Abstract
Objective
This study evaluated the validity of the HALO™ digital goniometer compared to the Cervical Range of Motion (CROM™) device for assessing cervical range of motion (ROM) in a sample of 20 patients with cervicogenic headache (CGH).
Methods
ROM was measured simultaneously using both devices across six movement planes at two time points, at baseline and 4 weeks later. Intraclass correlation coefficients (ICCs) were used to assess agreement, and Bland-Altman analyses evaluated measurement bias.
Results
Results showed excellent to good agreement between the HALO™ and CROM™ for most movements (ICC = 0.880 to 0.994), with flexion, extension, and left rotation demonstrating excellent reliability (ICC > 0.9). Left side flexion and right rotation showed good agreement (ICC = 0.888 and 0.880, respectively), while right side flexion had lower (moderate) agreement (ICC = 0.688, 95% CI [−0.073 to 0.902]). Bland-Altman analysis indicated that the HALO™ tended to slightly overestimate ROM in several directions, with statistically significant yet clinically negligible (<5° difference) biases for flexion, side flexion, and right rotation, indicating that the clinical interchangeability between the two devices may be limited for these movements.
Conclusion
The HALO™ demonstrated good validity compared to the CROM™ for measuring cervical ROM in individuals with CGH. Both its portability and ease of use make it a viable alternative. However, variability observed in side flexion and the modest sample size suggest that caution is warranted, and further validation in larger and more diverse populations is needed before it can be considered interchangeable with the CROM™.
Introduction
The prevalence of cervicogenic headaches (CGH) is estimated to affect between 0.17% and 4.1% of the general population1,2 and up to 20% among individuals suffering from chronic headaches. 1 Persons with CGH are often referred to physiotherapy, as this condition can cause significant physical limitations and emotional burden.3,4 The differential diagnosis of CGH includes the presence of cervical dysfunction, particularly originating from the craniocervical region, acting as a source of pain and headache.5,6 CGHs are defined as referred pain caused by irritation of cervical structures innervated by the C1, C2, and C3 spinal nerves, which converges to the trigeminal nucleus located in the brainstem.7,8 Consequently, any structure innervated by the C1-C3 spinal nerves may serve as a source of CGH.7–9 One objective finding supporting CGH diagnosis is restricted range of motion (ROM) in the upper region of the cervical spine.5,10 To identify this key characteristic, clinicians need to have appropriate and reliable assessment tools, such as the Cervical Range of Motion measurement device (CROM™).
The CROM™ tool, used in both research and clinical settings, is considered the gold standard for cervical ROM measurement, 11 as it has been widely validated in numerous studies, both in healthy individuals and those suffering from neck pain. 12 The CROMTM is easy to use to assess active movements, but presents some practical limitations when assessing passive ROM. The device is relatively bulky, requires the adequate placement of a magnet on the anterior and posterior upper thorax of the participant to measure rotations and can hinder certain procedures particularly those requiring precise manual control of the head, such as the Cervical Flexion-Rotation Test (CFRT). 13 The CFRT, which was developed to determine the presence of dysfunction at the C1-C2 segment, 14 where approximately 50% of cervical rotation occurs, 15 has high diagnostic accuracy for CGH.13,15,16 It involves passively flexing the neck to end range and then rotating it to each side while the patient is supine, aiming to isolate movement to the upper cervical spine. 14 Performing the CFRT with the CROM™ can be challenging due to its size and rigidity, and previous reliability studies have used a modified CROM™ for this reason. 13
With the advancement of digital tools, digital goniometers such as the HALO™ goniometer have emerged. The HALO™ goniometer is a lightweight, pocket-sized digital ROM assessment tool offering more versatility and maneuverability than the CROM™ to perform special tests such as the CFRT, and displays measurements digitally, with the option to store them internally — potentially reducing recall bias. 13 However, the HALO™ goniometer has not been studied as extensively as the CROM™, and its validity for measuring cervical ROM, especially in symptomatic populations, remains uncertain. In fact, to our knowledge, there is no study comparing measurements obtained with the HALOTM digital goniometer to those obtained with the CROM™ device.
Therefore, the aim of this study was to establish the validity of the measurements of cervical spine motion using the HALOTM device compared to the gold standard (CROM™ device) in a population of patients suffering of CGH, with measurements taken across six movement planes.
Methodology
Design
This is a descriptive, cross-sectional, correlational study comparing cervical ROM measurements obtained by the HALO™ device to those concomitantly acquired by the CROM™ device (gold standard) in patients with CGH. This study is a post-hoc analysis of data acquired during a prospective quasi-experimental design (pre-post without control group comparison), during which manual therapy and exercise interventions were provided to patients consulting in physiotherapy for CGH. Recruitment procedures, interventions and results of this primary study are detailed in Paquin et al. 2021. 17
Participants
Participants’ enrollment procedure is illustrated in Figure 1. Patients with CGH were recruited from advertisements and local physician referrals around the area of Drummondville (Quebec, Canada) from January to May 2018. Potential participants were first screened through phone interview. Thereafter, patients underwent a physical examination to confirm their eligibility. Inclusion criteria were: 1) age between 18 to 65 years old, 2) headache (HA) in temporal relation to the onset of the cervical disorder or appearance of the lesion, 3) HA aggravated or provoked by neck movements or postures, 4) HA associated with neck, shoulder and/or upper limb pain, 5) HA frequency ≥1 per week for ≥1 month, 5) average pain intensity ≥3/10 on the visual analog scale and 6) pain on palpation of paravertebral tissue of the cervical spine. Exclusion criteria were: 1) history of neck surgery, 2) reported diagnosis of another type of HA that causes two or more episodes per month (e.g. migraine), 3) currently receiving another form of physical intervention, 4) other chronic pain syndrome (i.e. fibromyalgia, systematic inflammatory disease), 5) inability to tolerate the CFRT, the SNAG mobilization, or the self-SNAG technique, and 6) presence of contraindications to cervical manual therapy. The initial assessment, including the physical examination, was performed by a single registered physical therapist. 17
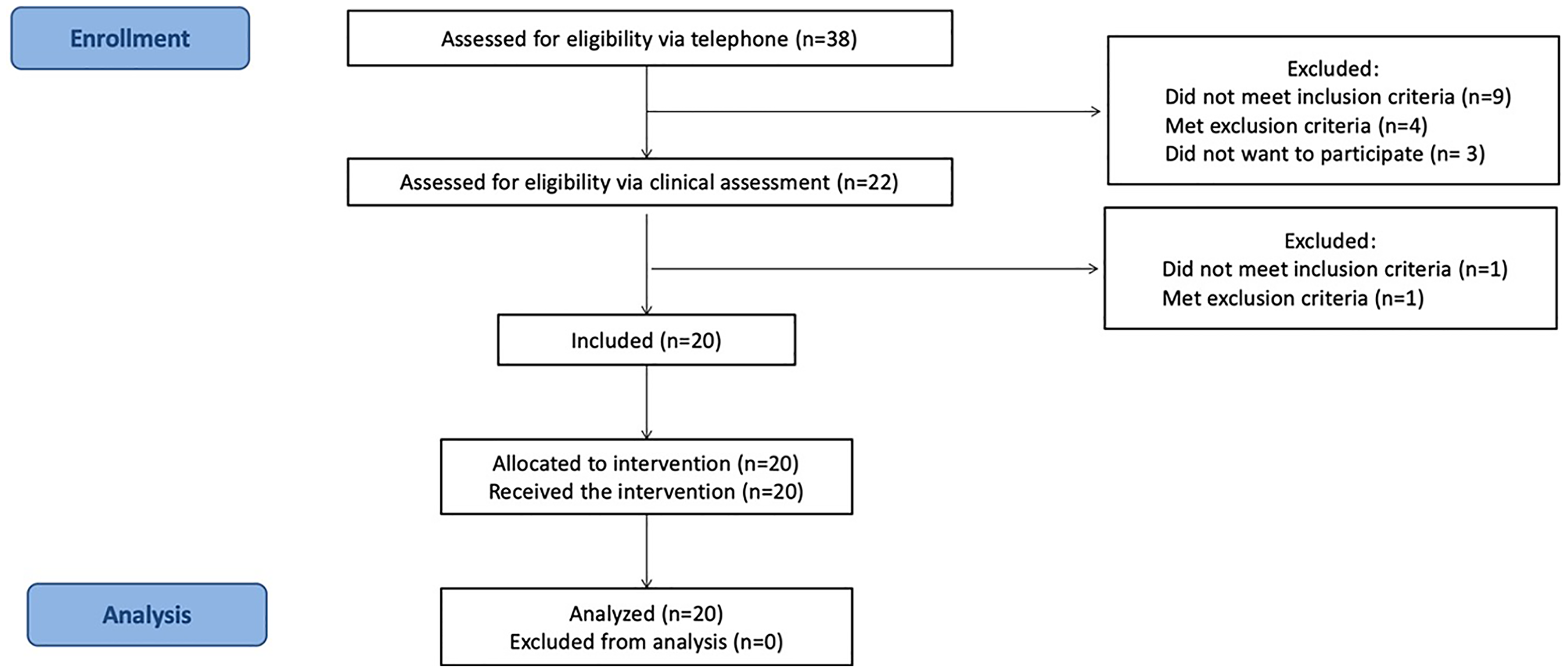
Participants’ enrollment flowchart.
Informed consent was obtained for all participants according to The Helsinki Accords. The study was approved by the Ethics Review Board of the Clinical Research Center of the Centre intégré universitaire de santé et des services sociaux de l’Estrie – Centre Hospitalier Universitaire de Sherbrooke (reference number: 2018-2642).
Instruments
The HALO™ goniometer (model HG1, HALO Medical Devices, Sydney, Australia): 18 This device is a digital ROM assessment tool that employs a laser-guided inclinometer system in place of the traditional needle of an inclinometer. The built-in laser of the HALO™ provides guidance for the therapist to align anatomical landmarks as the lasers intersect with the joint axis. This device can be used to measure movement in all three planes, as it uses inertial sensors to measure joint angles and movement, with built-in algorithms that account for gravitational effects. It can be used manually by the assessor or can be fixed directly to an anatomical region using a strap. It can also be used simultaneously with other measurement tools. The accuracy of the HALO™ was studied using a robotic arm and showed a margin of error of 1 degree. 19 The reliability and validity of this electronic device have been studied with various asymptomatic populations and on various anatomical locations, including the cervical and lumbar spine, the shoulder, the wrist, the hip, the knee and the ankle.20–24 Intra-rater reliability intraclass correlation coefficients (ICCs) have been found to be between 0.82–0.99 for shoulder ROM,20,21 0.994 for knee ROM 22 and between 0.5 to 0.82 for ankle ROM. 23 A recent study has also shown that the HALO™ has fair to good reliability and validity for cervical spine ROM measurement (intra-rater reliability ICC = 0.51–0.69) when compared to the universal goniometer (intra-rater reliability ICC = 0.49–0.58) in a healthy population, with higher reliability for the measurement of cervical flexion and extension. 24
The Cervical Range of Motion device (CROM™): This device uses magnets placed on the anterior and posterior upper thorax as well as three inclinometers to provide measurements for three independent planes of movement. The CROMTM is placed on the patient's head and its stability is ensured by its frame, nose piece and straps. A considerable number of studies investigated the psychometric properties of the CROMTM device and showed good reliability and validity for active neck movements.11,12 Studies have shown similar intraclass correlation coefficients, varying between 0.73 and 0.95, and similar inter-rater reliabilities ranging from 0.73 to 0.92.25–27 Criterion validity, where the CROMTM performance was compared to the gold standard, was found to be excellent for flexion (r = 0.97) and extension (r = 0.98), 28 very good for right (r = 0.84) and left (r = 0.82) lateral flexion, 29 and good to excellent for right (r = 0.89) and left (r = 0.94) rotation. 30 Its test-retest reliability has been rated good to excellent, with an ICC ranging between 0.89 and 0.98. 31 This device is also widely used in clinical practice to assess ROM in patients presenting neck disorders, including CGH. 32
Data collection procedures
Evaluator
The evaluator (co-author) was a physical therapist with expertise in advanced manual therapy (fellow of the Canadian academy of musculoskeletal therapy, FCAMPT) and a full-time clinician with 8 years of clinical experience treating musculoskeletal disorders.
Positioning of instruments
The HALOTM goniometer was first positioned on the participant's head using the original VelcroTM strap included with the device. The strap was modified prior to the experimentation to add an additional VelcroTM to position the device on the center of the top of the head. The strap was comfortably positioned on the head, at a height allowing for adequate placement of the CROMTM device above the strap. Afterwards, the CROMTM device was positioned on the participant's head according to the original instructions to help minimize error and get more accurate results. The three compasses and magnets were in place for all measurements. To facilitate reading of the results from both instruments, the HALOTM goniometer was positioned parallel and as close as possible to the compass for every movement: for flexion and extension, the goniometer was positioned above the external acoustic meatus (Figure 2); for side flexion, the goniometer was positioned on the forehead (Figure 3); and for rotation, the goniometer was placed at the center of the superior cranium (Figure 4).
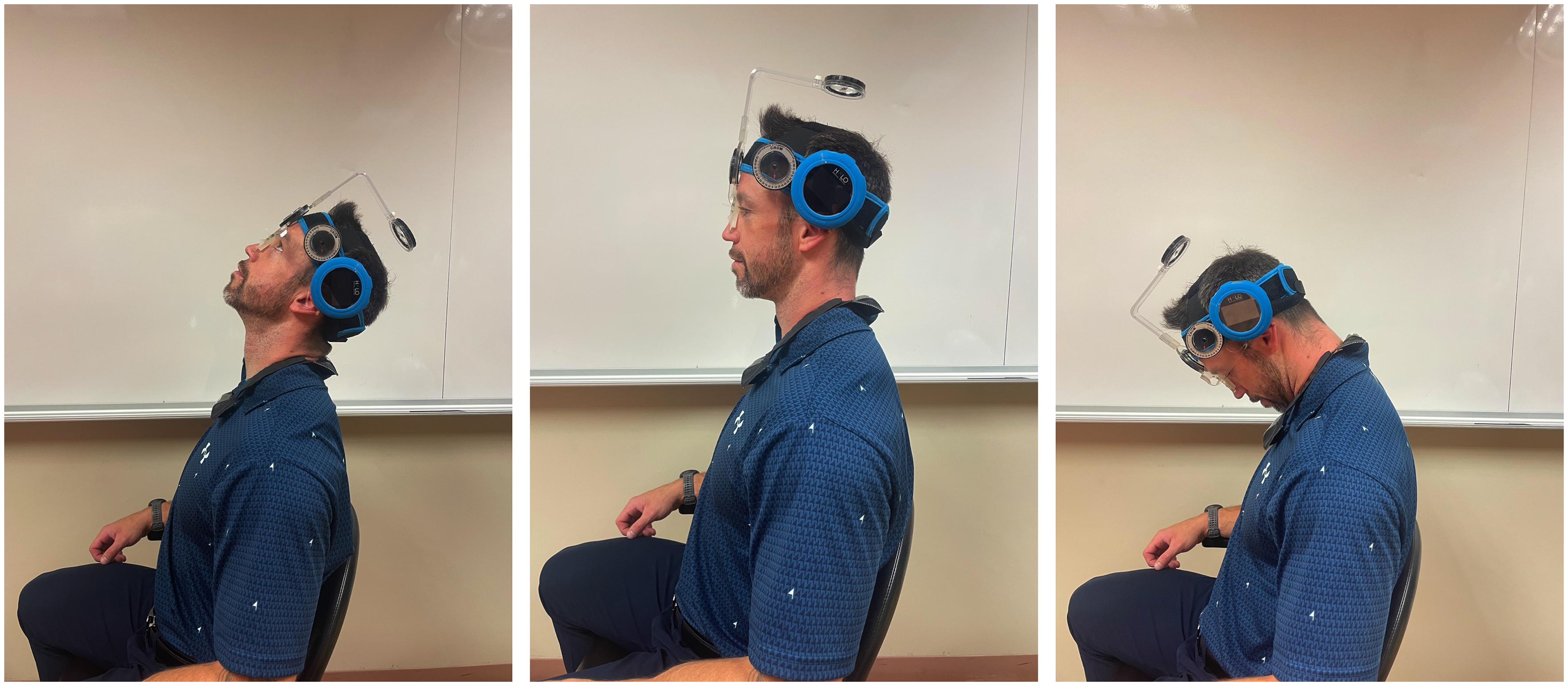
Basic positioning of the HALOTM goniometer and CROMTM during flexion and extension movements.

Basic positioning of the HALOTM goniometer and CROMTM during side flexion.

Basic positioning of the HALOTM goniometer and CROMTM during rotation.
The fact that both measures were taken concurrently reduced a potential measurement bias, allowing the evaluator to read results simultaneously on both instruments. It also allowed simultaneous calibration of the two instruments before taking measurements so that both instruments showed “0 degrees” before the patient was instructed to move.
Procedures and positioning of the participants
All measurements were taken with the participants sitting on the same chair (without armrests), positioned in the same location in the same room. Starting with both feet flat on the floor and back supported on the backrest, each participant had to actively perform one single repetition through their full available amplitude for every direction, in the same order. Measures were taken in the following order: Flexion (FLEX), extension (EXT), right side flexion (RSF), left side flexion (LSF), right rotation (RR), left rotation (LR). All measurements were taken by the same evaluator. The evaluator gave the following verbal indications to the participant:
FLEX: Lean your neck forward to look at the ground as far as you can without losing contact with the backrest EXT: Lean your neck backward to look at the ceiling as far as you can without losing contact with the backrest RSF: Keep looking forward and lean your neck to the right to touch your ear to your shoulder without elevating your shoulder LSF: Keep looking forward and lean your neck to the left to touch your ear to your shoulder without elevating your shoulder RR: Turn your neck to the right to look as far as you can without turning your shoulders and without losing contact with the backrest LR: Turn your neck to the left to look as far as you can without turning your shoulders and without losing contact with the backrest
To minimize measurement errors, the evaluator asked the patient to maintain the end-range position for a few seconds to read the measures on both instruments, waiting for the compass needle to stop moving and the number on the goniometer to be stable. For all movements, the evaluator identified and corrected compensations by visually monitoring the participant's shoulders and thorax to ensure minimal to no movement in the thoracic spine. In case of inadequate movement occurring lower in the spine, the evaluator would ask the patient to come back to neutral position and repeat the instructions for a second attempt.
Data analysis
Cervical ROM measurements for all planes of movement were taken at two different points in time, namely at the initial assessment and four weeks later, prior to any intervention at each time point, allowing us to get two sets of independent measurements for statistical analysis. Although the two measurement sets came from the same participants, they were considered independent for the purposes of agreement analysis because each session represented a separate, complete device comparison performed under identical conditions. The physiotherapy intervention provided between the two sessions was not expected to differentially affect one device over the other. This approach doubled the number of measurement pairs, improving the precision of ICC and Bland–Altman estimates, while maintaining consistency of evaluator, environment, and protocol.
To quantify the degree to which the measurements of the two instruments were related, ICC were calculated using a two-way random effect, absolute agreement, single rater model. According to Koo and Li (2016) this statistical model is appropriate for studies that assess “rater-based clinical assessment methods that are designed for routine clinical use by any clinicians”. 33 Single measures ICC values were used for analysis to reflect the reliability of a single measurement for a single rater. The interpretation of the ICC values was based on the following parameters: 1) <0.5: poor, 2) between 0.5 and 0.75: moderate, 3) between 0.75 and 0.9: good, and 4) >0.9: excellent. 33 While the ICC value provides information about how two instruments may be correlated with regards to the type of measurement, it does not provide information about agreement between the two instruments. 34 Therefore, mean level of agreement and 95% confidence interval (CI) between the CROM™ and the HALO™ active cervical ROM values were computed using the Bland-Altman analysis.
Results
Participants
Twenty patients, 18 females and 2 males, presenting with CGH, consented to participate in the study. Their mean age was 35.75 ± 11.48 years and they were all employed at the time of the study. Table 1 presents the sociodemographic characteristics of the participants.
Demographic characteristics of the participants.
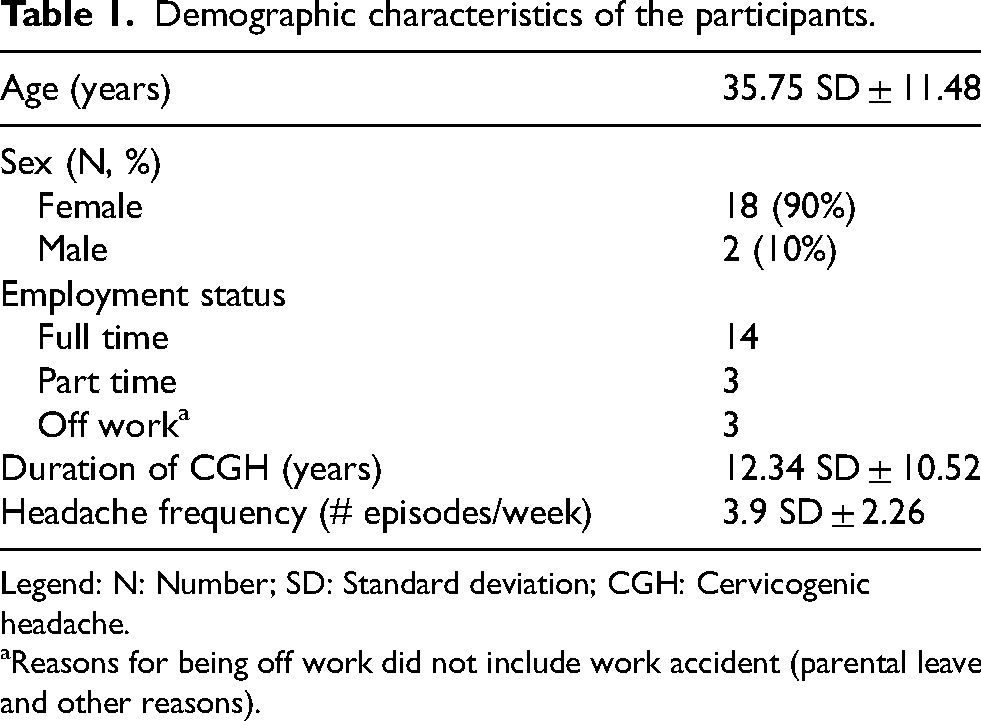
Legend: N: Number; SD: Standard deviation; CGH: Cervicogenic headache.
Reasons for being off work did not include work accident (parental leave and other reasons).
Active cervical ROM with HALOTM digital goniometer vs CROMTM device
Because measures were taken twice and at two different points in time (before and after receiving physiotherapy treatment), it was therefore possible to consider that the two sets of measures were independent, bringing our sample size to 40 measures for each plane of movement. The means and standard deviations for cervical ROM measures taken with the HALO™ and CROM™ device for all six planes of movement are detailed in Table 2.
Cervical ROM measured with CROM™ vs HALO™ (mean ± SD).
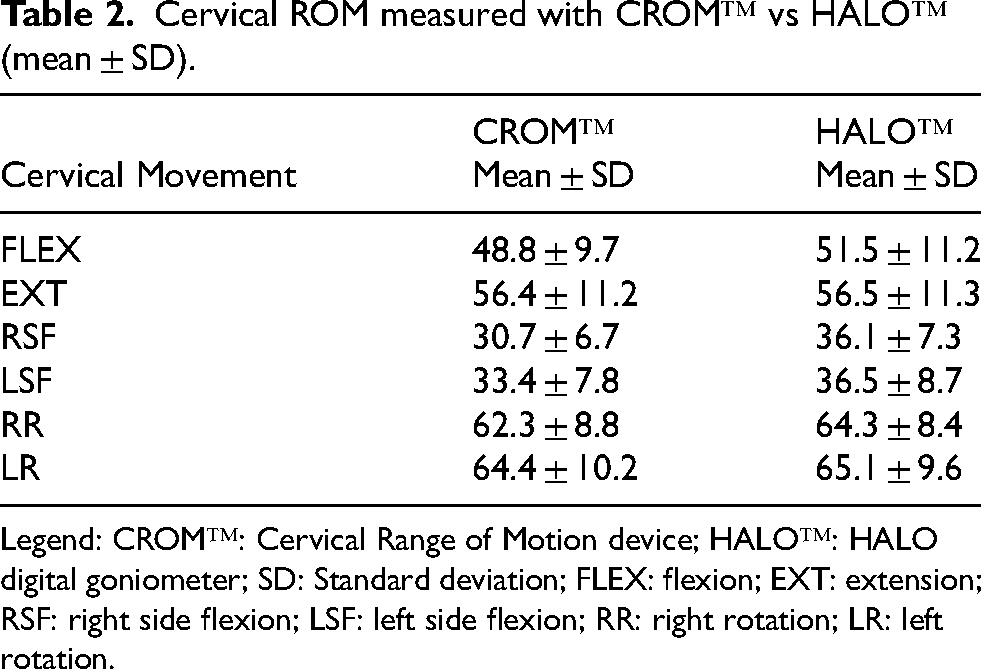
Legend: CROM™: Cervical Range of Motion device; HALO™: HALO digital goniometer; SD: Standard deviation; FLEX: flexion; EXT: extension; RSF: right side flexion; LSF: left side flexion; RR: right rotation; LR: left rotation.
ICC values for absolute agreement between the HALO™ digital goniometer and the CROM™ device are presented in Table 3. Concurrent validity was found excellent for FLEX (0.918, 95%CI[0.699–0.968]), EXT (0.994, 95%CI[0.989–0.997]) and LR (0.968, 95%CI[0.939–0.983]), good for RR (0.880, 95%CI[0.735–0.941]) and LSF (0.888, 95%CI[0.234–0.966]), and moderate for RSF (0.688, 95%CI[−0.073–0.902]).
Intraclass correlation coefficients for absolute agreement between HALO™ and CROM™.
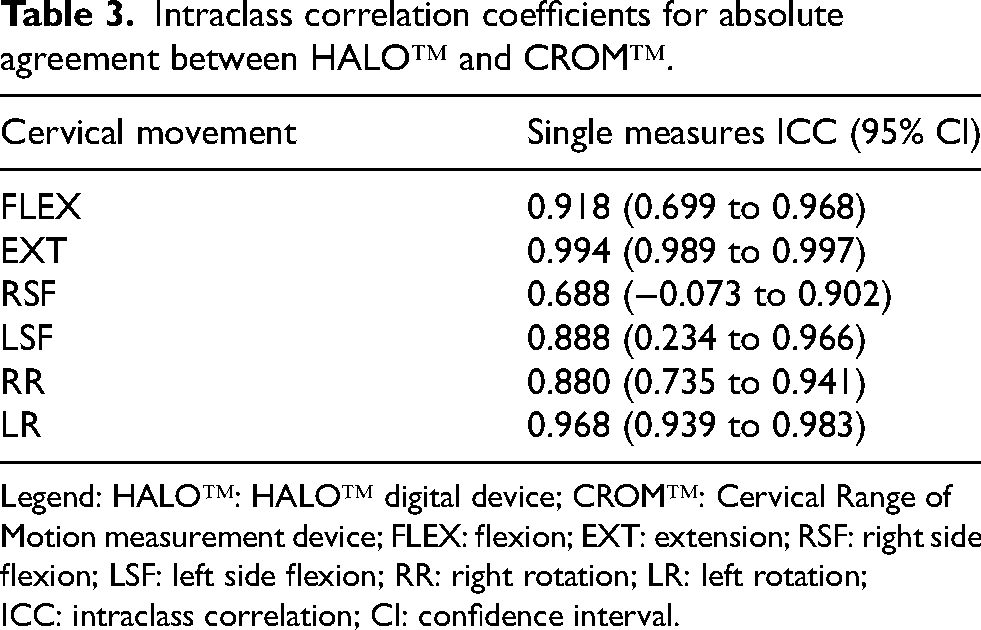
Legend: HALO™: HALO™ digital device; CROM™: Cervical Range of Motion measurement device; FLEX: flexion; EXT: extension; RSF: right side flexion; LSF: left side flexion; RR: right rotation; LR: left rotation; ICC: intraclass correlation; CI: confidence interval.
Means and mean differences between both instruments were compared using the Bland-Altman analysis and are presented in Table 4, as well as illustrated in a plot (Figure 5) for each one of the movement planes of the cervical spine. The differences were calculated considering the CROM™ as the reference value (Halo™ minus CROM™). Thus, a positive score indicates that the HALO™ overestimated compared to the CROM™, whereas a negative score indicates that the HALO™ underestimated compared to the CROM™.
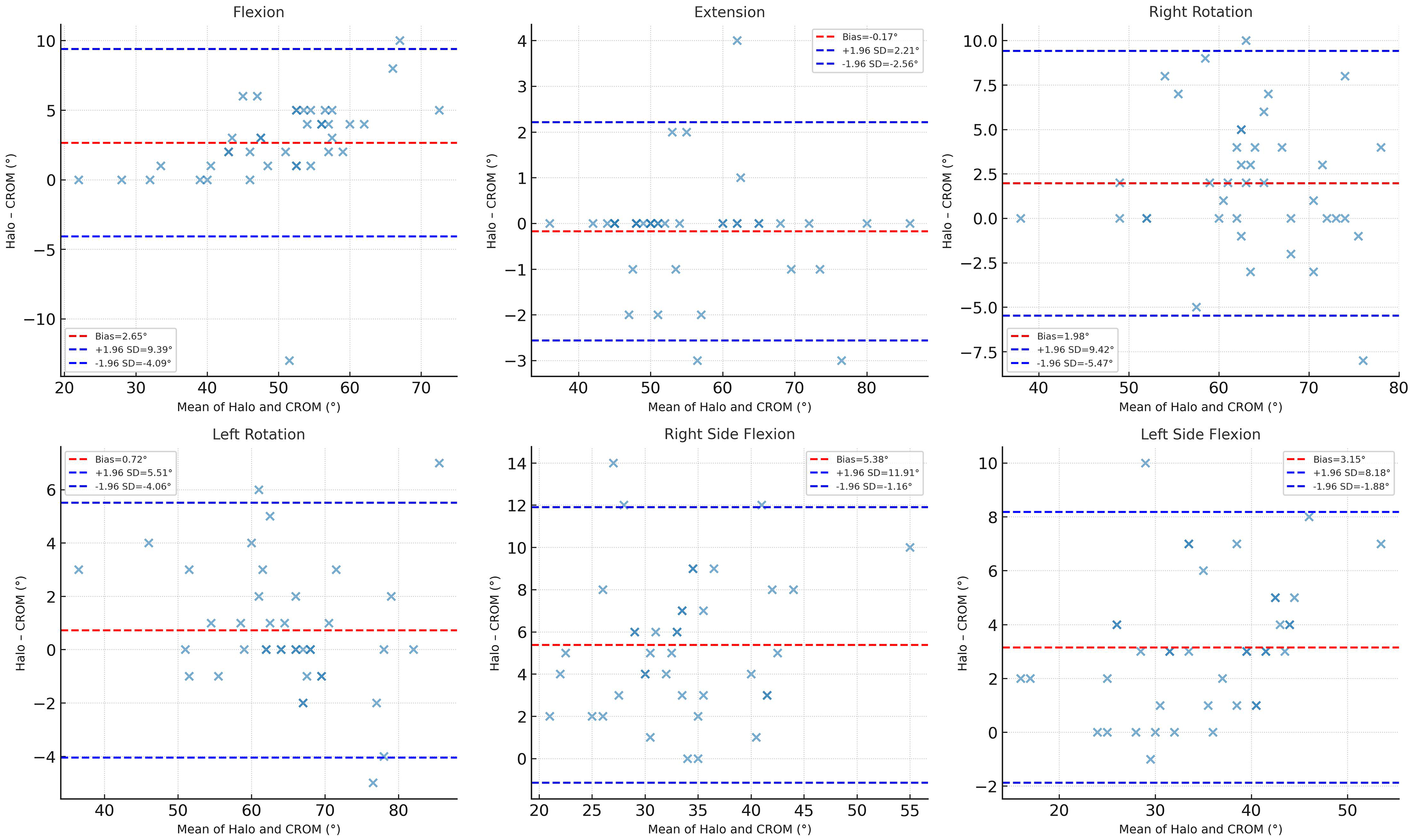
Bland-Altman analysis for each cervical movement. Legend: CROM™: Cervical Range of Motion device; HALO™: HALO™ digital device; FLEX: flexion; EXT: extension; RSF: right side flexion; LSF: left side flexion; RR: right rotation; LR: left rotation; SD: Standard Deviation.
Bland-Altman mean differences between CROM™ and HALO™ measurements.
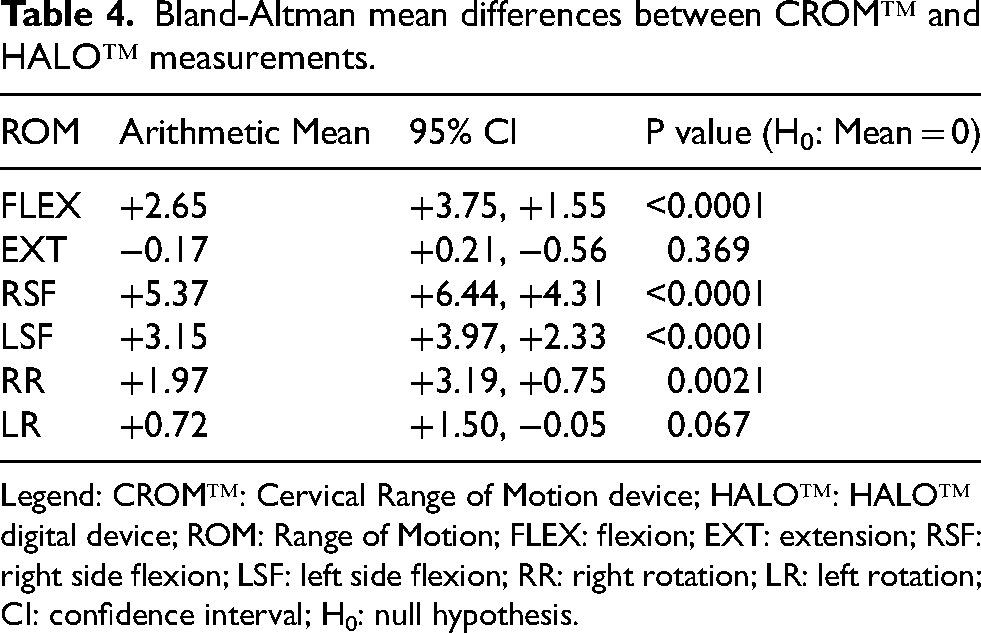
Legend: CROM™: Cervical Range of Motion device; HALO™: HALO™ digital device; ROM: Range of Motion; FLEX: flexion; EXT: extension; RSF: right side flexion; LSF: left side flexion; RR: right rotation; LR: left rotation; CI: confidence interval; H0: null hypothesis.
Flexion and extension
Figure 5(a) and (b) present the Bland-Altman plots for flexion and extension. The mean level of bias between the CROMTM and the HALOTM for FLEX was +2.65o (95% CI +3.75 to +1.55). This difference in agreement between the two devices was statistically significant (P = <0.0001). For EXT, the mean level of bias was −0.175 (95% CI +0.21 to −0.56) This difference was not statistically significant (P = 0.369) and did not reach a clinically meaningful difference.
Right and left side flexion
Figure 5(c) and (d) present the Bland-Altman plots for right and left side flexion. The mean level of bias between the HALOTM and the CROMTM for RSF was +5.37o (95% CI +6.44 to +4.31). This difference in agreement between the two devices was statistically significant (P = <0.0001). For LSF, the mean level of bias was +3.15 (95% CI +3.97 to +2.33). This difference was statistically significant (P = <0.0001), but it did not reach a clinically meaningful difference.
Right and left rotation
Figure 5(e) and (f) present the Bland-Altman plots for right and left rotation. The mean level of bias between the HALOTM and the CROMTM for RR was +1,97o (95% CI +3.19 to +0.75). This difference in agreement between the two devices was statistically significant (P = 0.0021). For LR, the mean level of bias was +0,72° (95% CI +1.50 to −0.05). This difference was not statistically significant (P = 0.067), and it did not reach a clinically meaningful difference.
Discussion
The primary objective of this study was to establish the validity of the measurements of cervical spine motion using the HALO™ device compared to the gold standard CROM™ in a population of patients suffering of CGH. In terms of correlation values between the two measurement tools (ICC), the results indicate a level of reliability that is primarily excellent to good, with only RSF remaining at a moderate level (ICC = 0.688, 95% CI [−0.073 to 0.902]). These findings allow us to conclude that the HALOTM is a valid tool for measuring active cervical ROM when compared to the gold standard CROM™.
To further document the agreement between measurements, we also conducted a Bland-Altman statistical analysis. This analysis determined that the HALOTM generally tended to overestimate measurement values compared to the CROM™. Indeed, aside from the EXT and the LR measurements, which showed non-statistically significant differences, measurements for all four other movements demonstrated an overestimation ranging from +1.97 [95% CI: +3.19, +0.75] degrees for RR to +5.37 [95% CI:+6.44, +4.31] degrees for RSF. This overestimation is statistically significant, suggesting that the HALOTM's measurements may not be as precise as those of the CROM™. When interpreting these results, it is important to note that the 95% limits of agreement were relatively wide for certain planes of motion, particularly side flexion. While ICC values indicated good to excellent correlation for most movements, the Bland–Altman analysis highlights that individual measurements from the HALO™ could differ from the CROM™ beyond thresholds often cited as clinically acceptable (±5°),11,27 which may limit the clinical interchangeability between the HALO™ and the CROM™ for these movements. Nonetheless, these values still compare favorably to the variability seen with visual estimation, which remains common in clinical settings.26,35
As mentioned previously, one of the criteria characterizing CGH is the presence of reduced ROM in the upper cervical region.5,10 A recent study demonstrated that the CROM™ is a tool with moderate to good reliability (ICC 0.65–0.9) for measuring upper cervical rotation. 32 Since the results of our study show a good level of agreement between the CROMTM and the HALOTM, the latter could therefore be a good option for measuring craniocervical rotations.
Strengths and limitations in relation to COSMIN guidelines
One of the study's strengths lies in the fact that both measures were taken simultaneously, which minimized potential information bias and procedural errors. Moreover, a single evaluator performed all measurements, reducing handling errors and harmonizing data collection for both the CROM™ and the HALO™. Additionally, we opted for two complementary statistical approaches (ICC and Bland–Altman analyses) to enhance robustness, and we included a symptomatic population rather than healthy volunteers, thereby improving external validity in clinical practice.
However, there are some limitations. According to the COSMIN guidelines for studies on reliability and validity, 36 several aspects of our methodology warrant consideration. First, our sample size (20 participants, yielding 40 independent device comparisons) remains below the ≥50 participants generally recommended. Although repeated measures increased statistical precision, the relatively modest participant number may limit external validity. Second, COSMIN recommends comparison with a true criterion standard such as 3D-motion capture. While such methods are difficult to apply in clinical contexts, we used the CROM™, which is widely regarded as the clinical reference standard, to provide a pragmatic and feasible comparator. Third, assessor blinding was not possible because measurements were taken concomitantly; while this may have introduced observation bias, it also reduced temporal and procedural variability and arguably provided a more robust estimate of device agreement. Additionally, potential device-related errors must be acknowledged: displacement of CROM™ magnets and the lower precision of its analog compass compared to the HALO™ digital readout could have affected data accuracy. Finally, our sample was restricted to patients with cervicogenic headache, which may limit generalizability. However, including a symptomatic clinical population enhances real-world applicability, as clinicians commonly evaluate patients with diverse cervical complaints rather than asymptomatic volunteers.
Future studies should therefore include larger and more diverse samples, apply COSMIN-recommended designs, and incorporate additional criterion standards such as 3D motion capture, in order to provide stronger evidence for the validity and potential interchangeability of the HALO™ with the CROM™.
Conclusion
The HALO™ digital goniometer demonstrates potential as an alternative to the CROM™ device for cervical ROM measurement, particularly given its versatility, usefulness, and practicality. Nonetheless, given the moderate agreement observed in certain planes, the relatively small number of participants compared to COSMIN recommendations, 36 and the absence of a true criterion standard, the device should be used cautiously. Future studies with larger and more diverse samples, and with additional gold-standard comparators, are needed to confirm its validity and determine its potential interchangeability with the CROM™.
Footnotes
Acknowledgements
n/a.
Ethical considerations
The study was approved by the Ethics Review Board of the Clinical Research Center of the Centre intégré universitaire de santé et des services sociaux de l’Estrie – Centre Hospitalier Universitaire de Sherbrooke (reference number: 2018-2642).
Consent to participate
Informed consent was obtained for all participants according to The Helsinki Accords.
Consent for publication
Written consent for publication of photographic content was obtained.
Author contributions
Nathalie Desmarais: validation, formal analysis, writing – original draft, review and editing.
Jean-Philippe Paquin: investigation, methodology, resources, writing – review and editing.
Yannick Tousignant-Laflamme: conceptualization, funding acquisition, project administration, supervision, writing – review and editing.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The dataset is available upon request from the corresponding author.
