Abstract
Background:
Free thyroxine (FT4) reference intervals (RIs) provided by many laboratories do not adequately represent the differences in FT4 levels observed across age groups, limiting their usefulness in the diagnosis and management of disease, most particularly at the extremes of age. Interpretive criteria specific to neonates, young children, and older adults are rarely provided. This work was undertaken to develop comprehensive age-based RIs from birth to age 100 to provide clinicians with precise context for result interpretation.
Methods:
RIs were calculated through multi-modal decomposition (MMD) analysis performed on de-identified retrospective FT4 results from specimens submitted for routine testing by direct dialysis at a commercial reference laboratory. Intervals were validated using a separate data set. The study population for MMD analysis included individuals from age 0 days to 100 years who submitted specimens for FT4 testing.
Results:
A total of 1,862,273 results were included in the analysis. MMD analysis yielded 14 distinct RIs by age. FT4 intervals were broadest, with higher upper reference limit (URL) and lower reference limit (LRL) at birth, narrowing toward adult ranges throughout childhood. A mild increase in the URL was observed in older adults.
Conclusions:
The development and validation of FT4 RIs provides interpretive criteria for FT4 results for patients throughout the lifespan. By providing RIs for distinct neonatal, pediatric, and adult age groups, this work enables clinicians to evaluate FT4 results in the appropriate context, allowing more accurate classification of abnormal results.
Introduction
Laboratory testing plays an essential role in the diagnosis of thyroid disease. In context with thyrotropin (TSH), measurement of FT4 provides an assessment of thyroid gland function. Although clinical presentation is helpful in guiding a clinician toward the thyroid as the cause of disease, to definitively establish a diagnosis of hypo- or hyperthyroidism, the clinician must compare a patient’s results against the reference interval (RI).
Children require RIs distinct from adults, and several intervals are often required to accurately reflect the distribution of expected results in a healthy pediatric population. While consenting adults volunteer to contribute their biological material toward the development of RIs, asymptomatic children usually do not have the occasion to get blood drawn, and the smaller the child, the more significant the impact of phlebotomy. For these reasons, scientists have sought out other methods for determining pediatric RIs. As an alternative, indirect statistical methods may be applied to large data sets of laboratory test results to ascertain an RI. The multi-modal decomposition (MMD) is an iterative indirect method that decomposes a mixture of multiple normal distributions into separate components using the expectation-maximization (EM) algorithm. The objective of the current study was to apply MMD to ascertain RIs for FT4 for the entire life span.
Methods
Cohort characteristics: Indirect analysis
The de-identified data used for MMD analysis constitute a limited data set (https://www.hhs.gov/hipaa/for-professionals/special-topics/research/index.html) with only patient sex and age information retained. Meta-analysis using our laboratory population de-identified data was approved by an IRB-consented protocol (WCG IRB # 1-1746845-1). Results from 1,862,273 patients (aged 0 days to 100 years) whose specimens were collected over the course of several years from locations across the United States and submitted to Quest Diagnostics for FT4 testing by equilibrium dialysis were retrospectively analyzed to establish RIs using MMD. FT4 testing by equilibrium dialysis is performed at two laboratories within the Quest Diagnostics network (San Juan Capistrano, CA, and Chantilly, VA, laboratories). Most results were consecutively and recently obtained. However, given the small number of results for individuals less than 1 year of age, it was necessary to retrieve results from a broader period to achieve an appropriate number of data points for analysis for that age group.
Cohort characteristics: validation cohort
For adults between the ages of 18 and 64, specimens were obtained from 132 ambulatory, community-dwelling, and nonmedicated adults employed at Quest Diagnostics San Juan Capistrano. Individuals with abnormal TSH or a history of thyroid disease were excluded. Sample collection was approved by an IRB-consented protocol (WCG IRB #20121940).
The remainder of the results for the validation cohort were from randomly collected and reanalyzed remnant specimens that were de-identified with only patient sex and age information retained. Remnant specimens had been originally submitted for non-thyroidal testing. All specimens (850 in total) were screened to confirm a TSH within the RI before consideration for inclusion in the validation cohort.
FT4 analysis
Testing was performed by equilibrium dialysis, which separates protein-bound T4 and FT4 first, before FT4 quantitation as previously described. 1 FT4 was directly measured by radioimmunoassay (RIA) (before 2017) or LC-MS/MS (liquid chromatography—tandem mass spectrometry) (after 2017). The limit of quantitation of both assays was determined to be 0.2 ng/dL. The analytical measurement range and clinical reportable range are 0.2–12.8 ng/dL. Method comparison by Deming regression demonstrated no clinically significant difference between RIA and LC-MS/MS (intercept = 0.004 [CI: −0.34–0.33]; slope = 1.16 [CI: 0.9–1.4]). The median yearly trend for FT4 for newborns is provided in Supplementary Figure S1.
MMD Data analysis
The MMD method is designed to discern subgroups of distribution within the mixed population without manually excluding data from unhealthy patients, which does not require specimens to be submitted from a presumed healthy control population. The normalmixEM function from the mixtools package in R was used to analyze the data. 2 No patient results were excluded in this analysis. Results reported as <0.2 ng/dL were converted to 0.19 ng/dL and results reported as >12.8 ng/dL were converted to 12.81 to allow for numerical analysis, accounting for 0.2% and 0.1% of all results, respectively. The FT4 median trend by age and patient sex was examined to determine the optimal age breaks for RI analysis.
Validation
Proposed RIs were validated following the Clinical Laboratory Standards Institute (CLSI) protocol for verification of RIs. 3 From the broader validation cohort, which included 850 specimens in total, 20 results were randomly sampled to validate each age-specific interval. The RI was considered validated if ≤2 results fell outside of the proposed interval.
Results
The final sample size for the ascertainment of indirect FT4 RIs was 1,862,273. More specimens from female (71%) than male (29%) patients were received. 784,449 results were obtained using the RIA method, and 1,077,824 results were obtained using the LC-MS/MS method. MMD analysis was used to generate RIs. The sample size used to generate the RI for each age group varies and is provided in Table 1. The RIs for the youngest individuals had the least data, with less than 10,000 results per MMD analysis. Intervals from age 10 through age 90 were computed from over 50,000 results per interval. RIs are based on combined data inclusive of both sexes. (Separate intervals based on patient sex are presented in Supplementary Table S1).
Reference Intervals for FT4 by Direct Dialysis Proposed by MMD Analysis and Reference Interval Validation Data
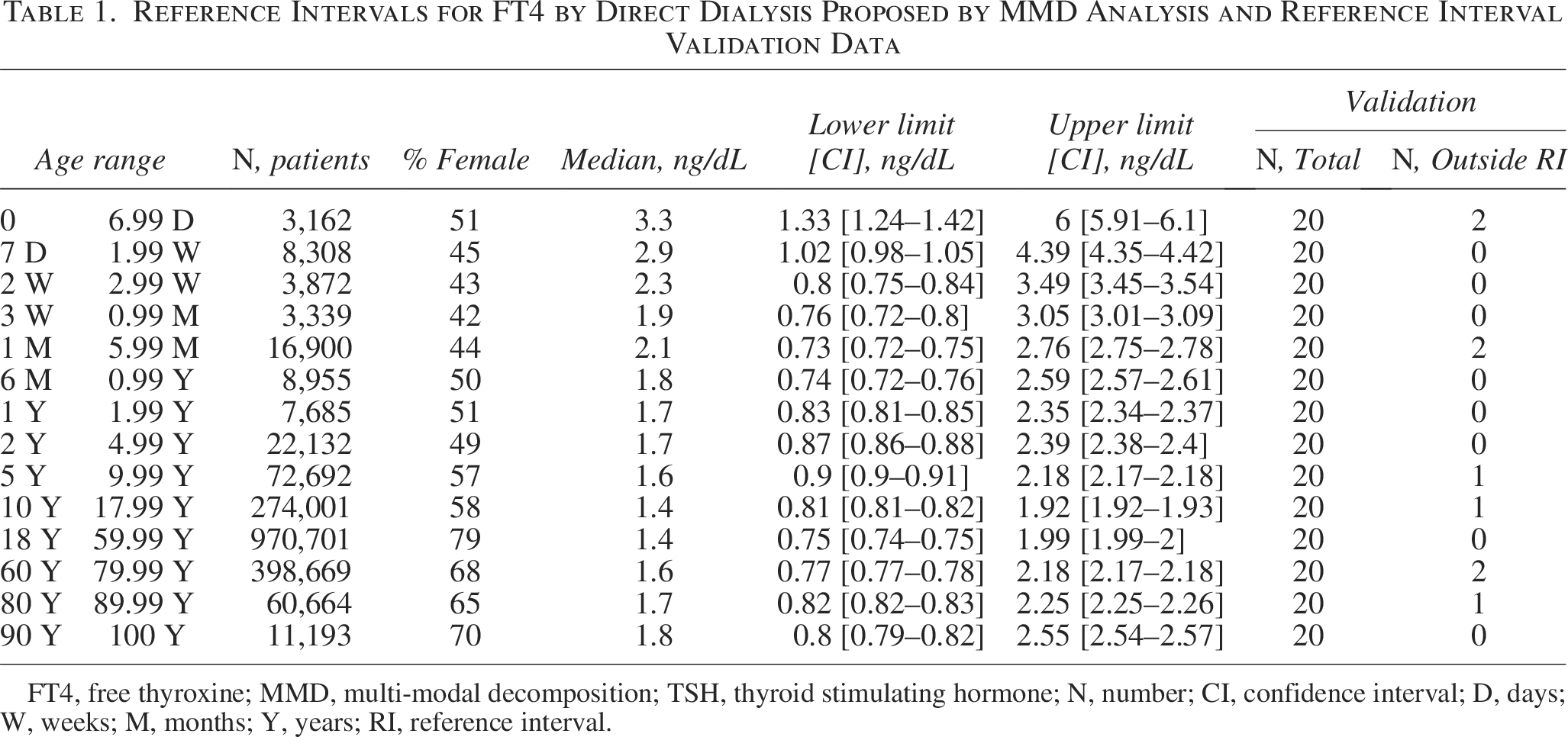
FT4, free thyroxine; MMD, multi-modal decomposition; TSH, thyroid stimulating hormone; N, number; CI, confidence interval; D, days; W, weeks; M, months; Y, years; RI, reference interval.
RIs were broadest at birth, with URL and LRL higher than what is observed in older age groups. RIs narrowed sharply from birth to age one year, with more modest narrowing through the rest of the pediatric intervals and more uniform adult ranges (Fig. 1). The URL leveled at around 2.0 ng/dL from age 10 up to age 60 and showed a slight trend upwards past age 60. The LRL was highest in the youngest patients and declined quickly toward adult values after two weeks of age. The LRL showed minimal variability above age 2 months. The validation data sets confirmed the RIs established through MMD. (Fig. 1 and Table 1).
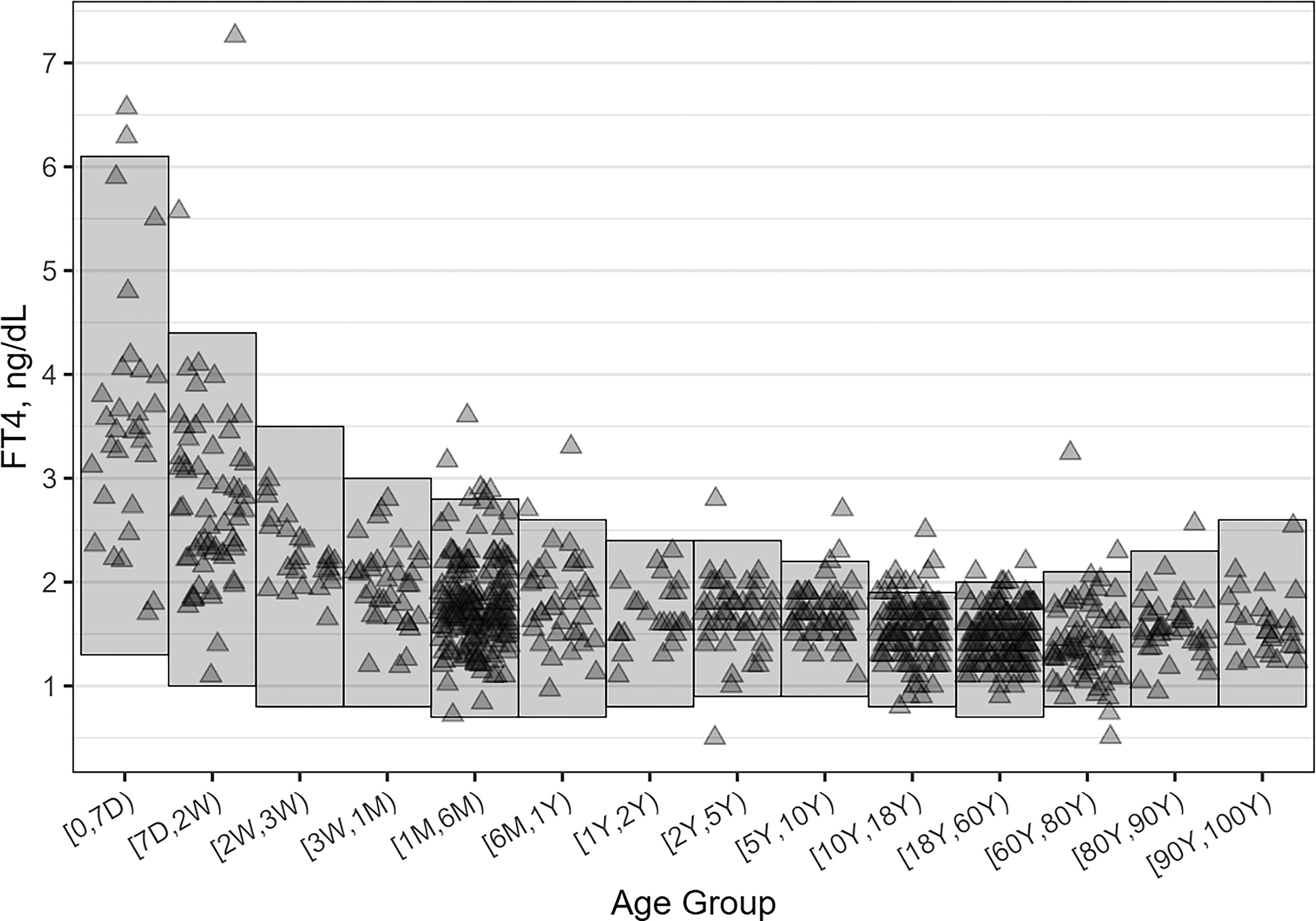
MMD derived reference intervals with data points of validation cohort results overlapped: shaded rectangles represent the RIs calculated for each age subgroup, and the triangles are the FT4 values for each of the 850 results in the validation pool, which were then randomly sampled for RI validation. MMD, multi-modal decomposition; RI, reference interval; FT4, free thyroxine; D, days; W, weeks; M, months; Y, years.
Discussion
A recent review that included many published RIs for directly measured FT4 found that (a) many studies did not consistently capture the neonatal surge in FT4, (b) did not cite the number of individual results used for RI determination, and (c) were inconsistent in the RI age grouping. 4 Furthermore, 7 of 9 clinical laboratories surveyed that compared pediatric RIs for TSH and FT4 from the same laboratory found inconsistencies in age grouping, suggesting that the RIs did not appropriately reflect the changing hypothalamic-pituitary-thyroid physiology as children age. 4 Our pediatric RIs show general agreement to directly ascertained FT4 direct dialysis RIs.5,6 However, an exact comparison cannot be made due to differences in the age cutoffs used. Soldin and colleagues analyzed results from 1426 specimens to directly ascertain two pediatric intervals: 1.3–2.8 ng/dL (ages 1–12 months) and 1.3–2.4 ng/dL (ages 1–18 years). 5 La’ulu and colleagues included 2213 results in their study to directly ascertain the following intervals: 1.40–2.70 ng/dL (ages 6 months to 6 years) and 1.10 to 2.0 ng/dL (ages 7–17 years). 6 Compared to the RIs recently published by Jansen and colleagues for ages 2 and up based off immunoassay results from various manufacturers, our RIs have a lower LRL, a lower URL, and are narrower. 7
Our adult RIs show a stable LRL with an increasing URL after age 60. This is a pattern that has been observed in other indirect studies, which break down RIs by age group throughout adulthood.7,8 However, there are several indirect studies that did not observe a change in the URL with increasing age.9–11 These differences may be attributable to variability in the population being studied, assay methodology, and/or the statistical tools used for analysis. Improved harmonization and standardization among FT4 assays are needed before RIs obtained using one method can be applied to other methods.12–14
FT4 by equilibrium dialysis is indicated in specific situations where FT4 by immunoassay may be inaccurate due to qualitative or quantitative abnormalities in binding proteins, as may occur in certain illnesses or due to medication. Interpreting patient results against age-specific RIs supports accurate diagnosis and monitoring for patients of all ages. The same statistical techniques applied in this study to establish indirect RIs for FT4 by direct dialysis could be used to ascertain RIs for other clinically significant thyroid-related analytes, including TSH and free T3.
Authors’ Contributions
C.B.: Conceptualization (equal); data curation (lead); formal analysis (lead); methodology (lead); visualization (lead); writing—original draft (supporting); writing—review and editing (supporting). E.S.: Conceptualization (supporting); writing—original draft (lead); writing—review and editing (lead), E.L.: Investigation (supporting). M.S.: Investigation (supporting). M.M.: Conceptualization (supporting). Writing—review and editing (supporting). E.W.: Conceptualization (supporting); writing—review and editing (supporting). N.C.: Conceptualization (supporting); writing—review and editing (supporting). Z.W.: Conceptualization (equal); investigation (lead); project administration (lead); data curation (supporting), visualization (supporting); writing—original draft (supporting); writing—review and editing (supporting). All authors have reviewed the final version of the article and accept responsibility for the content.
Footnotes
Funding Information
This study was funded by Quest Diagnostics.
Author Disclosure Statements
C.B.: Employee of Quest Diagnostics and receives employee salary. No other financial support or interests for the past 36 months. No patents to declare. No other relationships, interests, or activities from the past 36 months that relate to or could be perceived by others to influence the submitted work in any way. E.S.: Employee of Quest Diagnostics and receives employee salary. No other financial support or interests for the past 36 months. No patents to declare. No other relationships, interests, or activities from the past 36 months that relate to or could be perceived by others to influence the submitted work in any way. E.L.: Received no financial support related to this work. No other financial support or interests for the past 36 months. No patents to declare. No other relationships, interests, or activities from the past 36 months that relate to or could be perceived by others to influence the submitted work in any way. M.S.: Employee of Quest Diagnostics and receives employee salary. No other financial support or interests for the past 36 months. No patents to declare. No other relationships, interests, or activities from the past 36 months that relate to or could be perceived by others to influence the submitted work in any way. M.M.: Employee of Quest Diagnostics and receives employee salary. No other financial support or interests for the past 36 months. No patents to declare. No other relationships, interests, or activities from the past 36 months that relate to or could be perceived by others to influence the submitted work in any way. EW.: Employee of Quest Diagnostics and receives employee compensation and stock options. Receives royalties from Pediatric Reference Intervals, 8th edition (Elsevier) and royalties from Biochemical and Molecular Basis of Pediatric Disease, 5th edition (Elsevier). No other financial support or interests for the past 36 months. No patents to declare. No other relationships, interests, or activities from the past 36 months that relate to or could be perceived by others to influence the submitted work in any way. N.C.: Employee of Quest Diagnostics and receives employee salary. No other financial support or interests for the past 36 months. No patents to declare. No other relationships, interests, or activities from the past 36 months that relate to or could be perceived by others to influence the submitted work in any way. Z.W.: Employee of Quest Diagnostics and receives employee salary. No other financial support or interests for the past 36 months. No patents to declare. No other relationships, interests, or activities from the past 36 months that relate to or could be perceived by others to influence the submitted work in any way.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
