Abstract
Background:
Treatment of patients with thyroid cancer with Na[131I]I is routinely performed with empirical activity levels. Treatment success may be expected to correlate with the absorbed doses delivered to targets (thyroid remnants or metastatic lesions), but no systematic review or meta-analysis of absorbed dose–effect relationships has yet been performed.
Methods:
A systematic review and meta-analysis of reports published before August 22, 2025, was performed using PubMed, Web of Science, and OVID MEDLINE. Studies were included if they reported the proportion of patients achieving successful outcome as defined in individual publications and the absorbed doses delivered to targets. The study is registered with PROSPERO (CRD42024554956).
Results:
In total, 3723 studies were identified of which 18 were eligible for analysis. Number of patients in the included studies ranged from 4 to 509. For patients treated with Na[131I]I for thyroid remnant ablation, the reported success rates ranged from 60% to 100%, while lower success rates of 43–58% were found for patients with metastatic lesions. Success rates for patients with a thyroid remnant absorbed dose of 300 Gy or more ranged from 78% to 96%, while patients with metastatic lesions receiving at least 80 Gy had success rates ranging from 46% to 98%.
Conclusions:
While individual studies have demonstrated the importance of absorbed doses from Na[131I]I for differentiated thyroid cancer, no conclusive absorbed dose–effect relationship has been established in this review. A lack of standardization of dosimetry methodologies and follow-up criteria in the studies obscures the relationship. Large-scale observational prospective studies are required to determine the absorbed doses required for successful personalized treatments of patients with thyroid cancer with Na[131I]I.
Introduction
The optimal treatment for patients with differentiated thyroid cancer (DTC) remains controversial. 1 Several biomarkers have been proposed to be predictive of clinical outcomes in patients with DTC treated with Na[131I]I following thyroidectomy such as postoperative thyroglobulin (Tg) levels,2–5 the amount of radioiodine administered (in MBq), 6 and the absorbed dose (in Gy) delivered to thyroid remnants or metastatic lesions.7–14
The most common approach to the treatment of thyroid cancer with radioiodine is to administer empirical activities. If dosimetry is employed, this is often based on a maximum-tolerated activity (MTA) approach15,16 where the activity given is based on normal organ toxicity limits such as the absorbed dose to the bone marrow. Klubo-Gwiezdzinska et al. 15 reported a higher rate of complete response for the MTA approach when compared with empirical treatment with a similar safety profile.
Treatment optimization based on thyroid remnant or lesional dosimetry is rarely performed. Studies have provided initial indications that successful thyroid remnant ablation is dependent on the absorbed doses delivered, although absorbed dose thresholds were not clear and inconsistent between studies.7–10 Similarly, absorbed dose thresholds for successful treatments have been reported for the treatment of metastatic lesions.11–14
A systematic review of the literature covering studies of absorbed dose–response relationships in DTC has to our knowledge not yet been performed. A similar systematic review and meta-analysis for patients treated with radioiodine for Grave’s disease found a clear absorbed dose–response relationship, which could facilitate personalized treatment planning of Grave’s disease patients. 17
The aim of this review was to investigate the evidence for a relationship between radiation dosimetry and treatment outcomes in patients treated with Na[131I]I (radioiodine) for thyroid cancer. The review focused on the absorbed doses delivered to the thyroid remnant, residual disease and/or metastatic lesions, and relationship with clinical outcome of patients.
Methods
Search strategy and selection criteria
A comprehensive systematic review of published studies was performed to evaluate the clinical outcomes of Na[131I]I (radioiodine) therapy for thyroid cancer patients with respect to measurements of the absorbed doses delivered to thyroid remnants, recurrence, or metastatic disease. The systematic review was registered on PROSPERO (CRD42024554956).
PubMed, Web of Science, and OVID MEDLINE were searched on July 28, 2023, following the principles and checklist provided by preferred reporting items for systematic reviews and meta-analyses. 18 Only English language publications and publications published before July 28, 2023, were included. Two further searches were performed before starting the data extraction on July 10, 2024, and during the peer review on August 22, 2025, to ensure no records have been missed since July 28, 2023. No restrictions on the type of study design were applied.
Databases were searched for the following terms: (“radioiodine” OR “I131” OR “I-131” OR “131I”) AND (“thyroid”) AND (“cancer”) AND (“dosimetry” OR “absorbed dose”) NOT (Benign). Study authors were not contacted, and trial registries were not searched.
Only studies that reported the absorbed doses to the thyroid remnant and/or metastatic lesions and outcome (rate of successful treatment as defined in individual studies) at follow-up were included. Studies were included if they reported results for adults diagnosed with thyroid cancer (using any recognized diagnostic criteria) who had undergone a near-total or staged thyroidectomy (hemithyroidectomy followed by completion thyroidectomy) and were treated with Na[131I]I. Studies were excluded if they only reported results for adolescents (under 18 years of age) or if patients had undergone a partial thyroidectomy only.
Studies obtained from the initial search were reviewed by two blinded reviewers (J.T. and G.D.F.) based on title and abstract. After the initial screening, results were collated and discrepancies between the selected studies were resolved as a joint decision by both reviewers and by inclusion of a third reviewer (I.M.).
The remaining studies were then assessed based on the full text of the publication by all three reviewers (J.T., I.M., and G.D.F.). Studies were excluded at this stage if they did not report the absorbed dose to the thyroid remnant or metastatic lesions or the outcome at the end of the follow-up.
Data analysis
For any included study, the following data were recorded in a Microsoft Excel spreadsheet independently by the three reviewers: main author, journal/edition/pages, year of publication, title, type of study, number of patients in study group(s), patient cohort (remnant ablation and/or metastatic patients), disease type, lesion size [mL or g], lesion location, pretreatment Tg [ng/mL], TNM staging, mean age [in years], percentage of patients being male or female, TSH preparation protocol, follow-up period [in months], absorbed dose to thyroid remnant [in Gy] (if reported), absorbed dose to metastatic lesions [in Gy] (if reported) or absorbed dose thresholds [in Gy], outcome data (percentage of successful treatment at reported follow-up time), inclusion of patients with previous Na[131I]I treatments, and details of low iodine diets. Data were recorded for subcohorts if publications included results for both remnant and metastatic lesions or if different activities were given to individual subcohorts in the study.
In addition, details of the dosimetry methodologies were extracted from all publications, including whether the following items were reported: traceability of radionuclide calibrators to national standard; imaging or nonimaging study; scatter correction type; attenuation correction type; dead time correction type; partial volume correction type; number and timing of data points; fitting procedure to extract time-integrated activity; volume determination technique, and absorbed dose algorithm.
Risk of bias was assessed by all three reviewers, and disagreement between the three reviewers was resolved by consensus. The majority of studies were case series, and the Critical Appraisal Checklist developed by the Joanna Briggs Institute 19 was used to assess the methodological quality of the included studies (for details, see Supplementary Data S1).
Results
A total of 3723 studies were identified for the systematic review of which 697 were excluded due to presentation of duplicate data. A further 2985 studies were excluded for not satisfying the eligibility criteria based on title and abstract. Of the remaining 41 studies, a total of 185,7,8,10–14,20–29 full-text articles were deemed eligible for the systematic review following independent analysis (Fig. 1). The excluded 23 studies are listed in Supplementary Table S1.
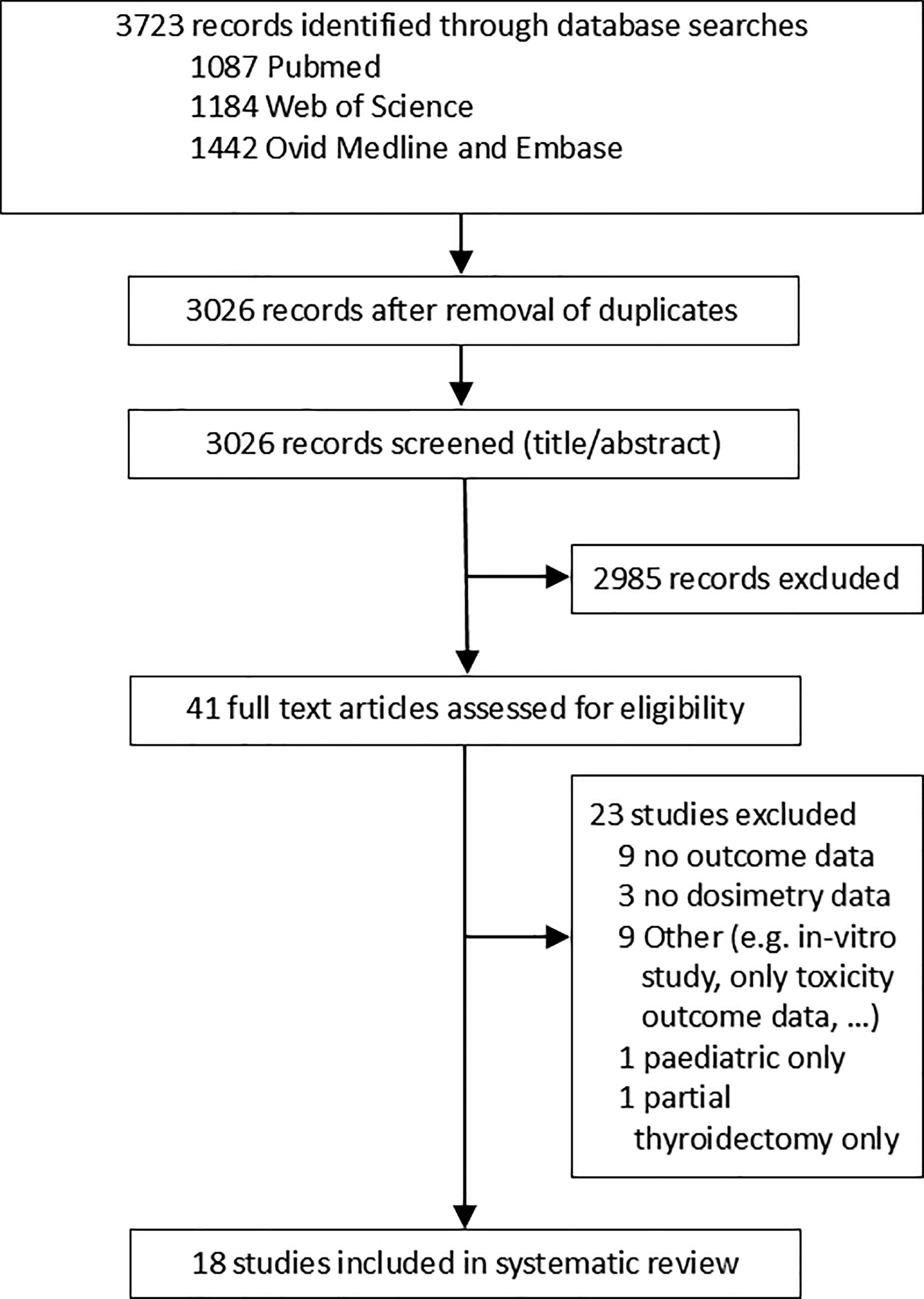
Flowchart for the systematic literature review.
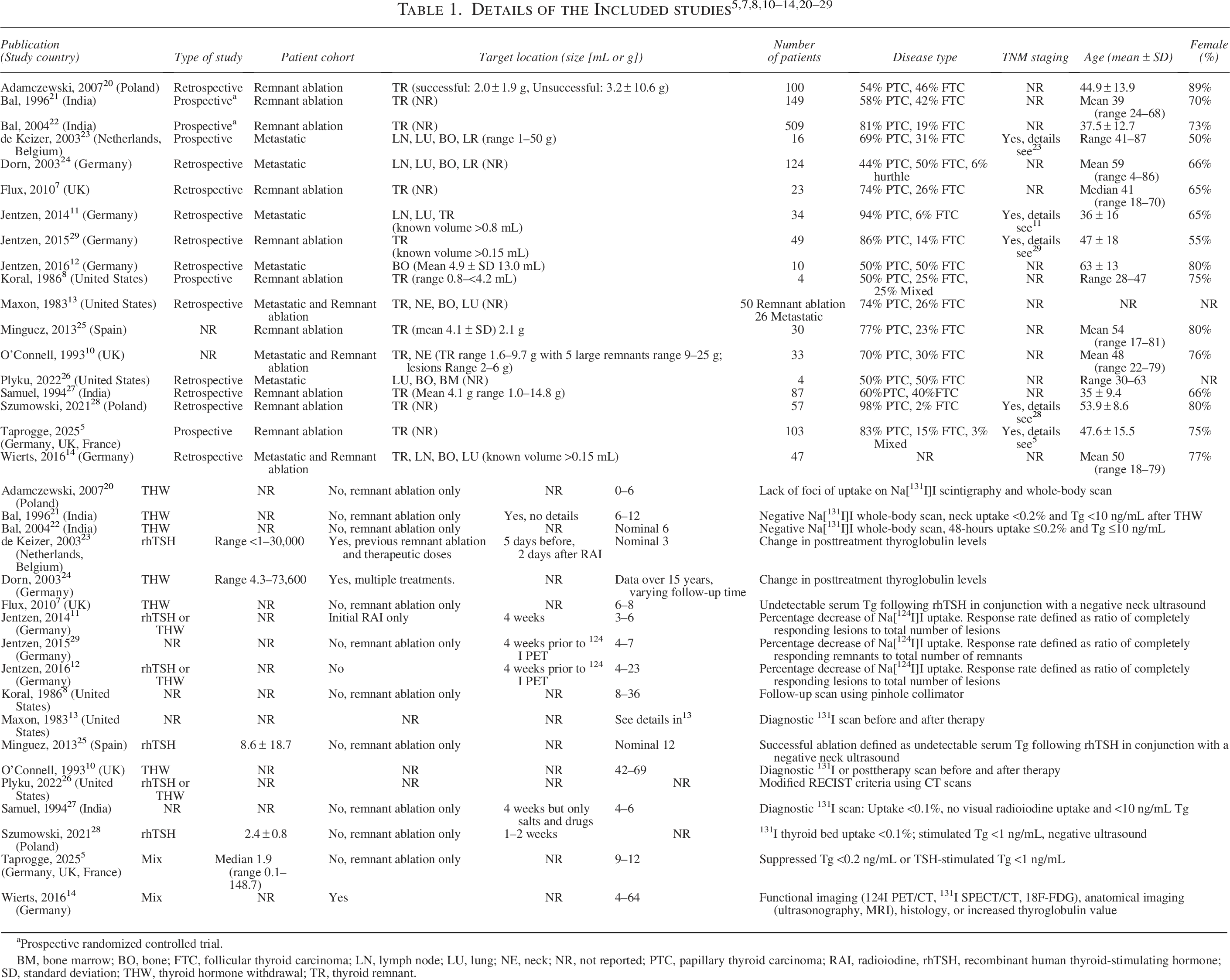
A summary of the study characteristics of the 18 included studies5,7,8,10–14,20–29 is presented in Table 1. Ten studies5,7,8,20–22,25,27–29 reported results from a remnant ablation patient cohort, three studies10,13,14 reported results for a mixture of remnant ablation and metastatic patients, and five studies11,12,23,24,26 considered only patients with metastatic disease. The number of patients included in individual studies ranged from 4 to 509 and from 4 to 124 for remnant ablation and metastatic cohorts, respectively. Table 1 also includes an overview of the different definitions of successful outcome used in the studies.
aProspective randomized controlled trial.
BM, bone marrow; BO, bone; FTC, follicular thyroid carcinoma; LN, lymph node; LU, lung; NE, neck; NR, not reported; PTC, papillary thyroid carcinoma; RAI, radioiodine, rhTSH, recombinant human thyroid‐stimulating hormone; SD, standard deviation; THW, thyroid hormone withdrawal; TR, thyroid remnant.
Dosimetry methodologies for each study are detailed in Table 2. Methodologies for thyroid remnant dosimetry in earlier publications mostly used planar imaging, rectilinear scanners, and probe measurements. Later publications have introduced imaging with SPECT or PET/CT. A similar trend is seen for lesional dosimetry with a move from planar or rectilinear imaging to mostly PET/CT in more recent studies. Imaging time points, corrections applied to ensure scans are quantifiable, volume determination techniques, and fitting methodologies vary between studies and are often not reported.
Dosimetry Methodologies Used in the Publications Ordered by Year of Publication
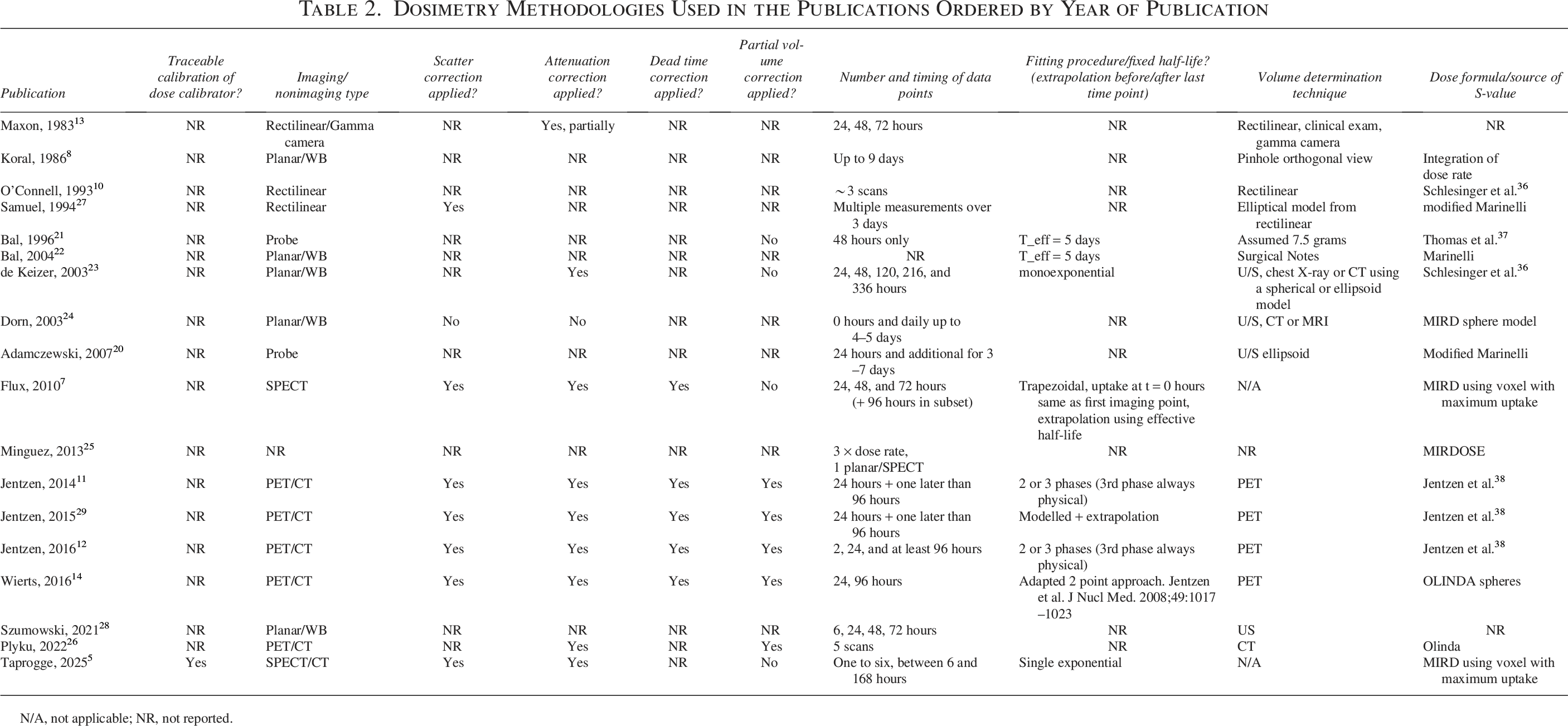
N/A, not applicable; NR, not reported.
The Critical Appraisal Checklist developed by the Joanna Briggs Institute can be found in the Supplementary Data S1. The checklist was extended by one additional question to assess the comparability of methodologies in the present studies. The results of the critical appraisal are summarized in Figure 2. All studies were either classed as having an intermediate or high risk of bias due to the lack of reporting of inclusion criteria in the studies and because treatment methodologies did not match other studies with respect to the dosimetry methodologies used and the assessment of treatment outcome.
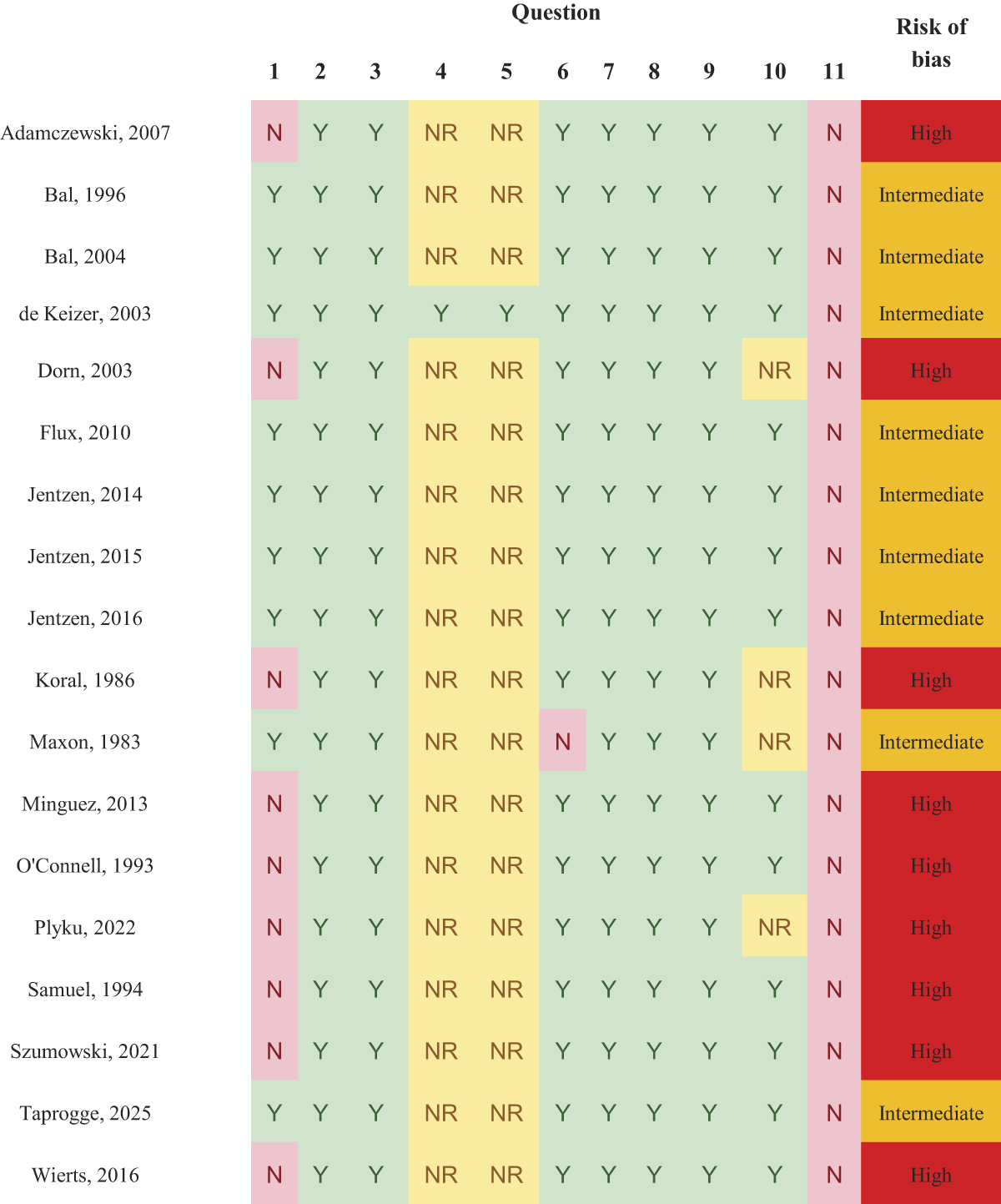
Risk of bias assessment following the Joanna Briggs Questionnaire (including additional question 11). Details of the questionnaire can be found in Supplementary Data S1.
Table 3 summarizes the administered activities, reported absorbed doses to targets (e.g., thyroid remnants or metastatic lesions), and the percentage of patients with successful treatment, as defined in each study, at the final follow-up.
Summary of Reported Administered Activities, Absorbed Doses Delivered to Targets, and the Percentage of Successful Treatments as Defined in the Respective Publications
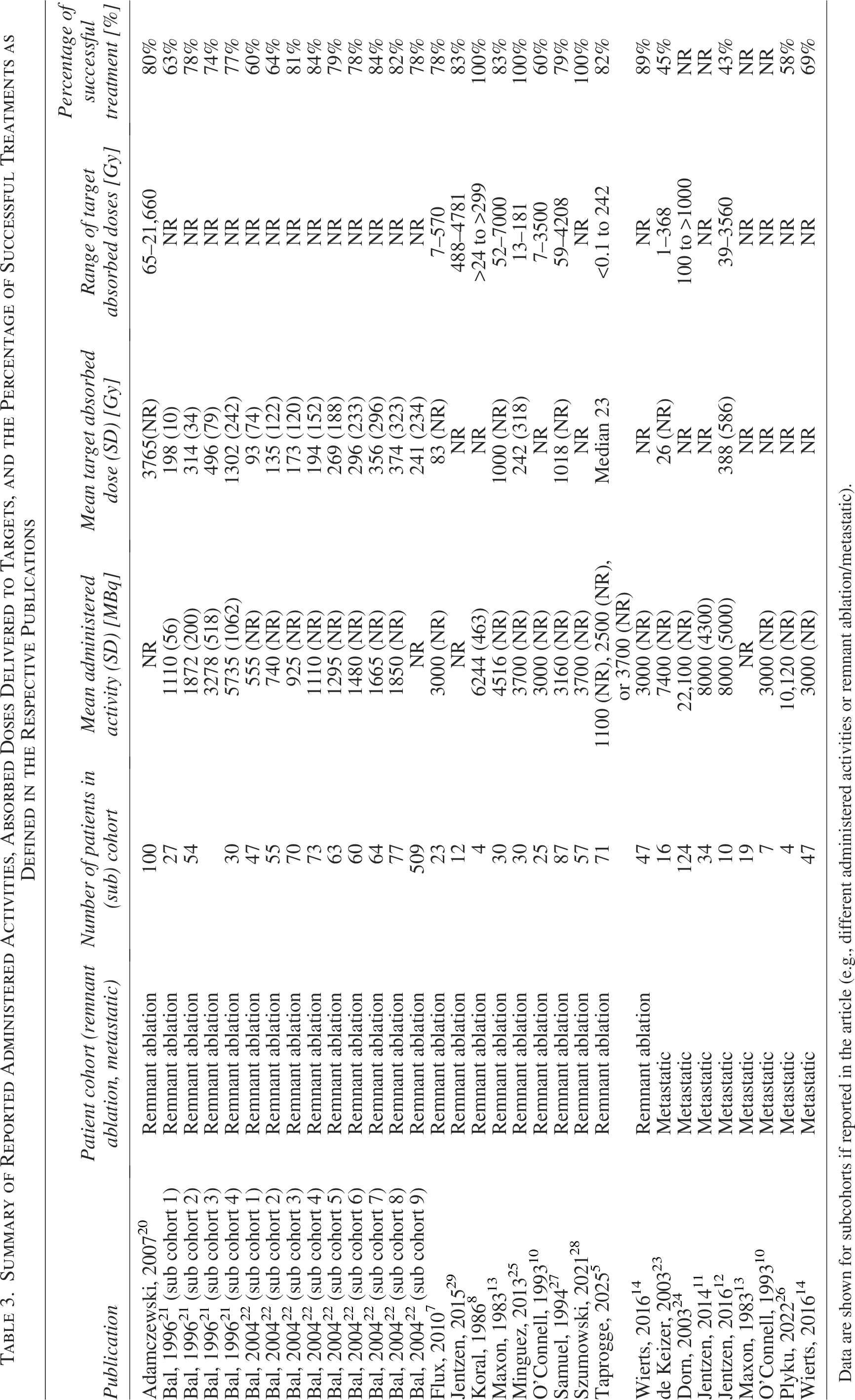
Data are shown for subcohorts if reported in the article (e.g., different administered activities or remnant ablation/metastatic).
Figure 3 shows the reported success rate as a function of the mean absorbed dose reported for the full patient cohort or subcohorts (if reported) in individual studies. Eight5,7,13,20–22,25,27 studies that report results for remnant ablation cohorts have included both the mean absorbed dose to the thyroid remnant and the percentage of successful treatments. In contrast, only two metastatic cohorts12,23 can be included in Figure 3 as the other studies only report success rates above absorbed dose thresholds defined in individual studies. The proportion of patients with successful remnant ablation ranges from 60% to 100% with an apparent plateau around 80%. One study reports a success rate of 100%. Reported success rates for metastatic patient cohorts are lower, ranging from 43% to 45% in Figure 3.
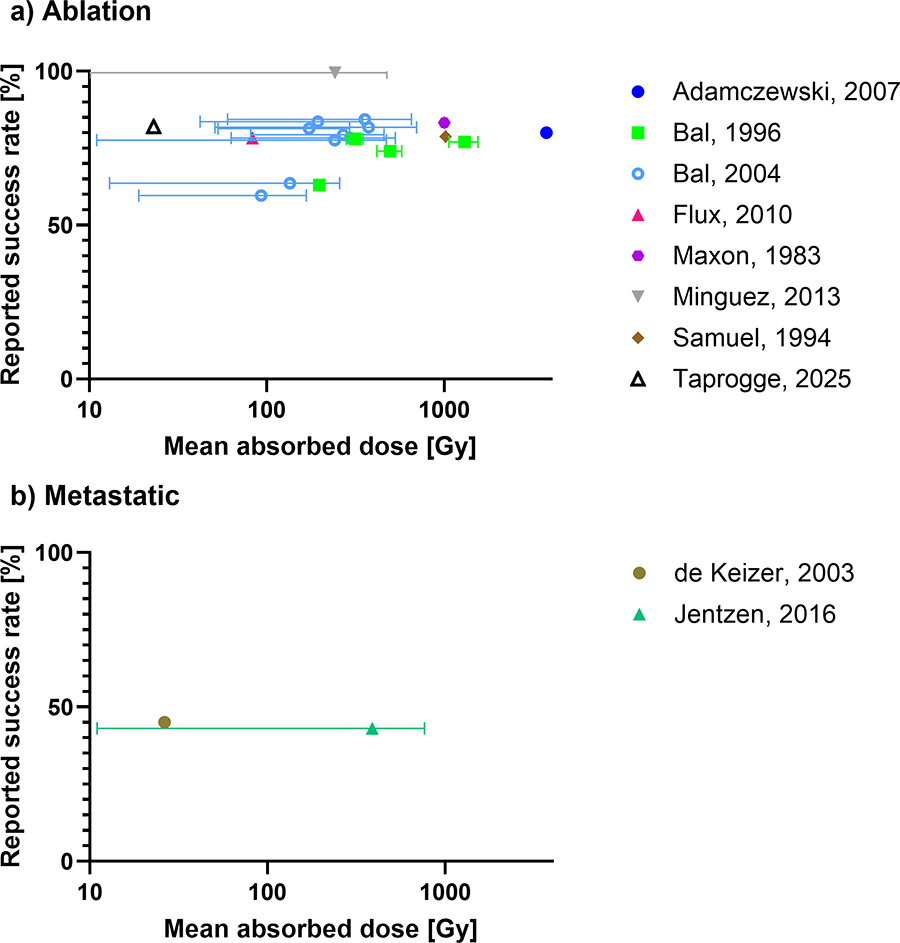
Reported success rate (according to success criteria defined in publication) against the mean (or median if mean not reported) absorbed dose reported for patient cohorts with
Both for remnant ablation and metastatic patient cohorts, Figure 3 does not appear to show the expected sigmoidal dose–effect relationship between the absorbed dose delivered and the proportion of patients with successful outcome.
In 105,7,10–13,20,26,27,29 out of the 18 studies, success rates were reported above absorbed dose thresholds summarized in Table 4. These absorbed dose thresholds were based on criteria defined in the individual studies and can probably not easily be compared. The absorbed dose thresholds for remnant ablation treatments ranged from 49 Gy with a 100% success rate to 4000 Gy with a success rate of 92%. At the absorbed dose threshold of 300 Gy, which was initially proposed by Maxon et al., 13 the success rate in different studies varies from 78% to 96%. For metastatic lesions, absorbed dose thresholds between 80 and 650 Gy were reported. At the absorbed dose threshold of 80 Gy, as proposed by Maxon et al., 13 the success rate in different studies varies from 46% to 98%. Reported absorbed dose thresholds and the corresponding proportion of patients with successful treatment are presented in Figure 4.
Reported Absorbed Dose Thresholds and Corresponding Success Rate Above the Absorbed Dose Threshold

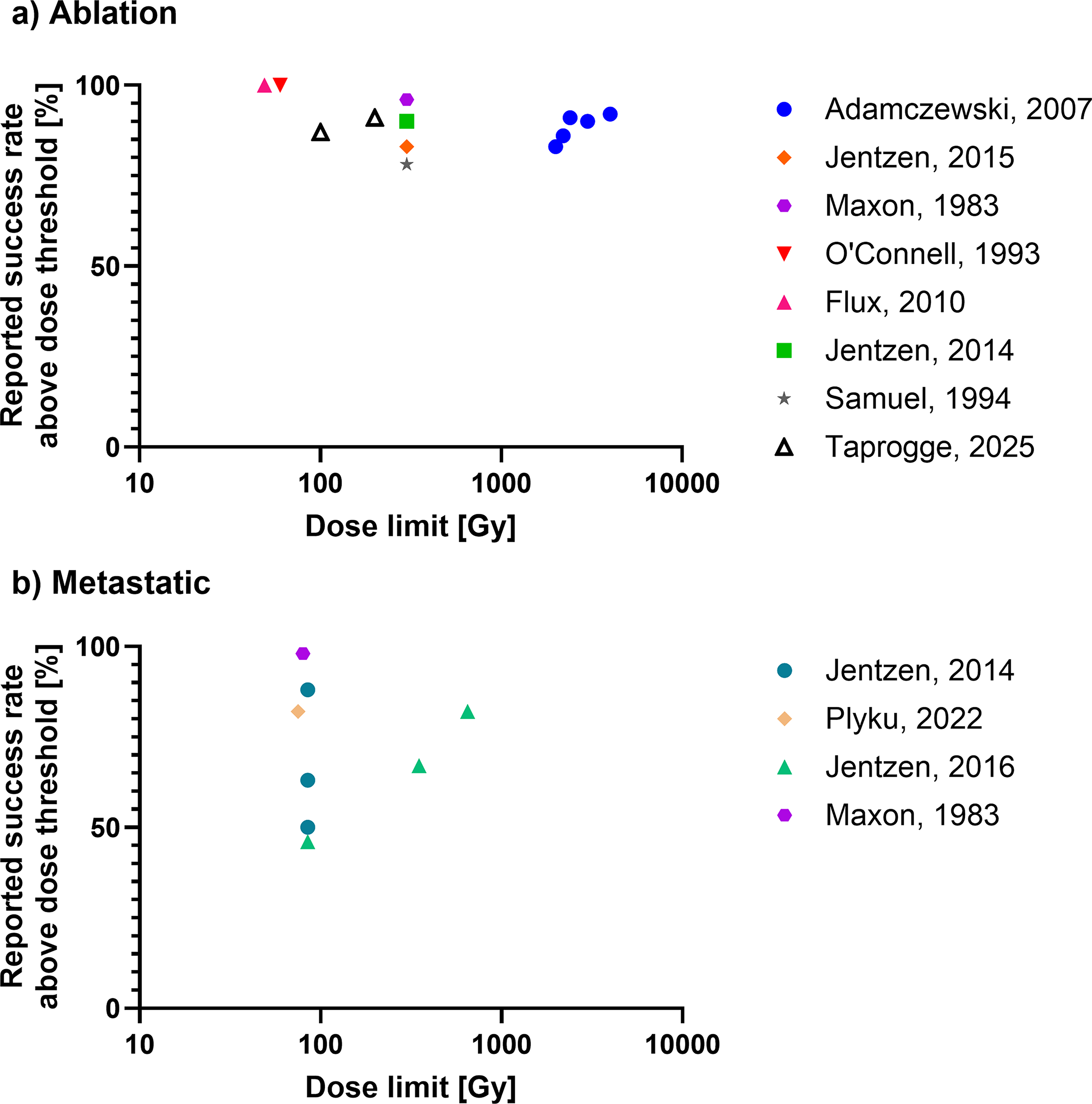
Reported absorbed dose thresholds in publications and corresponding success rate above absorbed dose threshold for patient cohorts with
Discussion
The aim of this review was to assess evidence for absorbed dose–response relationships for Na[131I]I treatments of DTC. While the review fails to find relationships for either ablation treatments of the thyroid remnant or therapy administrations for patients with recurrence or metastatic disease, the results of each individual study5,7,8,10–14,20–29 show that absorbed dose is a predictor of treatment response. Individual studies included in this review show absorbed dose–response relationships, but the data cannot be collated. A confounding factor that may explain this is that the reviewed studies present results from a small number of studies and patients treated over nearly four decades. Dosimetry methodologies and tools have evolved as have surgical techniques and clinical management approaches.1,30
The risk of bias assessment showed that all studies have an intermediate or high risk of introduction of a bias when collating the results due to differences in dosimetry methodologies, and their respective errors and uncertainties, and outcome assessment. The definition of treatment success and the respective follow-up time in individual studies in the present systematic review vary between studies and even in studies with similar dosimetry methodologies, the outcome criteria vary. There is a lack of consistent or standardized methodology to classify treatment response for metastatic disease and ablation treatments (see Table 1). Imaging assessment of therapy response on an individual lesion level has been used as biomarkers, particularly thyroglobulin, which does not give information relating to individual lesions.
In contrast to a previous systematic review of Na[131I]I for the treatment of Graves’ disease, 17 administered activities in the included studies were not prescribed to achieve a fixed target absorbed dose. In individual studies, a wide range of absorbed doses was delivered to targets (see standard deviations reported in Fig. 3). This likely results in the lack of a demonstrable absorbed dose–response relationship in Figure 3.
Four studies11–13,26 have shown that treatment success rates in patients with metastatic disease are significantly higher if lesions were treated with absorbed doses higher than 80–85 Gy. Nevertheless, this review indicates that reported success rates are not consistent between studies. For example, a value of 80 Gy was proposed by Maxon et al. as an absorbed dose threshold 13 and has subsequently been utilized by others as a reference for comparison, although this has resulted in varied success rates from 46% to 98%. Jentzen et al.11,12 have demonstrated that success rates may depend on the type/location of lesion, as success rates vary between lesions in bone, lungs, or lymph nodes. A further possible confounding factor is variations in radiosensitivity, whether due to genetic differences in the tumors themselves, the microenvironment, or other factors such as localized hypoxia. Genomic profiling of the tumor may be used for stratification or subcohort analysis in future trials and could potentially predict radiosensitivity and allow for personalized treatment planning. Other factors such as lesion size or histological subtype should also be considered when assessing absorbed dose–response relationships. As shown in Table 1, there are a range of potential confounding biological factors such as lesion size, pretreatment Tg levels, and lesion location that might explain the wide range of response rates observed.
The absorbed dose thresholds reported for thyroid remnant ablations vary from 49 to 4000 Gy and are, therefore, not currently clinically useful. While some of these differences might be due to variations in dosimetry methodologies, as discussed below, there is also a possibility that this may be due to changing practice of surgical management. 30 The studies included here were published from 1983 until 2025.
The reported success rates in the remnant ablation cohorts are in many cases lower than the success rates reported following empirical administration in recent large-scale prospective studies such as ESTIMABL2 and HILO of 96% 31 and 98% 32 at 3 years follow-up. A potential reason for this is the change in surgical and clinical management of patients from 1983 to today. Two recent studies by Wierts et al. 14 and Szumowski et al., 28 published in 2016 and 2021, had reported success rates of 89% and 100%, respectively, in line with the success rates reported in ESTIMABL2 and HILO. As shown in Table 1, the thyroid remnant volumes are likely larger than expected after a modern total thyroidectomy, potentially explaining the lower success rates. In addition, a small number of patients with incomplete thyroidectomy are included in some of the studies, which is a limitation of the present work.
As shown in Table 2, a wide range of dosimetry methodologies has been used. Imaging technology has evolved over time from rectilinear scanners to gamma cameras and 124I-PET for metastatic patients. In addition, a range of imaging and nonimaging techniques has been applied for remnant ablation studies. A major contribution to the dosimetry uncertainty is the target volume uncertainty, which is a particular issue in remnant ablations but also affects metastatic lesions. The thyroid remnant volume cannot accurately be assessed using PET, SPECT, ultrasound, CT, or MRI. Therefore, the absorbed dose delivered to the voxel with maximum uptake was calculated in some studies.5,7 Furthermore, a part of the thyroid remnant that is rarely removed in thyroid cancer surgery is accessory thyroid tissue such as the pyramidal lobe. 33
Validation of the accuracy of dosimetry methodologies was not consistently performed for the studies, and quantification of the respective errors and uncertainties is not retrospectively possible.
A further limitation is the small number of studies available for both remnant ablation and metastatic patients and the low number of patients included in many of the studies, ranging from 4 to 509 patients. Potential limitations of the present work are the exclusion of non-English studies and search strategy constraints due to the search terms used. In addition, the limited criteria for inclusion and exclusion of studies could have introduced heterogeneity.
A subset of low-risk patients has been shown not to benefit from Na[131I]I ablation of thyroid remnant tissue in the ESTIMABL2 31 and IoN 34 studies. Further work is required to establish the potential benefits for dosimetry in cohorts that still undergo Na[131I]I ablation, where a more tailored approach could potentially lead to patients receiving less Na[131I]I and therefore reducing absorbed doses to normal organs.
Dosimetry-driven treatment is likely to be of particular benefit for patients with metastatic disease where total eradication of target cells is observed less frequently compared with complete ablation of normal thyroid remnants. In addition to potentially achieving higher success rates due to treatment personalization, dosimetry would allow for organs-at-risk to be considered and potentially reduce activities if needed to reduce treatment-related toxicities. Pretherapy dosimetry using Na[123I]I has been shown to predict absorbed doses delivered after Na[131I]I therapy 35 in metastatic patients and could therefore be used for treatment planning. Pretherapy dosimetry uncertainties for lesions and healthy tissues would likely be smaller when using Na[124I]I PET pretherapy dosimetry.
The current review highlights the need for a large-scale multicenter clinical study to assess the radiation dosimetry with a view to treatment personalization, which will require standardization of methodologies used. This review emphasizes the urgent need for validated dosimetry methodologies and for internationally agreed response assessment criteria.
Conclusions
In this systematic review, no consistent relationship could be demonstrated between absorbed doses and outcomes in the treatment of thyroid cancer using Na[131I]I, due to inconsistent methodologies and a paucity of studies. Nevertheless, individual studies show the potential of radiation dosimetry and the potential for personalized treatment as absorbed dose was found to be a predictor of treatment response in many studies. Comprehensive and standardized data collection will form the basis for future studies, which are required to determine the clinical efficacy and cost-effectiveness of dosimetry-based patient-specific treatment planning.
Authors’ Contributions
J.T., I.M., K.N., K.G., J.W., and G.F. conceived the design of the systematic review. J.T., I.M., and G.F. did the abstract and full-text screening. J.T., I.M., and G.F. did the data extraction. J.T. performed the data analysis. J.T., I.M., and G.F. drafted the original article. All authors contributed to the edit and review of the final article.
Footnotes
Funding Information
NHS funding was provided to the National Institute for Health and Care Research (NIHR) Biomedical Research Centre at The Royal Marsden and the ICR. The RTTQA group was funded by the NIHR. This report is an independent research funded by the NIHR. The views expressed in this publication are those of the authors and not necessarily those of the NHS, the NIHR, or the Department of Health and Social Care.
Author Disclosure Statements
J.T., I.M., K.G., and G.D.F. have nothing to disclose with respect to the work carried out here. K.N. and J.W. have received speaker honoraria from Eisai.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.