Abstract
Background
Balance impairments and fatigue are common after stroke and impact physical therapy assessments and treatments. Reasons are multifactorial and include motor dysfunction and changes to cortical activation poststroke. The prefrontal cortex (PFC) is involved in motor control; yet, limited research has explored cortical activation during common physical therapy balance tasks or the link with fatigue.
Objectives
During standing balance tasks, the objective was to determine whether PFC activation levels: (1) change between tasks, (2) are asymmetric, and (3) are associated with fatigue.
Methods
Patients with hemiparesis were recruited from an inpatient stroke unit and functional near-infrared spectroscopy was applied bilaterally over the PFC to measure cortical activation during balance tasks. Fatigue was assessed using the Fatigue Severity Scale (FSS).
Results
Nine participants were included. PFC activation during semi-tandem stance showed greater amplitude than during double-leg stance, indicating more cortical activation. Bilateral PFC activation was observed during both tasks. Participants with greater fatigue (higher FSS score) showed more activation in the ipsilesional PFC than the contralesional PFC.
Conclusion
PFC activation may occur when performing more challenging balance postures, potentially indicating compensatory activation, and may be linked with greater fatigue.
Keywords
Introduction
A stroke causes cerebral damage by ischemia or hemorrhage, reducing the ability of the brain to generate the cortical activation needed to control functional daily tasks (Foucher & Faure, 2020; Lin et al., 2013). Post-stroke, individuals may experience hemiparesis, defined as weakness on one side of the body (Lin et al., 2013). Hemiparesis impacts up to 73% of people after a stroke and may affect the lower extremities, cause balance difficulties, and an increase in fall risk (Denissen et al., 2019; Wei et al., 2019). Falls can also result in hospitalizations, compromised independence, and ultimately worsen quality of life (Basso Moro et al., 2014; Cumming et al., 2016). Impaired postural control is recognized as a primary determinant of falls after stroke (Persson & Hansson, 2021). Although addressing standing balance during physical therapy interventions is crucial to reduce fall risk in people post-stroke, the link between cortical activation and standing balance after stroke is less clear and a better understanding may help determine response to physical therapy interventions.
Fatigue also significantly impacts people post-stroke, with prevalence rates ranging from 23% to 75% (Mutai et al., 2017; Wu et al., 2015). Fatigue, defined as a lack of physical or mental energy preventing an individual from performing activities of daily living (Cumming et al., 2016; Kuppuswamy et al., 2015; Mutai et al., 2017), hinders functional improvement during physical therapy (Glader et al., 2002; Wu et al., 2015). Poststroke fatigue has widespread impact on physical health and is associated with decreased range of motion, coordination, and altered cognition (Kutlubaev et al., 2015). While fatigue impacts cognition and motor function, it is unknown whether there is a relationship between cortical activation, balance and fatigue in people with subacute stroke who are admitted to inpatient rehabilitation.
Functional near-infrared spectroscopy (fNIRS) is used to investigate cortical activity during standing balance in healthy younger and older individuals (St George et al., 2021) as it detects prefrontal cortex (PFC) activation by monitoring changes in oxygenated and deoxygenated hemoglobin content (oxy-Hb and deoxy-Hb, respectively, Fujimoto et al., 2014; Pinti et al., 2020). When maintaining balance, younger adults have more right PFC activation, while older adults display bilateral PFC activity, potentially reflecting an age-related compensatory change due to reduced cognitive resources when maintaining balance (Teo et al., 2018). After stroke, postural perturbations increase ipsilesional PFC activation compared to the contralesional PFC during dynamic balance tasks (Fujimoto et al., 2014). What remains uninvestigated is PFC activation during static standing balance tasks poststroke (Fujimoto et al., 2014; Schulz et al., 2019; St George et al., 2021). Measuring PFC activation during standing balance may provide valuable information of the cortical activation required to control standing balance. Conventional imaging techniques, including functional magnetic resonance imaging or positron emission tomography, are not feasible due to the nature of standing tasks and these imaging techniques requiring participants to be supine (Fujimoto et al., 2014). fNIRS is a non-invasive neuroimaging technique suitable to assess cortical activation during standing balance (Fujimoto et al., 2014; Pinti et al., 2020) with good test-retest reliability during postural tasks and high tolerance of motion artifacts (Pinti et al., 2020; Rond et al., 2023; Suzuki et al., 2004). fNIRS has already been used to observe cortical activation patterns for healthy adults and patients during various tasks (Basso Moro et al., 2014; Chen et al., 2022; Fujimoto et al., 2014; St George et al., 2021).
Increased fatigue can negatively impact the cognitive control processes regulated by the PFC (Pires et al., 2018), and can be measured using fNIRS. Due to the PFC's role in execution of goal-directed movements, increased fatigue levels may impact its efficiency, resulting in a reduced ability to maintain balance (Pires et al., 2018). Investigating fatigue and its relation to the maintenance of balance may help further understand impairments post-stroke and potentially inform physical therapy interventions. Taken together, this study aimed to determine: 1) the potential changes in oxy-Hb and deoxy-Hb levels in the PFC measured with fNIRS during common standing balance tasks used in physical therapy interventions, 2) whether hemispheric PFC activation is symmetrical during these tasks, and 3) the potential link between self-perceived fatigue and task-based activation in people after stroke.
Material and Methods
Participants
Participants were recruited from the inpatient stroke unit at Parkwood Institute in London, Ontario, Canada. The study included individuals with lower limb hemiparesis who were medically stable, had the ability to understand and follow instructions, and could maintain a standing position for up to 20 s. Patients were excluded from the study if they had a cerebellar stroke due to the resultant impaired coordination limiting their ability to complete balance tasks. Individuals with any other chronic health condition that would affect participation (e.g., Parkinson's disease, multiple sclerosis, cancer) or any condition that directly affects hemoglobin (e.g., sickle cell anemia) were also excluded. Additional information, including age in years, sex, time since stroke, type of stroke, location of the stroke, and functional ability, were recorded. All participants provided informed written consent, and the study was approved by the Western University Health Sciences Research Ethics Board (HSREB#119880).
Balance Tasks
Participants were asked to perform two standardized balance tasks, a semi-tandem task to challenge postural stability, and a double leg stance to ensure all participants could complete the task (Bayouk et al. 2006). Participants were asked to focus on a fixation point during the tasks to maintain consistency between conditions. A licensed physiotherapist was present during all data collection sessions to ensure participant safety. For added safety and support, participants were positioned with a plinth in front of them and a chair behind them, ensuring that support was available in case they needed to rest during the session (Figure 1). The task order for semi-tandem and double leg stance was randomized. Each task included a five second baseline period, followed by 20 s of the balance tasks and an additional 15 s of rest, repeated five times, for a total of ten repetitions (five double leg and five semi-tandem).
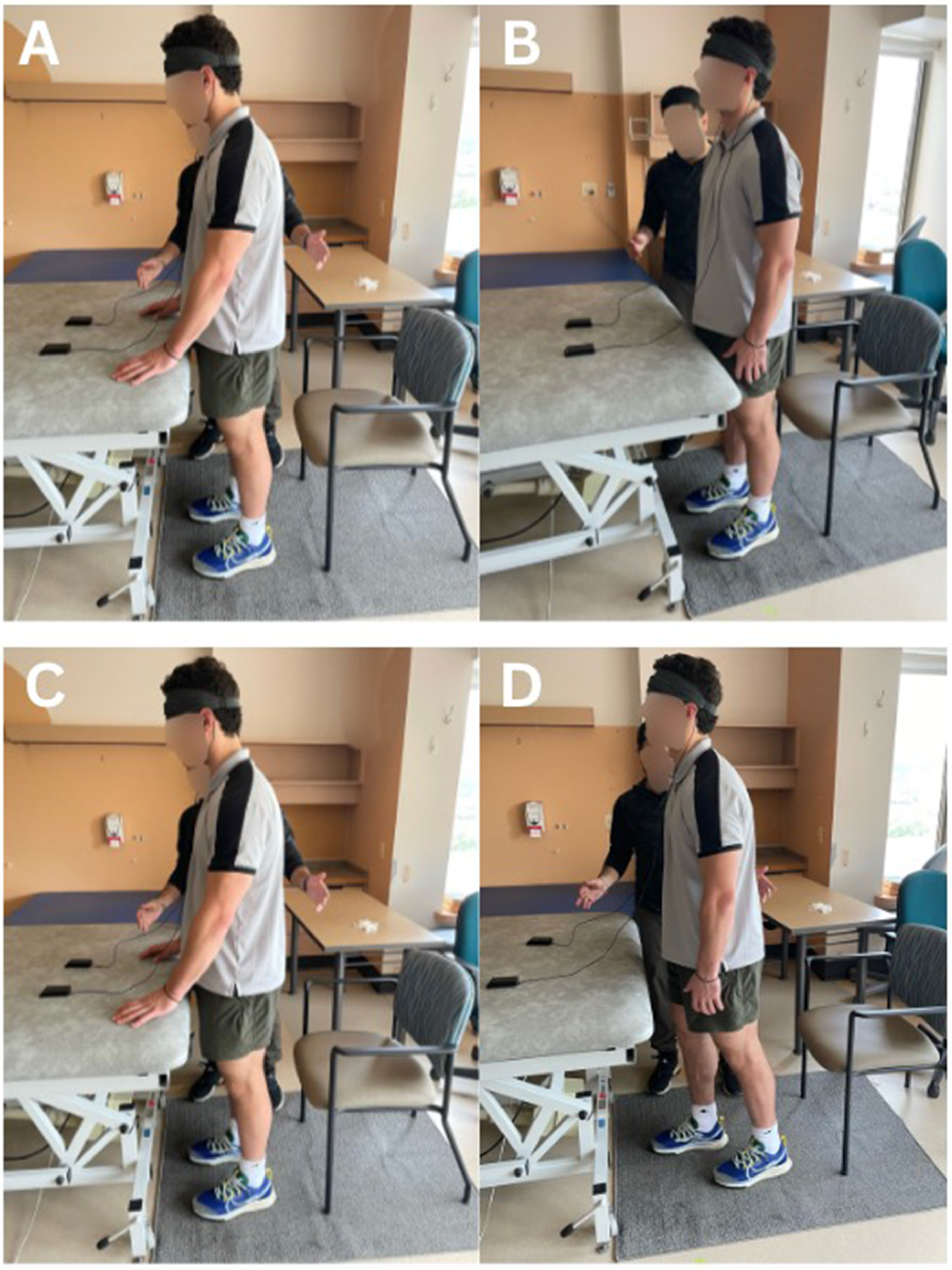
Representation of participant position during rest and task for the double-leg stance during fNIRS recording. A: Participant maintaining contact with a bed for support during baseline; B: Performing the double leg stance independently, C: Participant maintaining contact with a bed for support during rest; D: Participant performing the semi-tandem stance independently.
Clinical Outcome Measures
The Berg Balance Scale (BBS) provided a quantifiable measure of balance and is reliable and valid to assess balance for people post-stroke (Blum & Korner-Bitensky, 2008; Huang et al., 2020; Liao et al., 2021; Mao et al., 2002). The BBS has 14 items, with each scored from zero to four, where zero represents the inability to complete tasks, and four represents complete independence (Blum & Korner-Bitensky, 2008; Stevenson, 2001). The BBS was administered independent of the current study by a licensed physiotherapist upon hospital admission to the inpatient stroke unit. Following the fNIRS balance tasks, the Fugl-Meyer Lower Extremity (FM-LE) assessment was administered by a licensed physiotherapist to measure the level of motor impairment post-stroke. The FM-LE has good validity and reliability among individuals’ post-stroke (Gladstone et al., 2002).
Fatigue Severity Scale (FSS)
The FSS questionnaire is commonly used to measure subjective fatigue (Learmonth et al., 2013). The questionnaire is unidimensional consisting of nine items that participants rate on a seven-point Likert scale ranging from one (strong disagreement) to seven (strong agreement) and is then averaged. Higher FSS scores indicate greater self-perceived fatigue (Sharma & Sheth, 2019; Valko et al., 2008). The FSS has excellent test-retest reliability and internal consistency among individuals’ post-stroke (Nadarajah et al., 2017). To minimize the fatigue from balance tasks, the FSS was administered prior to the fNIRS.
fNIRS System
Changes in oxy-Hb and deoxy-Hb concentrations in the PFC were measured during both balance tasks using the PortaLite fNIRS system (Artinis Medical Systems, the Netherlands). The PortaLite has 3 light sources and 1 detector, with source-detector distances of 30, 35, and 40 mm. Two devices were positioned bilaterally, one over each PFC, and placed according to the international 10–20 EGG system at the height of 10% of the nasion-inion distance from the nasion and 10% of the head circumference to the left and right from the midline (Herbert, 1958; Mirelman et al., 2017). The devices were covered using a dark headband to prevent noise signals attributed to ambient light. Real-time neural activation in the form of oxy-Hb and deoxy-Hb was recorded at a frequency of 50 Hz. Oxysoft version 3.2.72 x64 (Artinis Medical Systems, the Netherlands) was used for data collection.
fNIRS Data Processing
The raw data of oxy-Hb and doxy-Hb was exported to open source Homer3 software for further data processing (Huppert et al., 2009). First, the raw intensity data was converted to optical density using hmR_Intensity2OD. A bandpass filter with frequencies of 0.01 to 0.14 Hz was then used to reduce physiological noise such as heartbeat, respiration, and signal drift (Maidan et al., 2016). The data were then converted from optical density to concentration changes using hmR_OD2Conc, which utilizes the modified Beer-Lambert law (partial pathlength factor = 1.0)(Hocke et al., 2018). To remove motion artifacts and improve the signal, a correlation-based signal improvement (CBSI) filter was used (Maidan et al., 2016). Finally, a block average filter was used by taking the baseline period (5 s before task onset) and the combined task and rest periods (0 s to 35 s) to calculate a hemodynamic response for both tasks (Hocke et al., 2018). Concentration changes for oxy-Hb and deoxy-Hb from the first channel (S1 to D1) was used from each device, resulting in four hemodynamic response functions (ipsilesional and contralesional) for each task. Task-based activation was determined by subtracting the average oxy-Hb concentration during baseline performance from the average oxy-Hb concentration during the task period (Maidan et al., 2016). This calculation allowed for the assessment of specific changes in oxy-Hb concentration attributed to task participation relative to the baseline (Maidan et al., 2016). Oxy-Hb concentration was used in this calculation since it is more sensitive than deoxy-Hb to motor tasks in the hemodynamic response function (HRF) (Borrell et al., 2023).
Laterality Index (LI)
The laterality index (LI) was used to evaluate the asymmetry between the ipsilesional and contralesional PFC. Negative and positive LI values represent contralesional and ipsilesional PFC activation, respectively (Jia et al., 2022). The predetermined LI threshold is usually set to −0.2 ≥LI ≤ 0.2, with values in this range indicating bilateral activation (Borrell et al., 2023). The formula for LI is as follows:
Statistical Considerations
The current study was conducted during the COVID-19 pandemic with people with stroke admitted to inpatient stroke rehabilitation; therefore, this frail population was difficult to recruit. Thus, the focus of this study was to meticulously define the sample population and describe in detail relationships which should be considered in future work.
Results
Demographics
Table 1 shows the clinical and demographical characteristics of the nine participants included in the study (five females and four males). All participants were in the sub-acute stage of stroke recovery, with an average of 39 days (SD = 28 days) since their stroke.
Participant Characteristics.
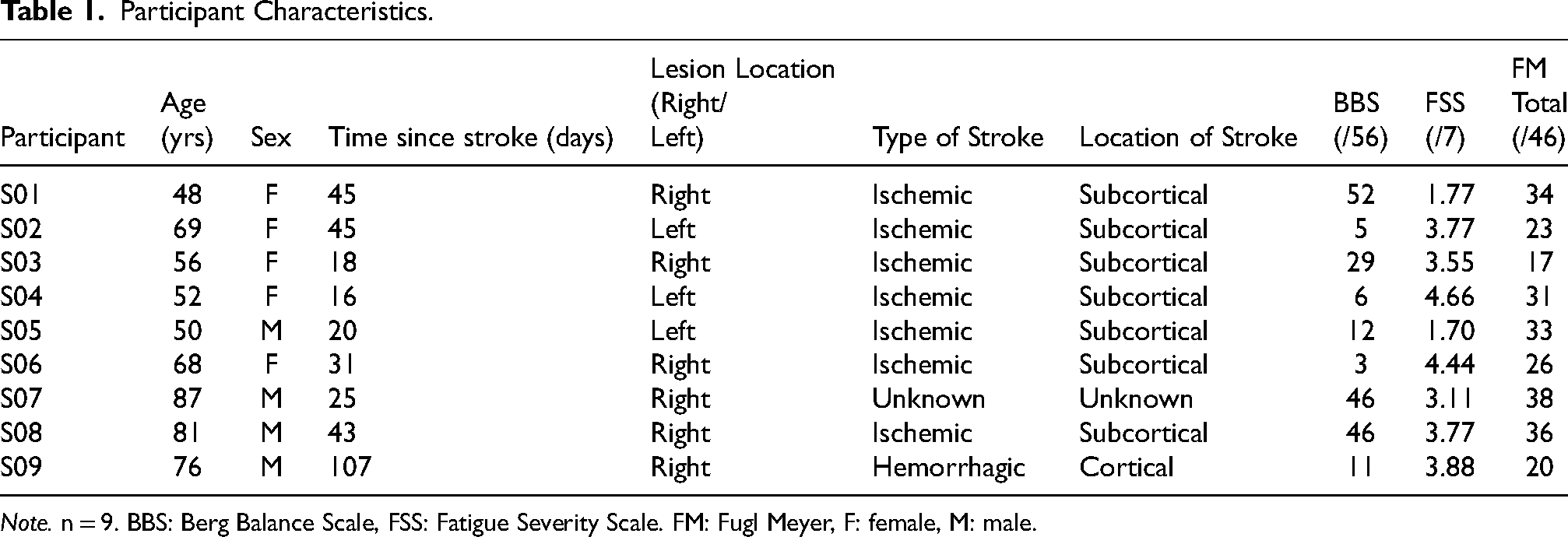
Note. n = 9. BBS: Berg Balance Scale, FSS: Fatigue Severity Scale. FM: Fugl Meyer, F: female, M: male.
Aim 1: Do oxy-Hb and Deoxy-Hb Levels Change for Standing Balance Tasks?
An inverse relationship between oxy-Hb and deoxy-Hb was observed for both tasks, indicating that as oxy-Hb increased, deoxy-Hb decreased, and vice versa (Figure 2). Both tasks (0 s to 20 s, Figure 2) produced an increase in activation (an increase in oxy-Hb concentration and a decrease in deoxy-Hb) compared to rest (−5 s to 0 s, Figure 2). During both tasks, the HRF took approximately 25 s after task onset to reach peak activation levels and approximately 5 s to 10 s to return to baseline levels. The semi-tandem stance displayed a greater increase in amplitude (oxy-Hb) in the ipsilesional and contralesional PFC compared to the double-leg stance (Figure 2). PFC activation was greater in the ipsilesional than the contralesional hemisphere during both tasks, although bilateral activation occurred. The contralesional deoxy-Hb concentration during the semi-tandem stances slightly increased, with an inverse relationship with oxy-Hb at approximately 12 s after task onset (Figure 2). Larger variability in the standard deviation of the oxy-Hb concentration of the ipsilesional PFC during the double leg stance was observed (Figure 2) as two participants had decreased oxy-Hb during the task (Figure 3, open circle and diamond symbols).
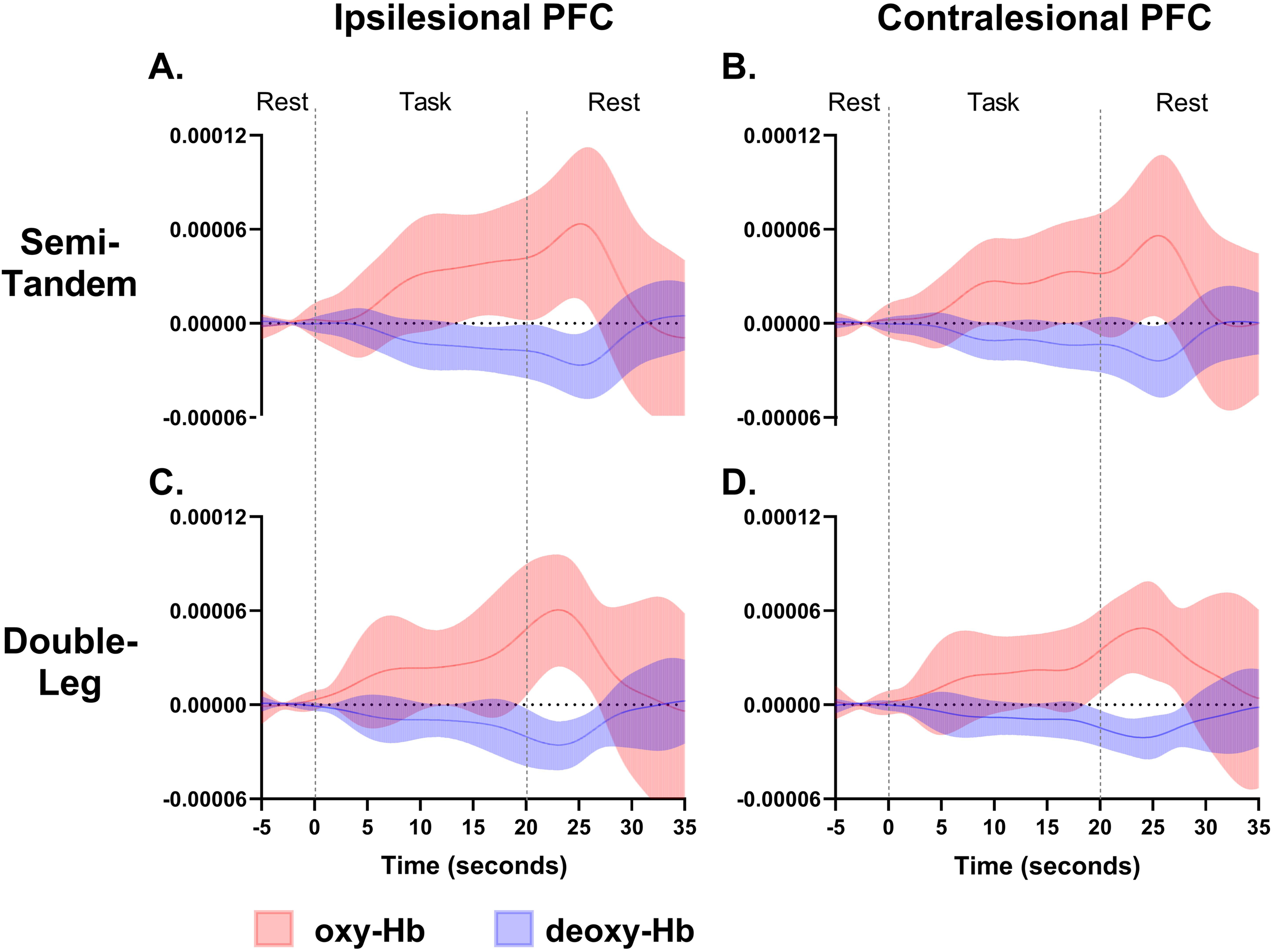
Average ipsilesional and contralesional PFC activation during both tasks. Panel A & B display PFC activation during semi-tandem stance, Panel C & D show PFC activation for double-leg stance. Solid lines represent average oxy-Hb and deoxy-Hb, with shaded areas displaying standard deviation. Oxy-Hb: oxygenated hemoglobin, deoxy-Hb: deoxygenated hemoglobin, SD: standard deviation, PFC: prefrontal cortex. X-axis: time (seconds), y-axis: concentration (μM/mm).
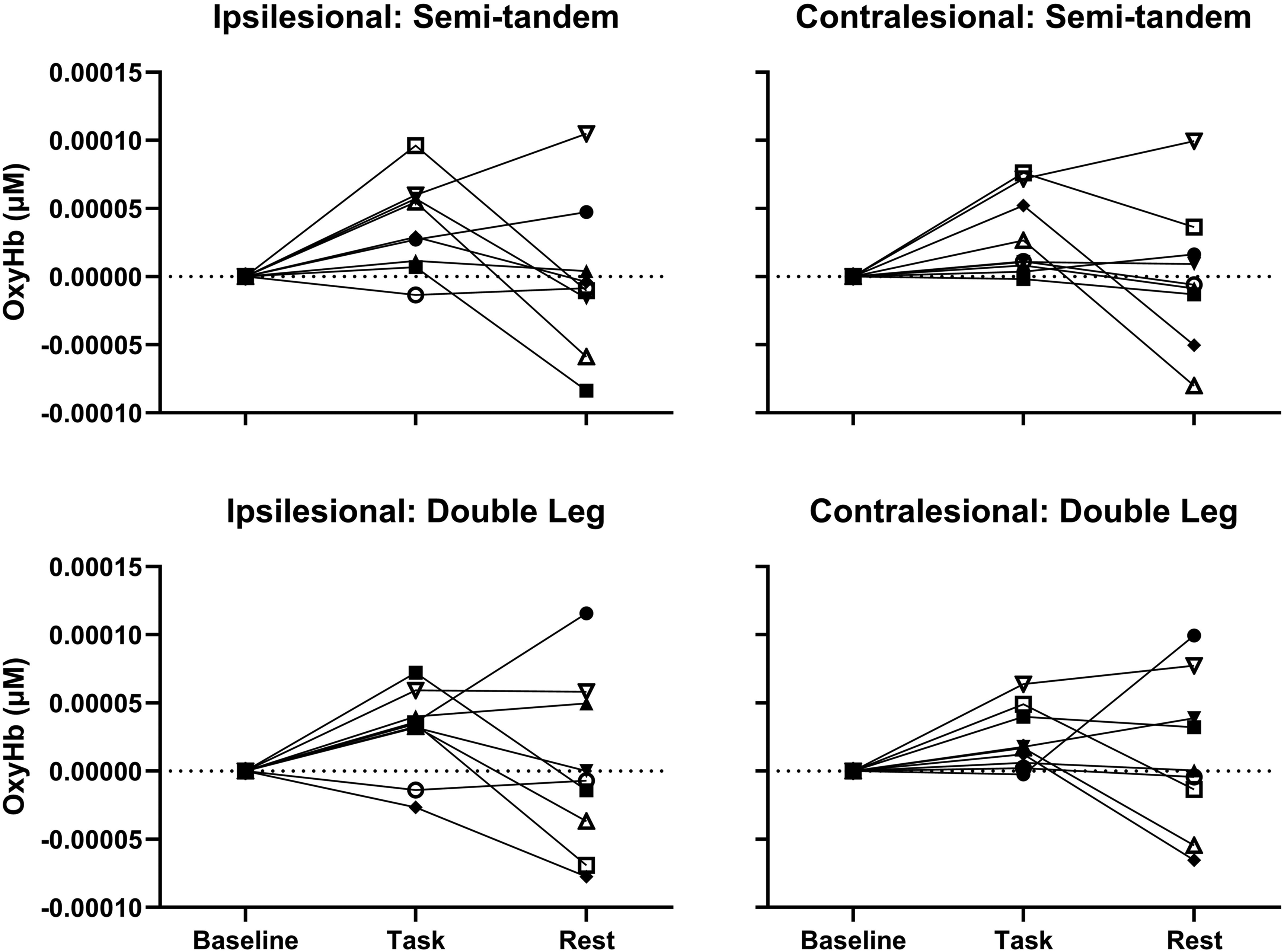
Change in PFC activation for all Participants During Both Tasks.
Aim 2: is Hemispheric PFC Activation Asymmetrical During Standing Balance Tasks?
Although there were no consistent patterns among participants for laterality, many patterns emerged and are reported below. As shown in Table 2, participants had a range of LI scores, with some showing bilateral activation during both tasks (−0.2 ≥ LI ≤ 0.2) (Borrell et al., 2023). The average LI during the double-leg stance was −0.201, indicating a slight preference for contralesional PFC activation. The average LI during the semi-tandem stance was 0.018, suggesting bilateral PFC activation.
Laterality index Values for Both Tasks.
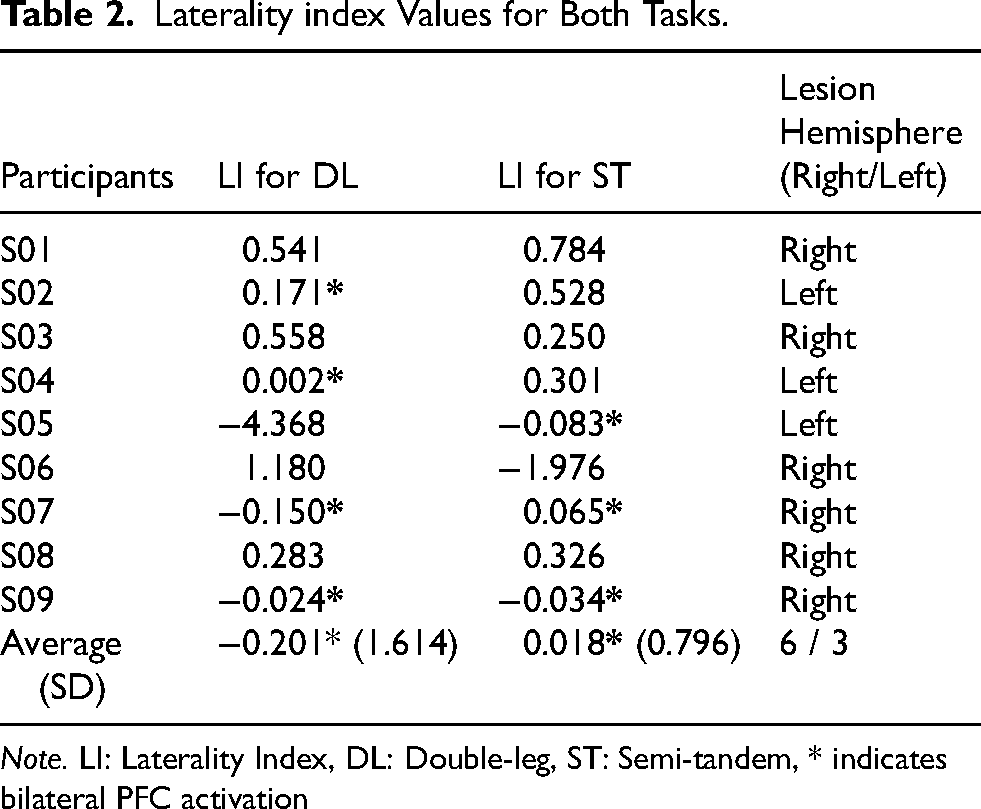
Note. LI: Laterality Index, DL: Double-leg, ST: Semi-tandem, * indicates bilateral PFC activation
During the double leg stance, four participants showed bilateral PFC activation, while during the semi-tandem stance, five participants showed more ipsilesional than contralesional PFC activation (Table 2). Participants 7 and 9 showed bilateral PFC activation during both tasks. Bilateral PFC activation was also evident during the double leg stance for Participant 4, while Participant 5 showed bilateral PFC activation during the semi-tandem stance.
Aim 3: Do Fatigue Levels Have a Relationship with Task-Based Activation?
As shown in Tables 1 and 2, and Figure 3, there was no consistent pattern of activation in relation to fatigue for either task.
Discussion
We sought to determine the potential changes in oxy-Hb and deoxy-Hb levels during common physical therapy standing balance tasks, the asymmetry of hemispheric PFC activation during these tasks, and the potential link between self-perceived fatigue and task-based cortical activation in people post-stroke. PFC activation during semi-tandem stance showed greater amplitude than during double-leg stance, indicating more cortical activation with bilateral PFC activation observed during both tasks. Participants with greater fatigue (higher FSS score) may show more activation in the ipsilesional PFC than the contralesional PFC. Thus, PFC activation may occur when performing more challenging balance postures, potentially indicating compensatory activation, and may be linked with greater fatigue.
Changes in oxy-Hb and Deoxy-Hb During Tasks
Our findings outline a potential increase in ipsilesional and contralesional PFC activation during the semi-tandem stance compared to the double-leg stance (Figure 2). The increase in PFC activation was characterized by an increase in oxy-Hb and the decrease in deoxy-Hb and is in line with other authors (Yap et al., 2017; Yeung et al., 2018). Consistent with previous research, visual inspection of the data shows that a more challenging balance task, like the semi-tandem stance, may evoke a higher level of PFC activation than a double-leg stance (Csipo et al., 2021; Moriarty et al., 2019). The increase in PFC activation may indicate that the PFC plays a role in the execution of more complex physical therapy balance tasks after stroke, potentially through an increase in cognitive control (Chen et al., 2022).
Similarities in activation patterns were found, however, variation existed. This variability may be due to several factors, including the location and severity of the lesion, individual differences in recovery potential, age, and pre-stroke functional levels (Goh & Stewart, 2019; Teo et al., 2018). Although statistics were not calculated due to the low sample size, Figure 2 indicates the potential for changes in oxy-Hb and deoxy-Hb between tasks which highlight the importance of considering task difficulty during physical therapy assessments and treatments post-stroke and the impact of task difficulty on PFC activation levels. Owing to the challenges associated with collecting participant data in hospital during the COVID-19 pandemic, the sample size remains small; yet, the research questions regarding PFC activation levels and fatigue are an important and understudied area of investigation.
Asymmetry of PFC Activation During Tasks
The average LI during the double leg stance indicated more activation towards the contralesional PFC compared to ipsilesional. However, this average may be skewed towards the contralesional PFC activation as the LI value for Participant 5 was larger than the other participants (LI for DL = -4.368, Table 2). For Participant 5, the left hemisphere was affected after stroke; thus, the negative LI value during the double leg stance indicated increased contralesional (right PFC for this participant) activation. Healthy young adults often display increased right PFC activation, likely due to a predominance of this brain area during challenging balance tasks (Teo et al., 2018). Since the PFC is also responsible for inhibition of unwanted movements, healthy young adults may require a greater involvement of the right PFC during balance to inhibit unwanted movements (Munakata et al., 2011; Teo et al., 2018). Similar to young healthy adults, younger patients’ post-stroke may display a dominance of the right PFC activation during balance. Considering that Participant 5 was relatively young (age 50 years, Table 1), their age could contribute to a larger LI score compared to the other participants.
Conversely, the average LI during the semi-tandem stance showed more bilateral PFC activation. The average LI value for the semi-tandem stance may also be influenced by the LI value for Participant 6 (−1.976, Table 2) which was also larger than the other participants. The varied LI values suggest that the extent of PFC contribution for balance may change depending on the task demands and difficulty. Participant 6's right hemisphere was affected by the stroke, and their negative LI value during the semi-tandem stance indicated increased activation in the contralesional hemisphere (left PFC in this case). Interestingly, Participant 6 experienced one of the highest fatigue levels, (FSS score of 4.44, Table 1) which could be associated with possible depression, and/or anxiety (Radman et al., 2012; Snaphaan et al., 2011; Wu et al., 2015). Structural changes like reduced gray matter volume have been observed in the PFC in relation with depressive symptoms (Savitz & Drevets, 2009) and some studies have targeted the left PFC with repetitive transcranial magnetic stimulation to alleviate symptoms of depression in adults (Leggett et al., 2015). This speculation is outside the scope of the current study, however it is plausible that Participant 6 may have been experiencing not only high fatigue levels but also co-occurring depression or anxiety, which could influence the LI values during both tasks. Future studies accounting for factors like depression and anxiety in addition to fatigue may help to further contextualize our findings.
Like aim 1, the variation within tasks may indicate that the activation level in ipsilesional and contralesional PFC might differ depending on the task difficulty and individual characteristics. In several of our participants, the increased ipsilesional PFC activation during the semi-tandem stance may reflect the additional cognitive processes essential for maintaining the more challenging standing balance position. People post-stroke display increased contralesional activation, associated with poorer functional outcomes (Calautti et al., 2007; Grefkes & Fink, 2020). However, challenging tasks like the semi-tandem stance increased ipsilesional activation in the current study, which has been linked to better functional outcomes (Dodd et al., 2017).
Fatigue and Task-Based Activation
Higher fatigue levels, as measured by the FSS, have been associated with worse balance performance on the BBS (Goh & Stewart, 2019), and poor functional outcomes post-stroke (Christensen et al., 2008). Our study shows that participants with higher fatigue levels on the FSS may have displayed more ipsilesional PFC activation related to the semi-tandem stance, indicating a potential link between PFC activation and fatigue during challenging balance tasks. Wolff et al., (2019) monitored changes in PFC activation during a strenuous physical task in people with multiple sclerosis and found increased PFC activation which may reflect fatigue level and increased effort required to manage the accumulating fatigue (Wolff et al., 2019). Our participants also demonstrated greater PFC activation which may be related to fatigue levels. However, due to the exploratory nature of this study which focuses on PFC activation between tasks, hemisphere asymmetry, and fatigue, the sample size is limited. Despite the small sample size, these findings provide insight into compensatory activations linked to fatigue, which to the best of our knowledge, has not previously been explored. Although, we did not capture the full range of fatigue levels possible, a larger sample size could examine these potential factors further.
Limitations
The current study was limited by a small sample size because of low recruitment from the inpatient stroke unit. This was due to the occurrence of several COVID-19 outbreaks during the participant recruitment period that prohibited the research team from interacting with patients on the unit. The fNIRS tool was restricted to the PFC region, excluding any additional brain regions from the measurement. Fatigue can be an everchanging phenomenon, but this study only assessed self-rated fatigue at one-time point. Measures of self-rated fatigue at multiple timepoints may provide insights into how changes in PFC activation may be linked with fluctuating fatigue levels and recovery.
Conclusion
Our study provides preliminary evidence of PFC activation during common physical therapy standing balance tasks in people post-stroke. We observed greater responses in oxy-Hb than deoxy-Hb concentration during the semi-tandem and the double leg stance, with some participants showing bilateral activation. The observed asymmetry in PFC activation between tasks may indicate task-specific activation patterns. The increase in ipsilesional PFC activation during the semi-tandem stance for some participants may suggest a potential involvement of the ipsilesional hemisphere in maintaining challenging balance positions. These observations are the first to be reported in literature and these findings contribute to our understanding of PFC activation patterns during balance control and its relationship with fatigue for people post-stroke.
Footnotes
Acknowledgements
We would like to thank all the participants for generously giving their time and energy.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the St. Joseph's Health Care Foundation through the London Life Stroke Rehabilitation Studentship, the Gray Centre for Mobility and Activity Seed Grant (St. Josephs Health Care London), and the Western Faculty Research Development Fund.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.

