Abstract
Objective
The objectives of this study were to evaluate functional outcomes of cervical motor control, strength and endurance following active transcranial direct current stimulation (tDCS) with exercise therapy (ET) compared to sham tDCS/+ET in patient with cervicogenic headache (CGH),
Design
This was a pilot sham-controlled, participant and assessor blinded, randomized controlled trial.
Methods
Thirty-two participants with CGH were randomized to active tDCS/+ET or sham tDCS/+ET. Participants completed 6-weeks of daily ET concurrently combined with 3 sessions per week of tDCS. Assessments included: craniocervical flexion test (mmHg), cervical isometric strength (N), cervical flexor and extensor endurance (seconds), and range of motion (degrees) pre-treatment, post-treatment, 6-weeks and 12-weeks post treatment. Linear mixed effect models evaluated group-time interactions at each follow-up while accounting for exercise program adherence and sex.
Results
There were significant group-time interactions for activation scores on the craniocervical flexion test from pre-treatment to post-treatment (β = 1.571; 95%CI[0.155, 2.988]; p = 0.030), 6-weeks (β = 1.571; 95%CI[0.155, 2.988]; p = 0.030), and 12-weeks (β = 1.954; 95%CI[0.465, 3.443]; p = 0.011) favoring the active tDCS/+ET group.
Conclusions
Active tDCS/+ET demonstrated significant benefits in deep cervical flexor motor control compared to sham tDCS/+ET that remained improved up to 12 weeks post-treatment. This suggests tDCS/+ET may improve of functional outcomes for patients with CGH.
Introduction
Cervicogenic headaches (CGH) are defined by the international classification of headache disorders 3rd edition (ICHD-3) criteria as a secondary headache resulting from dysfunction in the cervical spine or surrounding soft tissue which is often accompanied by neck pain (Sjaastad et al., 1998). Common mechanisms of CGH include mild traumatic brain injury, whiplash injury, osteoarthritis, degenerative disc disease, and musculoskeletal strain (Bogduk & Govind, 2009; Lord et al., 1994; Morin et al., 2016; Page, 2011). In the general population up to 4.4% of individuals meet criteria for CGH which is associated with positive findings on the craniocervical flexion test, cervical range of motion assessments, and pain on palpation of upper cervical vertebrae (Demont et al., 2022; Sjaastad & Bakketeig, 2008).
Symptoms of CGH include headache, neck pain, restricted neck mobility, and dizziness (Al Khalili et al., 2023). These symptoms can cause significant disability for individuals with CGH, as up to 35% fail to return to work 3-months post onset (Manchikanti et al., 2017). Despite the high prevalence and substantial disability burden, there are few evidence-based interventions showing significant improvement in function following CGH. Pharmacotherapy and facet joint interventions can provide temporary relief from pain and headaches but are not beneficial for all patients (Bogduk & Govind, 2009; Gabrhelík et al., 2011; Nagar et al., 2015; Provinciali et al., 1996). Conservative therapies such as manual therapy and exercise therapy (ET) have been shown to reduce headache frequency and intensity as well as neck pain intensity (Jull et al., 2002; Ylinen, Nikander et al., 2010; Ylinen, Takala et al., 2003). However, recent systematic reviews and meta-analyses of exercise, manual therapy, and dry needling primarily found short-term benefits with small effect sizes for headache and pain (Bini et al., 2022; Jung et al., 2024). Given the mixed results from ET and manual therapy, exploring other novel treatments is warranted in individuals with CGH.
Transcranial direct current stimulation (tDCS) is a potential intervention for chronic pain conditions including migraines (Andrade et al., 2017; Rahimi et al., 2020), neuropathic pain (Bonifácio de Assis et al., 2022), chronic post traumatic headaches (Charvet et al., 2023; da Silva Fiorin et al., 2024), and osteoarthritis (Sajadi et al., 2020). Anodal tDCS applies a direct current to areas of the brain involved with pain modulation to increase neuronal excitability thought to result in long-term potentiation leading to sustained pain relief (Kuo et al., 2007; Nitsche et al., 2003; Thirugnanasambandam et al., 2011). A recent systematic review demonstrated that ET following tDCS in chronic pain populations improved pain relief and functional outcomes when compared to ET alone (Cardenas-Rojas et al., 2020). It is hypothesized that tDCS over the primary motor cortex (M1), prior to ET, enhances motor learning and may be responsible for improvements (Cardenas-Rojas et al., 2020; Nitsche et al., 2003; Saucedo Marquez et al., 2013; Stagg et al., 2011). In a previous prospective cohort study, tDCS/+ET demonstrated improvement in deep neck flexor motor control. However, a sham tDCS/+ET group was not included and other measures of cervical function were not considered (Park et al., 2019). As such, research is needed evaluating a wider variety of functional outcomes in CGH patients receiving active tDCS/+ET compared with those receiving sham tDCS/+ET.
Preliminary studies evaluating tDCS/+ET have shown promise for the treatment of pain and function in other chronic pain conditions. We hypothesized tDCS/+ET may also improve function in CGH. The primary aim of this randomized double blind control trial was to evaluate motor control of the deep neck flexors (craniocervical flexion test) in individuals with CGH receiving active tDCS/+ET compared to sham tDCS/+ET. Exploratory outcomes included cervical isometric strength, cervical range of motion, and endurance of the cervical flexors and extensors in individuals with CGH receiving either active tDCS/+ET or sham tDCS/+ET. The numerous functional outcomes being investigated as well as the inclusion of an appropriate comparator group (sham tDCS/+ET) make these aims novel in the CGH patient population.
Methods
This randomized, sham-controlled, double-blind, pilot trial was nested within a parent trial evaluating the safety and feasibility of tDCS/+ET for the treatment of CGH symptoms (Jobin et al., 2025). Participants were recruited from (clinics described here in full version) clinics between January 2023 and February 2024. The methods of this study are described in a pre-print (Jobin et al., 2024). This paper focuses on functional outcomes from multiple physiotherapy assessments repeated post-treatment, 6- and 12-weeks after treatment. The 2010 CONSORT guidelines were used in reporting the findings of this study (Schulz et al., 2010). The study was approved by the (review board described here in full version) (REB22-0890) and was registered on October 4th, 2022 on clinicaltrials.gov (NCT05582616). Informed consent was obtained from all subjects and the rights of participants were protected.
Eligible participants were randomized 1:1 into active tDCS/+ET or sham tDCS/+ET via sealed sequentially numbered envelopes randomly allocated in blocks of ten by a researcher uninvolved in the study. Participants were assigned a number in the order of consent form completion by the researcher administering tDCS, concealment was maintained for the researcher administering tDCS until each participant's first treatment session and for participants until study completion. All participants underwent an initial assessment and 18 sessions of either active or sham tDCS combined with 6-weeks of daily ET. Assessors and care providers were blinded, however, for safety and logistics, the researcher administering tDCS was unblinded.
Participants
Eligible participants were between the ages of 18–65 years, fulfilling the ICHD-3 criteria for chronic CGH (Sjaastad et al., 1998). The ICHD-3 criteria include: clinical/imaging evidence of a cervical disorder or lesion known to be able to cause headache, the presence of two or more characteristics consistent with cervical involvement in headache including a positive diagnostic blockade, and have persisted more than 12 weeks to be considered chronic (Sjaastad et al., 1998; Treede et al., 2019). Cervicogenic headache diagnosis was established by a physiatrist, neurologist, or specialist physiotherapist with experience in treating CGH. Participants also had to have an average numeric pain rating scale (NPRS) score of ≥4/10 in the past week on days when a headache was experienced and a neck disability index score of >14/50 at the time of screening (Young et al., 2019). The neck disability index is a measure of neck functionality, it was only used as a screening tool for this study and a score of >14/50 is indicative of at least moderate disability (Young et al., 2019).
Participants were excluded for having known spinal pathology (tumor, fracture, etc), radicular symptoms or systemic muscular, joint (e.g., inflammatory), or neurological conditions. Furthermore, they were excluded if they had undergone recent surgery (within prior year) or had contraindications to tDCS. Contraindications to tDCS include exposed skin near electrode sites or other skin conditions such as dermatitis, metal or electronic implants, prior surgery on head or spinal cord, history of epilepsy or seizures, history of fainting spells or syncope, pregnancy or chance of pregnancy, and previous electrical stimulation.
Outcome Measures
Pre-treatment assessment included self-reported demographic information (age, sex, employment, education, medical history, medication use) and CGH characteristics (mechanism of onset, time since onset, and history of concussion and whiplash injuries). A physiotherapist blinded to group allocation performed a spine exam at pre-treatment and collected the following outcome measures at pre-treatment, post-treatment, 6-weeks, and 12-weeks post-treatment:
The craniocervical flexion test (CCFT) measured motor control of the cervical flexor muscles. Participants performed a nodding motion for ten seconds with a pressure biofeedback cuff (Chattanooga Group Inc., Hixson, TN, USA) placed under their neck. Activation score was ascertained from the maximum pressure (mmHg) participants were able to exert on the cuff. This test has been validated in healthy controls with an intra-rater reliability of 0.98 (James & Doe, 2010). In patients with chronic neck pain the minimal clinically important difference (MCID) is 2.00 mm-HG (Jørgensen et al., 2017).
The cervical flexor endurance test (CFET) and cervical extensor endurance test (CEET) were used to measure extensor and flexor endurance. Participants lifted their head off a plinth while lying prone (extensors) or supine (flexors) and maintaining craniocervical flexion. Participants were timed to a maximum of 120 s. Stopping points of 120 s were implemented based on safety concerns and normative data from individuals with CGH (Kumbhare et al., 2005; Ludvigsson et al., 2015; Peterson et al., 2018). These tests have been validated with high inter-rater reliability coefficients (κ = 0.68–0.8) in patients experiencing chronic neck pain (Harris et al., 2005; Sebastian et al., 2015). Furthermore, MCID of 19.15 and 73.00 s have been established in patients with chronic neck pain for the CFET and CEET respectively (Jørgensen et al., 2017; Lourenço et al., 2016).
The cervical isometric strength test (CIST) was used to measure the strength of cervical muscles in Newtons (N). A MicroFet 2 handheld dynamometer (Hoggan Scientific, Salt Lake City, UT, USA) was placed on the front, back, and each side of the head and then force was gradually applied (Versteegh et al., 2015). The participant was sitting and unrestrained but asked to maintain a neutral spine position during performance of the test. They were asked to meet the force being applied and the test was stopped when they were no longer able to resist the force being applied. The CIST has demonstrated validity with a high correlation (r = 0.94–0.97) in a test-retest setting for healthy controls and an MCID of 139.25 N when all four directions are added in patients with chronic neck pain (Versteegh et al., 2015).
The cervical range of motion assessment (ROM) was used to measure the range of motion in degrees for movements involving the cervical spine. Participants were asked to move their head as far as possible without pain in six different directions – flexion, extension, right and left side flexion and rotation respectively. This movement was measured using a cervical range of movement device CROM (Performance Attainment Associates, St. Paul, MN, USA) (Youdas et al., 1992). This test has been validated in healthy controls with a high correlation (r = 0.89–0.98) from test-retest and an MCID of 33° when all directions are added (Jørgensen et al., 2017).
Interventions
Transcranial Direct Current Stimulation
Transcranial direct current stimulation was administered using the NeuroConn DC-Stimulator Plus (Neurocare Group, Ilmenau, Germany) for 18 sessions over 6 weeks (3/week) via two 35 cm2 sponges. Stimulation was delivered to the M1 region anodally, and the cathode was placed over the supraorbital region. The M1 region was located through established head measurements (La Rocca et al., 2022; Purves et al., 2001). The M1 region was chosen as the treatment target based on positive results in previous chronic pain literature where tDCS was combined with ET (Chang et al., 2017; Park et al., 2019; Rahimi et al., 2021). The active tDCS/+ET group was provided 2 mA of current for 20 min while the sham tDCS/+ET group was administered current during the 30 s fade in period to a maximum of 2 mA and then was quiescent for 20 min (Brunoni et al., 2014). To determine blinding efficacy, participants were asked to identify their group after all treatment sessions were completed.
Exercise Therapy
Participants received an upper quadrant rehabilitation program, directed at physical impairments, recommended by a specialist physiotherapist with experience in treating CGH (Jull et al., 2002). The program focused on improving endurance and strength in scapulothoracic and neck muscles and was performed at a pain free level. Exercises were performed in the 20 min following tDCS therapy, and at home on days when they were not. Participants were also provided with videos for each of the assigned exercises and asked to self-report adherence.
Statistical Analysis
A sample size of 32 was chosen based on prior recommendations for pilot trials indicating between 24 and 50 participants is best practice (Julious, 2005; Sim & Lewis, 2012). Pre-treatment patient characteristics and assessment scores were reported through descriptive statistics. Wilcoxon rank sum tests were used to compare the active and sham tDCS/+ET groups where applicable. Linear mixed effect models with a group X time interaction adjusting for age and exercise adherence (number of days exercise was completed divided by 42) were used at post-treatment, 6-week, and 12-week follow-ups to determine if there were treatment (active vs. sham tDCS/+ET) by time effects for continuous outcomes using REML with Satterthwaite degrees of freedom. All data were checked for residual plots and homoscedasticity and p-values were determined through t-distributions of the estimates. Changes in outcomes measure were determined for each participant from pre-treatment to each follow-up and identified as meeting MCID for each functional outcome. The aims of collecting such a wide array of measures is to provide a basis for sample size calculations in future studies on this topic.
An a priori alpha level of 0.05 was set. Analysis was conducted with the guidance of a biostatistician (Author J-MG) using Rstudio (v4.3.3; Posit PBC, Boston, MA, USA) Individual de-identified data can be provided by request to qualified investigators.
Results
Participants and Tolerability
Sixty-two participants were screened and thirty-two participants with CGH were enrolled, randomized into either active tDCS/+ET (n = 16) or sham tDCS/+ET (n = 16), and included in the analysis (Figure 1). There were no significant group differences in demographic characteristics or symptom evaluation at pre-treatment (Tables 1 and 2). Pre-treatment information including additional medication usage information for the same population has been described in a separate manuscript (Jobin et al., 2025). Additionally, participants were required not to change treatment, including medications during the trial.
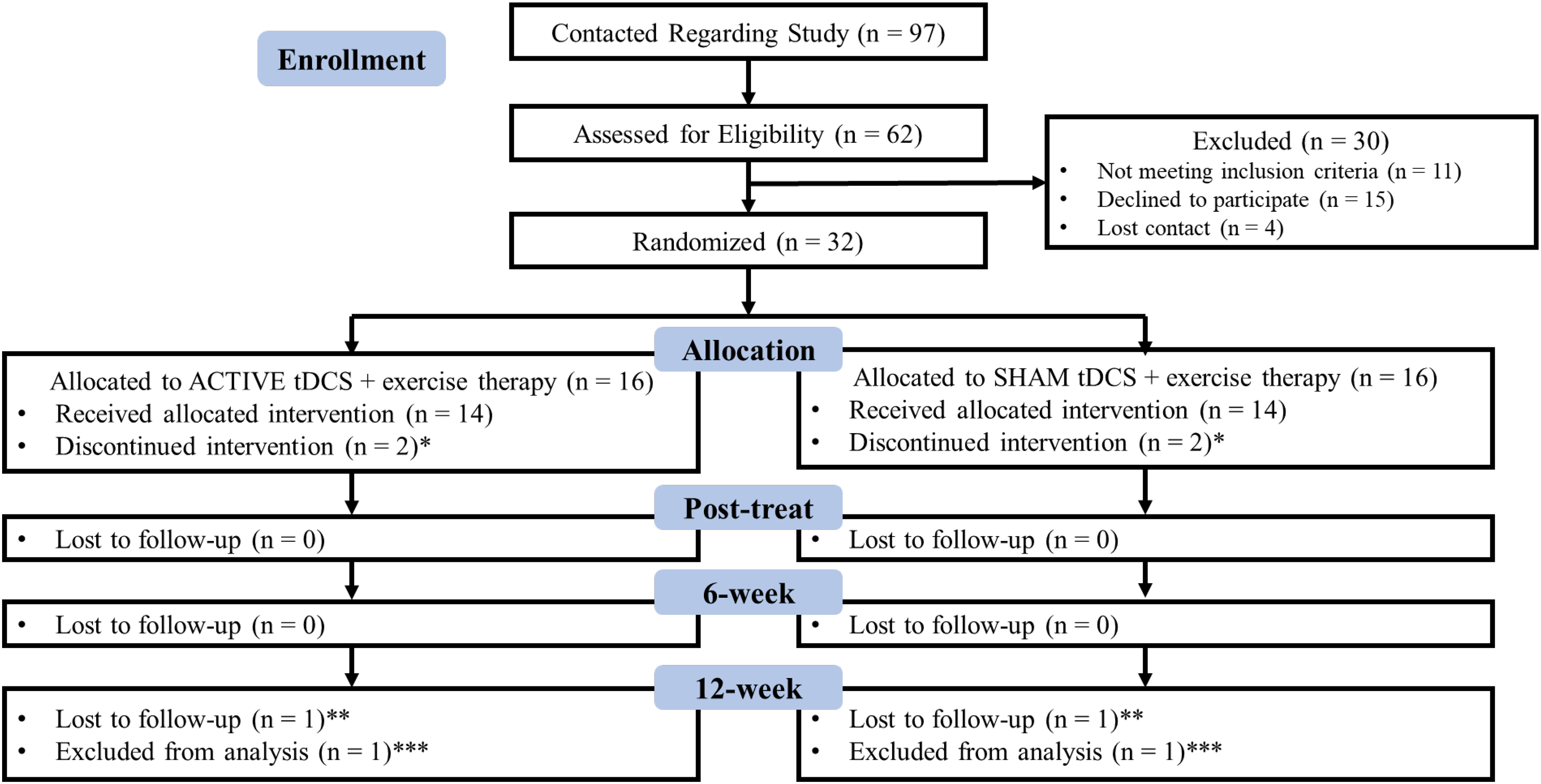
CONSORT diagram of participant flow through the study; *did not finish protocol due to time commitment (n = 2), amount of driving (n = 1), minor adverse event (n = 1); **lost to follow-up due to being on vacation for an extended period (n = 1), unrelated health concerns (n = 1); ***excluded from analysis due to being pregnant (n = 1) and re-aggravation of symptoms (n = 1).
Demographic Information and Headache Characteristics.
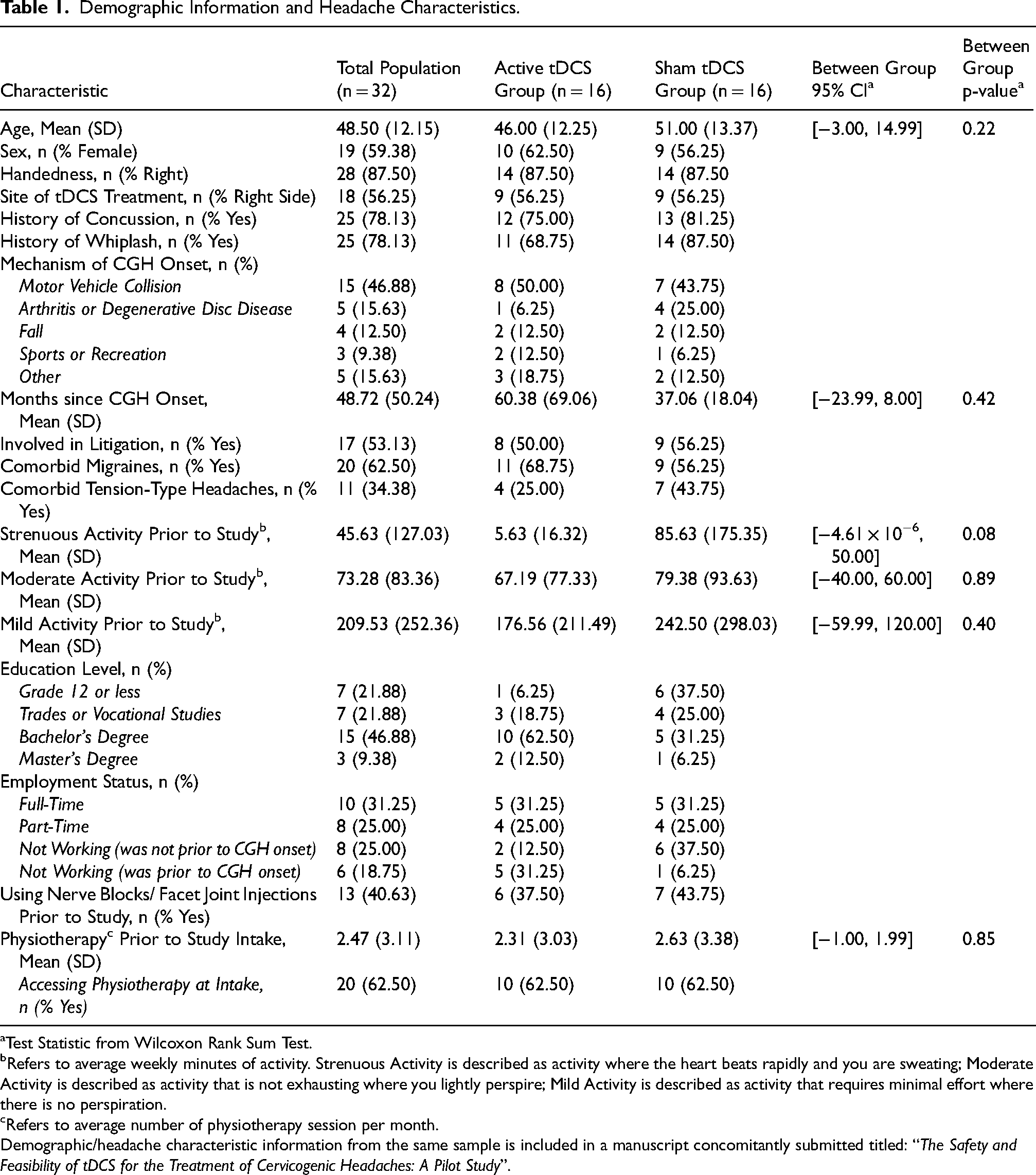
Test Statistic from Wilcoxon Rank Sum Test.
Refers to average weekly minutes of activity. Strenuous Activity is described as activity where the heart beats rapidly and you are sweating; Moderate Activity is described as activity that is not exhausting where you lightly perspire; Mild Activity is described as activity that requires minimal effort where there is no perspiration.
Refers to average number of physiotherapy session per month.
Demographic/headache characteristic information from the same sample is included in a manuscript concomitantly submitted titled: “The Safety and Feasibility of tDCS for the Treatment of Cervicogenic Headaches: A Pilot Study”.
Baseline Clinical Measures.
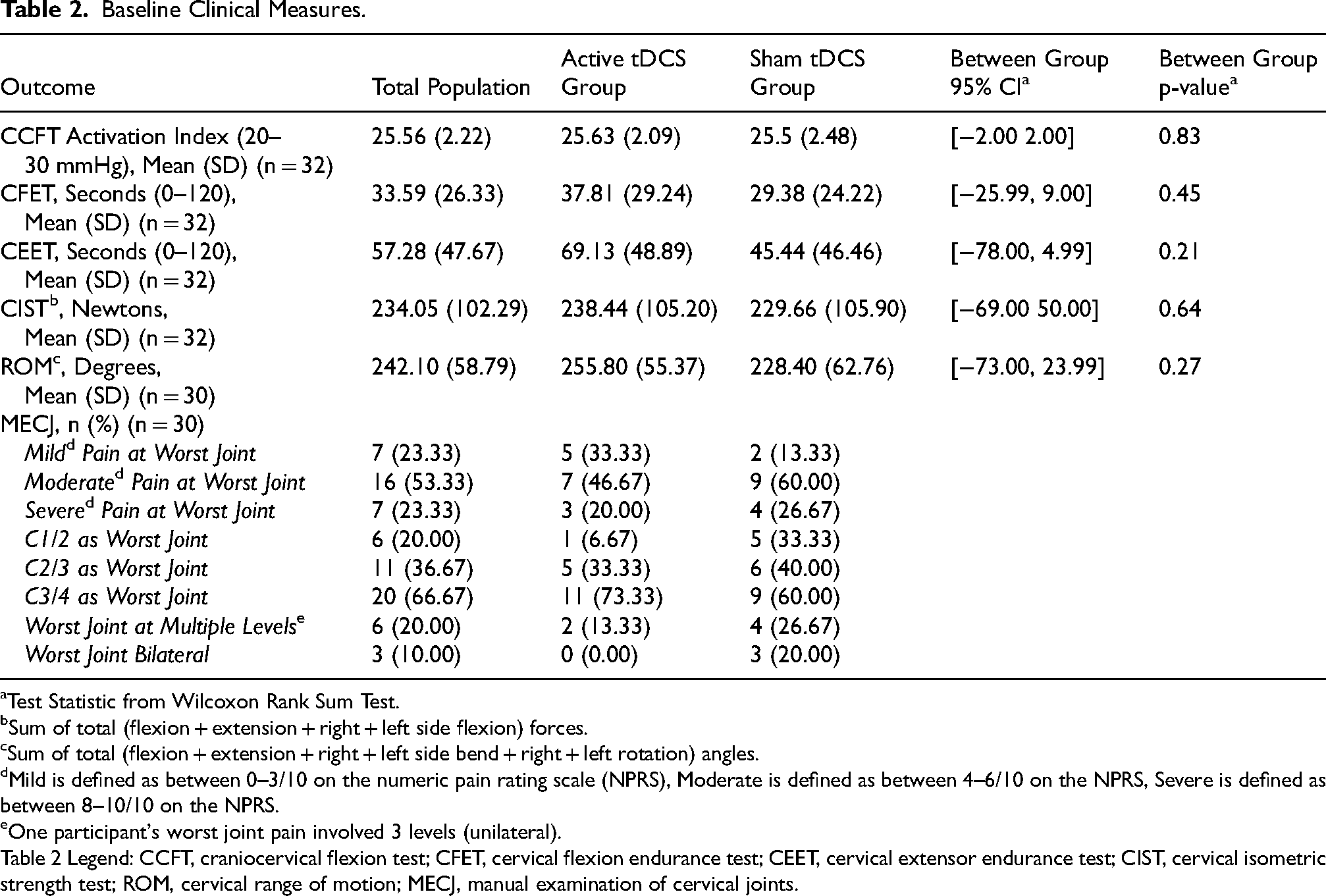
Test Statistic from Wilcoxon Rank Sum Test.
Sum of total (flexion + extension + right + left side flexion) forces.
Sum of total (flexion + extension + right + left side bend + right + left rotation) angles.
Mild is defined as between 0–3/10 on the numeric pain rating scale (NPRS), Moderate is defined as between 4–6/10 on the NPRS, Severe is defined as between 8–10/10 on the NPRS.
One participant's worst joint pain involved 3 levels (unilateral).
Table 2 Legend: CCFT, craniocervical flexion test; CFET, cervical flexion endurance test; CEET, cervical extensor endurance test; CIST, cervical isometric strength test; ROM, cervical range of motion; MECJ, manual examination of cervical joints.
Four participants did not complete the full treatment protocol and four participants did not complete the 12-week follow-up (Figure 1). There were no serious adverse effects from treatment, however, there was one minor adverse effect in the active tDCS/+ET group - a case of contact dermatitis that resolved shortly after tDCS treatment stopped. There was high adherence with 96% of tDCS sessions completed within 6-weeks and the remaining 4% completed within an additional week. Furthermore, during the 6-week treatment period, participants self-reported completing the ET program on an average of 87% of days. Lastly, 39% of participants were able to correctly identify their treatment group when asked to report upon the completion of treatment.
Between Group Differences in Outcomes
There were significant group-time interactions (difference of differences) for activation scores on the CCFT from pre-treatment to post-treatment (β = 1.571; 95%CI[0.155,2.988]; p = 0.030), 6-weeks (β = 1.571; 95%CI[0.155,2.988]; p = 0.030), and 12-weeks (β = 1.954; 95%CI[0.465,3.443]; p = 0.011) favoring the active tDCS/+ET group (Figure 2(a), Table 3). Group-time interactions tested for the CIST, CEET, CFET, and ROM were not significant at any of the follow-ups (Figure 2(b)-(e), Table 3). Furthermore, group-time interactions tested between different post-treatment timepoints were not significant.
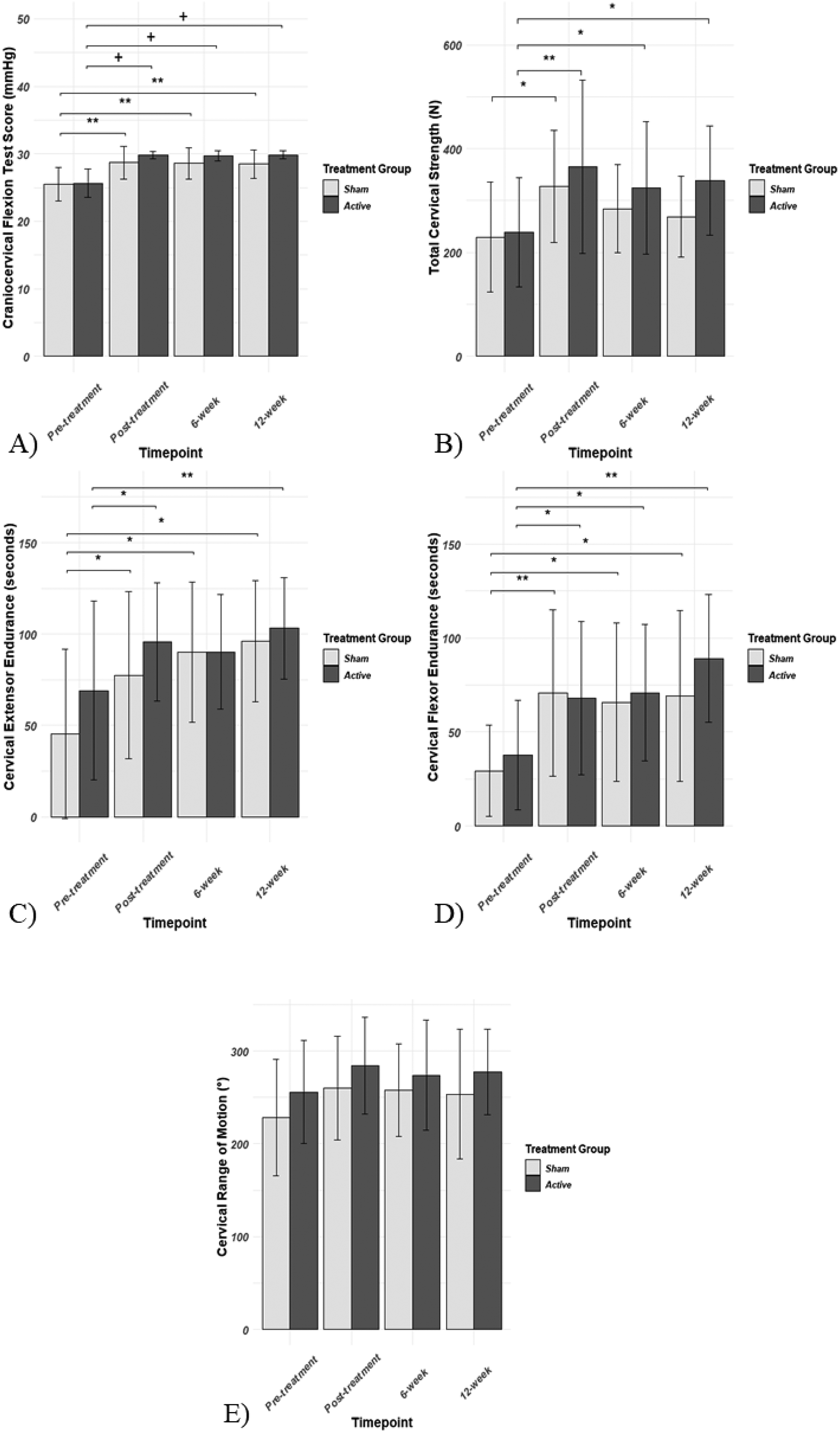
Functional outcome measures over time (a) craniocervical flexion test scores over time with “*” significance bars representing longitudinal improvements only and + significance bars representing both longitudinal improvements and group-time interactions (favored group is indicated) (b) cervical isometric strength test scores over time with significance bars representing differences in each group from pre-treatment to indicated follow-up (c) cervical extensor endurance scores over time with significance bars representing differences in each group from pre-treatment to indicated follow-up (d) cervical flexor endurance scores over time with significance bars representing differences in each group from pre-treatment to indicated follow-up (e) range of motion scores over time
Linear Mixed Effect Model Statistical Outputs for Functional Outcomes.
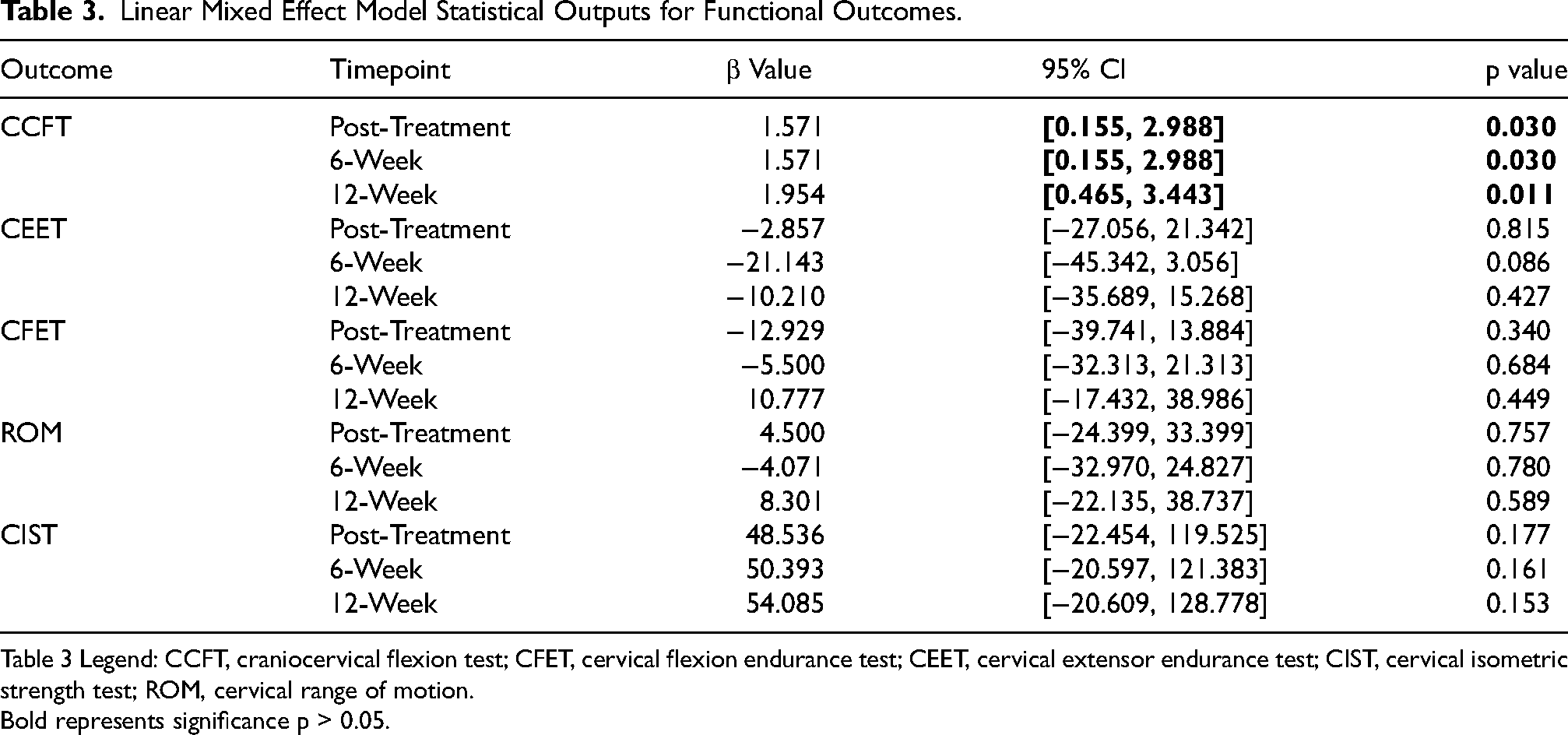
Table 3 Legend: CCFT, craniocervical flexion test; CFET, cervical flexion endurance test; CEET, cervical extensor endurance test; CIST, cervical isometric strength test; ROM, cervical range of motion.
Bold represents significance p > 0.05.
Longitudinal Changes Pre-Treatment to Post-Treatment Time Points
There were significant increases in CCFT activation scores for both the active tDCS/+ET and sham tDCS/+ET groups and for CIST scores in the active tDCS/+ET group from pre-treatment to post-treatment, 6-weeks, and 12-weeks (Figure 2(a) and (b), Table 4). Scores on the CEET increased significantly in the active tDCS/+ET group from pre-treatment to post-treatment and 12-weeks as well as in the sham tDCS/+ET group from pre- to post-treatment, 6-weeks, and 12-weeks (Figure 2(c), Table 4). Furthermore, scores on the CFET increased significantly in both the active tDCS/+ET and sham tDCS/+ET groups from pre-treatment to post-treatment, 6-weeks, and 12-weeks (Figure 2(d), Table 4). There were no significant changes from pre-treatment to any of the follow-ups in either group for ROM (Figure 2(e), Table 4).
Longitudinal Changes in Functional Outcomes.
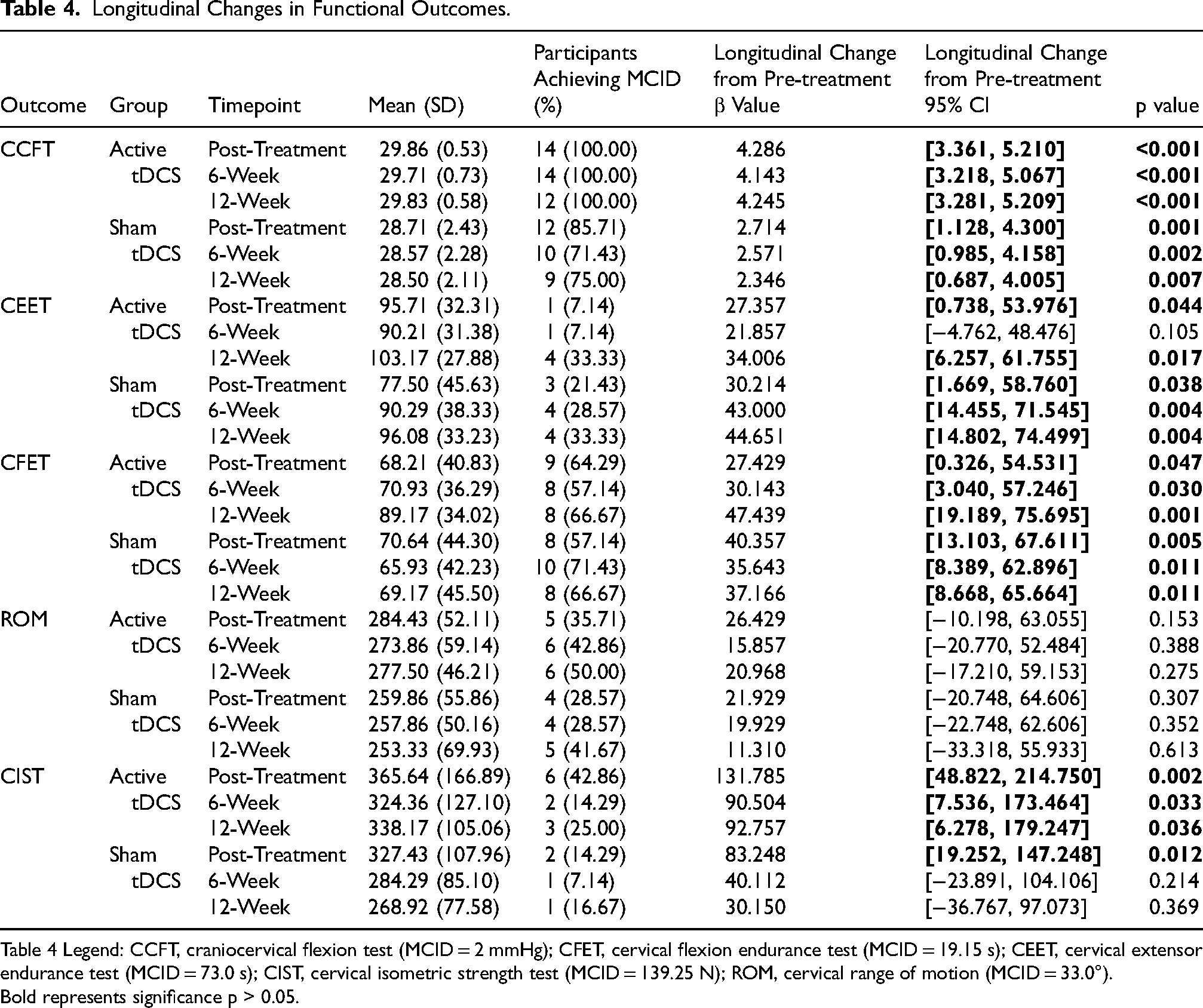
Table 4 Legend: CCFT, craniocervical flexion test (MCID = 2 mmHg); CFET, cervical flexion endurance test (MCID = 19.15 s); CEET, cervical extensor endurance test (MCID = 73.0 s); CIST, cervical isometric strength test (MCID = 139.25 N); ROM, cervical range of motion (MCID = 33.0°).
Bold represents significance p > 0.05.
Minimal Clinically Important Differences
The number of participants achieving MCID was determined from pre-treatment to each follow-up for both groups (Table 4). With respect to the CCFT 100% of participants exceeded the MCID of 2.00 mmHg (Jørgensen et al., 2017) in the active tDCS/+ET group at post-treatment, 6-weeks, and 12-weeks. In the sham tDCS/+ET group 86% of participants exceeded the MCID at post-treatment with 71% at 6-weeks and 75% at 12-weeks on the CCFT. For the CIST, 43% of participants exceeded the MCID of 139.25 N (Versteegh et al., 2015) in the active tDCS/+ET group at post-treatment with 14% at 6-weeks and 25% at 12-weeks. In the sham tDCS/+ET group for the CIST, 14% of participants exceeded the MCID at post-treatment with 7% at 6-weeks and 17% at 12-weeks.
Discussion
This randomized, sham-controlled, double-blind pilot trial evaluated the effects of tDCS/+ET treatment on functional outcomes of deep neck flexor activation, cervical strength, flexor and extensor endurance and range of motion in participants with CGH. A wide array of outcome measures were collected in order to provide insight into sample size calculations for future studies. There was a significant improvement in CCFT score for those receiving active tDCS/+ET treatment compared to those receiving sham tDCS/+ET which was sustained for 12-weeks post-treatment. For the CIST, CFET, and CEET both active and sham tDCS/+ET groups significantly improved up to 12-weeks post-treatment. Though preliminary, the outcomes from this study show promise for tDCS to enhance the benefits of ET to improve motor control in patients with CGH, yet ET alone can also benefit motor control, strength, and endurance but not ROM.
This study demonstrated that the effects of tDCS/+ET on deep neck flexor motor control can be maintained up to 12-weeks post-treatment without any significant increase or decrease following treatment. A previous open label observational study also demonstrated improvements of deep neck flexor motor control following the combination of tDCS/+ET, but lacked a sham tDCS/+ET control group or follow-up assessments (Park et al., 2019). In addition, studies in patients with chronic post-stroke symptoms have shown improvements in motor control in active tDCS/+ET compared to sham tDCS/+ET consistent with our findings (Ilić et al., 2016; Pavlova et al., 2020). However, neither of these studies evaluated motor control outcomes longer than 6-weeks post-treatment and as such, our findings are novel demonstrating a prolonged effect at 12-weeks post-treatment. These observed effects may be due to altered excitability of the M1 region with active tDCS, as the M1 and surrounding regions are primarily responsible for motor learning (Ammann et al., 2016; Dayan & Cohen, 2011; Saucedo Marquez et al., 2013). Furthermore, previous imaging studies in individuals with post-traumatic headache following whiplash have demonstrated changes in the thalamus (Obermann et al., 2009). Given the high degree of connectivity between the M1 region and thalamus (Bosch-Bouju et al., 2013), M1 tDCS may be correcting these structural changes resulting in improved motor control. Our findings suggest tDCS directed to the M1 may provide enhanced motor control when paired with ET, providing support for a larger phase 2 trial.
There were no significant group-time interactions at any follow-up for strength measures, endurance measures or ROM. Contrary to our study, research in patients with knee pain have shown improvements in strength following tDCS/+ET compared to sham/+ET up to 1-month post-treatment(F. Rahimi et al., 2021; Rodrigues et al., 2022) and improvements in isometric cervical strength have been associated with decreased pain and headache intensity (Ylinen, Nikander et al., 2010; Ylinen, Takala et al., 2003). Considering the similarity of our tDCS and ET protocols with these trials, the null result of our trial may reflect a smaller sample size, poor choice of outcome measures or the need for a longer tDCS/+ET protocol (Rahimi et al., 2021; Rodrigues et al., 2022). We may have seen an absence of group differences in CEET and CFET due to a ceiling effect of the chosen protocol. These tests were terminated at 120 s, as supported by previous literature based on safety concerns and normative data from individuals with CGH (Kumbhare et al., 2005; Ludvigsson et al., 2015; Peterson et al., 2018). Most of our participants exceeded the normative values for these measures at pre-treatment, however, it should be noted that 63% of participants were accessing physiotherapy prior to enrollment. For the CEET specifically, 38% and 25% of participants in the active and sham tDCS/+ET groups respectively achieved the maximum score at pre-treatment. Previous research have shown enhanced endurance with tDCS/+ET compared to sham/+ET in healthy controls using resistance training (Vieira et al., 2022; Wang et al., 2022), and improved endurance of the extensors and flexors has been shown to reduce symptoms of pain and headache in individuals with CGH (Ylinen, Nikander et al., 2010; Ylinen, Takala et al., 2003; Zito et al., 2006). These measures should be reconsidered in future studies with adaptations to avoid ceiling effects.
We found participants improved significantly from pre-treatment to most post-treatment follow-ups on the CCFT, extensor and flexor endurance, and strength. Additionally, we found that over 70% of participants and over 50% of participants achieved the MCID for CCFT and CFET scores respectively from pre-treatment to all follow-ups in both groups. This is consistent with previous exercise therapy studies and these improvements have been associated with a reduction in headache frequency and intensity, as well as neck pain intensity (Jull et al., 2002; Ylinen, Nikander et al., 2010; Ylinen, Takala et al., 2003).
There are strengths and limitations to this study. The study design was stringent allowing for a direct comparison of active versus sham tDCS/+ET. Second, adherence to the protocol was high, similar across groups, and controlled for in the analysis. This suggests the treatments and outcome assessments are appropriate and feasible interventions. Finally, the length of follow-up was longer than many other tDCS trials considering functional outcomes, shedding light on long-term outcomes. This trial was limited in several ways. First, the study was most likely underpowered, increasing the risk of type II error, however this was a pilot trial with a primary focus of evaluating safety and feasibility. Second, CFET and CEET outcome measures had ceiling effects. Third, adherence to ET was self-reported potentially leading to over-reporting. Finally, there was selection bias present as participants were recruited from specialized outpatient chronic pain clinics which may not be representative of all individuals with CGH. Future trials evaluating tDCS/+ET in individuals with CGH should be conducted with larger sample sizes and using measures without ceiling effect.
Conclusion
We showed active tDCS/+ET directed at the ipsilateral M1 was superior to sham tDCS/+ET over a 6-week period for improving CCFT score, suggesting tDCS has potential benefit in enhancing motor control. Improvements in CCFT score following active tDCS/+ET were sustained to 12-weeks post-treatment, showing promise in providing long term benefits. In both active and sham tDCS/+ET significant improvement was seen in strength and muscle endurance but not in ROM suggesting ET may be a beneficial stand-alone treatment in individuals with CGH. This study provides evidence to proceed with a phase 2 trial with a larger sample size and improved functional assessment tools for better understanding of the utility of tDCS/+ET in individuals with CGH.
Paper Highlights
This pilot study evaluated functional outcomes between individuals with cervicogenic headaches who received active transcranial direct current stimulation with exercise therapy and sham transcranial direct current stimulation with exercise therapy. We found that those receiving active transcranial direct current stimulation with exercise therapy improved significantly over those receiving sham transcranial direct current stimulation with exercise therapy on measures assessing the function of the deep cervical flexor muscles. No other functional outcome measures were significantly improved by the combination of active transcranial direct current stimulation with exercise therapy compared to sham transcranial direct current stimulation with exercise therapy, however, given the pilot nature of our study, future studies are needed on this topic.
Supplemental Material
sj-docx-1-nre-10.1177_10538135251325384 - Supplemental material for The Treatment of Cervicogenic Headache with Transcranial Direct Current Stimulation and Exercise Therapy: A Randomized Control Trial Evaluating Functional Outcomes
Supplemental material, sj-docx-1-nre-10.1177_10538135251325384 for The Treatment of Cervicogenic Headache with Transcranial Direct Current Stimulation and Exercise Therapy: A Randomized Control Trial Evaluating Functional Outcomes by Kaiden Jobin, Ashley Smith, Christina Campbell, Siobhan Schabrun, Jean-Michel Galarneau, Kathryn J Schneider and Chantel T Debert in NeuroRehabilitation
Footnotes
Abbreviations
ORCID iDs
Ethics Board Approval
University of Calgary Ethics Review Board (REB-0890).
Author Contribution Statement
KJ, AS, CC, SS, KJS, and CTD all contributed to the conceptualization of the study and design/ methodological choices. KJ, AS, and CC all contributed to collection of the data and study logistics. KJ and J-MG both contributed to statistical analysis for the study. KJ drafted the initial manuscript with all other authors providing edits and approving the final manuscript for submission. CD acquired funding for the project and takes responsibility for the integrity of the study. All authors have read and agreed to the submitted version of the manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Hotchkiss Brain Institute at the University of Calgary (Calgary, Alberta, Canada), by a Canadian Institute of Health Research Master's Studentship Award, and by the Department of Clinical Neurosciences at the University of Calgary (Calgary, Alberta, Canada). These institutions had no role in directing or guiding the research project.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability
Data are available from the corresponding author upon reasonable request. The methodology is published as a pre-print available at: (given in full unblinded version).
Data Sharing Statement
De-identified data regarding assessment scores is available upon request by qualified investigators. The corresponding author should be contacted regarding requests for data. Data may be reused for the purposes of sample size calculation or in systematic reviews/meta-analyses.
Trial Registration
Clinicaltrials.gov (NCT05582616), please note that additional outcomes are listed for safety, feasibility, headache outcomes, and pain outcomes. These are not included in the current manuscript as they are being published in a separate manuscript.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
