Abstract
Palliative Care Consultation (PCC) is an established intervention that prioritizes goals of care conversations and assists patients with complex medical decision-making. Evidence suggests that PCC can reduce the amount of non-goal concordant care patients receive such as fewer invasive procedures, less mechanical ventilation, and quicker transition to hospice and referral to appropriate resources, if that aligns with patient wishes. This results in decreased resource utilization, reduced anxiety for patients and their family members, and decreased moral distress for the health care team. Studies generally agree that benefits of PCC are greater the quicker the time to intervention, but results are mixed regarding hospital and intensive care unit (ICU) length of stay (LOS). LOS is a surrogate marker for goal-concordant care, and shorter LOS has been associated with decreased health care costs. This systematic literature review seeks to clarify the effects of early PCC versus standard PCC on length of stay. From an extensive search of CINAHL, EMBASE, Scopus, and PubMed, 17 articles met criteria to be reviewed. These articles include 2 randomized controlled trials (RCTs), 6 prospective cohort studies, and 9 retrospective cohort studies. Nine studies found statistically significant results linking early PCC with shorter length of stay. These results indicate that a change in practice could be considered given the multiple positive outcomes associated with early PCC. Further research is recommended with larger sample sizes across multiple institutions to improve generalizability of these initial findings, and to examine the different modalities of implementing early PCC into existing ICU workflows.
Introduction
In the United States, an estimated 1 in 5 Americans will die while utilizing ICU services that they may not have wanted. 1 Palliative care consultations (PCC) are a known tool that help clinicians provide goal concordant care, alleviating anxiety and moral distress for patients, their families, and the care team. 2 Palliative care (PC) is an interdisciplinary specialty that emphasizes prevention and relief of suffering by focusing on symptom management, psychosocial well-being, shared-decision making, and patient-directed goals of care.1,3,4 The current body of literature shows PCC is associated with numerous positive outcomes, such as improved quality of life; increased patient and caregiver satisfaction; fewer emergency room visits; reductions in unplanned hospitalizations; and lower rates of caregiver burden.1,2
Research indicates involvement of PC is beneficial early in the disease course, but many factors affect whether a patient will receive PC interventions. In 2017, a multidisciplinary, multicenter set of surveys indicated that ICU clinicians, including nurses, physicians, and advanced providers, agree PC is underused. 5 The same survey highlighted difficulties with ordering PCC, citing confusion or disagreement over what factors necessitate one. 5 There is no standard screening or assessment tool to determine which patients would benefit from PC involvement – PCC typically come from a subjective assessment by the primary care team responsible for the patient or from meeting a list of institution-specific triggers. 6 Many of these institution-specific screening tools are based on data and recommendations from the Improving Palliative Care in the ICU (IPAL-ICU) Project in 2013. 7
In some cases, patients and/or clinicians are not aware that palliative care can be offered concurrently with curative treatments, and so clinicians may not offer palliative consults especially in critical care units for fear it is seen as undermining life-saving efforts. 8 Patients in the ICU have high rates of unmet symptom needs and complex medical decision-making8-10 and may be missing valuable services that could guide goal-concordant care.
Purpose
There is no specific timeline for PC involvement for critically ill patients, although the Center to Advance Palliative Care urges proactive PCC. 7 The American College of Surgeons’ Palliative Care Best Practices Guidelines 11 recommend evaluation for PCC within 24 hours of admission and a structured family meeting for critically ill patients within 72 hours. As a result, 72 hours is widely accepted as the target time-to-intervention for PCC.
Amidst many positive outcomes associated with PCC, the data regarding the effects of early PCC on ICU patients has been mixed and studies have not consistently shown statistically significant results. 12 Articles included in this review consider the embedded PC model as well as referral-based PC, both with the intent to decrease time from admission to PC intervention. The embedded model consists of a PC provider or team that is based within the unit where they work and are part of the established care team. A referral-based model aligns with the typical consult service model seen in hospitals, where the primary team refers patients to receive PC from a separate palliative service that may work with patients across the entire hospital.
Length of stay was the primary outcome chosen because it is related to several secondary outcomes of interest to the healthcare system at large, such as decreased costs for patients and hospitals.1,3,10,12,13 This review also considers the rate of invasive procedures, code de-escalation, and transition to hospice care. Evidence suggests that shorter LOS in the ICU and fewer invasive interventions are surrogate markers of goal-concordant care and decreased patient suffering, which should also reduce clinician moral distress related to providing care that is medically futile or not aligned with patient wishes.3,6,9,12 While previous research supports the use of LOS as a marker of goal-concordant care, it is understood that there may be individual patient scenarios where prolonged care in the ICU and invasive procedures align with patient wishes. Therefore, this literature review aims to determine the effect of early PCC on length of stay in the hospital and/or ICU compared with usual PCC for critically ill adults.
Methods
Search Strategy
Literature Search Strategy for Palliative Care Timing in Critical Care, all Searches Completed Between May 20th and September 23rd, 2025

Review Process
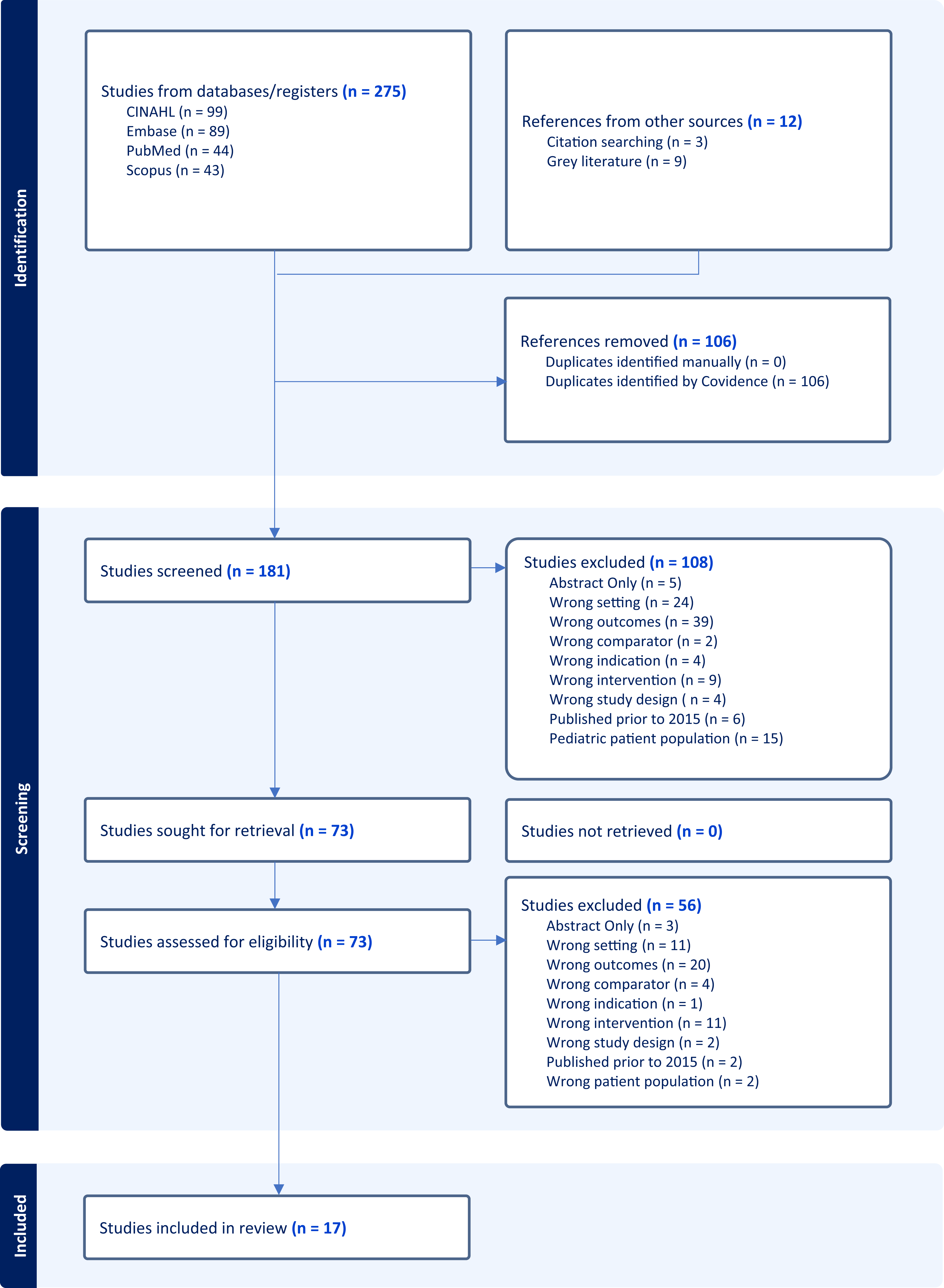
Two hundred and eighty-seven articles met criteria for review between the 4 databases and additional searches. Once the articles were consolidated, duplicates were removed, leaving 181 sources for review. The titles and abstracts of these articles were screened by a single reviewer. Based on the original filters from the database searches, 108 studies were excluded. Primary reasons for article exclusion beyond the established parameters included studying patients outside of ICUs or failing to consider length of stay as an outcome. This resulted in 73 full text articles that were then assessed for eligibility. Seventeen studies were included in the final review, consisting of: 2 Randomized Control Trials (RCTs), 6 prospective cohort studies, and 9 retrospective cohort studies. For a detailed view of the process of article selection during each phase of the review, see the PRISMA diagram, Figure 1. PRISMA Diagram
14
Data Extraction
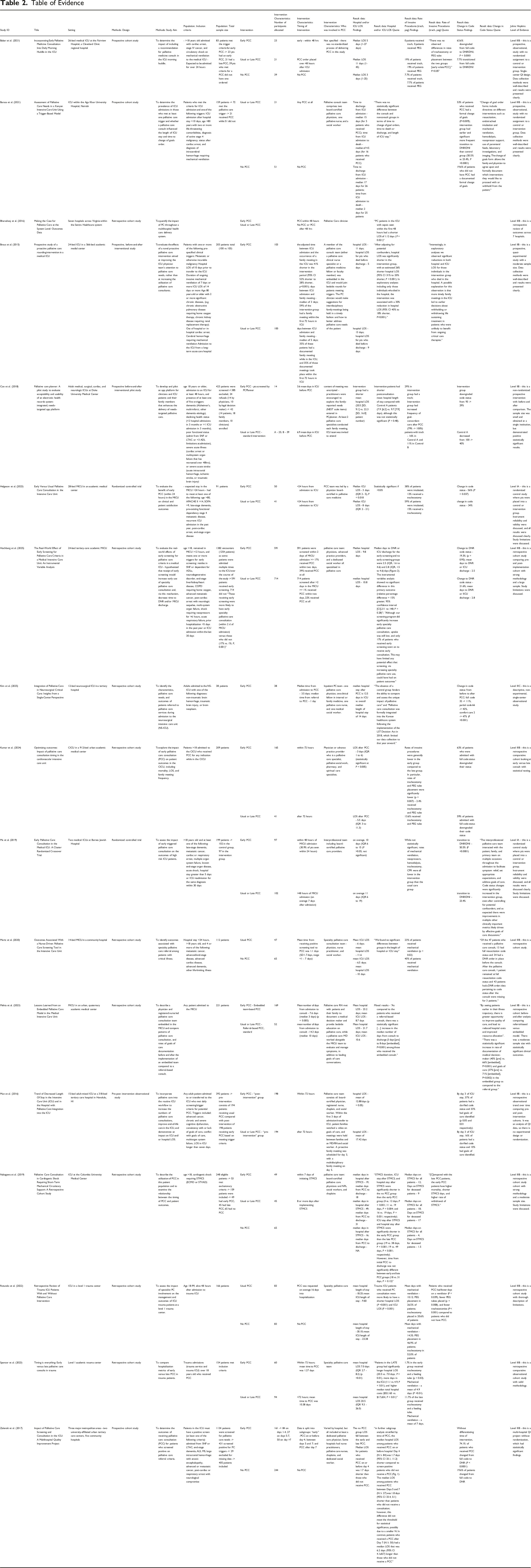
Table of Evidence
Critical Appraisal
Each study was critically analyzed for the quality of its evidence and graded according to the Johns Hopkins Evidence-Based Practice Model and can be found in the final column of the TOE. 15 Two articles were rated high quality, 14 medium quality, and 1 low quality. No articles were excluded for quality reasons.
Results
Patient Populations
Sample size ranged from 38 16 to 1,035 17 patients. The mean age across studies was 60-77 years old, barring 2 studies. Barasa et al 18 included a younger patient population, with a mean age of 56 and 58 years old in the intervention and control groups, respectively. Nakagawa et al 6 also had a younger population with mean ages of 55.3-60.9 years old. Race and gender varied more widely across studies but were well-matched between intervention and control groups within each study. No studies reported notable group differences. Details regarding group demographics can be found in Table 2.
Inclusion and Exclusion Criteria
Inclusion criteria varied across the 17 studies. Each study included patients over the age of 18 who were admitted to the ICU. Four studies12,18-20 referenced the IPAL-ICU criteria 7 and used that screening tool as a base for their individual criteria, opting to include chronic and severe cognitive dysfunction, metastatic cancer, cardiac or respiratory arrest, multiple organ system failure, known end-stage organ disease, and recurrent hospital and ICU admissions within a specific time frame among the list of PC triggers.
Seven studies12,17,21-25 relied on triggers determined in prior RCTs taken in consideration with internal data collected from their institution and suggestions made by experienced ICU clinicians. For example, Cox et al 24 evaluated PC triggers that clinicians flagged as valuable, and which had evidence of association with readmission, symptoms, and mortality. Ultimately, Cox et al 24 included patients receiving mechanical ventilation or in shock for greater than 48 hours after ICU admission, in addition to experiencing 1 of the following: dementia, declining health status, poor functional status, severe acute illness, or severe acute stroke.
All studies excluded patients younger than 18 years old, patients or surrogates who refused a PCC, and patients who already had a PCC prior to admission to the ICU. Multiple authors excluded patients who died before PCC or who were expected to be discharged from the ICU within <24 hours because it was deemed inadequate time for a PCC to take place.21,22,24 Other exclusion criteria included patients already having their code status as do-not-resuscitate and do-not-intubate (DNR/DNI) prior to ICU admission or within 6 hours of transfer12,17 and insufficient data from medical records for the retrospective studies.
Study Design and Methods
All studies reviewed were published between 2015 and 2025. The only studies published from outside of the United States were conducted in a university hospital in Kenya 18 and a tertiary hospital in South Korea. 16
Thirteen studies occurred in a single ICU within 1 institution. Ma et al 12 included patients from 2 ICUs in the same hospital. Cox et al 24 included patients from adult medical, surgical, cardiac, and neurological ICUs within the same institution. Zalenski et al 25 collected data from ICUs in 2 university-affiliated hospitals and 5 community hospitals. Bharadwaj et al 26 examined multiple ICUs across 7 hospitals in 1 health system. Of note, Bharadwaj et al (2016) conducted 5 different retrospective studies reported in 1 article. Studies 2 and 4 specify that all patients involved were in the ICU, 26 so only results from those studies are considered in this portion of the review.
Early PCC was the independent variable in all the studies conceptually. However, early PCC was defined differently across studies. Four studies19,23,27,28 in this review considered early PCC as intervention within 72 hours of admission to the ICU; 1 study 21 considered consultation within 24 hours of admission to the ICU; and 3 studies12,22,26 considered consultation within 48 hours of admission to the ICU. Hochberg et al 17 screened patients within 48 hours of admission to the ICU, but only 17% of those patients received PCC within 48 hours. PCC placed outside of these windows were considered late PCC, usual intervention, or standard of care. Four studies did not define early PCC by a specific time frame because they considered PCC for patients who met the triggers as the intervention and usual intervention was considered no PCC at any point during the ICU stay.8,16,18,20
Helgeson et al 21 and Ma et al 12 conducted RCTs by randomizing eligible patients to either a control group or an intervention group by a random number generator. In the intervention groups, patients received early PCC within 24 and 48 hours of admission to the ICU, respectively. In the control group, PCC was ordered as needed at the discretion of the ICU intensivists or as requested by patients or surrogate decision makers, if at all.
There was no standardization of who conducted the PCC and what a PCC consisted of from study to study. Thirteen of the studies had a PC team consisting of at least a board-certified PC physician, often also including advance practice providers, registered nurses, and dedicated social workers and chaplains.6,9,12,16-21,23-25,27 Four studies8,22,26,28 did not define the members of the PC team or give insight as to their level of expertise or specialty certification. Priorities in PCCs included discussing advance care planning and completing an advance directive, identifying a surrogate, addressing code status, identifying what needs remain unmet, facilitating family meetings, and updating goals of care based on prognosis.
Outcomes
Nine studies6,8,19,21,23,24,26-28 found a statistically significant decrease in length of stay for patients who received early PCC. Helgeson et al 21 examined the shortest time to intervention and found ICU LOS was shorter for patients who received PCC within 24 hours of ICU admission (P = 0.018) at 3 days, versus 8 days for patients in the control group who received late or no PCC. Bharadwaj et al 26 examined PCC within a 48 hour window found patients had significantly shorter ICU LOS. Four studies19,23,26,28 found hospital and ICU LOS was significantly shorter for patients who received PCC within 72 hours of ICU admission. Nakagawa et al 6 examined the longest time to intervention, considering early PCC as intervention within 7 days of initiating short term mechanical circulatory support, usual or late PCC as intervention after eight or more days on short-term mechanical circulatory support, and compared both groups to those who did not receive any PCC in the ICU. Even considering the longest time to intervention in this review, Nakagawa et al found median days from PCC to discharge significantly shorter (P < 0.001) for those who received early PCC compared to those who received usual care, at 18 and 31 days respectively.
While not statistically significant, 5 studies12,16-18,25 showed a decrease in hospital and/or ICU length of stay for patients who received early PCC. Barasa et al found ICU LOS to be 2 days shorter for patients who received PCC, 18 while Hochberg et al and Ma et al found patients who received early PCC had a shorter hospital stay by about 1 day compared to groups who received usual or late PCC.12,17 Kim et al 16 did not have a control group but found that the median hospital LOS for those who received PCC while in the neurosurgical ICU was 12.5 days, which was shorter than the overall average hospital LOS of 14 days. Zalenski et al 19 compared 4 distinct sets of data: PCC on or before day 4, between days 5 and 7, and PCC after day 7, all compared to the control group who did not receive PCC at all while in the ICU. Results indicated patients who received PCC between days 1 and 7 had a shorter median length of stay than those who did not receive PCC, however the patients who received PCC after day 7 had a median length of stay many days longer than those without PCC.
The final 3 studies in this review found mixed results. Babar et al 22 reported a shorter median ICU LOS of 5 days for early PCC and 11 days for late PCC, however the cohort that did not receive PCC at all had the shortest median LOS at 3 days. Mehta et al 9 compared patients who received an embedded team-based PCC that was generally 3 days after ICU admission, against patients who received the standard referral-based PCC a median of 10 days after admission to the ICU and found that mean ICU LOS was shorter than in the usual PCC group. However, in this study, the median number of days from PCC to discharge was significantly longer in the embedded team-based cohort that received early PCC (8 days vs 3 days, P < 0.001). 9 Martz et al 20 found that patients who received PCC had a shorter mean ICU LOS but longer hospital LOS than the cohort of patients who did not receive PCC. This was the only study in this review where hospital and ICU LOS did not correlate after intervention.
Overview of Pertinent Findings for the Intervention Group (Patients Who Received Early/Any PCC) Compared to the Control Group (Patients Who Received Usual/Late or No PCC) Within Each Study
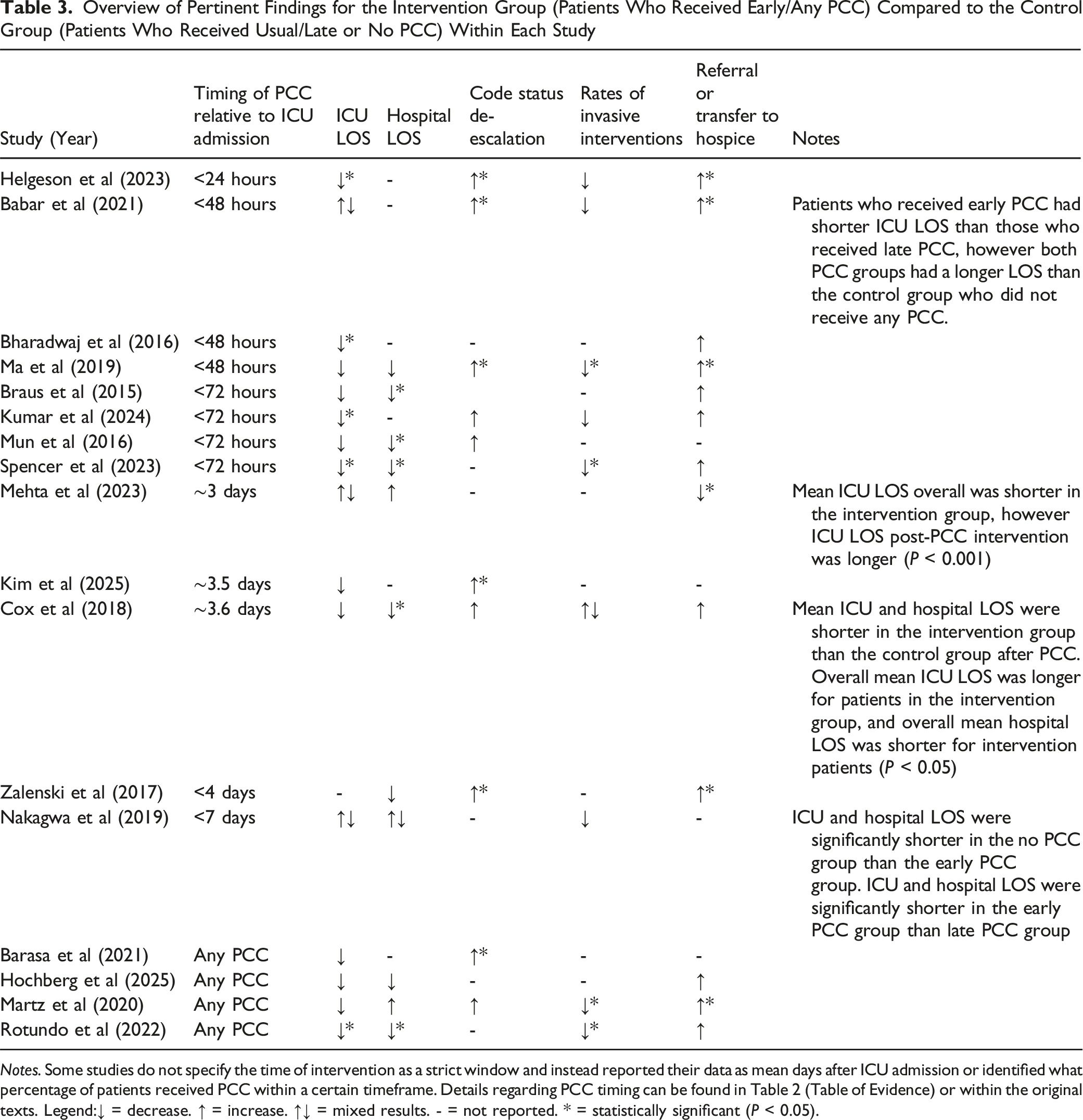
Notes. Some studies do not specify the time of intervention as a strict window and instead reported their data as mean days after ICU admission or identified what percentage of patients received PCC within a certain timeframe. Details regarding PCC timing can be found in Table 2 (Table of Evidence) or within the original texts. Legend:↓ = decrease. ↑ = increase. ↑↓ = mixed results. - = not reported. * = statistically significant (P < 0.05).
Helgeson et al 21 found that 56% of patients who received early PCC changed their code status to DNR/DNI, compared to 34% of patients in the control group (P = 0.037). Ma et al 12 found patients who received early PCC de-escalated their code status more frequently and earlier in the hospital stay than patients in the control group (50.5% vs 23.4%, P = <0.0001). Babar et al 22 determined patients who received early PCC were more likely to transition to DNR/DNI than those who did not have PCC (63.6% vs 7.7%, P < 0.0001). While more patients who received early PCC transitioned, patients who received PCC at any point during their hospital visit also transitioned to DNR/DNI significantly more often than patients who did not receive PCC (P = 0.0001). 22
Referral or transfer to hospice care occurred much more often in intervention groups than control groups. For example, Helgeson et al 21 found 42% vs 15% of patients enrolled in hospice (P = 0.004); Ma et al 12 found 18.6% vs 4.9% patients entered hospice (P = 0.0026); and Babar et al 22 found 36% compared to 2.6% patients transitioned to hospice (P = 0.0001).
Discussion
Primary findings in this literature review indicate that early PCC has an impact on length of stay in the ICU and the hospital overall. The methods of implementing early PCC ranged from an embedded model and a trigger-based referral model to an integrated electronic health-record app that triggers targeted PC. Regardless of the specific delivery model, multiple studies found that early screening for unmet PC needs led to earlier PCC in the ICU, ultimately decreasing LOS.
Of note, data for multiple variables was reported by median and mean, which makes comparison of results difficult across studies. The median was more likely reported due to significant variation or standard deviation that would skew results if reported as the mean, and so it gives a more appropriate sense of the effect of the intervention. The data was notable for other findings related directly to components of a comprehensive PCC and thus show a positive relationship between PCC and patient-centered goals of care being met.
The findings of this literature review underscore the persistent lack of standardization in both the implementation and operationalization of PCC in the critical care setting. Across these studies, there was considerable variability in the tools, criteria, and triggers used to initiate PCC. As detailed in the Results section, the most common approach to initiating PCC relied on institution-specific triggers, often shaped by individual clinician experience and informed by broader recommendations like those from the Improving Palliative Care in the ICU (IPAL-ICU) Project. 7 While these guidelines have provided a valuable framework, they have not been universally adopted in a standardized manner.
This variability introduces significant opportunities for bias in determining which patients receive PC, particularly in observational or non-randomized study designs. Studies demonstrated that without random assignment to PCC, clinician discretion played a central role in consult initiation, leaving room for subjective factors to influence patient selection. For example, Babar et al. 22 found the most common reason that eligible patients did not receive early PCC was due to clinical decisions made by their primary team. Data was not collected regarding why teams opted not order PCC in most studies. Future studies would be beneficial to determine reasoning behind the choice not to order PCC for patients who meet validated triggers. Such bias complicates the interpretation of outcomes and limits the generalizability of findings across institutions.
Furthermore, several studies lacked clarity regarding the composition of the PC team delivering the intervention and the specific objectives of PCC for each patient. Specialty trained PC clinicians should be able to thoroughly assess patients and conduct goals-of-care discussions, beyond non-specialty trained providers performing primary PC. Without consistent reporting on whether PCC focused on goals-of-care discussions, symptom management, or psychosocial support, it becomes difficult to assess the true impact and efficacy of these interventions. This gap highlights the need for greater transparency and standardization in both the initiation and documentation of PC involvement in the ICU setting.
Of note, 2 of the studies included in this literature review were conducted outside of the United States. As such, there are cultural, political, and epidemiological differences that may impact the generalizability of the findings from those studies to the critically ill patients in the United States. The Barasa et al study was conducted in Kenya, while the Kim et al study was based in South Korea. Kim et al 16 noted that the Life Sustaining Treatment Decision Act, passed in 2018, widely expanded the availability of PC and hospice in services in South Korea, but patient and family knowledge and acceptance of these services was still in the early stages. Barasa et al 18 enrolled a younger patient population, possibly due to the difference in life expectancies in Kenya as compared to the United States, with a life expectancy of 79.61 years in the United States vs 64.01 years in Kenya. 29 Age of the patient is often a factor considered along with overall health status, as people tend to accumulate comorbidities over time. The difference in age may have impacted study findings in a way that was not seen in the other studies.
Nakagawa et al 6 also had a younger population with mean ages of 55.3 – 60.9 years. Because this study looked specifically at PCC for patients on short-term mechanical circulatory support, there were likely other factors like comorbidities and protocols, that reduce the likelihood older adults will receive short-term mechanical circulatory support, resulting in a younger population.
Strengths and Limitations
In this review, 2 studies included patients across multiple institutions8,25 and 2 studies evaluated more than 1 unit within a single institution.12,24 Due to the limited sample sizes and the predominance of single-unit, single-institution designs, the findings of most studies in this review are not broadly generalizable. However, this review encompasses multiple studies involving patients from a wide range of critical care subspecialties, including mixed ICUs, surgical, medical, trauma, neurosurgical, and cardiovascular ICUs. This diversity in patient populations helps mitigate subspecialty-specific bias and enhances the applicability of findings across the broader critical care setting.
The patients in all included studies, while balanced within their intervention and control groups, were not reflective of the larger population in the United States and this is a source of sample bias and minimizes generalizability of the results of this review. It is possible that lack of racial diversity and limited data collection on religious and spiritual beliefs may have also obscured bias regarding the willingness of patients and families to participate in PCC or extensive life-sustaining therapies like mechanical circulatory support or receiving blood products.
A key limitation of this review is the heterogeneity in PC intervention design and delivery. Timing varied widely, with no consistent definition of ‘early’ vs ‘late’ PCC, complicating conclusions about optimal consultation timing but underscoring its potential impact when provided within specific time frames. Many studies also lacked detail on the personnel delivering PCC; while some described team composition, none specified which members provided bedside care. Similarly, intervention goals and objectives were rarely reported. This variability in structure and intent limits comparability and interpretation of outcomes across studies.
Conclusion
The results of this literature review are promising, but not conclusive for changing practice at this time. Further research is needed to determine the best way to implement earlier PCC amid already over-extended PC providers and busy shifts in the ICU. PC can be provided by any clinician, and while the results are not as well studied as interventions provided by a PC specialist, patients still derive benefits from primary PC. Decreased hospital and ICU length of stay, reduced rates of invasive procedures and non-goal concordant care, and code status de-escalation are only a limited example of the appealing outcomes tied to early PCC. These outcomes are reasonable proxies for an expected increase in patient and caregiver satisfaction, decreased moral distress for health care providers, and reduced healthcare resource utilization and costs. The goal of providing early PC, as evidenced by the literature, is to have more time to intervene and thus benefit from having a greater opportunity to tailor care to a patient’s needs and wishes. Effective PC lends itself to improvement in many clinically important metrics 9 because of the patient-centered nature of the specialty and emphasis on communication.
To advance the field, additional research is recommended with larger sample sizes across multiple institutions to produce statistically meaningful and generalizable results. There was also no consensus across the literature of the best way to integrate PC into the workflow in the ICU – Mehta et al. 9 was the only study that compared an embedded team-based model and a referral-based model, which showed mixed results.
Footnotes
Ethical Considerations
Not applicable as there is no original research in this manuscript. All references have been properly cited throughout this work.
Author Contributions
All authors meet the ICMJE criteria for authorship.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data used in this review comes from published and peer-reviewed works. This literature review cites all the studies referenced appropriately.
