Abstract
Keywords
Introduction
Dyspnea (breathlessness) is a common and highly distressing symptom of chronic cardiorespiratory conditions. Over 90% of patients with Idiopathic Pulmonary Fibrosis (IPF), the most common form of fibrosing lung diseases, present with dyspnea. 1 Dyspnea is associated with mortality and poor health-related quality of life in IPF.2,3 Despite the availability of several validated dyspnea scales, in clinical practice, systematic assessment of dyspnea is not routinely performed, and there is no universally accepted approach to managing dyspnea in patients with IPF.3-6
The Medical Research Council (MRC), or modified MRC (mMRC), is the most commonly used dyspnea scale worldwide because of ease of use and brevity. 4 The scale measures disability due to dyspnea but does not assess dyspnea severity directly. The American Thoracic Society (ATS) defines dyspnea as “a subjective experience of breathing discomfort that consists of qualitatively distinct sensations that vary in intensity.”. 7 Hence, assessment of severity is essential for comprehensive dyspnea evaluation. In one study, 90% of US-based hospitalists stated that awareness of dyspnea severity rating influences their management decisions, and 65% reported it influences their decision to prescribe opioids. 8 The American College of Chest Physicians (ACCP) statement on dyspnea management in patients with advanced lung or heart disease recommends that patients should be asked to rate their dyspnea, and this rating should be used to guide management. 9 Yet, only 2.3% of practitioners report using a scale in clinic to rate dyspnea severity. 8
Several multidimensional dyspnea tools exist including one that measures dyspnea severity, but they are typically used only in research settings,and it is unclear whether or how their results effect dyspnea management. 10 Patients with interstitial lung disease (ILD) and various stakeholders also recommend the use of a numeric ratings scale to measure dyspnea and its impact on physical function and daily living. 11 We developed the Edmonton Dyspnea Inventory (EDI), a simple, easily-administered patient-reported outcome measure to assess dyspnea severity in the clinic. In this study, we examine the validity of EDI scores for assessing dyspnea in patients with IPF. We hypothesized that EDI is a unidimensional tool with acceptable internal consistency and that its scores possess validity for assessing dyspnea severity across activities of varying intensity in patients with IPF; specifically, item scores would be associated with MRC scores, measures of IPF severity and mortality.
Methods
This is a retrospective study of IPF patients seen in the Multidisciplinary Collaborative ILD clinic, Edmonton, Canada. The study population includes all consecutive IPF patients with baseline MRC and EDI (Nov 2012 – Jun 2018) who were eligible for inclusion. IPF diagnosis was made in accordance with the ATS 2011 guidelines. 12 IPF patients without baseline MRC and EDI were excluded. All data were extracted from the medical record and included demographics, disease history, pulmonary physiology (percent predicted forced vital capacity or FVC%, diffusion capacity or DLCO%), submaximal exercise (6-minute walk test or 6MWT) and questionnaire response data. Pulmonary physiology and 6MWT were performed within 7 days of the baseline visit and according to ATS standards. Gender-Age-Physiology (GAP) stages were calculated using baseline data. 13
Edmonton Dyspnea Inventory (EDI)
Development
We developed the EDI to assess dyspnea in patients with IPF. We aimed to create a dyspnea severity profile across three phases (with activities of daily living (ADL), exercise and rest) to inform the systematic application of dyspnea therapies.14,15 The original EDI had 8 items, ordered by metabolic equivalents, from low to high intensity, and response options on a numeric rating scale, ranging 0-10 (0: no breathlessness to 10: worst possible breathlessness). 16
The items were chosen for their relevance to daily life and include rest, eating, talking (low metabolic demand), showering, dressing, bowel movement (moderate metabolic demand), stairs and exercise (high metabolic demand). 17 Based on item review and informal patient feedback, walking was recently added as a new item to make the current 9-component EDI. Scores for walking were not included in this analysis due to limited data collected during this period (n = 17).
In addition, we ask about dyspnea crises as these episodes result in frequent hospitalizations and lead to panic and anxiety amongst patients and their informal caregivers. This is a free text response detailing triggers for acute onset distress, event details and resolution or outcomes.
Administration
The clinic nurse or physician administers the instrument in person or by phone. Wording for items is as follows: “Please score your breathing when (state context)”. The question about crisis dyspnea is phrased as follows: “Have you had episodes where your breathing worsened to point of needing urgent help (felt like calling or called 911 or going to hospital)?" If yes, number of episodes and context are documented.
Scoring
Each item (except crisis dyspnea response) yields a score; there is a total score. We use the individual scores for each item to determine therapies, track progression and assess responses to intervention. Per convention, item scores ≥4/10 are considered moderate to severe.18,19
Statistical Analysis
We report means (SD) or median (IQR) for continuous variables and counts and percentages for categorical variables.
Dimensionality, Structure and Internal Consistency
We conducted several psychometric analyses to examine the structure of the EDI instrument and its individual items; these included exploratory factor analysis, item correlational analysis (item-item and item-total [without the given item] using Pearson correlation), and an examination of internal consistency using Cronbach’s coefficient alpha.
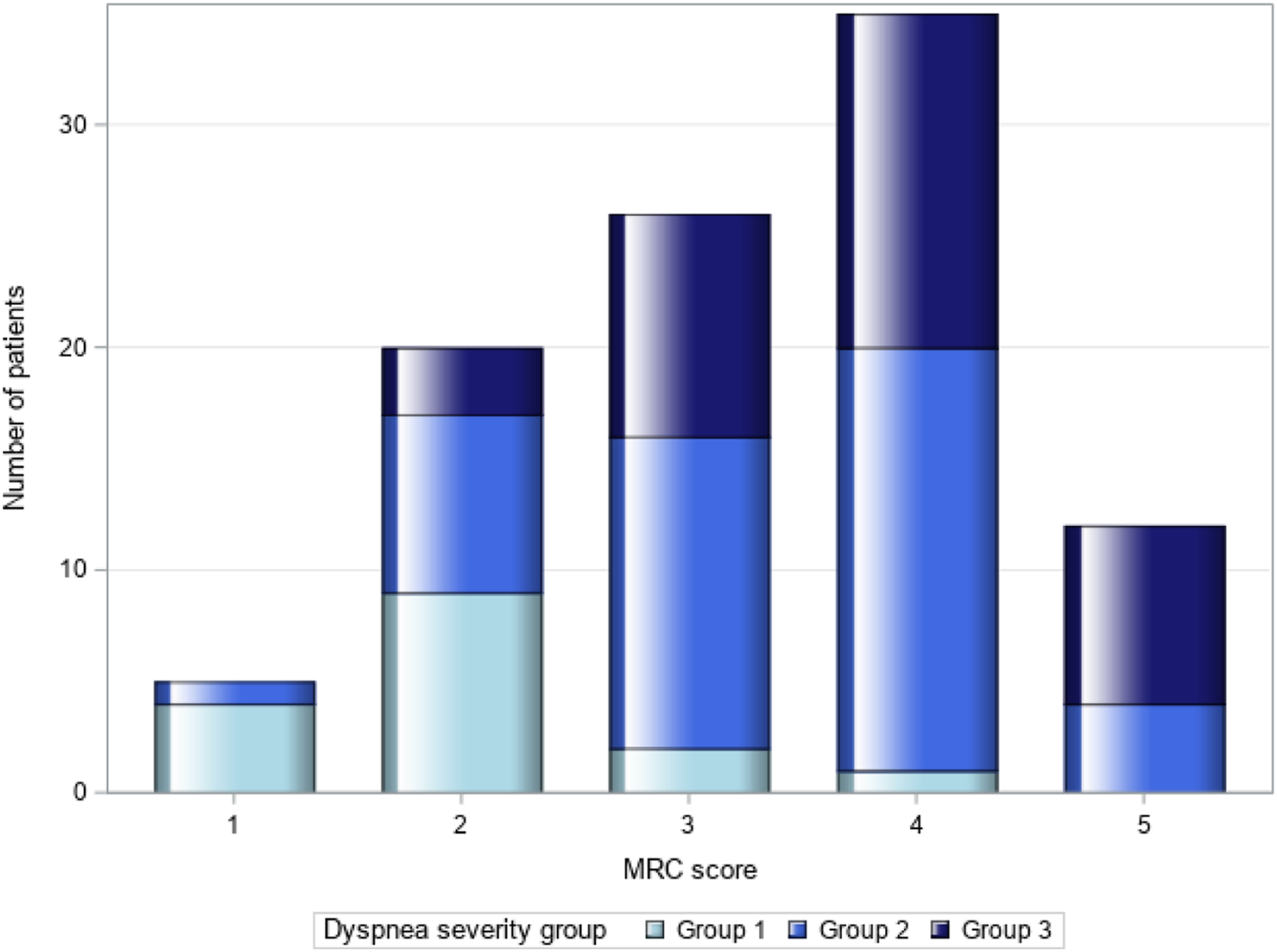
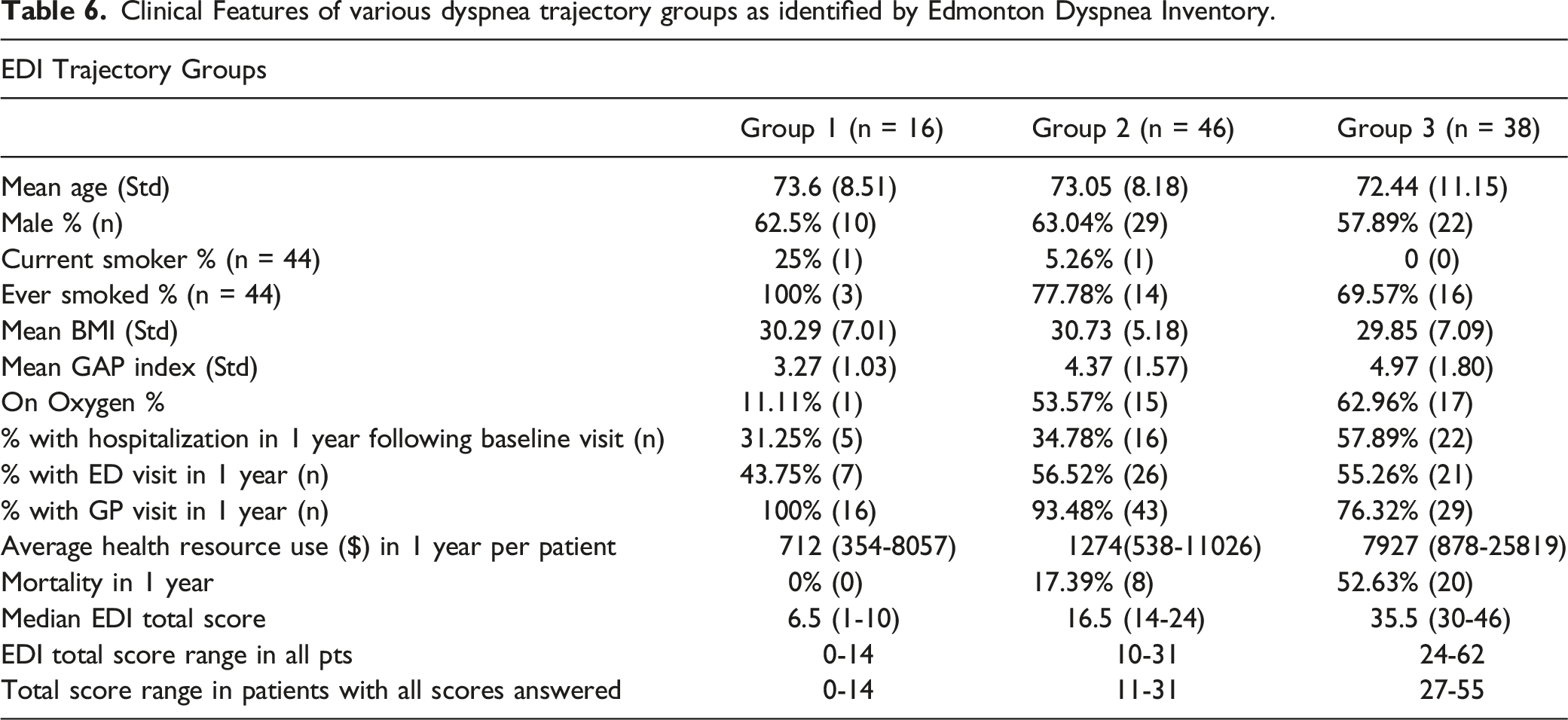
We used group-based trajectory modeling (GBTM) on EDI scores to group patients with similar patterns of dyspnea behavior and to further explore the response structure. We ordered the components of EDI by the metabolic equivalents from low intensity to high intensity in order (rest, talking, eating, bowel movement, dressing, showering, exercise and stairs) and applied the GBTM to identify groups of patients following similar patterns of dyspnea scores with the increasing intensity of activities. The order of exercise and stairs is arbitrary (some patients treated walking around house as exercise), so the model was also tested by switching these two activities. 20 We also compared the baseline clinical features and survival amongst the 3 groups identified by GBTM.
Validation
We examined the relationship between EDI scores, MRC grade and IPF severity (FVC%, DLCO%, distance walked during 6MWT) using one-way ANOVA and Spearman correlation coefficients to establish convergent validity.
Survival Analysis
In order to explore the relationship responses to the EDI we created a series of logistic regression models. In order to quantify the impact of adding the EDI to the other predictors of mortality we calculated net reclassification improvement (NRI) and pseudo-c indices. 21 NRI was calculated comparing the 1-year mortality model with MRC as the only predictor to a model with both MRC and EDI as predictors. Subsequently we compared the model using age, gender and FVC as predictors to one with the additional EDI variable. Missing Data were considered missing at random.
All statistical analyses and visualisations were conducted using SAS 9.4 (Cary, NC.), R 4.0.0 (Vienna), and Tableau (Seattle, WA.). This study received ethics approval from the University of Alberta Health Ethics Research Board (Pro00082981).
Results
Demographic Characteristics and Patient Profile.
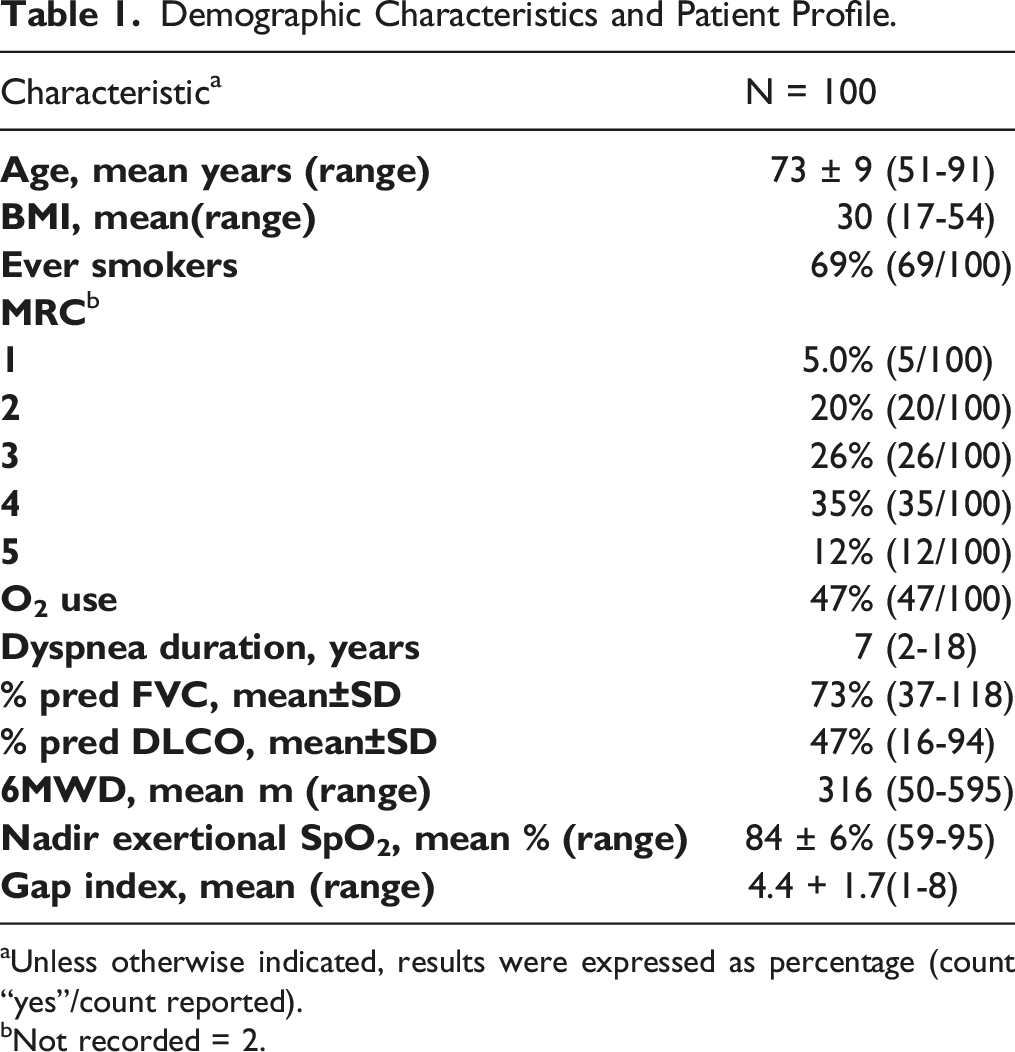
aUnless otherwise indicated, results were expressed as percentage (count “yes”/count reported).
bNot recorded = 2.
Edmonton Dyspnea Inventory Component Scores.

aBaseline: rest, talk, eat
bIncident or episodic dyspnea: bowel movement, dress, shower, exercise, stairs.
cCrisis dyspnea was reported by 11 patients across MRC grades (MRC 1 = 1, 2 = 1, 3 = 2, 4 = 3, 5 = 4 patients).
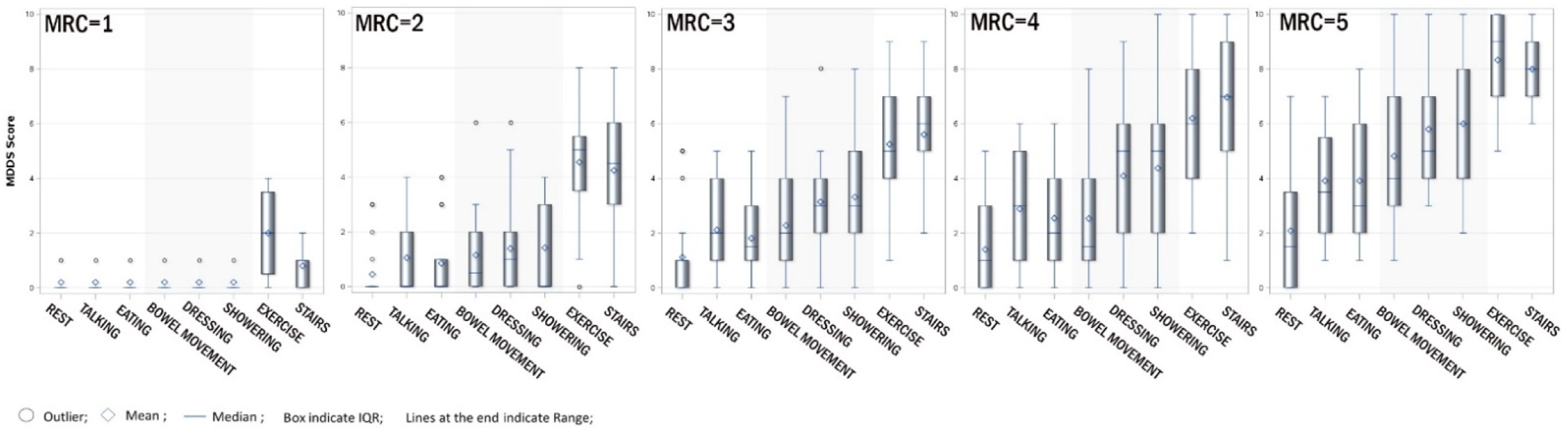
Distribution of MDDS scores within each MRC grade.
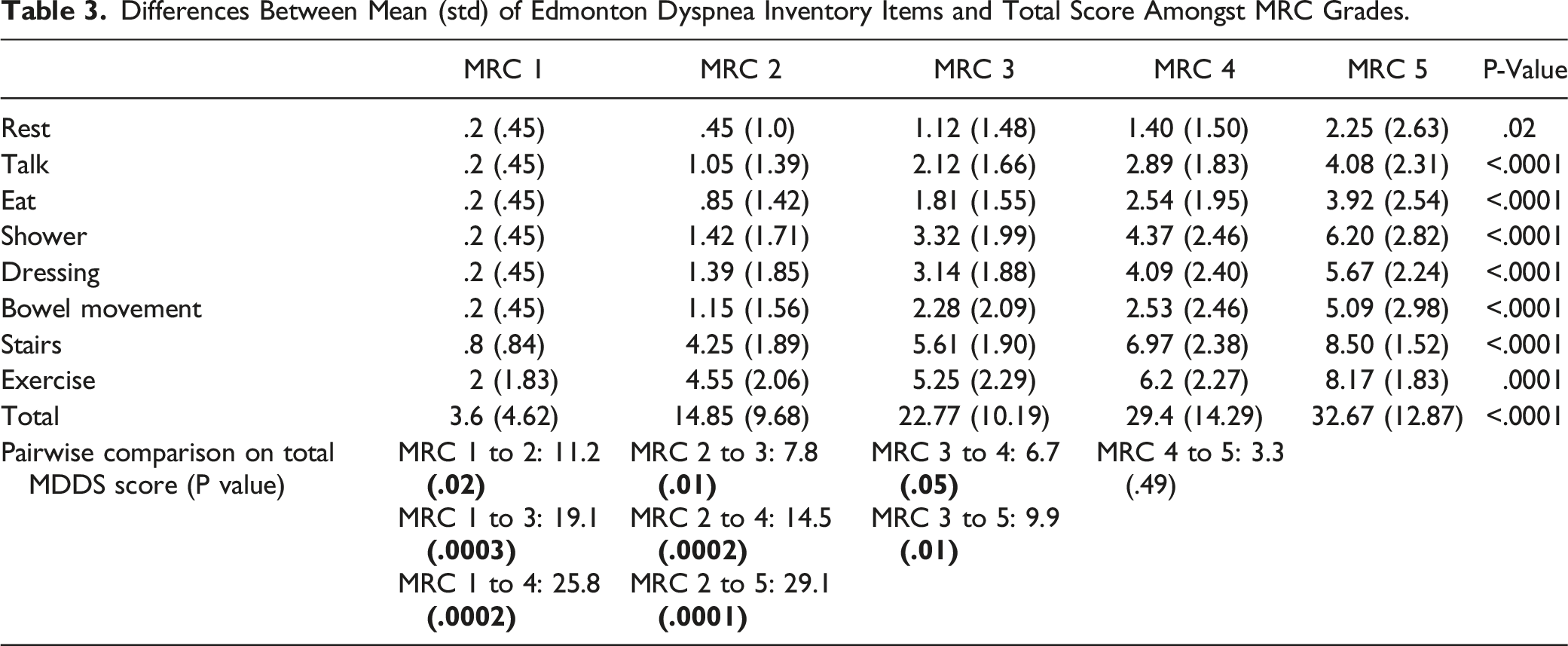
Differences Between Mean (std) of Edmonton Dyspnea Inventory Items and Total Score Amongst MRC Grades.
Dimensionality, Structure and Internal Consistency
Item Analysis and Exploratory Factor Analysis Results for Edmonton Dyspnea Inventory.

Note: The Cronbach alpha is .915.
Correlations Among Dyspnea Instruments and Lung Function Measures.
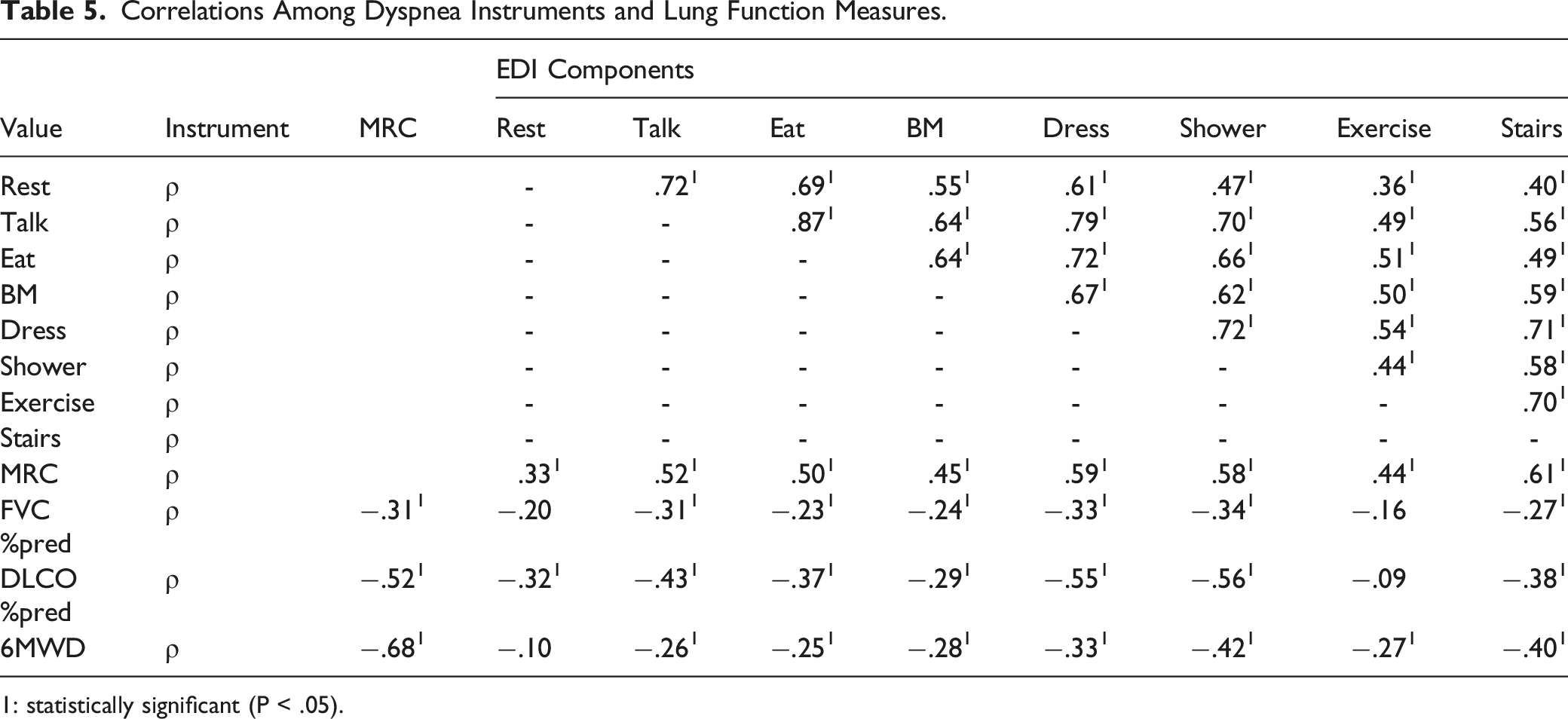
1: statistically significant (P < .05).
Examination of Response Structure
The GBTM was optimized with three groups of dyspnea behaviour based on the EDI item scores (Figure 2). Group 1 patients reported no or very low dyspnea during their daily activities and dyspnea was only rated higher during the higher intensity activities. Group 2 patients were grouped around increased dyspnea with moderate intensity activities and Group 3 patients had mild to moderate dyspnea when at rest, and with increasing activity intensity. All three EDI trajectory groups are distributed within MRC grade 2-4, and in general, the proportion of patients in Group 3 increased with increasing MRC grade (Figure 3). Figure 1A-B (Supplement) shows the individual MDDS responses for each patient in MRC 2 and MRC 5 as examples, the individual scores are coloured by their trajectory group. Trajectories of dyspnea behavior. Distribution of Dyspnea trajectory grouping within MRC grades.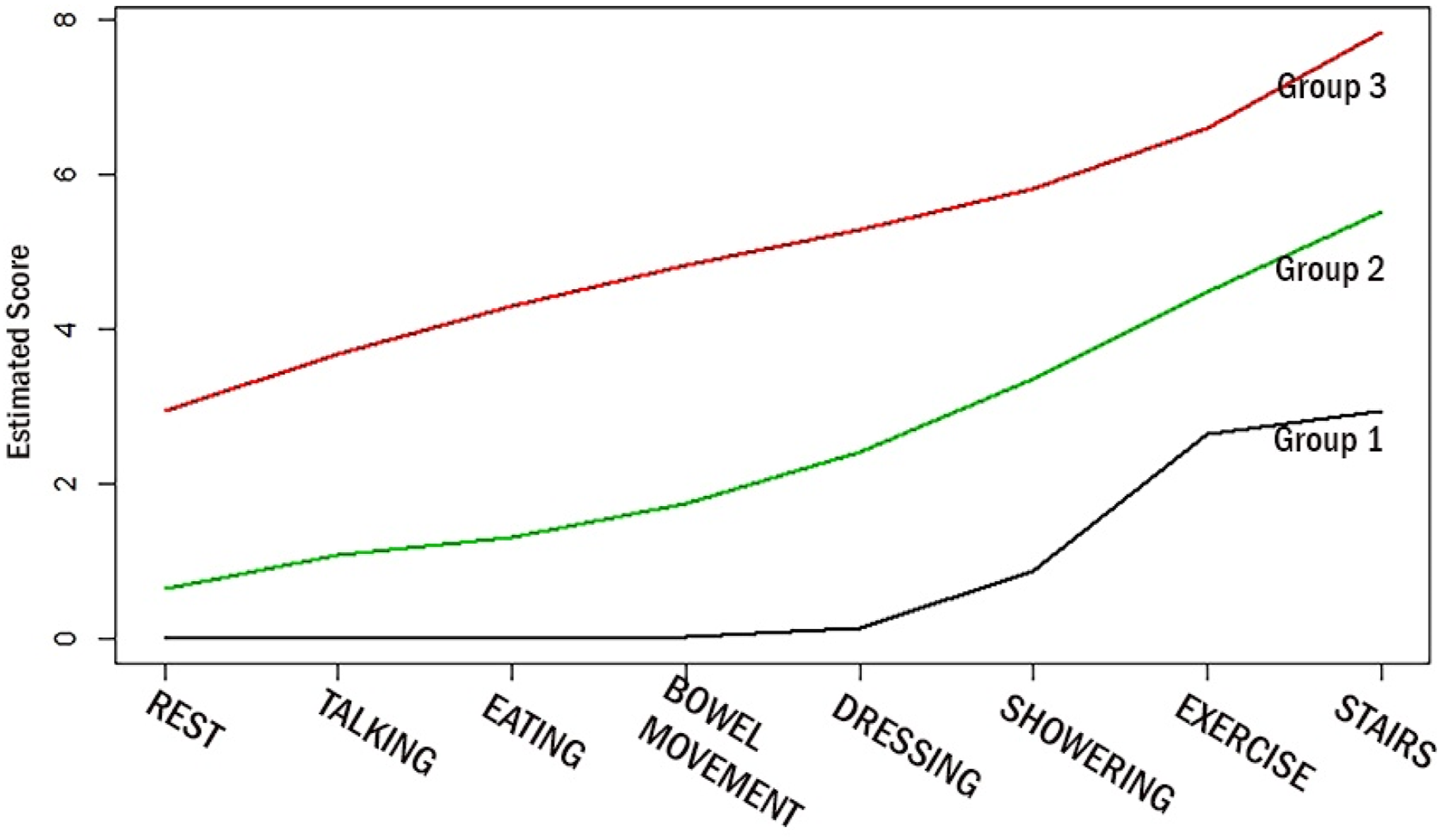
Clinical Features of various dyspnea trajectory groups as identified by Edmonton Dyspnea Inventory.
Validation
We examined the EDI score between the levels of MRC grade, each item demonstrated a significant increase in EDI Score across the levels of MRC (Table 3). In post-hoc analysis we found that the total differed for each level except between MRC 4 and 5. Correlation between all EDI items and the MRC was moderately strong (Table 5). Correlations between EDI items and FVC% or DLCO% were generally weaker, but all were in the hypothesized direction, and many were statistically significant. For FVC%, DLCO% and 6MWD, the strongest correlation was with showering component of EDI (ρ = −.34, −.56 and −.42 respectively, Table 5) establishing convergent validity.
Survival Analysis
One-year mortality was increased for patients in group 2 and 3, P = .0091 (Figure 4) suggesting worse survival with increasing dyspnea severity. To examine the additional benefit of dyspnea severity rating on mortality prediction we compared 2 sets of logistic models on predicting 1-year mortality in IPF. First, we assessed improvement by adding EDI grouping to MRC grade. Logistic model with MRC grade only had a c-statistic of .86 for 1-year mortality prediction. The addition of the EDI grouping to MRC score improved the c-statistic to .90 and had an NRI of .66 (95% CI -.11-1.11). Mortality distribution amongst MRC grades and EDI groups is shown in Supplement Table 1A-B. Second, we assessed improvement by adding EDI grouping to the logistic model predicting 1-year mortality using age, gender and FVC at baseline. Model without EDI grouping had a c-statistic of .78. Addition of EDI grouping improved the c-statistic to .86 and had an NRI of .88 (95% CI 0.45-1.11) indicating EDI aided dyspnea severity rating may improve survival estimates in IPF. Kaplan-Meir Survival Curves based on dyspnea severity.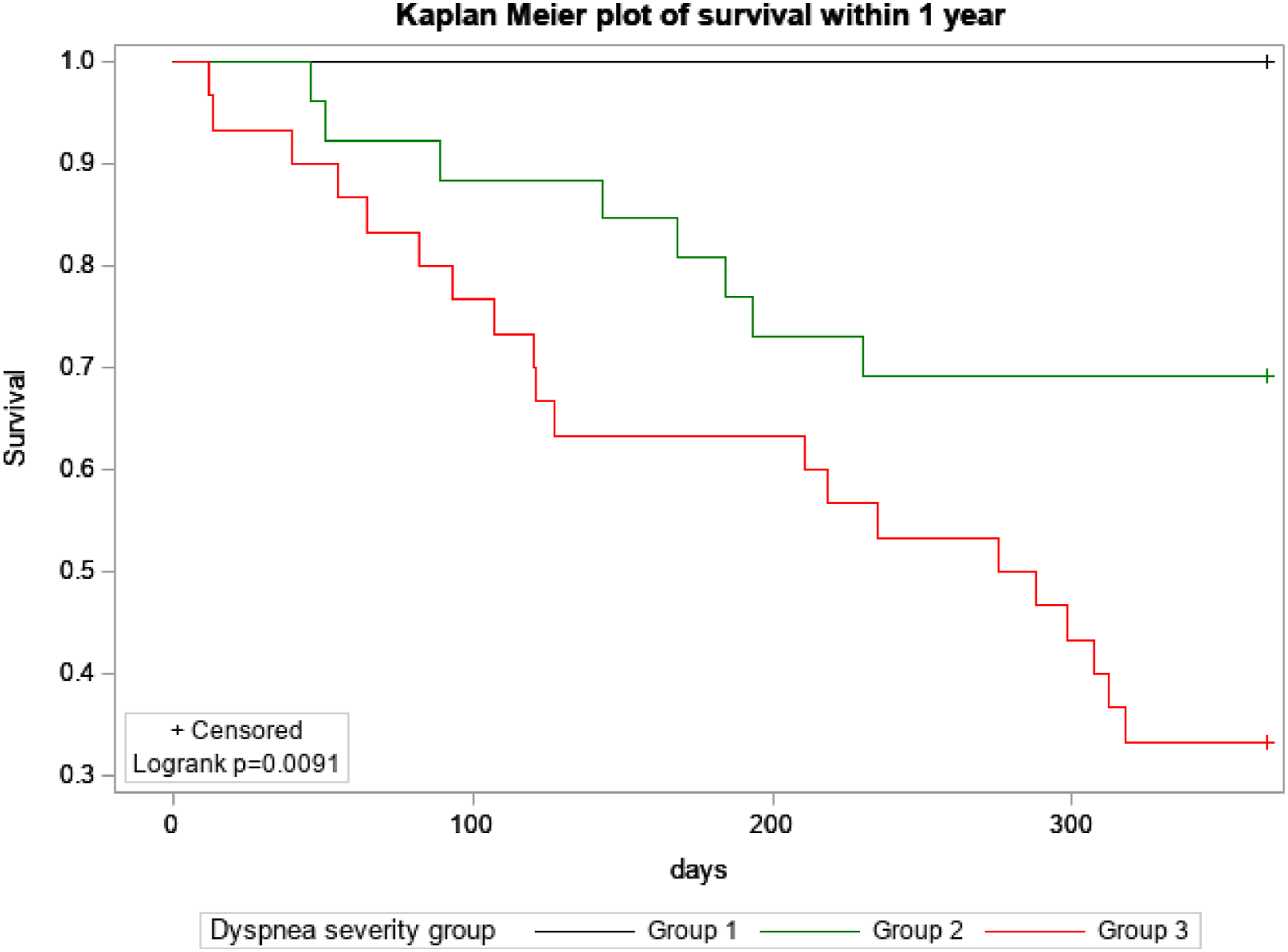
Discussion
We have developed the Edmonton Dyspnea Inventory, a brief, easily-administered, clinically relevant, patient-centered unidimensional tool whose scores possess the validity to assess dyspnea severity in the clinical (or research setting) in patients with IPF. Dyspnea is a prevalent, highly disabling symptom and a strong driver of quality of life impairment in patients with IPF. Despite the availability of myriad scales to assess it, dyspnea remains under-assessed and under-treated in practice. Many professional societies including a recent publication specific to ILD has highlighted the need to use a numeric rating scale to assess dyspnea severity and its impact on daily living and physical function. 11 The EDI answers this call.
Our analyses confirm that the EDI is a unidimensional questionnaire whose items, importantly, tap activities that most (if not all) patients with IPF perform on a daily basis – and thus, can respond to directly without having to guess what their level of dyspnea might be while doing it (as suggested in the directions for other dyspnea instruments). The exploratory factor analysis supported the use of a single total score, and the internal consistency analysis showed that the EDI exceeds the psychometric benchmark (0.7). 22
Validity of EDI scores for assessing dyspnea severity in IPF is supported by the general pattern of correlations with another dyspnea measure (MRC) and with metrics hypothesized and previously shown to be associated with dyspnea in this patient population (FVC%, DLCO% and 6MWD): a number of correlations (28 of 32) were statistically significant, and all were in the hypothesized direction. Interestingly and somewhat surprisingly, MDDS scores for exercise was not correlated with FVC or DLCO. This may be due to the non-standardized nature of exercise as reported by patients. Some patients followed a standard regime of home exercises (treadmill, exercise bike etc.), others did gentle stretches and for some walking to their mailbox constituted exercise. Our results reinforce the observation by Gronseth and colleagues that lung function explains only a minimal amount of variability in dyspnea ratings, thus we do not expect EDI scores to correlate strongly with pulmonary physiology. 23 Dyspnea is a complex perception influenced by cognitive factors, illness beliefs, and expectations or priors; chronic breathlessness results in various forms of psychological and physical adaptions, therefore it is expected that dyspnea will be influenced by many other factors rather than physiology alone. 24 Finally, validity of EDI is also supported by the GBTM analysis, which identified three groups of patients solely from trends in EDI scores at baseline, who also showed differences in disease severity and treatments including supplemental oxygen needs and prognosis. Higher dyspnea severity as measured by EDI was not only associated with worse survival in IPF but appeared to improve the predictive ability of a model that included baseline age, gender and FVC. This needs further examination in a prospective controlled study but does support its clinical utility and relevance. On top of these, the total score of EDI items varied among the MRC grades and covered different ranges; this may provide the opportunity to use EDI total score as single point dyspnea assessment for research purposes.
MDDS May Improve Care and Research
An aspect of dyspnea in patients with IPF that the EDI captures but one that does not contribute to or influence the current set of validation analyses is dyspnea crisis [30, 31]. From a clinical perspective, identifying the frequency, severity, context and triggers of dyspnea crisis is essential for personalized management. 15 Sixty-four % of crisis dyspnea episodes detected by EDI were in MRC grades 4 and 5; however, patients in the lower MRC grades 1-3 also reported at least one crisis episode signaling the need to explicitly ask about these events regardless of disability level as indicated by MRC. 15 This has not been previously reported in IPF and highlights a missed opportunity that, if addressed, could potentially reduce hospitalizations and related costs.25,26 Additionally, knowledge of crises can inform development of personalized action plans to improve care. 27 Thus EDI answers the call from an ATS workshop report to assess dyspnea intensity using an instrument to facilitate patient centered care. 15 Unlike, other dyspnea scales our tool can also aid dyspnea management. We have previously shown that EDI use in our clinic led to early and systematic implementation of non-pharmacological intervention, initiation and/or adjustment of oxygen therapy, and the prescription of opiates.28-30 Systematic dyspnea assessment and management has been identified as an urgent need. We believe that EDI can be easily adapted to digital application-based symptom assessment for point-of-care use or home monitoring by patient for tracking symptoms and thus enable self-management.
Research trials of dyspnea interventions in IPF have used MRC or mMRC grade as inclusion criteria or outcomes.31-33 Our work demonstrates the heterogeneity of dyspnea among patients in any one MRC grade; therefore, we propose that the concurrent use of EDI may facilitate subject stratification and cohort enrichment for more severe dyspnea and its response structure could promote better signal detection in trials of therapeutic efficacy.
Limitations of this study include its single center, retrospective design. However, we used a battery of analyses whose results support the EDI as a tool that generates scores with requisite validity for assessing dyspnea severity in patients with IPF. The EDI was amended to include “walking across the room”, and additional studies are needed to support the validity of the 9-item version, to assess test-retest and generate minimal important change thresholds. Also, more research is needed to examine the applicability of EDI in clinics other than ours and to assess how its scores influence clinical decision-making. A multicenter prospective validation study is underway.
Despite these limitations, we have shown that EDI scores are valid estimates of dyspnea severity in patients with IPF. Its scores are correlated with other direct and indirect measures of dyspnea; they distinguish subgroups of patients with differing levels of dyspnea severity; and they are associated with longitudinal disease behavior. In the clinical arena, EDI scores can facilitate personalized dyspnea management.
Supplemental Material
Supplemental material - Validation of a Novel Clinical Dyspnea Scale – A Retrospective Pilot Study
Supplemental material for Validation of a Novel Clinical Dyspnea Scale – A Retrospective Pilot Study by Meena Kalluri, Ying Cui, Ting Wang, and Jeffrey A. Bakal in American Journal of Hospice and Palliative Medicine®
Footnotes
Acknowledgments
The authors wish to acknowledge Dr. Janice Richman-Eisenstat for her clinical contributions and Ms. Sarah Younus for data collection.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
