Abstract
Background:
No international consensus exists on the selection and reporting of outcomes after differentiated thyroid cancer (DTC) surgery, hindering assessment of new treatments and guideline formation. Therefore, we aimed to develop an international core outcome set (COS) to be measured and reported in future studies investigating surgical treatment for DTC, as well as in clinical practice.
Methods:
COS development consisted of three phases: (1) collecting an initial outcome list through a literature review, (2) a two-round international Delphi process with experts and patient representatives, and (3) international expert panel meeting to finalize the COS. A steering committee including experts from varying medical (sub-)specialties and a patient representative from the Dutch Thyroid Patient Organization advised on the study protocol, Delphi rounds, and expert panel meeting. Experts were identified through scientific associations, international guidelines on DTC, ClinicalTrials.gov, and last authors of key studies and suggestions from the steering committee. The outcomes from the literature review were presented in successive rounds to experts and patient representatives to assess their importance for inclusion in the DTC surgical COS. Delphi results were analyzed for each stakeholder group on a 1–9 Likert scale. Consensus-in was defined as 70% or more of participants in both stakeholder groups rating the outcome 7–9 or 90% in one group. Consensus-out was defined as 70% or more in both groups rating the outcome 1–3. Consensus-out required agreement across both groups.
Results:
A total of 125 experts and 7 patient representatives from 35 countries across 5 continents completed all rounds. After two rounds, consensus was reached for 17 outcomes. Of these, 13 outcomes were ratified during the expert panel meeting: recurrence, persistent disease, location of metastatic lymph nodes, number of retrieved metastatic lymph nodes, postoperative thyroglobulin levels, surgical completeness, permanent recurrent laryngeal nerve paralysis due to surgery, permanent postoperative hypoparathyroidism, 30-day postoperative complication rate, accidental intraoperative injury to adjacent organ, unplanned reoperation rate, 30-day postoperative mortality, and quality of life.
Conclusions:
This international consensus on the COS for DTC surgery promotes consistent and appropriate outcome selection in clinical practice and research and may be incorporated into future study designs. Future steps include defining some outcomes.
Introduction
Differentiated thyroid cancer (DTC) is the most prevalent malignancy of the endocrine system, affecting a growing number of patients worldwide.1,2 Surgery is the mainstay of treatment and plays a central role in disease management. However, there is notable variation in the outcomes reported after surgical treatment, which complicates the comparison of studies and the synthesis of evidence for clinical decision-making. For instance, five highly cited recent systematic reviews on the surgical treatment of DTC all highlighted inconsistencies in the outcomes reported across eligible trials.3–7 To properly compare treatment strategies, researchers and clinicians should select and report similar and relevant outcomes. A core outcome set (COS) is an agreed-upon collection of outcomes that should be measured and reported within specific areas of health care. 8 A COS offers a method to standardize outcome selection in both research and clinical practice, enabling consistent measurement and reporting, enhancing the accuracy of treatment outcome determination, facilitating data synthesis, and ultimately improving patient outcomes.8,9 Currently, there is no COS for the surgical treatment of DTC. Therefore, the aim of this study was to develop an international COS to be measured and reported in all future studies on surgical treatments for DTC, as well as in clinical practice.
Methods
This study was registered with the Core Outcome Measures in Effectiveness Trials initiative (registration number 2597) on March 21, 2023. 10 A systematic approach for the development of the COS was applied, consisting of three phases: (1) collection of the initial list of outcomes for the Delphi study by an extensive literature review, (2) a two-round international Delphi study to identify a set of core outcomes for the surgical treatment of DTC, (3) an international expert panel meeting with experts and patient representatives to ratify the final COS. 11 Our published protocol provides full details on study design, rationale, and methods, including adherence to the Core Outcome Set-STAndards for Development recommendations checklist (COSTAD), Guidance for Reporting on Involvement of Patients and Public short form reporting checklist (GRIPP2), and the Core Outcome Set-STAndard for Reporting guidelines (COS-STAR).11–13 This study is approved by the institutional review board of Amsterdam University Medical Centers (UMCs) (registration number #2023.0677). Consent to participate in the survey was declared by answering and returning the survey.
(1) Literature review
A systematic literature review was performed to identify reported outcomes of surgical interventions for DTC. MEDLINE and EMBASE were searched for studies published in English from January 2020 to March 2023, with additional relevant studies identified through reference lists (see Supplementary Data S1, Supplementary Table S1 for the full search strategy). We limited our search to studies published from January 2020 to ensure inclusion of the most up-to-date evidence, with the expectation that outcomes from earlier studies still relevant to current practice would also be reflected in more recent literature. Titles, abstracts and full texts were screened by D.J.B. using predetermined inclusion/exclusion criteria. 11 One experienced reviewer (D.J.B.) screened the studies, as the purpose was solely to extract outcomes exactly as reported, minimizing the potential for bias. Both adult and pediatric populations were included, while studies on nonsurgical treatments (i.e., laser ablation) or other thyroid cancers were excluded. In case of doubt, a second independent reviewer (J.P.M.D.) made the final decision.
(2) Delphi procedure
Steering committee
A steering committee was established, comprising eight medical experts on DTC from various medical (sub-)specialties, including endocrinology, pediatric endocrinology, endocrine surgery, pediatric surgery, and radiology and nuclear medicine, affiliated with UMCs in The Netherlands, along with a patient representative from the Dutch Thyroid Patient Organization (SchildklierNL). The committee approved the final study protocol and reviewed the results of each round, and approved subsequent rounds and the expert panel meeting. Steering committee members did not participate in the Delphi study. A smaller study management group (D.J.B., J.P.M.D., E.J.M.N.D., and A.F.E.) oversaw the study progress.
Participants
The primary users of this surgical COS will be clinicians and researchers from various medical fields worldwide, as well as patient representatives. Involving patients as a stakeholder group for the development of a COS is imperative as patients may value outcomes differently than physicians.14,15 We made efforts to ensure broad expert representation from various medical disciplines, recognizing that treatment and research of DTC involve collaboration among various medical professionals. Therefore, we invited a diverse group of professionals involved in DTC management globally. Invitations were distributed by the European Thyroid Association (ETA), EU Reference Network Rare Endocrine Conditions (Endo-ERN), European Organisation for Research and Treatment of Cancer-Endocrine Tumor Group (EORTC-EnTG), the European Reference Network on Rare Adult Solid Cancers (ERN-EURACAN) and the African Head and Neck Society (AfHNS). Additionally, experts involved in international thyroid guidelines (ESMO, ATA, ETA), those identified via clinicaltrials.gov, and the last authors of key studies from our literature review were invited, along with recommendations from the steering committee.
Study design
The Delphi methodology is distinct in relying solely on the structured input of a predefined expert panel; neither authors nor external stakeholders may retrospectively add or remove components once the process has begun. The list of outcomes from the literature review was presented to the experts and patient representatives to assess the importance of each outcome to be included in the surgical COS of DTC. The outcomes were accompanied by plain language questions written in layman’s terms for the patient representatives, which were piloted by the patient representative from SchildklierNL (A.S.) and two other laypersons to assess ambiguity and readability. Participants rated each outcome on a Likert scale from 1 to 9, with 7–9 indicating critical importance, assessing how important they considered each outcome for inclusion in the definition, following the recommendations of the Grading of Recommendations Assessment, Development, and Evaluation working group.11,16 In addition, participants could select “not my area of expertise” per question if they did not feel equipped to score certain questions. 11 Each round had to be completed within 4 weeks, with two reminder emails sent to participants who had not yet finished. Participants who completed a round were invited to participate in the next, and only participants who completed all rounds were included in the final collaboration group (Thyroid Surgery COS collaboration group) for publication. In round one, participants could suggest additional outcomes not included in the initial list of outcomes derived from the literature review. The steering committee then determined if these suggestions constituted new outcomes to be added in the subsequent round. Participants could see the scoring distribution of the other participants answers and a reminder of their individual answer from the previous round, in accordance to the Delphi principle. Participants were then asked to rescore the remaining outcomes in the same manner as the previous round.
Delphi results were analyzed collectively and by stakeholder group to account for potential differences between patients and professionals. 17 Consensus was defined using commonly accepted thresholds: outcomes were included (“consensus-in”) if ≥70% of participants in both groups rated them 7–9, with <15% rating 1–3, or if ≥90% of one group rated them 7–9. Outcomes were excluded (“consensus-out”) if ≥70% in both groups rated them 1–3, with <15% rating 7–9.8,11.
(3) International expert panel meeting
On October 17, 2024, an online expert panel meeting was organized, accommodating multiple time zones, to ratify the final list of outcomes for the surgical COS of DTC. All experts and patient representatives who completed both rounds received an invitation. Outcomes were introduced by patient representatives, whose personal narratives underscored their importance and strengthened the incorporation of patient perspectives in the COS. 11 The outcomes were then evaluated by the participants based on the following predetermined criteria: (1) is the outcome relevant; (2) is the outcome measurable by an accepted tool or instrument; (3) is the outcome specific to be improved by surgical interventions for DTC. 11 Following moderated discussion, experts and patients revoted on each outcome to determine inclusion in the final COS. Consensus was predefined as ≥80% agreement. Voting was possible immediately after discussion or within 48 hours of the meeting. 11 The final COS is categorized according to the four core areas of the OMERACT filter 2.0,18,19 to maximize data harmonization.
Results
Literature review
The literature search yielded 2585 studies. After review of titles and abstracts, 165 studies were selected for full-text review. A total of 149 studies were eligible according to the predefined criteria, of which 116 retrospective studies, 17 systematic reviews/meta-analysis, 9 prospective studies, and 7 randomized controlled trials. In total, 521 unique outcomes were found. These outcomes were grouped into 38 outcomes by the study management group. The full list of the included studies with references and unique outcomes, the list of grouped outcomes and the PRISMA flowchart can be found in Supplementary Table S1, Table S2, and Supplementary Figure S1.
Delphi study
On January 10, 2024, invitations were sent by email for the first round. There were 222 respondents, of which 208 experts and 14 patient representatives. Of these, 172 experts and 10 patient representatives completed the round. On April 2, the second round was sent by email. A total of 132 participants completed this round, of which 125 experts and 7 patient representatives. The experts included endocrinologists, oncologists, surgeons, pathologists, radiologists and nuclear medicine physicians, pediatricians, and researchers from 35 countries across 5 continents (Fig. 1). In the first round, the initial list of 38 outcomes was presented, with 7 outcomes reaching consensus-in and none reaching consensus-out. Based on suggestions from experts and patient representatives in the first round and after discussion with the steering group, 2 outcomes were added. In round two, 10 additional outcomes reached consensus-in and 1 reached consensus-out. After two rounds, 17 outcomes reached consensus-in (Table 1). The full rounds can be found in Supplementary Table S4.

Geographical distribution of experts.  Delphi participants;
Delphi participants;  No participants.
No participants.
Consensus-in and out after Two Delphi Rounds
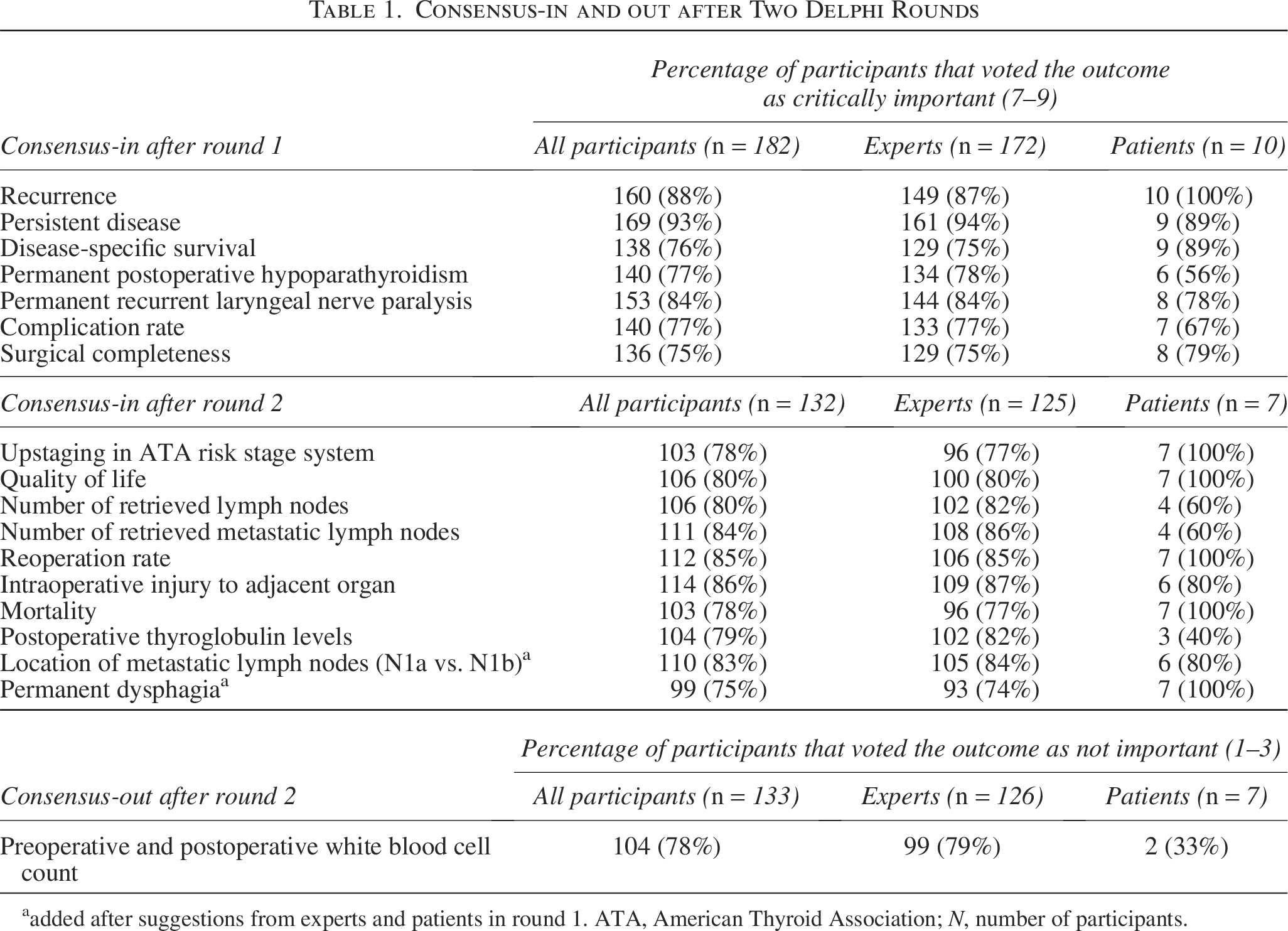


added after suggestions from experts and patients in round 1. ATA, American Thyroid Association;
Expert panel meeting
A total of 52 experts and 3 patient representatives participated in the expert meeting, excluding the study management group. All outcomes were introduced and discussed in accordance with the established procedures. Supplementary Table S5 lists the outcomes discussed and the number of votes for their inclusion in the COS. After discussion, the outcomes “mortality” and “complication rate” were further specified as “30-day postoperative mortality” and “30-day postoperative complication rate.” “Intraoperative injury to adjacent organ” was further clarified as “accidental intraoperative injury to adjacent organ.” “Reoperation rate” was further specified as “unplanned reoperation rate.” “Permanent recurrent laryngeal nerve paralysis” was further specified as “permanent recurrent laryngeal nerve paralysis due to surgery.” In total, 13 outcomes reached consensus for inclusion in the final COS (Table 2).
Final Core Outcome Set for Reporting Surgical Treatment of Differentiated Thyroid Carcinoma

Final core outcome set
The three phases of this COS development process resulted in a final set containing 13 outcomes in 4 core areas (Table 2). One outcome was categorized under the core areas of death and life impact: 30-day postoperative mortality and quality of life. The following outcomes were grouped under pathophysiological/clinical manifestations: recurrence, persistent disease, location of metastatic lymph nodes, number of retrieved metastatic lymph nodes, postoperative thyroglobulin levels and surgical completeness. The following outcomes were categorized under adverse events: permanent recurrent laryngeal nerve paralysis due to surgery, permanent postoperative hypoparathyroidism, 30-day postoperative complication rate, accidental intraoperative injury to an adjacent organ and unplanned reoperation rate.
Discussion
Through an international two-round Delphi process and expert panel meeting, we identified 13 outcomes to be included in the COS for surgical treatment of DTC. This COS was developed using a rigorous, protocol-driven Delphi consensus methodology, involving patient representatives and a diverse group of medical experts in the field of DTC from around the world. We intentionally brought together experts from various medical disciplines in the field of DTC, recognizing that in many countries, treatment and research in this area involve collaboration among various medical professionals. This COS is intended for use within the research community to standardize outcome selection and reporting in future studies related to surgical treatment of DTC, thereby facilitating data synthesis, and in clinical practice, improving the accuracy of treatment outcome assessment.
The next essential step is to focus on defining some of these outcomes, as this is beyond the scope of this COS development. Streamlining the definitions and measurement methods of the selected outcomes will further standardize outcome reporting. We recommend that decisions on how to best measure the outcomes from this COS are informed by criteria from consensus-based standards for the selection of health Measurement Instruments (COSMIN) guidelines. 20 During the expert panel meeting, participants suggested some clarifications for the outcomes. First, a postoperative period of 30 days was added to “mortality” and “complication rate.” This ensures that the mortality and complications are more accurately attributed to the surgical treatment, rather than to additional therapies such as radioiodine treatment. Likewise, “permanent recurrent laryngeal nerve paralysis” was further specified with “due to surgery” to ensure that future research is focused on the effect of surgery on this complication, excluding the permanent recurrent laryngeal nerve paralysis caused by tumor infiltration. “Reoperation rate” was further specified to “unplanned reoperation rate.” We suggest that this is defined as the rate of unplanned (re-)operations, which are executed for DTC, excluding a two-phase thyroidectomy. Furthermore, as authors, we suggest the following: “Permanent recurrent laryngeal nerve paralysis due to surgery” should refer to accidental injuries, excluding cases in which the nerve is deliberately sacrificed due to tumor-related encasement of a functional nerve. “Accidental intraoperative injury to an adjacent organ” should refer to structures in the neck such as the trachea, large blood vessels, and esophagus. The cutoff between temporary and permanent recurrent laryngeal nerve paralysis and hypoparathyroidism should be set at 1 year, as some studies suggest recovery may still occur beyond 6 months.21–23 .
Our study has several limitations. First, the majority of responses came from Europe and North America. Despite efforts to include more experts from Africa, Asia, Australia, and Latin America through invitations distributed by AfHNS and contacting lead authors of key studies, of which many were from Asia, these regions remained underrepresented in our Delphi process. Second, selection bias might have influenced our study, as the final group of expert clinicians consisted of those who accepted our invitation, potentially leading to overrepresentation of certain viewpoints. Despite efforts to invite a diverse range of experts, those more engaged in DTC research may have been more likely to participate, possibly affecting the generalizability of the findings. Third, a general limitation in the development of a COS through Delphi consensus is that potential outcomes are often assessed by experts without standardized definitions, which can lead to varying interpretations of the importance of including a potential outcome in the COS. Paradoxically, a COS identifies the most important outcomes for future studies, thereby providing a selection of outcomes that need to be defined first. In the meantime, we attempted to clarify most outcomes according to the suggestions of the experts. However, the clinical applicability of this COS is limited until the selected outcomes are further defined via international discussion and agreement. Last, we included only Dutch patient representatives in this study, as including patient representatives from other countries would require obtaining ethical approval from each treatment center involved, which we deemed as unfeasible. Nonetheless, we recognize that including patients from all participating countries might have led to slightly different results, as cultural differences might influence which outcomes achieve consensus.
In conclusion, an international consensus was reached on 13 outcomes to be included in the COS for the surgical treatment of DTC. The systematic approach of this study warranted the involvement of patient representatives and a relevant representation of DTC experts from around the world. The value of this research lies in establishing an internationally agreed set of outcomes for the surgical treatment of DTC, identified as most relevant for both research and clinical practice. Incorporating internationally standardized outcomes into the design of future studies and into daily clinical practice will promote consistent and appropriate outcome selection, facilitate comparison of treatment strategies, and ultimately improve patient care. At present, however, the clinical applicability of this COS remains limited, as the selected outcomes still need to be precisely defined. Therefore, the next step is to determine how these outcomes should be defined and measured, further harmonizing the way we assess the effect of surgical treatments for DTC.
Thyroid Surgery COS Collaboration Group
Adekunle Daniel: Department of Otorhinolaryngology, College of Medicine, University of Ibadan, Ibadan, Nigeria.
Agnes van Dijken: Patient.
Agnieszka Czarniecka: 3rd Department of Oncological Surgery, M. Sklodowska-Curie National Research Institute of Oncology, Gliwice Branch, 44-101 Gliwice, Poland.
Alessandra Cassio: Department of Medical and Surgical Sciences, University of Bologna, Bologna, Italy.
Alexis Vrachimis: Department of Nuclear Medicine, German Oncology Center, European University Cyprus, Limassol, Cyprus.
Anca Sirbu: Endocrinology Department, Carol Davila University of Medicine and Pharmacy, 020021 Bucharest, Romania.
Andrea Frasoldati: Endocrinology Unit, Azienda USL-IRCCS di Reggio Emilia, Reggio Emilia, Italy.
Andrea Contarino: Endocrinology Unit, Fondazione IRCCS Ca' Granda Ospedale Maggiore Policlinico, Milan & Department of Clinical Sciences and Community Health, University of Milan, Milan, Italy.
Andrej Belančić: 1. Department of Clinical Pharmacology, Clinical Hospital Centre Rijeka, Krešimirova 42, 51000 Rijeka, Croatia;
Andrew J. Bauer: Andrew J. Bauer, MD, Director, The Thyroid Center, Division of Endocrinology and Diabetes, The Children's Hospital of Philadelphia, Philadelphia, PA, United States. Professor of Pediatrics, Perelman School of Medicine, The University of Pennsylvania, Philadelphia, PA, United States.
Anna Crescenzi: Department of Radiological, Oncological and Pathological Sciences, Sapienza University of Rome, Rome, Italy.
Anna Konney: Eye, Ear, Nose and Throat Department, Komfo Anokye Teaching Hospital, Kumasi, Ghana.
Anna M Sawka: Division of Endocrinology, Department of Medicine, University Health Network and University of Toronto, Toronto, Canada.
Annamária Erdei: Division of Endocrinology, Department of Internal Medicine, Faculty of Medicine, University of Debrecen, Debrecen, Hungary.
Antje Redlich: Paediatric Oncology Department, Otto von Guericke University Children's Hospital, Magdeburg, Germany.
Antonio Matrone: Endocrine Unit, Department of Clinical and Experimental Medicine, University of Pisa, Pisa, Italy. Electronic address.
Marialuisa Appetecchia: Oncological Endocrinology Unit, IRCCS Regina Elena National Cancer Institute, Via Elio Chianesi 53, 00144 Rome, Italy.
Arseny Semenov: Department of Endocrine surgery, St. Petersburg State University Hospital.
Ashok R. Shaha: Head and Neck Service, Department of Surgery, Memorial Sloan Kettering Cancer Center, New York, NY, USA.
Berna O. Evranos: Ankara Yildirim Beyazit University, Bilkent City Hospital, Endocrinology and Metabolism Department, Ankara, Turkey.
Britt Verrips: Patient.
M.C. Burlacu: Department of Endocrinology Diabetology and Nutrition, Cliniques Universitaires St-Luc, Université Catholique de Louvain, Brussels, Belgium.
Pietro G. Calò: Affiliation Department of Surgical Sciences University of Cagliari, Policlinico D. Casula, 09042 Monserrato (Cagliari) Italy.
Camille Buffet: Thyroid and Endocrine Tumors Department, Pitié-Salpêtrière Hospital, Thyroid Tumors Clinical Research Group n°16, Sorbonne University, Cancer Institute, Inserm U1146, CNRS UMR 7371, Paris, France.
Carmine De Bartolomeis: Barbantini Hospital – Endocrine Surgery Unit – Lucca (Italy).
Catherine A. Dinauer: Department of Pediatrics, Division of Endocrinology & Diabetes, Yale School of Medicine, New Haven, CT, USA.
Corina E. Andreescu: Department of Endocrinology, UZ Brussel, Vrije Universiteit Brussel, Brussels, Belgium.
Dario Tumino: Endocrinology Unit, Department of Clinical and Experimental Medicine, Garibaldi Nesima Hospital, University of Catania, Catania, Italy.
Doina Piciu: Faculty of Medicine, “Iuliu Hațieganu” University of Medicine and Pharmacy, 400012 Cluj-Napoca, Romania.
Domenico Albano: Nuclear Medicine Department, Università degli Studi di Brescia and ASST Spedali Civili of Brescia, Italy.
Elena Chobankova: Department of Endocrinology, University Hospital Kaspela, Plovdiv, Bulgaria.
Ellen Kapiteijn: Department of Medical Oncology, Leiden University Medical Center, Leiden, the Netherlands.
Esther Willemse: Department of Head and Neck Surgery, Jules Bordet Institute-HUB, Université Libre de Bruxelles, 93 rue Meylemeersch, Brussels, 1070, Belgium.
Ferenc Gyory: Department of Surgery, Faculty of Medicine, University of Debrecen, Debrecen, Hungary.
Flavia Magri: Department of Internal Medicine and Therapeutics, University of Pavia, 27100, Italy; Istituti Clinici Scientifici Maugeri IRCCS, Unit of Internal Medicine and Endocrinology, Laboratory for Endocrine Disruptors, Pavia 27100, Italy.
Florence van Ryckeghem: Department of Medical Oncology, AZ Groeninge Kortrijk, Belgium and AZ Glorieux Ronse, Belgium.
Francesco Ferraù: Department of Human Pathology of Adulthood and Childhood “G. Barresi”, University of Messina, Messina, Italy; 3 Endocrine Unit, “G. Martino” University Hospital, Messina, Italy.
Françoise Borson-Chazot: Department of Endocrinology, Diabetes and Metabolic Diseases, Hospices Civils de Lyon and Claude Bernard University, 69394 Lyon, France.
Furio Pacini: Section of Endocrinology, University of Siena, Siena, Italy. Electronic address: pacini8@unisi.it.
Gabriella Pellegriti: 1. Endocrinology Division, Garibaldi Nesima Hospital, Catania Italy. 2. Medical Oncology, Department of Internal and Experimental Medicine, University of Catania, Italy.
Gerlof D. Valk: Department of Endocrine Oncology, University Medical Center, Utrecht University, Utrecht, Netherlands.
Gerasimos P. Sykiotis: Service of Endocrinology, Diabetology and Metabolism, Lausanne University Hospital and University of Lausanne, 1011 Lausanne, Switzerland.
Gerdi Tuli: 1. Department of Pediatric Endocrinology, Regina Margherita Children's Hospital, Turin, Italy. 2. Department of Pediatrics, University of Turin, Turin, Italy.
Gesthimani Mintziori: Unit of Reproductive Endocrinology, 1st Department of Obstetrics and Gynecology, Aristotle University of Thessaloniki Medical School, Thessaloniki, Greece.
Gianni Bocca: Division of Paediatric Endocrinology, Department of Paediatrics, Beatrix Children's Hospital, University Medical Centre Groningen, University of Groningen, Groningen, The Netherlands.
Giulia Brigante: 1. Unit of Endocrinology, Department of Biomedical, Metabolic and Neural Sciences, University of Modena and Reggio Emilia, Modena, Italy; 2. Unit of Endocrinology, Department of Medical Specialties, Azienda Ospedaliero-Universitaria of Modena, Modena, Italy.
Giuseppe Costante: Departments of Endocrinology and Medical Oncology, Institut Jules Bordet Comprehensive Cancer Center – Hôpital Universitaire de Bruxelles, Université Libre de Bruxelles (ULB), Brussels, Belgium.
Giuseppe Fanetti: Division of Radiation Oncology, Centro di Riferimento Oncologico di Aviano (CRO) IRCCS, Aviano, Italy.
Gloria Marquina: Department of Medical Oncology, Hospital Clínico San Carlos, School of Medicine, Complutense University (UCM), IdISSC, 28040 Madrid, Spain. Electronic address: gloria.marquina@salud.madrid.org.
Gordana Horvatić-Herceg: Department of Nuclear Medicine and Radiation Protection, University Hospital Center Zagreb, Zagreb, Croatia.
François Gorostidi: Service d'oto-rhino-laryngologie et chirurgie cervico-faciale Lausanne University Hospital, Lausanne Switzerland.
Greg Randolph: Division of Thyroid and Parathyroid Endocrine Surgery, Department of Otolaryngology-Head and Neck Surgery, Harvard Medical School, Boston, Massachusetts.
Haykanush Hambardzumyan: Wigmore Women`s and Children`s Hospital, Armenia.
Heike Odermatt: Patient.
Ineke Bonder: Patient.
James Suliburk: Michael E. DeBakey Department of Surgery, Baylor College of Medicine, Houston, Texas.
Jan Krátký: Clinic of Endocrinology and Metabolism, First Faculty of Medicine, Charles University and General University Hospital in Prague, Czech Republic.
Jan Podoba: Department of Endocrinology, Slovak Medical University and St. Elisabeth Cancer Institute, 812 50 Bratislava, Slovak Republic.
Jan Zedenius: Department of Molecular Medicine and Surgery, Karolinska Institutet, Stockholm, Sweden & Department of Breast, Endocrine Tumors and Sarcoma, Karolinska University Hospital, Stockholm, Sweden.
Jolanda Bikkel: Patient.
Jolanta Krajewska: Nuclear Medicine and Endocrine Oncology Department, M. Sklodowska-Curie National Research Institute of Oncology, Gliwice Branch, 44-101 Gliwice, Poland.
Jon Wadsley: Weston Park Hospital, Sheffield, United Kingdom.
Jonathan D. Wasserman: Division of Endocrinology, The Hospital for Sick Children, Toronto, Canada.
Juan C. Galofré: Department of Endocrinology, Clínica Universidad de Navarra, Pamplona, Spain & Instituto de Investigación Sanitaria de Navarra, Pamplona, Spain.
Katerina Saltiki: Endocrine Unit and Diabetes Center, Department of Clinical Therapeutics, Alexandra Hospital, School of Medicine, National and Kapodistrian University of Athens, Athens, Greece.
Kenneth Kojo Baidoo: EAR, NOSE AND THROAT DEPARTMENT, KORLE-BU TEACHING HOSPITAL , ACCRA, GHANA.
Kerstin Lorenz: Medical Faculty, Department of Visceral, Vascular and Endocrine Surgery, Martin Luther University Halle-Wittenberg, Ernst-Grube-Str. 40, D-06097, Halle (Saale), Germany.
Koen M.A. Dreijerink: Department of Endocrinology and Metabolism, Amsterdam UMC Location VU University, De Boelelaan 1117, Amsterdam, The Netherlands; Amsterdam Center for Endocrine and Neuroendocrine Tumours (ACcENT), Cancer Center Amsterdam, The Netherlands.
Laszlo Hegedüs: Department of Endocrinology, Odense University Hospital, Odense, Denmark.
Laura Fugazzola: Department of Endocrine and Metabolic Diseases and Laboratory of Endocrine and Metabolic Research, Istituto Auxologico Italiano, Istituto Di Ricovero e Cura a Carattere Scientifico (IRCCS); Department of Pathophysiology and Transplantation, University of Milan, Milan, Italy.
Laura D. Locati: 1. Department of Internal Medicine and Therapeutics, University of Pavia (Italy); 2. Medical Oncology Unit, Istituti Clinici Scientifici Maugeri IRCCS, Pavia (Italy).
Livia Lamartina: Department of Imaging, Endocrine Oncology Unit, Gustave Roussy, University Paris Saclay, Villejuif, France.
Luca Giovanella: Department of Nuclear Medicine, Gruppo Ospedaliero Moncucco, Lugano, Switzerland & Clinic for Nuclear Medicine, University Hospital of Zürich, Zürich, Switzerland.
Luca Persani: 1. Department of Medical Biotechnology and Translational Medicine, University of Milano. 2. Department of Endocrine and Metabolic Diseases, IRCCS Istituto Auxologico Italiano, Milano, Italy.
Luisa de Sanctis: Department of Pediatric Endocrinology, Regina Margherita Children's Hospital, Turin, Italy.
Department of Pediatrics, university of Turin, Turin, Italy.
Maaike Versluis: Patient.
Malek Mnejja: Department of Otorhinolaryngology Head and Neck Surgery, Habib Bourguiba University Hospital, Sfax, Tunisia & Sfax Medical School, University of Sfax, Sfax, LR23ES01 Tunisia.
Malgorzata Trofimiuk-Müldner: Chair and Department of Endocrinology, Jagiellonian University Medical College, Kraków, Poland.
Małgorzata Waśniewska: Pediatric Unit, Department of Human Pathology of Adulthood and Childhood, University of Messina, 98166 Messina, Italy.
Marcin Barczynski: Department of Endocrine Surgery, Jagiellonian University Medical College, Krakow, Poland.
Marco Raffaelli: 1. UOC Chirurgia Endocrina e Metabolica, Fondazione Policlinico Universitario Agostino Gemelli IRCCS, Rome Italy; 2. Centro di Ricerche in Chirurgia delle Ghiandole Endocrine e dell'Obesità (C.R.E.O.), Università Cattolica del Sacro Cuore, Rome, Italy.
Marek Niedziela: Department of Pediatric Endocrinology and Rheumatology, Institute of Pediatrics, Karol Jonscher Clinical Hospital, Poznan University of Medical Sciences, Poland.
Maria C Vigone: Department of Pediatrics, IRCCS San Raffaele Scientific Institute, Milan, Italy.
Maria Grazia Maratta: 1. Comprehensive Cancer Center, Fondazione Policlinico Universitario Agostino Gemelli IRCCS, Largo A.Gemelli, 8, Rome, Italy. 2. Università Cattolica del Sacro Cuore, Largo F.Vito, 1, Rome, Italy.
Maria João Bugalho: Endocrinology Department, Northern Lisbon University Center, Lisbon, Portugal; Endocrinology University Clinic, Lisbon Medical School, Lisbon, Portugal.
Maria Sandström: Department of Diagnostics and Interventions, Oncology, Umeå University, Umeå, Sweden.
Marie-Louise Healy: St James's Hospital, (Department of Endocrinology), Dublin, Ireland.
Mario Salvi: Endocrinology Unit, Graves' Orbitopathy Center, Fondazione IRCCS Ca' Granda Ospedale Maggiore Policlinico, Milan, Italy.
Mark Gruppetta: Department of Diabetes and Endocrinology, Mater Dei Hospital, Msida, Malta.
M.T. Stegenga: Academic Center for Thyroid Diseases, Department of Internal Medicine, Erasmus Medical Center, Rotterdam, the Netherlands.
Michele N. Minuto: Unit of Surgery 2 (Endocrine Surgery), San Martino University Hospital, Genoa, Italy & Department of Surgical Sciences and Integrated Diagnostics (DISC), University of Genoa, Genoa, Italy.
Miklós Tóth: Department of Internal Medicine and Oncology, Semmelweis University, Faculty of Medicine, Budapest, Hungary.
Miloš Žarković: a. University of Belgrade Faculty of Medicine, Belgrade, Serbia;. b. Clinic of Endocrinology, Diabetes and Diseases of Metabolism UKCS, Belgrade, Serbia.
Monica L Gheorghiu: Carol Davila University of Medicine and Pharmacy and C.I. Parhon National Institute of Endocrinology, Bucharest, Romania.
Montserrat Negre Busó: Department of Nuclear Medicine, Dr Josep Trueta University Hospital, Girona, Spain.
Brooke Puttergill: Department of Surgery, Buckinghamshire Healthcare Trust, Stoke Mandeville, United Kingdom.
Nicholas Reed: Consultant Clinical Oncologist, Beatson Oncology Centre, Gartnavel General Hospital, 1089 Great Western Road, GLASGOW, G12 0YN, Scotland, UK.
Nicole D Bouvy: Department of Surgery, Maastricht University Medical Center, 6229 HX Maastricht, The Netherlands & NUTRIM School for Nutrition and Translational Research in Metabolism, Maastricht University, 6211 LK Maastricht, The Netherlands.
Nikola Besic: Department of Surgical Oncology, Institute of Oncology, Ljubljana, Slovenia; Faculty of Medicine, University of Ljubljana, Ljubljana, Slovenia.
Oleksandr Nechai: UKRAINIAN SCIENTIFIC and PRACTICAL CENTER of ENDOCRINE SURGERY, TRANSPLANTATION of ENDOCRINE ORGANS and TISSUES of MOH of UKRAINE, Klovsky descent, 13A, Kyiv, 01021, Ukraine.
Özer Makay: Özel Sağlık Hospital, Centre for Endocrine Surgery, Izmir, Türkiye.
Pablo Valderrabano: Department of Endocrinology and Nutrition, Hospital Universitario Ramón y Cajal, IRYCIS, Carretera Colmenar Km 9,100, 28034 Madrid, Spain. ORCID: 0000-0001-8379-2709.
Paul A. Onakoya: Department of Otorhinolaryngology, University College Hospital, University of Ibadan, Ibadan, Nigeria.
Peter Angelos: Department of Surgery, University of Chicago & MacLean Center for Clinical Medical Ethics, University of Chicago.
Petra Petranović Ovčariček: Department of Oncology and Nuclear Medicine, University Hospital Center Sestre Milosrdnice, Zagreb, Croatia & School of Medicine, University of Zagreb, Zagreb, Croatia.
Petros Perros: Institute of Translational and Clinical Research, Newcastle University, Newcastle upon Tyne, United Kingdom.
Kristien Boelaert: Department of Applied Health, School of Health Sciences, College of Medicine and Health, University of Birmingham, Birmingham, UK.
Thomas J. Musholt: Section of Endocrine Surgery, Department of General, Visceral and Transplantation Surgery, University Medical Centre, Johannes Gutenberg University Mainz, Mainz, Germany.
Radu Mihai: Endocrine Surgery Unit, Churchill Cancer Centre, Oxford University Hospitals NHS Foundation Trust, Oxford, UK. Electronic address: radumihai@doctors.org.uk.
Ramon RJP van Eekeren: Department of Surgical Oncology, Rijnstate Hospital, Arnhem, The Netherlands.
Renuka P. Dias: 1. Institute of Applied Health Research, University of Birmingham, UK; 2. Department of Paediatric Endocrinology and Diabetes, Birmingham Children’s Hospital, UK.
Roman Chernikov: Department of Endocrine Surgery, Medical Institute, St Petersburg University, St Petersburg, Russia.
Ronald R. de Krijger: Princess Maxima Center for pediatric oncology, Heidelberglaan 25, 3584 CS Utrecht, The Netherlands & Department of Pathology, University Medical Center Utrecht, Heidelberglaan 100, 3584 CX Utrecht, The Netherlands.
Rosé Lemeer: Patient.
Patrick O. Rosselet: Cabinet Médical 2, rue Bellefontaine, Lausanne, Switzerland.
Roussanka Kovatcheva: Department of Endocrinology, Medical University Sofia, University Hospital of Endocrinology “Acad. Ivan Penchev”, Sofia, Bulgaria.
Salvatore Cannavò: Department of Human Pathology of Adulthood and Childhood “G. Barresi”, University of Messina, Messina, Italy & Endocrine Unit, “G. Martino” University Hospital, Messina, Italy.
Sam Van Slycke: Department of General and Endocrine Surgery, Onze-Lieve-Vrouw (OLV) Hospital Aalst, Aalst, Belgium & Department of Head and Skin, University Hospital Ghent, Ghent, Belgium & Department of General Surgery, AZ Damiaan, Ostend, Belgium.
Sandra Nuyts: Laboratory of Experimental Radiotherapy, Department of Oncology, KU Leuven, 3000 Leuven, Belgium; Department of Radiation Oncology, Leuven Cancer Institute, University Hospitals Leuven, 3000 Leuven, Belgium.
Silvia A. Eskes: Department of Internal Medicine, Franciscus Gasthuis and Vlietland, Rotterdam, The Netherlands.
Steven I. Sherman: Department of Endocrine Neoplasia and Hormonal Disorders, The University of Texas MD Anderson Cancer Center, Houston, TX 77030, USA.
Steven G. Waguespack: Department of Endocrine Neoplasia and Hormonal Disorders, University of Texas MD Anderson Cancer Center, Houston, TX, USA.
Susan J. Mandel: Perelman School of Medicine, University of Pennsylvania, Philadelphia, Pennsylvania; Division of Endocrinology, Diabetes, and Metabolism, Department of Medicine, Hospital of the University of Pennsylvania, Philadelphia, Pennsylvania.
Thera P. Links: Department of Internal Medicine, division Endocrinology, University of Groningen, University Medical Center Groningen, Hanzeplein 1, 9700. RB Groningen, The Netherlands.
Tessa Malaika van Ginhoven: Academic center for Thyroid disease, Erasmus MC, department of surgical oncology, Rotterdam, The Netherlands.
Tiago Nunes da Silva: Department of Endocrinology and Molecular Pathobiology Research Unit, Instituto Português de Oncologia de Lisboa Francisco Gentil, Lisbon, Portugal.
Tiago Pimenta: Department of Surgery of Centro Hospitalar, Universitário de São João, Porto, Portugal.
Tijana Ičin: University of Novi Sad, Faculty of Medicine in Novi Sad, Novi Sad, Serbia; Diabetes and Metabolic Disorders, Clinic for Endocrinology, Clinical Center of Vojvodina, Novi Sad, Serbia.
Timm Denecke: Department of Diagnostic and Interventional Radiology, University Hospital Leipzig, Leipzig, Germany.
Tobias Zingg: Department of Visceral Surgery, Lausanne University Hospital and Lausanne University, Lausanne, Switzerland.
Ulla Feldt-Rasmussen: Department of Nephrology and Endocrinology, Copenhagen University Hospital Rigshospitalet, Copenhagen, Denmark.
Valeriano Leite: Serviço de Endocrinologia, Instituto Português de Oncologia de Lisboa Francisco Gentil, Lisbon, Portugal.
Authors’ Contributions
D.J.v.d.B. coordinated the Delphi procedure under the supervision of A.F.E., J.P.D., E.J.N.v.D., and A.P.v.T. D.J.v.d.B. performed the analysis under the supervision of A.F.E., J.P.D., E.J.N.v.D., and A.P.v.T. Together with D.J.v.d.B., A.F.E., J.P.D., E.J.N.v.D., and A.P.v.T. also had access to the data and verified the results of each round. H.M.V.S., S.C.C., M.R.V., E.B., S.K., R.P.P., F.A.V., A.S., and R.N.-M. all critically reviewed and approved the study protocol, assessed the data of each Delphi round, and approved each Delphi round. D.J.v.d.B. took the lead in writing the final article under the supervision of A.F.E., J.P.D., E.J.N.v.D., and A.P.v.T. H.M.V.S., S.C.C., M.R.V., A.P.v.T., E.B., S.K., R.P.P., F.A.V., R.N.-M., E.J.N.v.D., A.S., J.P.D., and A.F.E. All critically reviewed and improved the final article. All authors agreed to the publication of the article in its current form.
Footnotes
Acknowledgments
The authors thank the European Thyroid Association, the EU Reference Network Rare Endocrine Conditions, the European Organisation for Research and Treatment of Cancer-Endocrine Tumor Group, the European Reference Network on Rare Adult Solid Cancers, and the African Head and Neck Society for distributing the invitation to participate in this study among their members. The authors thank the Dutch Thyroid Patient Organization ‘Schildklier NL’ for their collaboration, providing a patient representative for the study steering group, and inviting patients to participate in this study. The authors thank all the experts and patient representatives who participated in this Delphi study and the expert panel meeting.
Author Disclosure Statement
F.A.V. has received consultancy fees from GE Healthcare, Immedica (all money paid to employer) and speaker fees from Bayer, AstraZeneca and GE Healthcare (all money paid to employer). For the remaining authors, none were declared.
Funding Information
This work was supported by the Foundation Children Cancer Free (KiKa) grant number 441. The funders of the study had no role in the study design, data collection, data analysis, data interpretation, or writing of the report. The corresponding author and last author had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Data Access and Sharing Statement
D.J.B. and A.F.E. had full access to all the data in the study and took responsibility for the integrity of the data and the accuracy of the data analysis. The data that support the findings of this study are available on request from the corresponding author, D.J.B., after approval of a proposal, with a signed data access agreement. The data are not publicly available as they contain information that could compromise the privacy of the participating experts and patients’ representatives.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
