehabilitation, resulting in increased chronic disability following a hip fracture, increasing the risk of a poor long-term prognosis, and including higher mortality (Nho et al., 2014; Wang et al., 2022). Feng et al. (2009) also highlighted that these patients typically have more severe health conditions, alongside multiple comorbidities, diminished cognitive and walking abilities, and longer hospital stays, all of which emphasize the need for the assessment of these concurrent conditions in orthogeriatric care.
The results revealed two distinct comorbidity clusters of highly interlinked diseases. Notably, patients exhibited co-occurrence with 10 illnesses, including diseases of the cardiovascular/circulatory system (e.g., heart failure), renal disease, pulmonary disease, peptic ulcer disease, other metabolic diseases (e.g., diabetes mellitus), and dementia. The findings highlight the co-occurrence of both physical and cognitive comorbidity among hip fracture patients. In addition, metastatic carcinoma and mild liver disease, each of which is well-known to independently heighten the risk of surgical complications (Forssten et al., 2022; Frost et al., 2011; Kannegaard et al., 2010), were most densely connected out of all disease pairs and also clustered together. Identifying such clusters can help providers take preemptive actions related to the health of a patient.
Importantly, the occurrence of the diseases in a distinct cluster implies that the patient population may be markedly prone to serious complications associated with these conditions. Because a cluster has the maximum possible interactions between nodes, its presence in a patient can be critical due to the high risk (Kalgotra, 2017). Such a cluster indicates a trap state in a patient where escape is challenging (Kalgotra, 2017; Van Borkulo et al., 2015). An important research question is related to the influence of these clusters on health outcomes: “Can we identify a cluster as a trap state, where its presence in a patient increases mortality risk, hospital stay duration, or other critical outcomes?” The influence of multiple concurrent diseases and their underlying interactions can be significantly more intricate and synergistic than the sum of their effects for each disease.
Furthermore, the results based on the clustering algorithm underscore a need for a multidisciplinary orthogeriatric team approach to patient care for patients undergoing hip fracture surgery, which has recently gained greater literature support (Henderson et al., 2017; Pfeufer et al., 2020; Santy-Tomlinson et al., 2021). A multidisciplinary approach to identifying and optimizing comorbidities perioperatively enables better perioperative periods and long-term functional outcomes.
Limitations
This study is subject to several limitations. First, the cohort in the current study was based on a single hospital system, which can result in a lack of generalization of the study findings and selection bias. Thus, data interpretation should be treated with caution. Second, the undirected graph provides a limited insight into the causality of the observed interactions. It should be noted that to use a CNA appropriately to guide intervention strategies and understand disease progression, longitudinal designs in which we can model changes in variables across time are needed. Third, we acknowledge that critical risk factors that influence comorbidities patterns such as age, sex, race, or socioeconomic factors have not adequately been accounted for in the networks, such as performing analyses by stratifying the cohort by these factors. Indeed, prior studies based on CNA have demonstrated differences in the strength of comorbidities in patients of different sexes and races (Qiu et al., 2022; Zhou et al., 2022), possibly reflecting variations in biological processes, environmental factors, or the quality of healthcare provided to each population. Although the overall sample size was not small, the dataset displays an imbalanced distribution of age, sex, race, and socioeconomic factors (e.g., insurance status), and therefore, conducting rigorous stratification analyses was not feasible in the current study. In particular, due to the small sample sizes for some diseases, using these imbalanced stratified samples might lead to the non-detection of multimorbid disease pairs that are disproportionally represented in the dataset, which could result in an underestimation of the complexity of the comorbidity network. With our study as a starting point for the application of the CNA approach to older patients undergoing hip fracture surgery, it is our hope that future studies integrate more samples and investigate how diseases co-occur differently and form distinct network structures across diverse patient populations. Finally, psychiatric illnesses (e.g., depression and anxiety disorders) were not considered in this study. The extension of network models to include these diseases could significantly contribute to deepening our understanding of patient comorbidities. For instance, depressive disorders were reported to be prevalent among older hip fracture surgery patients (Nikkel et al., 2012) and even lead to an increase in postoperative complications after hip fracture surgery (Broggi et al., 2021), but are often overlooked and not incorporated into the clinical care of patients with hip fractures.
Conclusions
Using a network approach and diagnostic codes from EHR data, the complex relationships between chronic conditions were explored in depth, which has important implications for the integrated management of patients undergoing hip fracture surgery. We hope this study can serve as a preliminary guideline for many other nursing studies; thus, we share the R code in Supplemental Table S5.
Supplemental Material
sj-docx-1-cnr-10.1177_10547738231209367 – Supplemental material for Comorbidity Patterns in Older Patients Undergoing Hip Fracture Surgery: A Comorbidity Network Analysis Study
Supplemental material, sj-docx-1-cnr-10.1177_10547738231209367 for Comorbidity Patterns in Older Patients Undergoing Hip Fracture Surgery: A Comorbidity Network Analysis Study by Chiyoung Lee, Sijia Wei, Eleanor S. McConnell, Hideyo Tsumura, Tingzhong (Michelle) Xue and Wei Pan in Clinical Nursing Research
Footnotes
Acknowledgements
None.
Credit Authorship Contribution Statement
Availability of data,code,and materials
Deidentified participant data is available upon reasonable request from the corresponding author. The R code is shared in the Supplementary file.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by Sigma Theta Tau International’s Psi-at-Large Chapter.
Supplemental Material
Supplemental material for this article is available online.
Author Biographies
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.