Abstract
Background:
Medullary thyroid carcinoma (MTC) is a rare neuroendocrine malignancy with a 10-year mortality rate up to 50%. Current therapies for metastatic MTC comprise various tyrosine-kinase inhibitors, but resistance often appears due to the need for lifelong treatments. Like in other tumors, genetic, epigenetic, post-transcriptional, post-translational, and cell–cell interaction events influence drug response. However, progress in understanding MTC biology is limited by the lack of reliable in vivo and in vitro models. This study aims to develop a patient-derived model faithfully reproducing the microenvironmental alterations present in MTC.
Methods:
We applied a two-step protocol consisting of a first phase in which primary cells are cultivated as multicellular spheroids and a second phase in which they are switched to adherent cultures. After evaluation of the genetic background by targeted Next Generation Sequencing, we characterized our cells phenotype by examining a panel of stem/progenitor-related markers, the secretory abilities by ELISAs, the drug response by proliferation assays, the in vivo angiogenic and invasiveness by the use of zebrafish model, the in vitro invasivity by Matrigel Dome assays and the spatial variation of stem/progenitor marker in both 3D cell models and tissue samples by confocal microscopy.
Results:
Our model allowed the establishment of eight MTC patient-derived cell lines with different genetic backgrounds. The cultures faithfully reproduced the changes in stem and progenitor markers that we detected in our cohort of MTC tissue samples and could be successfully xenotransplanted in zebrafish model, showing both angiogenetic and invasive properties. Drug screening assays revealed the potential of our model for the study of patient-specific responses, as we were able to identify different candidate regulators of the sensitivity to currently available therapies for MTC.
Conclusions:
Our two-step protocol successfully generated primary MTC lines that maintain high plasticity, can be cultivated for several passages, and recreate the heterogeneity observed in patients’ tissues. Our model will offer a robust platform for preclinical drug testing and mechanistic studies, addressing a longstanding gap in MTC research. It enables exploration of tumor microenvironment interactions and personalized therapeutic responses, supporting progress beyond current genomic-driven frameworks.
Keywords
Introduction
Medullary thyroid carcinoma (MTC) is a rare neuroendocrine malignancy originating from thyroid C cells. 1 Surgery is the gold standard treatment, but unresectable distant metastasis is detected in up to 40% of patients.2–4 Current therapies for advanced MTC comprise multi-kinase inhibitors (multi-KIs) and Proto-oncogene tyrosine-protein kinase receptor Ret (RET)-selective inhibitors. Unfortunately, since all KIs are lifelong treatments, the development of therapy resistance is common. Known general KI resistance mechanisms include acquisition of alternative genetic alterations, on-target5,6 and off-target,7–9 and non-genetic mechanisms such as epithelial-to-mesenchymal transition and microenvironmental alterations.10–13 Consequently, MTC still has a 10-year mortality rate up to 50%.1,3,14
Indeed, genetic drivers are not the only determinants of MTCs’ behavior, but epigenetic, post-transcriptional, post-translational, and cell–cell interactions events may contribute. Multi-omics approaches revealed different MTC subtypes that may evolve one into each other under selective pressure, with predominant mesenchymal and basal/stem characteristics associated with the worst outcome.15–18 In this setting, the historically limited availability of reliable in vivo and in vitro MTC models is striking. Different murine models of sporadic MTC, created by tissue-specific expression of mutated Ret or Ras, exhibited strong variations in tumor penetrance between strains with different backgrounds, implicating a significant role of other species-specific factors in carcinogenesis that may hamper the translatability of the results.19–21 The few available spontaneously immortalized MTC lines harbor multiple genetic and chromosomal alterations rarely found in MTC,22,23 indicating that only exceptionally aggressive MTC cells can survive in long-term continuous culture conditions. Consequently, immortalized lines not only misrepresent the genetic background of MTC, consisting of a few genetic alterations and virtually no defects in DNA repair systems, 24 but also lack all the microenvironmental factors that may influence drug sensitivity, raising concerns about the translational potential of the in vitro preclinical drug studies performed until today. For many solid tumors, this scientific impasse has been overcome by the creation of reliable three-dimensional patient-derived models. Unfortunately, standardized MTC models that faithfully reproduce MTC genetic and the complexity of microenvironmental conditions are still under development. Only recently, patient-derived organoids from RET-mutated MTCs were established by two different groups, although loss of differentiation at early passages may limit their utility in preclinical pharmacological studies.25,26
Here we propose an alternative patient-derived MTC model that allowed the creation of primary lines with different genetic background and response to external stimuli, permitting the identification of different candidate genes involved in drug-specific responses. We believe that this model represents a promising tool to study MTC biology and patient-specific therapeutic responses.
Methods
Patient-derived MTC lines and immortalized cell line culture
Samples of 9 MTCs (Supplementary Table S1) were collected, dissociated, and processed within two hours, except for MTC3 and MTC9, processed the day after. After mechanical dissociation with a surgical scalpel, enzymatic dissociation was performed with 2 mg/mL Collagenase II (Merk, Darmstadt, Germany)/DMEM (Thermo Fisher Scientific) solution at 37°C for 2 hours, with gentle disaggregation by pipetting every 20 minutes. The solution was filtered through a 100 µm strainer to remove debris and cells pelleted by centrifugation at 800 g for 5 minutes; 125 cells/µL were seeded in ultra-low attachment plates (Corning, New York, USA) in Sphere Medium (SM, 1:1 DMEM/F12:Neurobasal medium supplemented with 1:50 B27 (Thermo Fisher Scientific), 20 ng/mL b-FGF (Thermo Fisher Scientific), 20 ng/mL EGF (Thermo Fisher Scientific). Penicillin/Streptomycin (Merk) was added for the first week. The media was partially refreshed every 3 days. Multicellular spheroids appeared after 3–4 days. After three weeks of suspension culture, the larger multicellular spheroids started to show typical microscopy features of a hypoxic core, thus the cells were switched to adherent culture in order to avoid replicative senescence and/or necrosis. The multicellular spheroids were collected by gravity, washed in PBS (Euroclone, Milan, Italy) and disaggregated with incubation in Accutase:Trypsin 3:1 mixture (Merk, both) for 10 minutes. The cells were seeded on 0.2% gelatin (Merk) coated dishes in Adherence Medium (AM, 1:1 DMEM/F12:Neurobasal medium supplemented with 1:50 B27, 20 ng/mL b-FGF, 20 ng/mL EGF, 2 ng/mL IGF-1 (Thermo Fisher Scientific), and 1% FBS (Thermo Fisher Scientific) and grown until confluence. The cultures were obtained by spontaneous growth, without any specific selection for cell types. Adherent cultures were screened for mycoplasma contamination with VenorGeM Classical Mycoplasma Detection Kit (Minerva Biolabs, Berlin, Germany) and routinely replated at 1:2 ratio. For long-term storage, cells were cryopreserved in a solution composed of 50% Adherence Medium, 40% FBS, and 10% DMSO (Merk). After thawing, MTC lines required at least one week of culture before getting back to exponential growth.
Western blotting, next-generation sequencing (NGS), Sanger sequencing, primary and secondary sphere forming assay, Extreme Limiting Dilution Analysis (ELDA), Doubling time calculation, Calcitonin, Chromogranin A, and thyroglobulin dosage, real-time quantitative PCR, drug screening assays, matrigel dome invasion assay, immunofluorescence and confocal microscopy, zebrafish care, and tumor xenograft experiments are reported in Supplementary Data and Supplementary Tables S2, S3, and S4.
Institutional review board approvals
Informed consent was obtained from all participants. Institutional review board approval was obtained (Ethical Committee of the Istituto Auxologico Italiano IRCCS, THY-CANC 2022_03_08_03; code 05C212 and ZEBRANET 2017_03_28_19; code 05C402). Research was completed in accordance with the Declaration of Helsinki as revised in 2024.
Quantification and statistical analysis
Blot densitometry, integrated density, and zebrafish vessels quantification were performed with FIJI software. 27
Differences in the number of variants with minor allele frequency <1% and read depth >3028,29 between MTC tissues and lines were evaluated by chi-squared test.
Gene expression and dose-response parameters relationship was assessed using Pearson’s correlation.
Stemness parameters at early and late passages were calculated with the ELDA webtool (http://bioinf.wehi.edu.au/software/elda/). 30
At least five independent biological replicates were performed for each experiment, except for patient-derived multicellular spheroids (PDS, Fig. 1A), in which a total of 100 spheroids were analyzed from at least 4 technical replicates. After evaluation of data distribution, homoscedasticity, and normality by D’Agostino-Pearson, Shapiro–Wilk normality tests, and Bartlett’s test for equal variances, the appropriate statistical test (One-way and two-way analysis of variance (ANOVA), paired t-test, Pearson’s correlation), Spearman correlation) was performed by either GraphPad Prism 5 or R software version 4.4.2. All data are represented as mean ± standard error (SEM) or median with interquartile range (IQR). Minimal statistical significance was set at p < 0.05 for all experiments.
Results
MTC cells can be cultivated as multicellular spheroids expressing stem and progenitor markers
We successfully cultivated PDS for 8 out of 9 patients (Fig. 1A and B and Supplementary Table S1). PDS showed a significant increase in previously reported stem and neuro/enteroendocrine progenitor markers,31–33 and a significant reduction of p53 levels. In agreement with the literature,34,35 we detected variable levels of Prominin-1 (CD133). No variations in general levels of E-cadherin and Vimentin were found, but, except for MTC9, the increase in PDS size significantly correlated with the increase in E-cadherin/Vimentin ratio, suggesting the maintenance of an epithelial-like phenotype (Fig. 1C and D and Supplementary Fig. S1A). A significant enrichment in stem-like cells was also confirmed by the high rate of primary and secondary clonal sphere formation (Supplementary Fig. S1B).
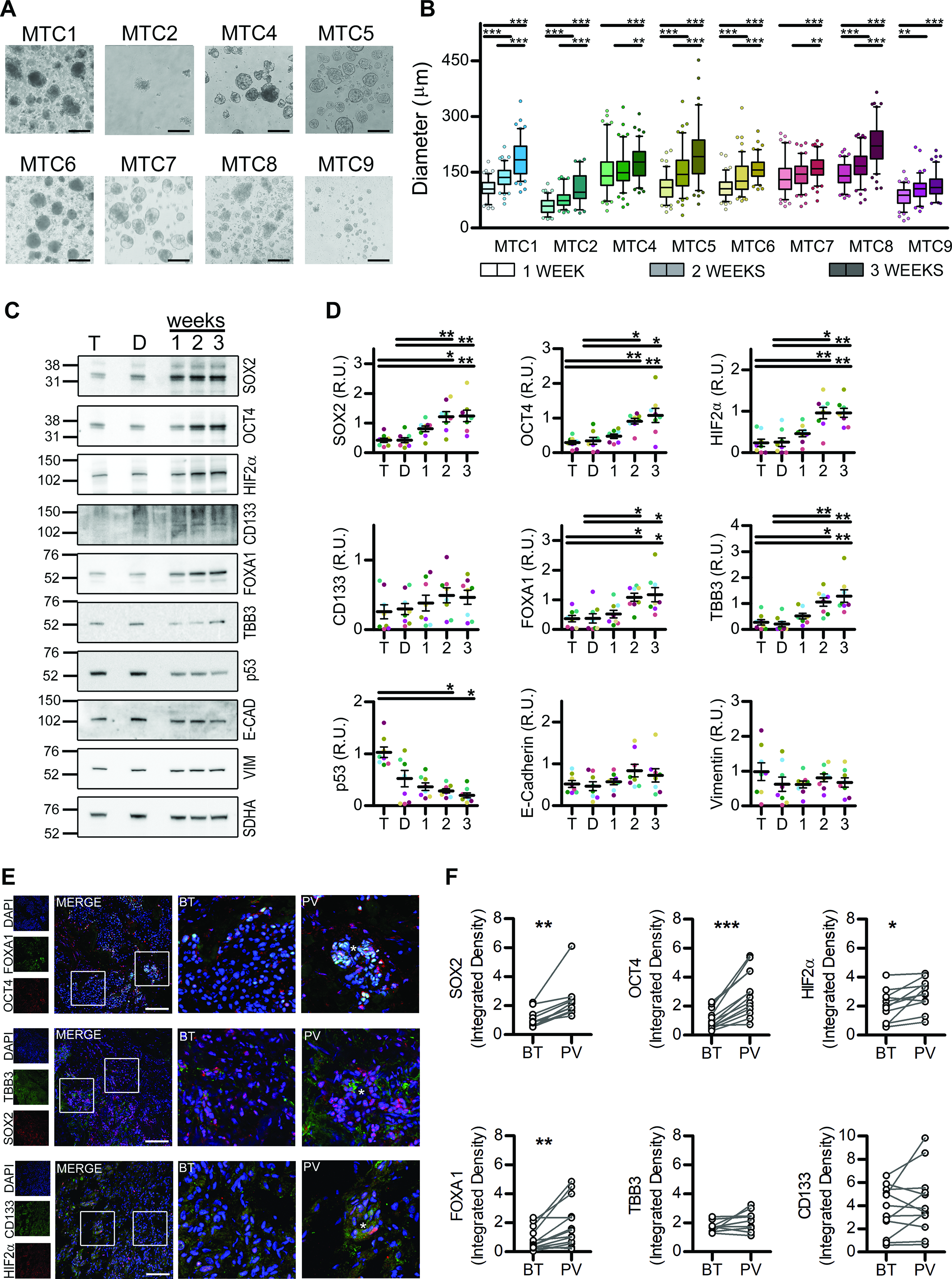
Medullary thyroid carcinoma (MTC) primary cells can be cultivated as multicellular spheroids enriched in stem-like cells.
Since little is known about stem-like cells in MTC, we validated our PDS data in a small cohort of 17 MTC tissues (Supplementary Table S1), confirming a significant increase in the expression of Transcription factor SOX-2 (SOX2), POU domain, class 5, transcription factor 1B (also known as Octamer-binding transcription factor 4, OCT4), Endothelial PAS domain-containing protein 1 (HIF2α), and Hepatocyte nuclear factor 3-alpha (FOXA1) in perivascular areas (Fig. 1E and F), niches usually enriched in stem-like cells. CD133 expression was significantly higher in tumor harboring RET mutations, irrespective of the area (Supplementary Fig. S2).
PDS propagated as adherent cultures retain MTC tissue characteristics
To avoid the development of replicative senescence or necrosis typical of larger spheroids, after three weeks, PDS were switched to adherent monolayer culture. On the basis of the scarce literature on C-cell differentiation and MTC organoids,25,32,36 the similarities between neuro/enteroendocrine and neural crest cells,32,37 and the fact that neuroendocrine organoids that undergo p53 suppression-mediated reprogramming acquire independence from niche factors, 38 cultures were propagated in Sphere Medium supplemented with 2 ng/mL IGF-1 and 1% FBS. All PDS produced adherent cell lines that could be cultured for at least 15 passages, with a doubling time of 2.113–6.904 days (MTC1 3.197 ± 0.556, MTC2 3.509 ± 0.519, MTC4 6.904 ± 0.601, MTC5 2.754 ± 0.621, MTC6 2.113 ± 0.463, MTC7 5.254 ± 0.553, MTC8 2.99 ± 0.477, MTC9 4.337 ± 0.893).
NGS confirmed maintenance of the genetic background (variants tissues:185, lines:183, Chi-squared p value = 0.917), with 4 RET-mutated patients/lines and two RAS-mutated patients/lines (Table 1). One case (MTC2) had a CDKN1B mutation previously associated with MEN4 39 and never reported in sporadic MTC, while in one case (MTC9), no pathogenic variants were found, even in other noncanonical genes previously described in MTC such as NF1, BRAF, CTNNB1, VHL and APC. Variants predicted to be pathogenic by ClinGen, CGC and VICC consortium criteria 40 were also identified in other 16 cancer-related genes (Fig. 2A, Table 1 and Supplementary Table S3). Interestingly, although the mutation burden was similar in each tissue-line couple, the three cases with no clear genetic driver showed lower mutation burden than the RET or RAS driven ones (Table 1, p < 0.001 for RET vs. NonRETnonRAS and for RAS vs. NonRETnonRAS).
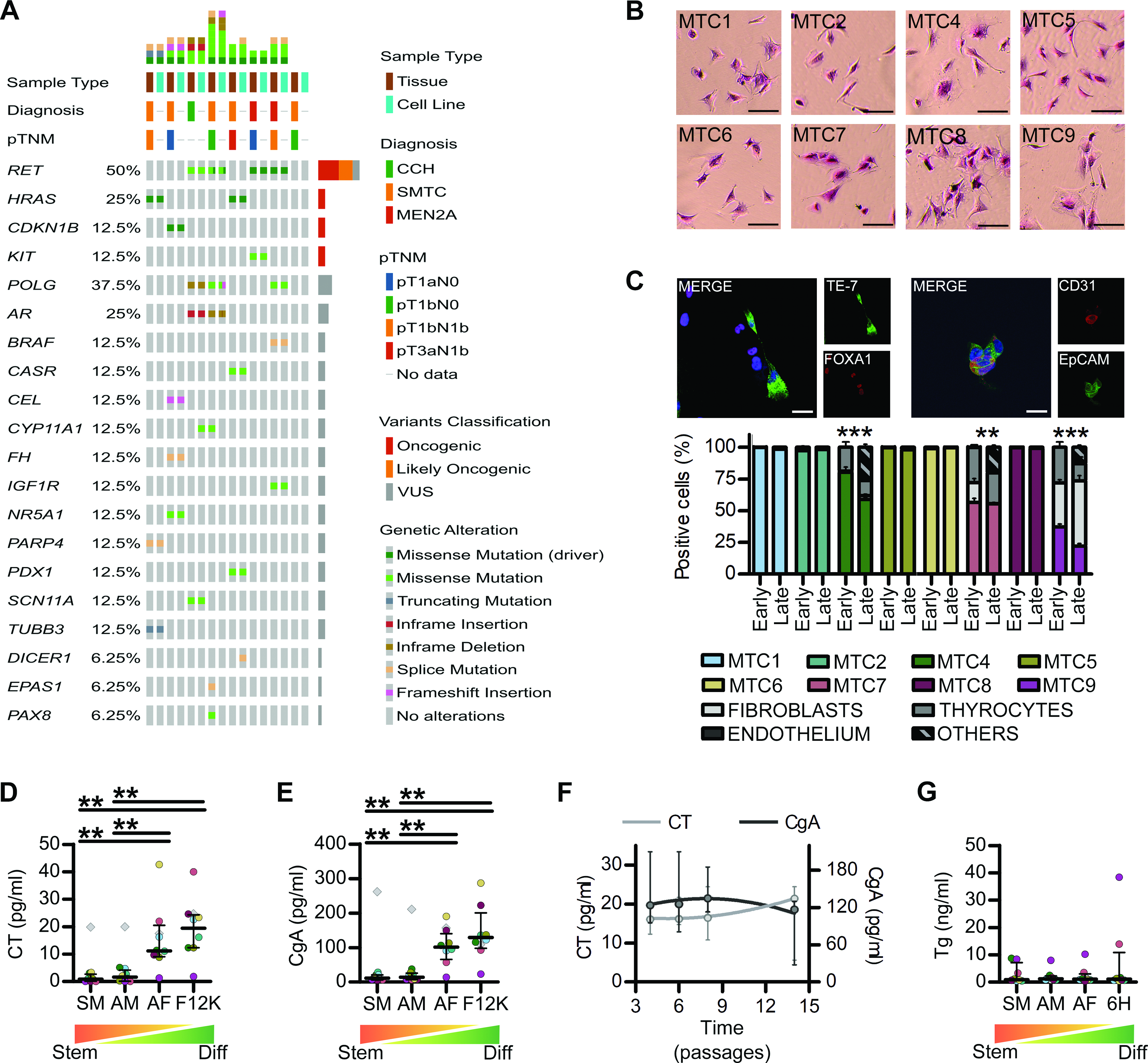
Adherent MTC cell lines recapitulate MTC tissues characteristics.
Clinical and Molecular Data for the Medullary Thyroid Carcinoma Tissues and Relative Cell Lines at Early and Late Passages
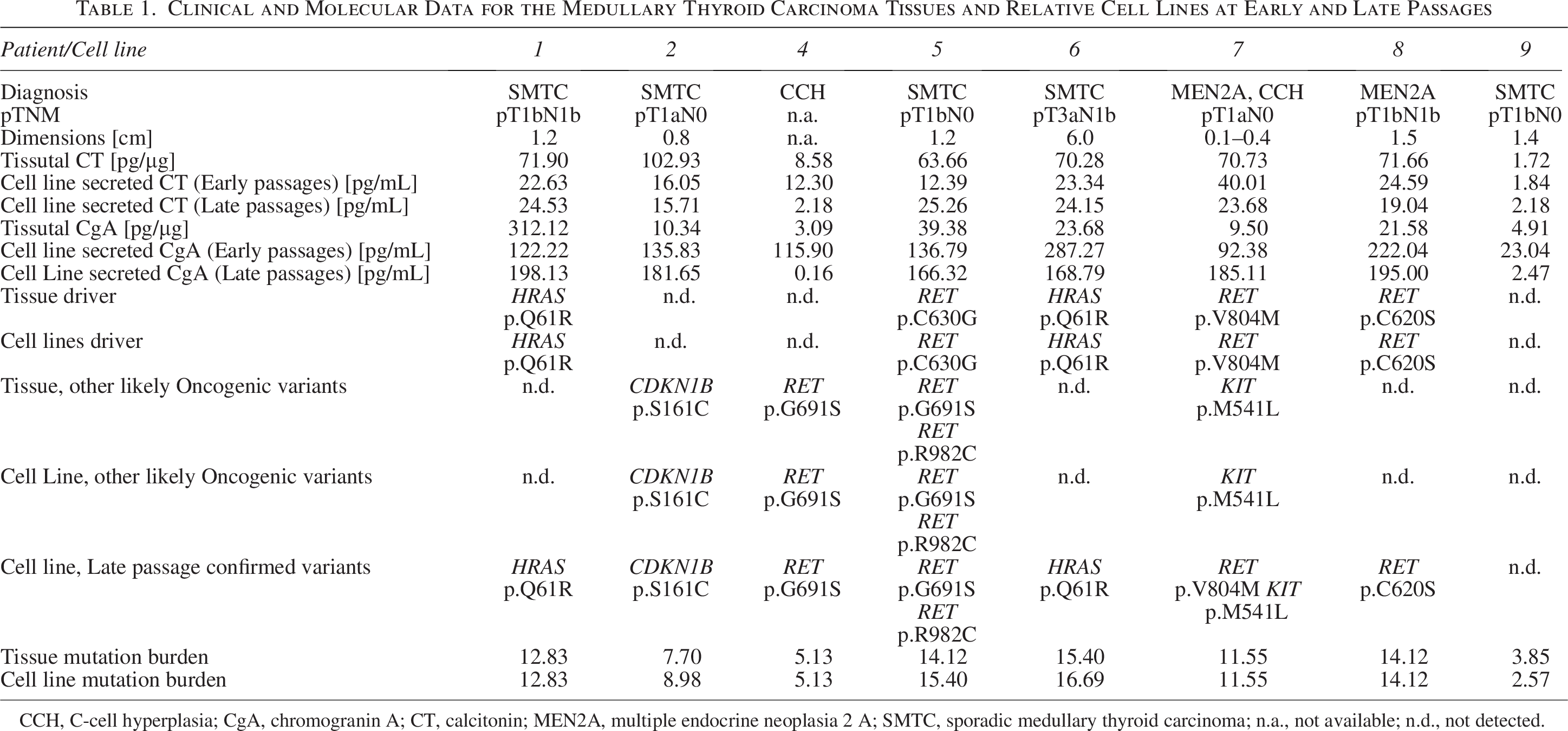
CCH, C-cell hyperplasia; CgA, chromogranin A; CT, calcitonin; MEN2A, multiple endocrine neoplasia 2 A; SMTC, sporadic medullary thyroid carcinoma; n.a., not available; n.d., not detected.
All cells showed a predominant epithelial-like phenotype, but in MTC4, MTC6, and MTC9 fibroblasts, thyrocytes, and endothelial cells were present (Fig. 2B and C). Fibroblasts and thyrocytes were maintained even at later passages, when the appearance of a significant percentage of cells without clear lineage markers appeared (Fig. 2C, Supplementary Table S5), probably because of primary cell lines reaching replicative senescence, as also indicated by significant alterations in the phosphorilated Histone H2AX (γH2AX), Cyclin-dependent kinase inhibitor 1 (p21), Cyclin-dependent kinase inhibitor 1B (p27), and p53 phosphorylation (Supplementary Fig. S3). In differentiation-promoting conditions, all lines, except MTC9, were able to secrete significant levels of Calcitonin (CT) and Chromogranin A (CgA), with values similar to the baseline TT ones, which were instead unaffected by stem-promoting conditions (Fig. 2D and E, Supplementary Fig. S4A and B). The secretion of both CT and CgA was maintained for several passages (Fig. 2F), with the exception of MTC4, whose secretion of both markers seemingly declined after p9 (Table 1, Supplementary Fig. S4D). Thyrocytes contamination in MTC7 and MTC9 was also confirmed by the secretion of Thyroglobulin (Fig. 2G, Supplementary Fig. S4C).
MTC lines have angiogenic and invasive abilities
The pro-angiogenic and invasive potential was first assessed in vivo using zebrafish model, with three representative MTC cell lines. All lines showed pro-angiogenic responses (Fig. 3A and B) comparable with previous studies on immortalized MTC cells.41–44 The presence of tumor cell clusters far from the injection site revealed a significant invasive behavior (Fig. 3C and D), confirmed by Matrigel Domes invasion assay, where significantly different patterns of invasion were also detected (Supplementary Fig. S5). The invasive phenotypes reflected dynamic shifts in stem/progenitor markers expression, with Tubulin beta-3 chain (TBB3) and FOXA1 being significantly higher in cells during budding and lobular invasion, while SOX2 and OCT4 were generally more expressed on the edges. The variations of stem/progenitor markers were generally similar to those observed in the TT cells, except for FOXA1, where all primary lines exhibited higher levels. Spheroids with smooth surface and no peculiar local invasion showed very few cells positive for TBB3, FOXA1, and nuclear SOX2 and OCT4 (Fig. 3E and F). Similar significant variations were also confirmed in MTC tissues (Fig. 3G and H).
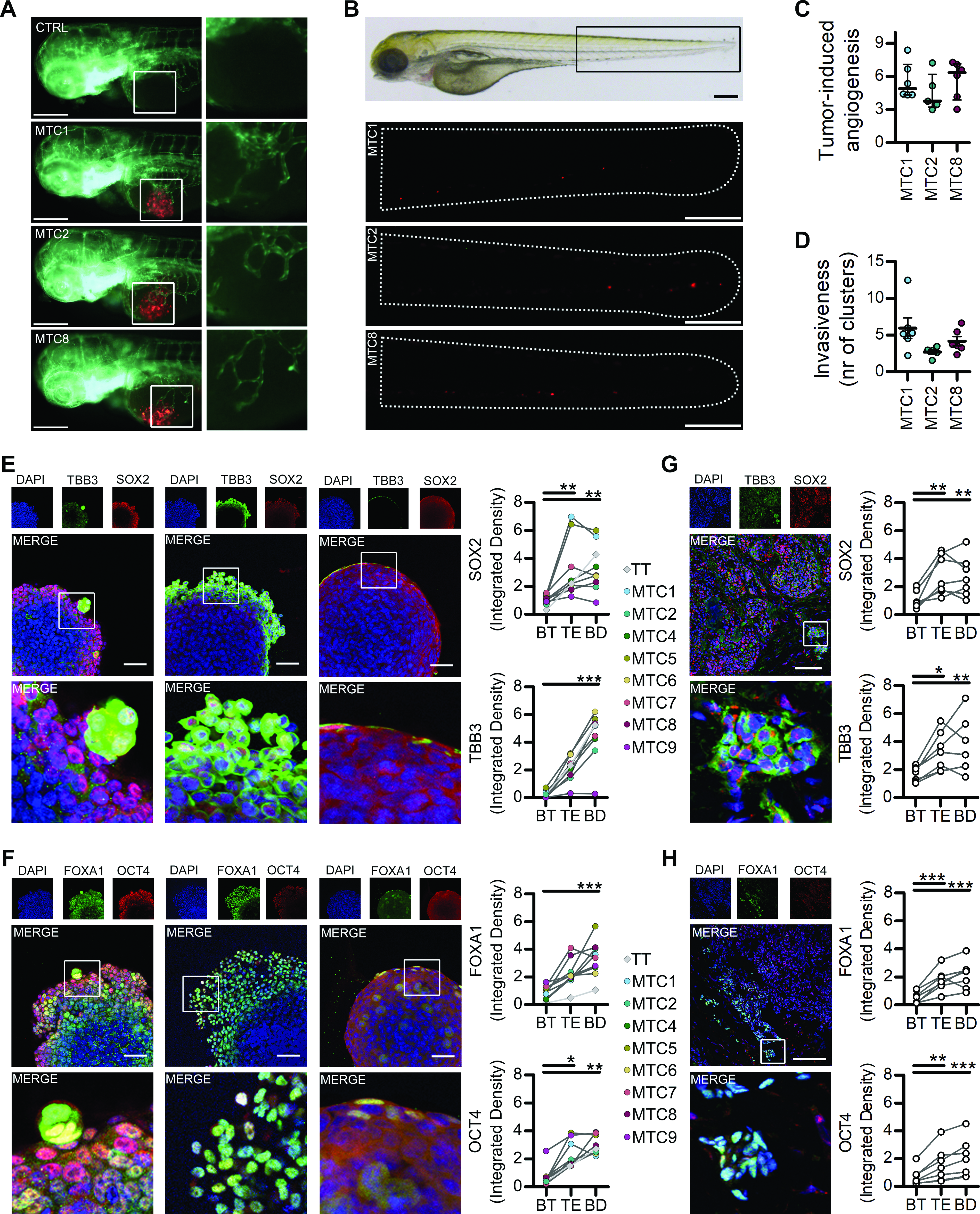
MTC lines have pro-angiogenic and invasive behavior in vivo and in vitro.
MTC cell lines maintain a stem-like phenotype and high plasticity
We proceeded to characterize the plasticity of our primary cell lines to define whether they are a suitable model for advanced MTC tumor biology studies. A high but variable enrichment in stem-like cells was detected, with a stem-like fraction more stable than the TT cells one (Fig. 4A and B, Supplementary Table S6) and still similar to the original PDS one (Fig. 4C), accompanied by variable expression of stem/progenitor markers (Fig. 4D).
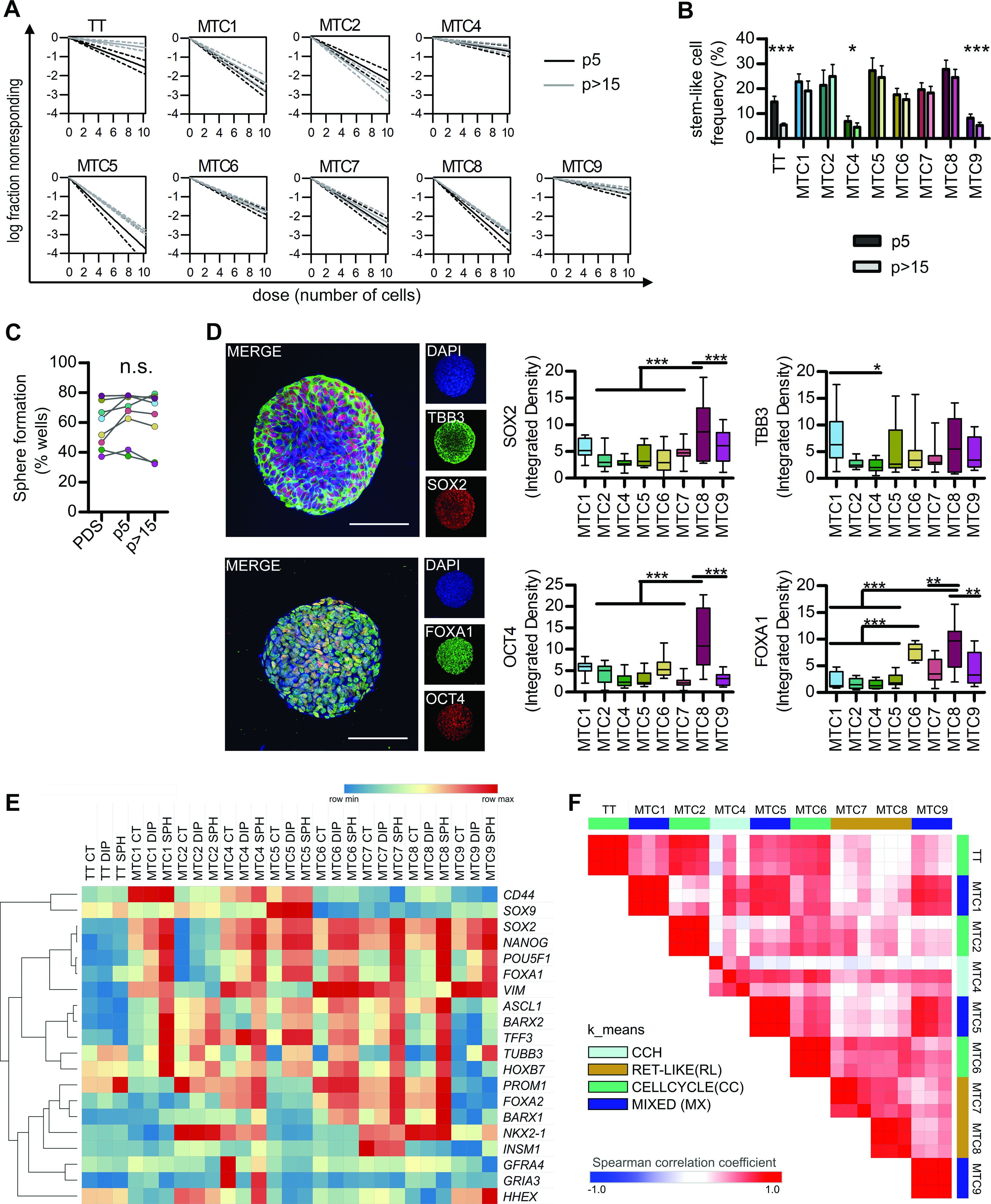
MTC cell lines maintain a stem-like phenotype and high plasticity.
As the second step, we evaluated how the lines reacted to two stem-promoting conditions: hypoxia induced by DIP treatment and selective cultivation as clonal spheres.
Our data showed significant differences in the expression of an extended panel of known classical stem markers (SOX2, NANOG, POU5F1, CD44, PROM1), endoderm and C-cell progenitor markers (FOXA1, FOXA2, SOX9, HHEX, NKX2-1, BARX1, BARX2, HOXB7, ASCL1, INSM1, TFF3) and MTC-associated markers (VIM, TUBB3, GFRA4, GRIA3).34,45,46 First of all, TT cells had lower levels of almost all stem-related genes, with only significant induction in TUBB3 and PROM1 (coding for CD133), while the primary MTC cells showed a variable but significant pattern of induction (Fig. 4E). Similarity analysis revealed that MTC6, aggressive tumor, and MTC2, with genetic cell cycle dysregulation, are similar to TT line (Cell Cycle group), MTC7 and MTC8 are grouped together (RET-like) as well as MTC9, MTC1 and MTC5 (Mixed, MX). MTC4, derived from C-cell hyperplasia (CCH), was different than all the other ones (Fig. 4F).
MTC lines unveil patterns of tyrosine-kinase inhibitor resistance
We investigated how the differences detected in the genetic background and in the cells phenotype may influence the response to current MTC therapies. Treatments with Vandetanib, Lenvatinib, Cabozantinib, Selpercatinib, and Pralsetinib showed a variable drug response (Supplementary Table S7), with genetic and gene expression grouping confirming a lower sensitivity in the lines not harboring classical RET or RAS mutations (Fig. 5A) and lower sensitivity to multi-KIs for those belonging to the RET-Like group (Fig. 5B). The most significant differences were detected between normal growing cells and those in stem-promoting environments, irrespective of the individual line background and sensitivity (Fig. 5C and D). The analysis of the IC50 and EMAX values in respect to the gene expression panel revealed interesting correlations (Fig. 5E and Supplementary Table S8). Vandetanib and Cabozantinib showed a significant negative correlation between the maximal effect and genes involved in the maintenance of stem and C-cells progenitor plasticity. Selpercatinib and Pralsetinib showed a significant positive correlation of IC50 with PROM1 expression (Fig. 5E and F). Four out of five drugs showed a significant negative correlation between their maximal effect and expression of TFF3, revealing for the first time that Trefoil factor 3 protein (TFF3) may be involved in MTC therapy resistance (Fig. 5E–G).
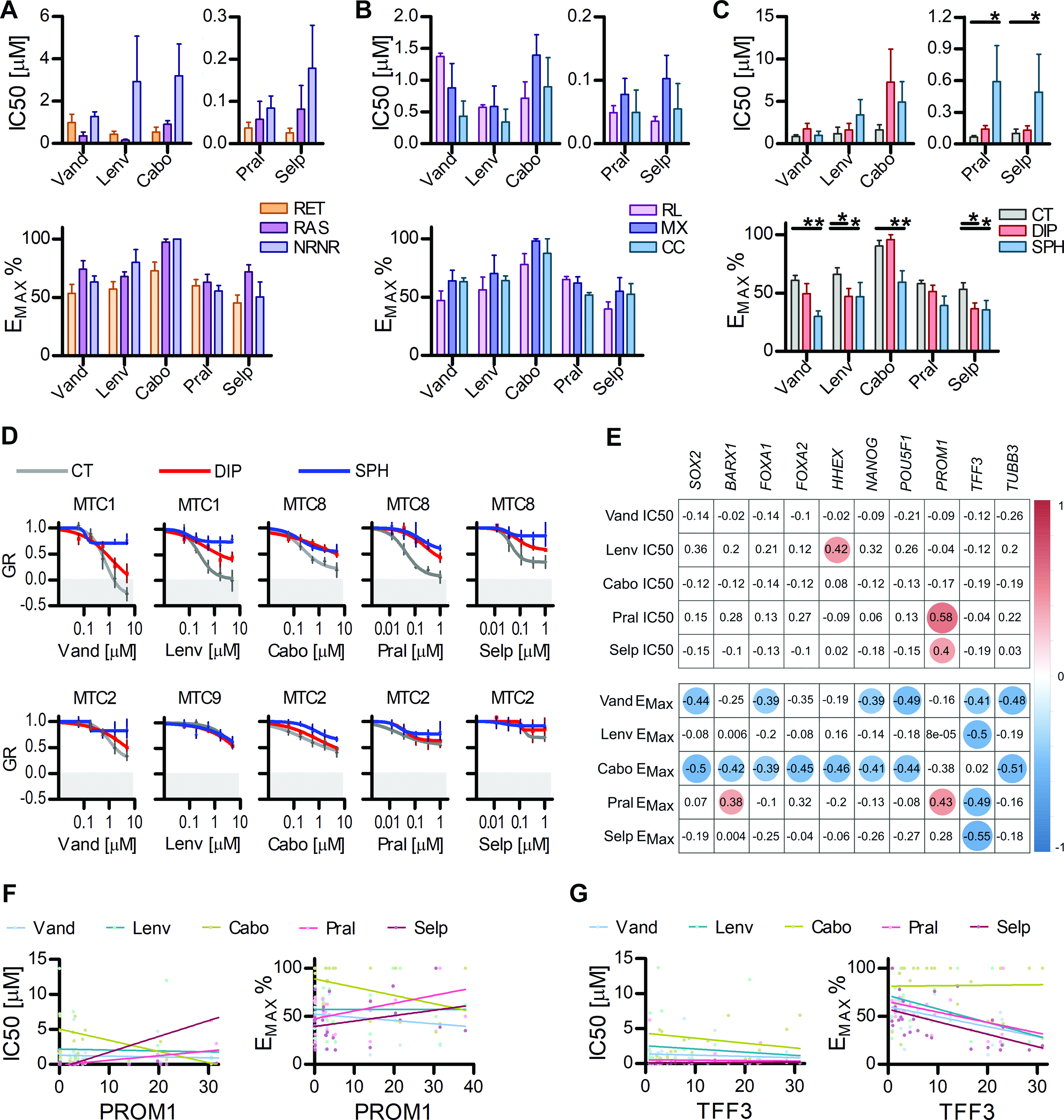
MTC lines unveil patterns of tyrosine-kinase inhibitors resistance.
Discussion
In this study, we propose a two-step protocol to establish patient-derived MTC cell lines that maintain a high plasticity, can be cultured for several passages, and have significant potential for preclinical drug testing.
Our model involves initial selection as PDS, followed by a switch to adherent culture, an approach already successful for stem-like glioblastoma lines. 47 Differently from the model recently proposed by Jager et al, 26 our strategy aimed at setting experimental conditions optimal for spontaneous MTC cells proliferation. This implied reducing to the bare minimum the use of supplements, forcing stem-like pluripotent replicative status such as Nogging, R-spondin, and Rho-associated protein kinases (ROCK) inhibitors that are necessary for MTC organoids establishment from biopsies. Our procedure allowed longer in vitro culture time without loss of cell plasticity, maintaining both stem-like properties and C-cell characteristics.
Of nine patients, we obtained five pure MTC lines (Fig. 2B and C), a success rate of 56%, comparable to other neuroendocrine tumor models.48–50 This also increases to 71% when not considering the samples processed 24 hours after surgery, suggesting prompt processing as crucial for MTC. In two out of three cases of lines containing multiple lineages, the proportion of cells with MTC remained predominant even at later passages (Fig. 2B, Supplementary Table S5). The MTC lines presented relatively stable characteristics until passage 12–14, after which variable degree of replicative senescence was detected, together with loss of Calcitonin and Chromogranin secretion in the CCH-derived cell line and followed by a reduction in stem-like properties in the CCH-derived line and in the atypical MTC line (Supplementary Figs. S3 and Figs. S4 and Fig. 4A and B). PDS showed spontaneous changes in p53 and in stemness markers (Fig. 1A–D, Supplementary Fig. S1A) similar to those of induced Pluripotent Stem Cells reprogramming in neuroendocrine organoids, 38 but without need of numerous supplements to maintain the line,25,50 thus allowing spontaneous proliferation, maintenance of plasticity and ability to differentiate into calcitonin-secreting cells (Figs. 2 and 4). A positive correlation between PDS growth and the E-cadherin/Vimentin ratio indicated maintenance of an epithelial-like phenotype despite the increase in stem marker expression (Supplementary Fig. S1A). This hybrid phenotype was reported as a key factor in the maintenance of high stem-like properties in different cancers and is characteristic of cells undergoing collective invasion,51,52 a pattern we described for the first time both in vivo and in vitro in invading spheroids and in MTC tissues (Fig. 3). These findings also confirm presence of stem-like cells in the peritumoral and capsular regions 18 and possibly explain the differences with what previously detected. 31
Indeed, our lines successfully adapted to different microenvironments mimicking the heterogeneity of MTC tissues (Figs. 1E and 3E–H). The cultivation of cell lines in different stem-promoting conditions revealed different patterns of gene expression regulation, only partially linked to the presence of RET or RAS genetic drivers and similar to what was previously reported.15,18,53 Of the three main groups, one included variable genetic drivers (RET, RAS, and undetermined) and the other included two RET drivers. MTC2 resembled immortalized TT cells, as both have cell cycle dysregulation, while the CCH-derived cells showed unique behavior.
Finally, drug-response studies provided new insights into the role of stem-like phenotypes in therapeutic responses (Fig. 5). Two multi-KIs showed reduced maximal effects in case of strong stem-like phenotype, while the IC50 of Pralsetinib and Selpercatinib positively correlated with PROM1, remarking that the relationship between RET-related biological alterations and RET-selective KI effects are not univocal, and RET-specific inhibitors may not equally affect all the processes altered by RET mutations. A novel finding was the link between TFF3 expression and decreased effects for four out of five drugs. TFF3 is expressed in C-cell progenitors 45 and in different tumors, regulating key processes, such as proliferation, metastasization, angiogenesis, and apoptosis, through modulation of MAP kinases, phosphoinositide 3-kinase/RAC-alpha serine/threonine-protein kinase (PI3K/AKT), and hypoxia-inducible factor 1-alpha (HIF1) signaling pathways. 54 In recent years, TFF3 has emerged as a promising candidate biomarker and drug target, and has been proven to be a driver of both intrinsic and acquired resistance to EGFR-TKI in lung adenocarcinoma. 55 For these reasons, TFF3 may be a promising study target for MTC, at the crossroad between regulation of stem-like phenotype, therapy resistance, and clinical outcome prediction, remarking the potential of reliable patient-derived in vitro models in bottom-up bench-to-bedside drug discovery approaches.
In conclusion, our patient-derived MTC model will offer a robust platform for preclinical drug testing and mechanistic studies, which together with other recently proposed protocols25,26 will address a longstanding gap in MTC research. It enables exploration of tumor microenvironment interactions and personalized therapeutic responses, supporting progress beyond current genomic-driven frameworks.
Authors’ Contributions
Conceptualization: E.S.G., V.G., and L.P. Data curation: E.C. and D.G. Formal analysis: E.C., E.S.G., and D.G. Funding acquisition: L.P. and L.F. Investigation: E.S.G., V.G., G.Gaudenzi, E.M., S.U., and V.C. Methodology: E.S.G., V.G., and G.Gaudenzi. Project administration: E.S.G., L.P., G.V., L.F., C.C., G.Gazzano, and G.D. Resources: L.P., G.V., L.F., C.C., G.Gazzano, and G.D. Software: E.C. and D.G. Supervision: L.P. Validation: E.S.G., V.G., and E.C. Visualization: E.S.G. and G.Gaudenzi. Writing—original draft: E.S.G., V.G., G.Gaudenzi, and L.P. Writing—review and editing: E.S.G., V.G., L.P., V.C., C.C., G.V., and L.P.
Footnotes
Acknowledgments
The authors thank Dr. Anna Maria Di Blasio and Dr. Fabiana Guizzardi from the Molecular Biology facility of IRCCS Istituto Auxologico Italiano for the Next Generation Sequencing on tissues and cell line samples. The authors also thank Dr. Gabriele Pogliaghi for assisting in late passage lines genetic validation.
Funding Information
The present study was supported by the Italian Ministry of Health—Ricerca Corrente 05C212 (P.I.: L.F.) and 05C402 (P.I.: G.V.) and by European Thyroid Association (ETA) Research Grant for Basic Science 2025—MINERVA 05A501 (P.I.: V.G.).
Availability of Data and Materials
Raw data, NGS FASTQ, and uncropped western blot gels have been deposited at Zenodo doi: 10.5281/zenodo.15594056.
The primary cell lines generated in this study are available upon reasonable request, depending on the availability of aliquots for each cell line.
Author Disclosures Statement
All authors declare no competing interests.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
