Abstract
Background:
The International Medullary Thyroid Carcinoma Grading System (IMTCGS) is a two-tier score that classifies high-grade medullary thyroid carcinoma (MTC) by the presence of at least one of the following features: mitotic index ≥5/2 mm2, Ki-67 proliferation index ≥5%, or tumor necrosis. Cases lacking all three features are classified as low-grade. This study aimed to validate the prognostic role of the IMTCGS in patients with metastatic MTC. The prognostic significance of a high proliferative index (Ki-67 index ≥20%) was also investigated.
Methods:
We conducted a monocentric retrospective study of 99 metastatic MTC patients treated at Gustave Roussy between 2000 and 2024, in whom the IMTCGS was assessed on the primary tumor.
Results:
IMTCGS high-grade tumors were found in 67 patients (67.7%), who were older (p = 0.009) and had larger primary tumors (p < 0.001) compared with 32 patients with low-grade tumors. Postoperative calcitonin levels, number of metastatic sites/patient, prevalence of synchronous metastases, and RET-M918T mutation were similar between groups. Median overall survival (OS) was shorter in patients with IMTCGS high-grade than low-grade (4.8 vs. 13.9 years; p = 0.01), as was time to systemic treatment initiation (TTI) (1.0 vs. 4.8 years; p < 0.001). However, among the 75 patients who received systemic therapy, OS from treatment initiation was similar between the two groups (2.8 vs. 3.89 years; p = 0.865). RET-M918T mutation was not associated with worse OS. On multivariable analysis, IMTCGS high-grade and bone metastases were independently associated with both shorter OS and TTI (p < 0.05 for both). Patients with Ki-67 index ≥20% had worse OS (2.6 years) compared with those with Ki-67 index <5% (10.5 years; hazard ratio [HR] = 6.11; p < 0.001) and 5–19% (6.5 years; HR = 3.29; p = 0.001).
Conclusions:
The IMTCGS is a strong independent prognostic factor in patients with metastatic MTC. Patients with IMTCGS high-grade tumors and Ki-67 index ≥20% represent a high-risk subgroup with the poorest prognosis.
Keywords
Introduction
Medullary thyroid carcinoma (MTC), a neuroendocrine tumor that represents <5% of all thyroid cancers, 1 originates from the parafollicular C cells of the thyroid and secretes calcitonin. 2 MTC can exhibit aggressive behavior, including distant metastases at diagnosis or during the course of the disease in up to 30% of patients. 3 Given the heterogeneous survival of patients with metastatic MTC, the absence of curative options at the metastatic stage, and the potential adverse events of systemic interventions, prognostic information is critical for identifying the optimal timing for initiating systemic therapy.4–6
Among the strongest prognostic factors reported so far, the International Medullary Thyroid Carcinoma Grading System (IMTCGS) has been recently implemented. 7 This two-tier system classifies MTC based on proliferative activity and the presence of tumor necrosis on histopathology. Tumors with at least one feature among mitotic index ≥5/2 mm2, Ki-67 proliferation index ≥5%, and tumor necrosis are classified as high-grade, whereas tumors lacking all three features are classified as low-grade. IMTCGS high-grade status was independently associated with shorter overall survival (OS), disease-specific survival (DSS), and disease-free survival compared with low-grade status in a pivotal study encompassing patients across all TNM stages of MTC. 7 Although the presence of some RET mutation (e.g., M918T) has been associated with more aggressive disease,8,9 the prognostic value of the IMTCGS was shown to be independent from RET and RAS somatic mutation status. 7
While the IMTCGS has been validated in MTC overall, its prognostic role in patients with distant metastases remains unclear. The aim of this study was to validate the prognostic role of the IMTCGS in metastatic MTC patients, focusing on survival outcomes from the time of distant metastases diagnosis. As a secondary objective, we examined the association between the IMTCGS and OS after the initiation of systemic treatments. In addition, we investigated the prognostic role of highly proliferative MTC, defined as primary tumors exhibiting a Ki-67 index ≥20%, a threshold that has recently been recognized in gastrointestinal and pulmonary neuroendocrine tumors.10,11
Patients and Methods
Patients and data collection
In this retrospective study, we assessed the eligibility of 224 consecutive patients with metastatic MTC and available follow-up, who were treated and followed up at Gustave Roussy between January 2000 and April 2024. Inclusion criteria were histologically confirmed diagnosis of MTC (sporadic or hereditary) with synchronous or metachronous distant metastases and availability of IMTCGS assessment on the primary tumor tissue. Patients without IMTCGS on the primary tumor were excluded.
Patients were followed every 3–12 months after surgery, based on stage, carcinoembryonic antigen and calcitonin doubling times, clinical status, and imaging findings according to a standardized routine clinical practice. 12 Clinical charts were reviewed to extract patients’ general characteristics, pathological features of the primary tumor and metastatic disease, and details on systemic treatments. Metastases were classified as synchronous if diagnosed within six months of the primary tumor diagnosis; otherwise, they were considered metachronous.
Informed consent was obtained from all patients. The study was conducted in accordance with the Declaration of Helsinki and was approved by the Institutional Review Board of Gustave Roussy (protocol number 2024-451).
Pathological and tumor mutational analysis
Formalin-fixed paraffin-embedded (FFPE) tissue for IMTCGS analysis was available for 99 cases. All initial thyroidectomy specimens were reviewed by two expert pathologists (M.-A.B. and A.A.G.) blinded to clinicopathological and follow-up data. Immunohistochemical staining was performed on unstained whole tissue sections 4 µm thick generated from FFPE blocks using a Ventana Automated Immunostainer (BenchMarker Ultra; Ventana Medical System, Inc.), according to the manufacturer’s instructions. The Ki-67 index was assessed as proposed for gastrointestinal neuroendocrine tumors. 10 The mitotic index was counted in 10 high-powered fields and reported per 2 mm2. Tumor necrosis was considered in the presence of degenerating cytoplasm and punctate karyorrhectic nuclear debris, regardless of whether it was focal or extensive.
All patients underwent genetic screening for germline RET mutation. Somatic RET and RAS mutations were analyzed in selected cases, at the discretion of the treating physician. Somatic molecular alterations were detected either by Sanger sequencing 13 or through various next-generation sequencing platforms as part of personalized medicine studies conducted at Gustave Roussy, including MOSCATO 02 (NCT01566019), MATCH-R (NCT02517892), and STING (NCT04932525), as previously described. 14
Serum calcitonin measurement
Serum calcitonin levels were measured between 2000 and 2002 using the ELSA-CT kit, which had a functional sensitivity of 10 pg/mL. In 2002, this was improved to 2 pg/mL with the ELSA-hCT kit (CIS Bio International), with a normal range of <10 pg/mL. 15 Since 2021, serum calcitonin has been measured using a chemiluminescence assay: the Atellica IM Calcitonin Assay (Siemens Healthineers) with a measuring range of 1.89–2000 pg/mL. Calcitonin doubling time was calculated with the Kuma Hospital calculator (https://www.kuma-h.or.jp/english/about/doubling-time-progression-calculator) only in patients who initiated systemic treatment, provided that at least three measurements were available over a 6- to 12-month period following initial surgery and prior to the systemic treatment initiation.
Systemic treatment management
The selection criteria for initiating systemic treatment were based on tumor burden, symptom presence, and/or morphologically significant disease progression according to RECIST v1.0 16 before 2009 or v1.1 17 thereafter and were systematically discussed by a dedicated tumor board. Biomarker levels or their doubling times were not considered in treatment decisions, as recommended. 18 Locoregional therapies were applied, whenever clinically appropriate, for slowly progressing, localized metastases before and during systemic therapy, regardless of tumor grade. 19 The choice between first-line cytotoxic chemotherapy and tyrosine kinase inhibitors (TKIs) was guided by available clinical trials20,21 and/or approval by the French High Authority for Health for vandetanib (February 17, 2012) and cabozantinib (December 3, 2014). The first patient treated with a TKI received therapy in February 2006. Systemic treatments were continued until disease progression according to RECIST, unacceptable toxicity, and/or the patient’s decision to discontinue treatment.
Outcome measures
OS and DSS were calculated from the date of distant metastases diagnosis to the date of death. For patients alive at the time of last follow-up, OS and DSS were censored at the date of the last visit. Time to systemic treatment initiation (TTI) was calculated from the date of distant metastases diagnosis to the initiation of first-line systemic therapy. For patients who did not receive any systemic therapy, TTI was censored at the date of the last visit. In addition, for patients who received systemic therapy, OS was also analyzed from the start of systemic treatment to the date of death or last follow-up.
Statistical analysis
Standard descriptive statistics were used to characterize the cohort: mean ± standard deviation and range, or median with interquartile range (IQR: 25th percentile; 75th percentile) and range for numerical variables; absolute frequency with percentages for categorical variables. Between-group comparisons were performed using the t-test for independent samples, the Mann–Whitney U test, and the chi-square test (or the Fisher exact test, as appropriate).
Time-to-event analyses were based on the Kaplan–Meier estimates of the survival function. Univariate and multivariable Cox proportional hazards regression models were used to evaluate the prognostic value of IMTCGS, with results expressed as hazard ratios (HRs) and confidence intervals [CIs]. Variables included in the multivariable models were as follows: age at distant metastases diagnosis, primary tumor size, gross extrathyroidal extension (yes vs. no), extensive vascular invasion (yes vs. no), presence of synchronous distant metastases (yes vs. no), metastatic sites (bone, yes vs. no), and number of metastatic sites per patient (>2 vs. ≤2). Age at distant metastases diagnosis and primary tumor size were treated as continuous variables. For DSS analysis, cause-specific Cox models were used, treating competing events as censored. All statistical analyses were performed using R version 4.4.0 (R Core Team, 2024).
Results
Patient characteristics and pathological findings
Ninety-nine consecutive patients with metastatic MTC were included in the study (Fig. 1). Patient characteristics and pathological findings for the entire cohort, as well as comparisons between patients with IMTCGS low-grade and high-grade tumors, are summarized in Table 1. Features of IMTCGS in primary tumors within the study population are detailed in Supplementary Table S1. Compared with excluded patients, the study population was older and had a higher proportion of sporadic cases and of cases with synchronous distant metastases (p < 0.05 for all comparisons) (Supplementary Table S2).
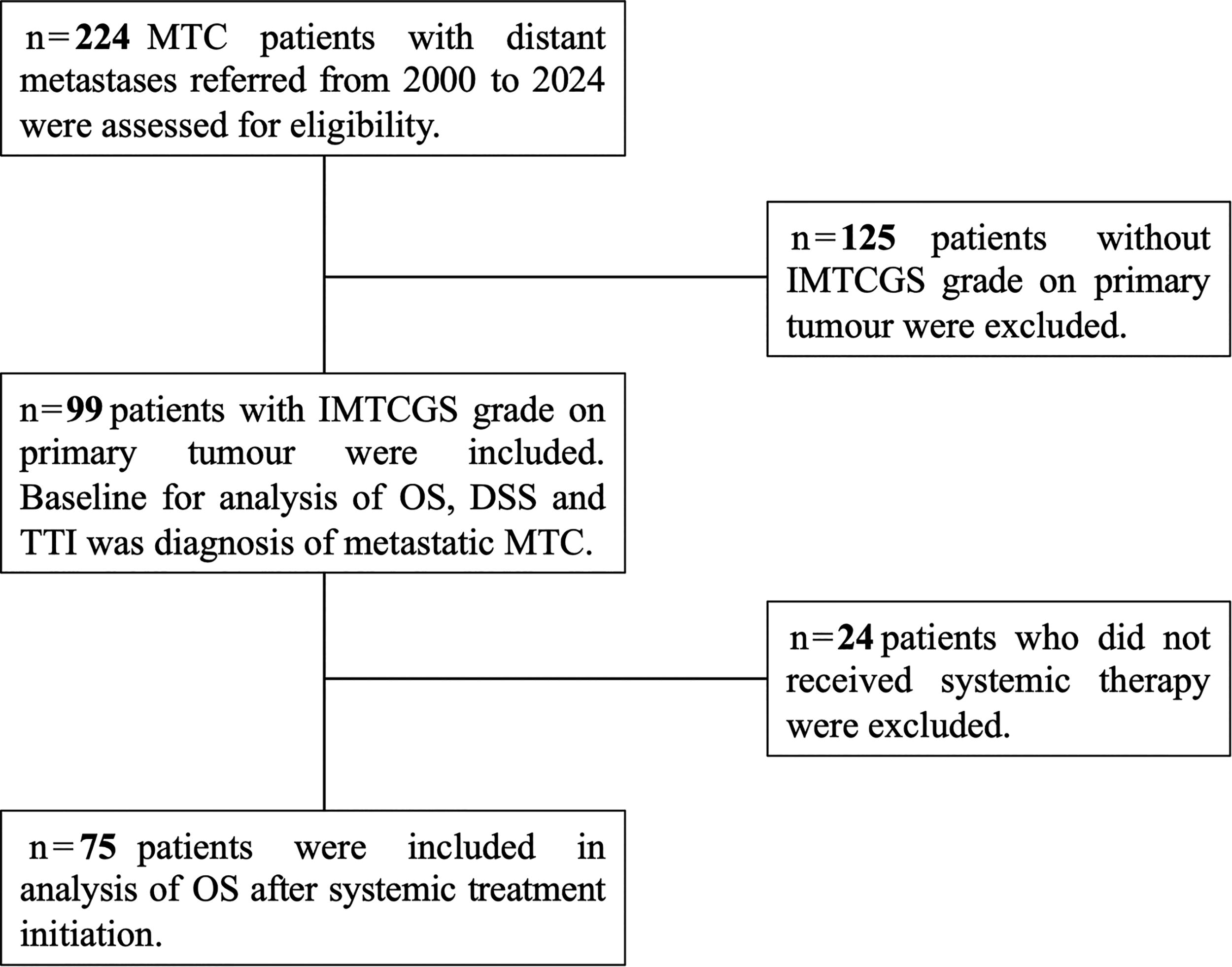
Study participant flow diagram. DSS, disease-specific survival; IMTCGS, International Medullary Thyroid Carcinoma Grading System; MTC, medullary thyroid carcinoma; OS, overall survival; TTI, time to treatment initiation.
Clinical and Pathological Characteristics of the Study Population According to the International Medullary Thyroid Carcinoma Grading System
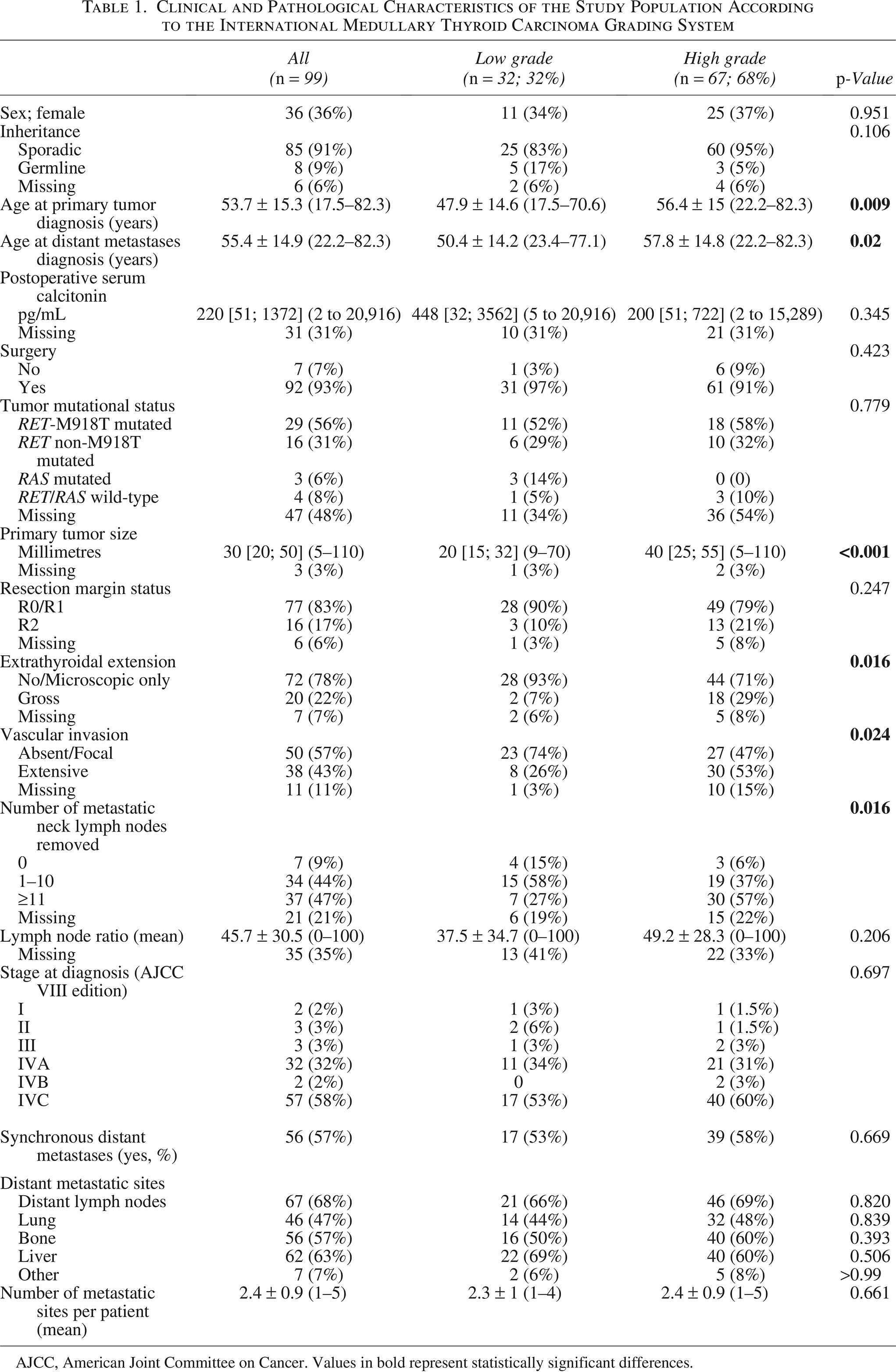
AJCC, American Joint Committee on Cancer. Values in bold represent statistically significant differences.
Calcitonin doubling time was assessable in 25 out of the 75 patients who initiated systemic treatment. In the remaining patients, it could not be assessed due to initiation of systemic therapy within 6 months of initial diagnosis (n = 30), lack of postoperative determination (n = 12), or absence of measurement prior to systemic therapy initiation (n = 8). Patients with IMTCGS high-grade tumors exhibited shorter calcitonin doubling times compared with those with low-grade tumors (median: 1.04 years vs. 3.24 years, respectively; p = 0.21). However, this difference did not reach statistical significance, likely due to the limited sample size (n = 8 for low-grade, n = 17 for high-grade).
Systemic treatment management
Patients with IMTCGS high-grade tumors had a higher probability of initiating systemic treatment compared with those with low-grade tumors. Systemic treatment was administered to 56 out of 67 patients (83.6%) with IMTCGS high-grade versus 19 out of 32 patients (59.4%) with IMTCGS low-grade (p = 0.01) (Table 2). Of the 75 patients who received systemic treatment, eight did not receive TKIs: two died before February 2006, four received cytotoxic chemotherapy between February 2006 and February 2012 due to trial ineligibility, and two were treated after February 2012 due to contraindications to antiangiogenic TKI treatment. Among patients with RET mutation-positive tumors, 11 out of 22 (50%) with IMTCGS high-grade and 4 out of 8 (50%) with low-grade received a selective RET inhibitor as further-line treatment, with no significant difference in the proportion of patients receiving RET inhibitors between the two groups.
First-Line Treatments According to the International Medullary Thyroid Carcinoma Grading System
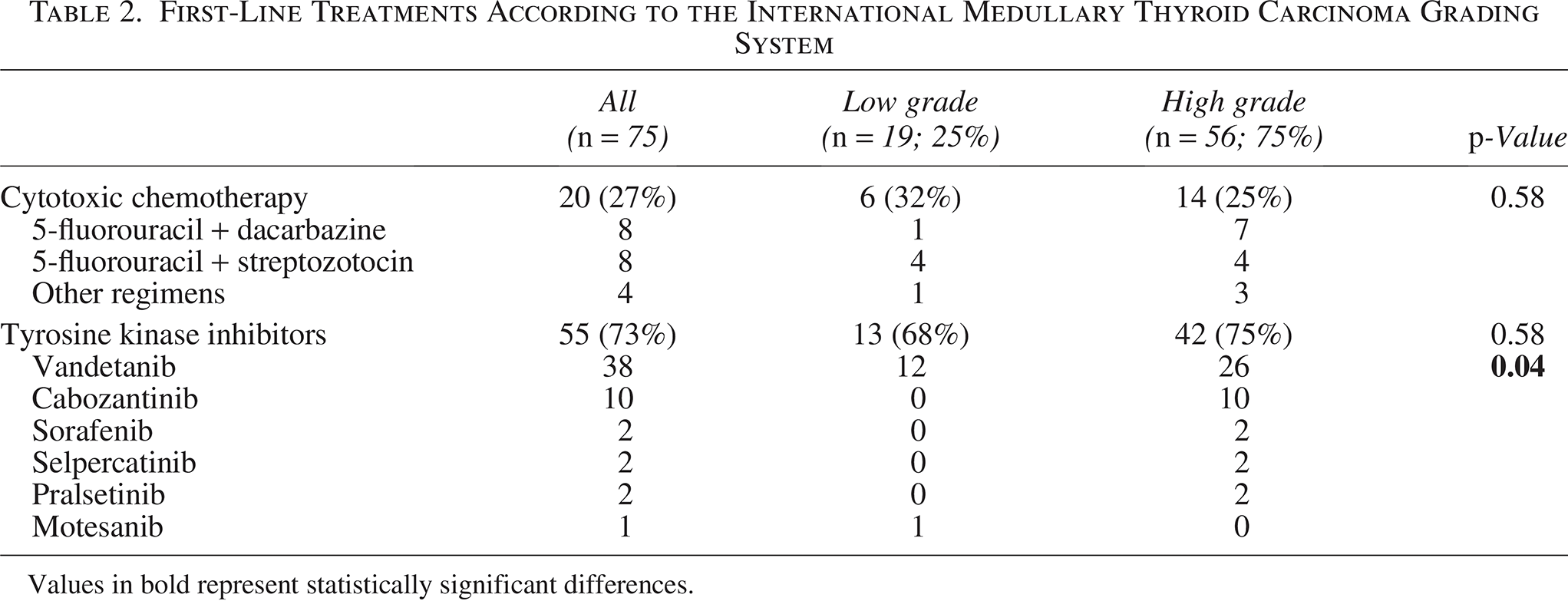
Values in bold represent statistically significant differences.
Survival analyses
During a median follow-up of 6 years (IQR: 2–11.5), death occurred in 61 patients. Disease-related death occurred in 51 patients (51.5%), while 10 patients (10.1%) died from other causes (2 from grade 5 treatment-related adverse events, 1 from a second cancer, 1 from Alzheimer’s disease, 1 from acute heart failure, 1 from colonic perforation, and 4 from unknown causes while in stable disease).
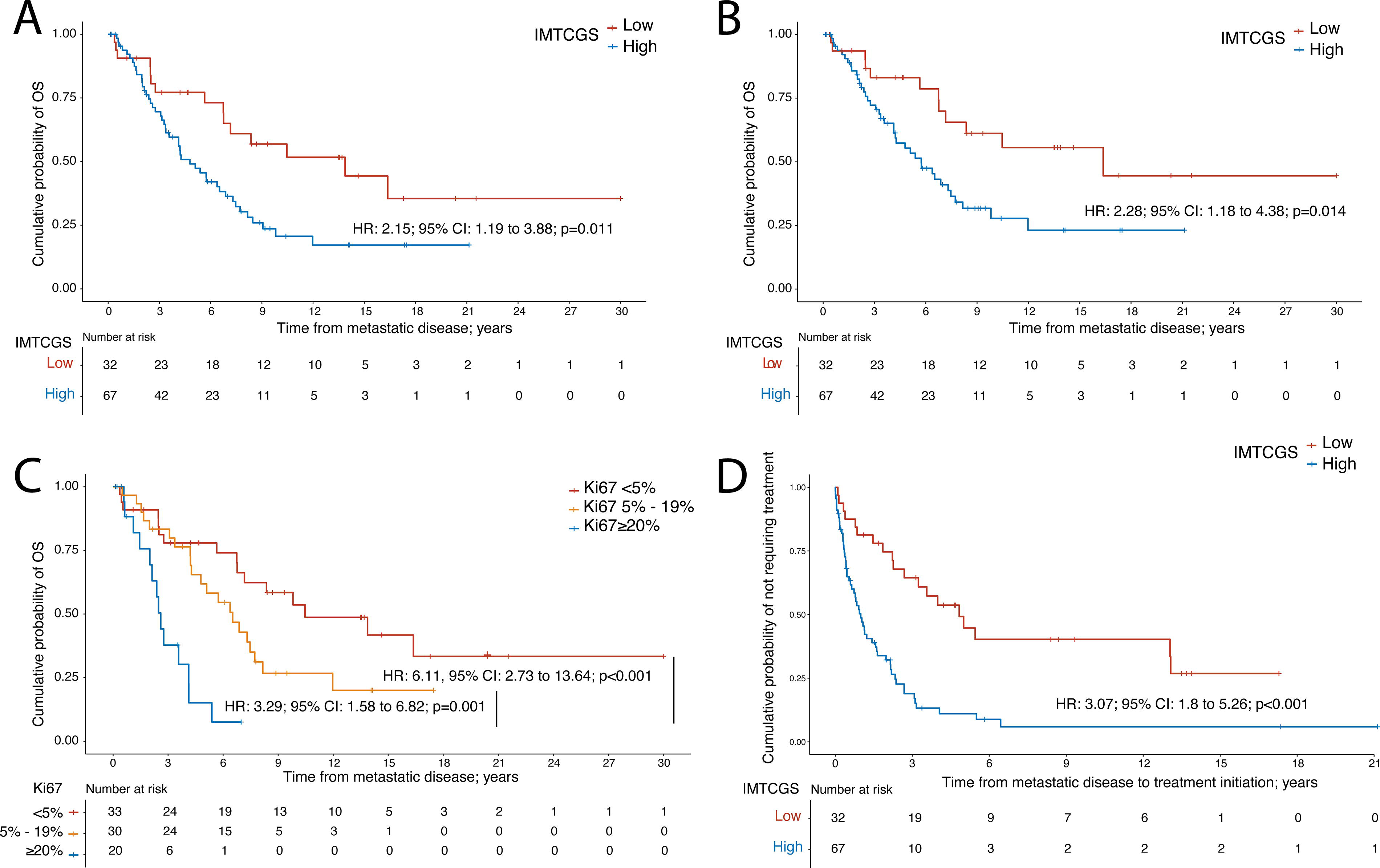
On univariate analysis, IMTCGS high-grade was significantly associated with worse survival. Median OS was 4.8 years [CI: 3.6 to 7.3] in patients with IMTCGS high-grade, compared with 13.9 years [CI: 6.8 to not estimable] in those with low-grade (HR = 2.15; [CI: 1.19 to 3.88]; p = 0.011) (Fig. 2A). Similarly, median DSS was 5.7 years [CI: 4.1 to 8.2] in the high-grade group, compared with 16.4 years [CI: 7.2 to not estimable] in the low-grade group (cause-specific HR = 2.28; [CI: 1.18 to 4.38]; p = 0.014) (Fig. 2B). Further stratification of MTC patients by Ki-67 index (<5%, 5–19%, and ≥20%), showed that patients with Ki-67 index ≥20% had a median OS of 2.6 years, which was significantly shorter than that of patients with Ki-67 index between 5% and 19% (6.5 years; HR = 3.29; [CI: 1.58 to 6.82]; p = 0.001) and those with Ki-67 index <5% (10.5 years; HR = 6.11; [CI: 2.73 to 13.64]; p < 0.001). Patients with Ki-67 index <5% showed a trend toward longer median OS compared with those with 5–19%, although the difference did not reach statistical significance (p = 0.066) (Fig. 2C).
In a multivariable Cox regression model, IMTCGS high-grade and the presence of bone metastases were independently associated with worse prognosis (Table 3). IMTCGS high-grade remained significantly associated with both worse OS (HR = 2.51; [CI: 1.05 to 6.02]; p = 0.039) and DSS (cause-specific HR = 2.41; [CI: 1.12 to 5.17]; p = 0.024). Additionally, bone metastases were independently associated with both OS and DSS in the multivariable analysis (HRs, CIs, and p-values provided in Table 3). When the individual components of the IMTCGS—Ki-67 index, mitotic index, and tumor necrosis—were analyzed separately in the multivariable model, only Ki-67 index ≥5% (HR = 2.38; [CI: 1.07 to 5.31]; p = 0.034) and mitotic index ≥5 (HR = 2.96; [CI: 1.36 to 6.43]; p = 0.006) were independently associated with OS.
Multivariable Analysis of Prognostic Factors for Overall Survival and Disease-Specific Survival
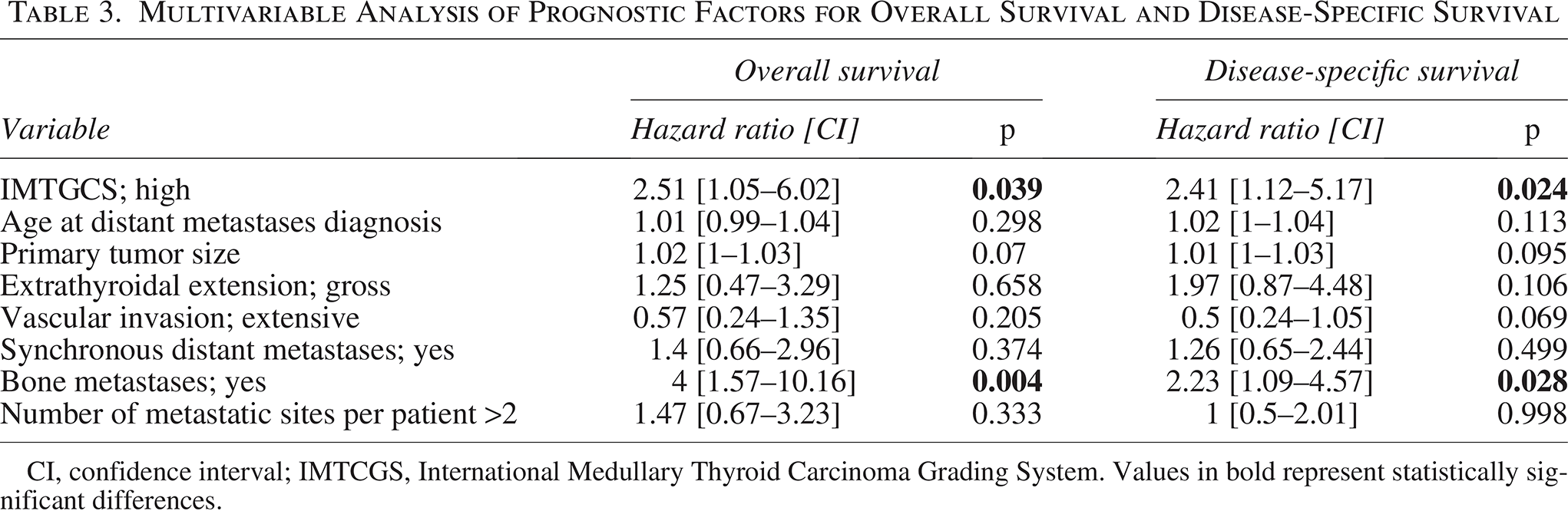
CI, confidence interval; IMTCGS, International Medullary Thyroid Carcinoma Grading System. Values in bold represent statistically significant differences.
IMTCGS high-grade was also significantly associated with a shorter TTI in univariate analysis (HR = 3.07; [CI: 1.8 to 5.26]; p < 0.001), with a median TTI of 1.0 year [CI: 0.6 to 1.6] in high-grade patients versus 4.8 years [CI: 2.7 to not estimable] in low-grade patients (Fig. 2D). In a multivariable Cox regression model, both IMTCGS high-grade (HR = 3.5; [CI: 1.74 to 7.06]; p < 0.001) and bone metastases (HR = 2.42; [CI: 1.26 to 4.65]; p = 0.008) were independently associated with a shorter TTI (Table 4). To further assess the prognostic relevance of the IMTCGS in patients who initiated systemic treatment, OS was compared between patients with IMTCGS low-grade (n = 19) and high-grade (n = 56) from the start of systemic treatment. No significant difference in OS was observed between the two groups (2.8 vs. 3.89 years, respectively; HR = 1.06; [CI: 0.57 to 1.96]; p = 0.865).
Multivariable Analysis of Prognostic Factors for Time to Treatment Initiation
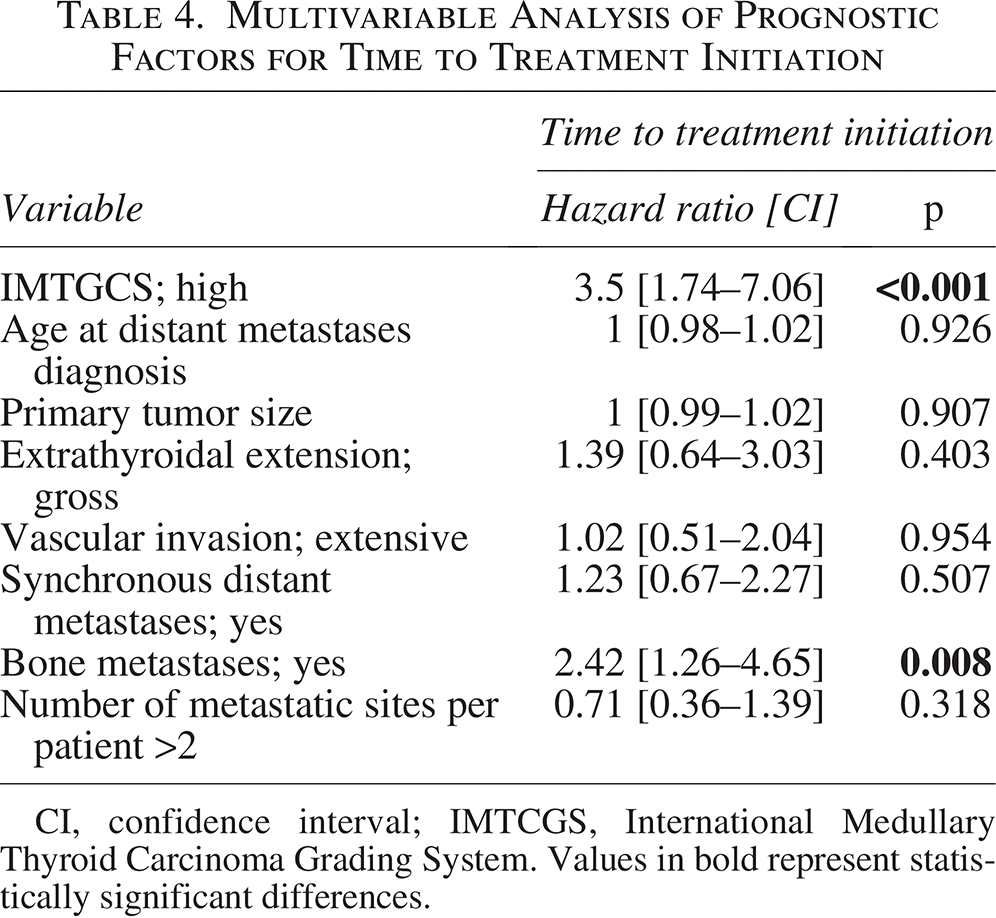
CI, confidence interval; IMTCGS, International Medullary Thyroid Carcinoma Grading System. Values in bold represent statistically significant differences.
Discussion
We analyzed 99 consecutive patients with metastatic MTC to investigate whether the IMTCGS could serve as a prognostic factor for survival in advanced disease. For the first time, we found that IMTCGS high-grade was independently associated with worse OS and DSS, compared with low-grade, in this subset of MTC patients. High-grade IMTCGS status was also associated with more aggressive tumor characteristics, a higher likelihood of initiating systemic treatment, and a shorter interval between metastasis diagnosis and systemic treatment initiation, all parameters supporting its prognostic relevance. Establishing IMTCGS as an independent prognostic factor marks a significant step toward more tailored management of patients with metastatic MTC, potentially guiding both follow-up strategies and therapeutic decisions.
Importantly, patients with metastatic MTC and a Ki-67 index ≥20% in the primary tumor—representing 20% of our cohort—had significantly worse OS compared with patients with Ki-67 index <20%, in line with observations reported in other neuroendocrine neoplasms.10,11 Based on our findings, we suggest that patients with a Ki-67 index ≥20% may benefit from a more intensive follow-up. A clinically meaningful difference in OS, though not statistically significant, was observed between patients with Ki-67 index <5% (10.5 years) and those with IMTCGS high-grade tumors and Ki-67 index between 5% and 19% (6.5 years), suggesting that the optimal grade categorization may require further validation in larger cohorts of patients with stage IV MTC.
The development of distant metastases marks a critical turning point from a locoregional to a systemic disease and shifts the treatment goal from cure to palliation. In our study, we used the time of distant metastases diagnosis as the baseline reference. While synchronous metastases may imply faster disease progression, the typically indolent course of MTC makes this assumption uncertain. The presence of synchronous distant metastases was not independently associated with survival outcomes. Furthermore, there was no significant difference in the proportion of patients with synchronous distant metastases between the IMTCGS low-grade and high-grade groups. Notably, patients with low-grade tumors demonstrated better outcomes than those with high-grade even when OS, DSS, and TTI were calculated from the date of primary tumor diagnosis (data not shown; p < 0.05 for all comparisons). Importantly, we found that bone metastases were independently associated with worse survival, consistent with previously published findings. 22
The tumor mutational status was available in approximately half of the patients, as genetic profiling was not routine prior to the availability of selective RET inhibitors. The RET-M918T mutation was not associated with worse survival compared with other RET mutations. However, its prognostic significance was assessed solely against other RET mutations, as the number of RET wild-type tumors was insufficient for analysis. The use of selective RET inhibitors may have impacted this result; however, the proportion of treated patients was similar between those with RET-M918T and other RET mutations, and the distribution of RET-M918T was similar between IMTCGS low- and high-grade cases. Notably, the prognostic significance of the IMTCGS has been shown to be independent of RET mutation status. 7 This finding aligns with data from phase 3 trials in metastatic MTC, where RET somatic mutations had no prognostic impact in placebo arms. 23 RAS mutations were identified in only 6% of tumors, all classified as IMTCGS low-grade, supporting previous evidence that RAS-mutated MTC tends to follow a less aggressive clinical course.8,24 Interestingly, a shorter calcitonin doubling time was observed in a subgroup of high-grade MTC patients. However, the sample size was too limited for meaningful conclusions.
As expected, the IMTCGS was strongly and independently associated with TTI. Changes in treatment availability and evolving clinical practices over time may have influenced the timing of systemic therapy initiation. However, both IMTCGS low-grade and high-grade patient groups were managed uniformly using the same decision-making criteria, ensuring consistency throughout the study period. By contrast, no survival difference was observed between IMTCGS high- and low-grade patients when measured from systemic treatment initiation. This finding supports the role of IMTCGS as a prognostic rather than a predictive factor.
This study has several limitations. The design was retrospective, but all patients with available primary tumor samples suitable for IMTCGS evaluation by highly experienced pathologists were included. As our Institution is a national referral center for cancer, there is a potential selection bias favoring the inclusion of patients with more aggressive disease. Nonetheless, the systematic case review by a dedicated tumor board with high expertise in endocrine tumors, along with the inclusion of multiple patients enrolled in clinical trials, ensured a standardized treatment approach across the cohort. Another limitation is the lack of standardized criteria for quantifying tumor burden in metastatic MTC. Bone metastases are frequently osteolytic and nonmeasurable, and lung metastases can also be nonmeasurable when they present in a miliary pattern. To address this issue, we considered both the location and number of distant metastatic sites per patient as surrogate indicators of tumor burden.
In conclusion, this study demonstrates, for the first time, that the IMTCGS is an independent prognostic factor for both OS and DSS in a large cohort of patients with metastatic MTC, after adjustment for tumor burden. These findings support the routine assessment of the IMTCGS grading in primary tumor specimens from all patients with distant metastatic MTC, as it may inform follow-up strategies and guide therapeutic decision-making. Patients with IMTCGS high-grade and a Ki-67 index ≥20% represent a high-risk subgroup with the poorest prognosis. Further validation in larger, multicenter, prospective cohorts and refinement of the grading categorization for this patient population are warranted.
Authors’ Contributions
T.P.: Investigation (lead), methodology (equal), writing—original draft (lead), and writing—review and editing (equal). S.M., J.H., D.H., I.B., and N.S.K.: Investigation (supporting) and writing—review and editing (supporting). M.-A.B.: Investigation (equal) and writing—review and editing (supporting). D.B.: Formal analysis (lead) and writing—review and editing (supporting). D.S. and D.D.: Writing—review and editing (supporting). M.S.: Investigation (supporting), supervision (supporting), and writing—review and editing (supporting). E.B.: Investigation (supporting), methodology (equal), supervision (supporting), and writing—review and editing (equal). A.A.G.: Conceptualization (equal), investigation (supporting), and writing—review and editing (supporting). L.L.: Conceptualization (lead), investigation (supporting), methodology (equal), and writing—review and editing (equal).
Footnotes
Author Disclosure Statement
T.P.: Honoraria: Eisai Europe. S.M.: None declared. M.-A.B.: None declared. J.H.: Consulting or advisory role: Eisai Europe, Lilly, Roche, PharmaMar, HRA Pharma; research funding: Novartis (Inst); travel, accommodations, expenses: Roche. D.B.: None declared. D.S.: Honoraria: Eisai Europe; consulting or advisory role: Eisai Europe. D.D.: None declared. M.S.: None declared. D.H.: Consulting or advisory role: Medtronic. I.B.: None declared. N.S.K.: None declared. E.B.: Honoraria: Ipsen, AAA/Endocyte/Novartis; consulting or advisory role: Ipsen, AAA HealthCare; research funding: Pfizer (Inst), Novartis (Inst), HRA Pharma (Inst), AAA/Endocyte/Novartis (Inst). A.A.G.: None declared. L.L.: Honoraria: Eisai Europe, Lilly, Roche; consulting or advisory role: Bayer, Eisai Europe, Ipsen; travel, accommodations, expenses: AAA Novartis, Ipsen.
Funding Information
No funding was received for this article.
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
